Abstract
Background
This study aimed to explore the expression and clinical implication of guanine nucleotide-binding protein alpha 13 (GNA13) in esophageal squamous cell carcinoma (ESCC).
Methods
We first employed western blot analysis to test the GNA13 protein expression level in ESCC tissues. Subsequently, we used immunohistochemistry assays to detect the GNA13 in ESCC specimens from 173 patients who underwent esophagectomy. Survival analysis was performed to define the impact of GNA13 expressions on the prognosis of the ESCC patients based on the clinical and follow-up data.
Results
The GNA13 protein was shown to be considerably higher in ESCC tissues than in normal esophageal tissues. The level of expression was closely related to the tumor, node, TNM stage, and tumor size. More importantly, ESCC patients with high GNA13 expression carried an increased risk of tumor recurrence compared to those with low GNA13 expression. In addition, a high GNA13 expression level could independently predict worse overall survival and disease-free survival in ESCC.
Conclusions
GNA13 could be a novel prognostic biomarker for ESCC patients after esophagectomy.
Introduction
Esophageal carcinoma is a highly aggressive malignancy, which is the sixth cause of cancer-related mortality in the world. 1 Esophageal squamous cell carcinoma (ESCC) is a predominant pathologic type that accounts for over 70% of all cases.1,2 Most ESCC diagnoses are made at an advanced stage, and only 20% of patients are eligible for radical esophagectomy. 3 However, patients who underwent surgery alone still have poor outcomes, with a local recurrence rate ranging from 20.5% to 43.0%. 4 The tumor, node, metastasis (TNM) staging system remains the standard for treatment options and prognostic prediction. 5 However, due to several limitations, patients with ESCC with the same TNM stage and treatment modality may present different therapeutic effects and prognosis. Therefore, it is necessary to find new biomolecular markers that can predict the tumor malignancy, as well as the prognosis of ESCC patients.
G protein-coupled receptors (GPCRs) are the largest family of cell surface signal receptors, which play critical roles in multiple physiological processes in human diseases, including cancer.6,7 The GPCRs function by interacting with heterotrimeric G proteins, which are categorized into Gα, Gβ, and Gγ subunits. Indeed, the G12 family (comprising GNA12 and GNA13), one of the subfamilies of the Gα subunit, has proved to be closely associated with oncogene transformation and tumorigenesis. 8 In our previous study, we found that increased guanine nucleotide-binding protein alpha 13 (GNA13) would contribute to tumor invasion and metastasis in hepatocellular carcinoma (HCC), and it could be a biomolecular marker for poor prognosis of HCC patients after surgery. 9 Also, we identified that higher expression of GNA13 could promote proliferation and tumorigenicity in gastric cancer through modulation of G1/S cell cycle transition, thus resulting in a poor prognosis. 10 We also revealed that GNA13 could promote tumor metastatic spread in colorectal cancer by binding to epithelial-mesenchymal transition (EMT) activators. 11 In conclusion, existing data has demonstrated that GNA13 has become a new oncogene and could be an independent prognostic biomarker in multiple cancers. However, data on the expression and clinical implication of GNA13 in ESCC remain scant.
In this study, we performed western blot assays and immunohistochemistry (IHC) to explore the GNA13 protein expression in ESCC tissues. Further, the prognostic value of GNA13 in ESCC patients was analyzed in combination with the clinical data.
Methods
Patients and samples
ESCC and para-cancerous normal esophageal tissues were collected from 173 patients diagnosed with ESCC, who underwent surgery from 2007 to 2008 at the First Affiliated Hospital, Sun Yat-sen University, Guangzhou, China. Enrolled patients had no distant metastasis and had not received any antitumor treatment before surgical excision. The postoperative pathologic stage was I–III (T1N0M0-T4aN2M0). Tumor differentiation was graded according to the World Health Organization (WHO) classification
Western blot analysis
The ESCC protein lysates were collected and then separated using SDS-PAGE. After that, the proteins were transferred onto a polyvinylidene difluoride (PVDF) membrane and incubated with primary mouse monoclonal antibodies (1:1000 dilution; Abcam, Cambridge, MA, USA) against GNA13 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) overnight. Then the membrane was washed with tris-buffered saline (TBS) 3 times and finally incubated with the secondary anti-mouse antibody (Santa Cruz Biotechnology, CA, USA).
IHC assays
The paraffin-embedded ESCC specimens (5-μm) were dewaxed and rehydrated with xylene and different graded ethanol. Thereafter, the antigenicity of ESCC tissue was retrieved with EDTA (pH 6.5), then the endogenous peroxidase activity was blocked. Subsequently, a primary antibody (1:400 dilution; Abcam) against GNA13 was added and incubated overnight. The samples were washed with PBS, followed by incubation with the secondary antibody. Next, 3,3'-diaminobenzidine (DAB) was used to stain the sample, which was further counterstained with hematoxylin. The sections were washed under running water for 5 min, then dehydrated and mounted. Finally, a light microscope was used to analyze the tissue samples. The GNA13 expression was determined by IHC score, which was assessed manually by two independent pathologists. The final score was determined as the intensity score multiplied by the proportion scores. The scoring criteria were defined as: 0 = no staining, 1 = weak staining, 2 = moderate staining, and 3 = strong staining. Tumor cells were randomly chosen from five filed in each sample, and proportion scores were depended on the percentage of cells that were favorably stained: 0 (no cell stained), 1 (1–10%), 2 (11–50%), 3 (51–80%), and 4 (81–100%). The cut-off value for the final grouping was determined by the median of the score.
Statistical analysis
We used SPSS (Standard version 16.0) for statistical analyses in this study. The connection between GNA13 and clinicopathologic parameters was investigated using the Pearson chi-square test/Fisher exact test, Kaplan–Meier analysis, and log-rank test, which were performed to compare the prognostic differences between the groups. In addition, the impact of various factors on clinical outcomes was assessed using a univariate and multivariate Cox proportional hazard regression model. A P-value less than 0.05 was regarded as statistically significant.
Results
GNA13 expression in ESCC by western blot
We randomly choose 10 fresh ESCC tissues and adjacent normal esophageal tissues from 10 different patients. Our results demonstrated that GNA13 protein expression was substantially higher in ESCC tissues than in normal esophageal tissues (Figure 1(a)).
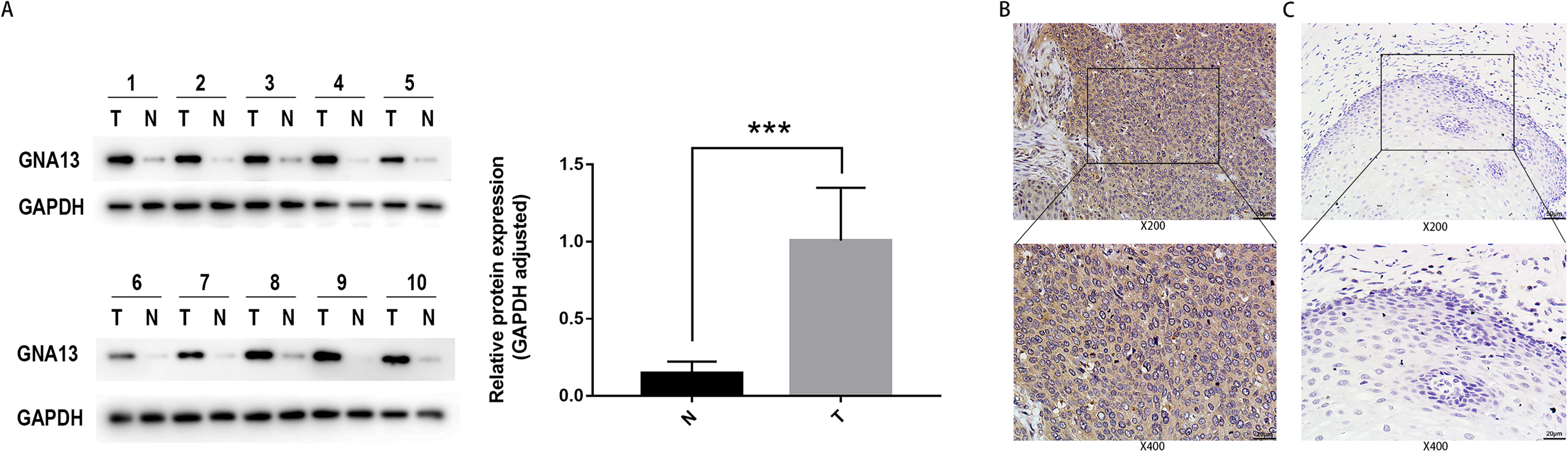
GNA13 protein is overexpressed in ESCC by western blot and immunohistochemical analysis. (a) Western blot analysis of GNA13 protein expression in 10 pairs of ESCC tissues (T) with their neighboring non-tumor esophageal tissues (N). The expression levels were normalized by GAPDH. ***P < 0.001 (b) IHC assay of the expression of GNA13 in ESCC tissues. (c) IHC assay of the expression of GNA13 in non-tumor esophageal tissues.
GNA13 expression in ESCC by IHC
We further detected the GNA13 expression of 173 postoperative ESCC patients using IHC. As the results show, high expression of GNA13 was expressed in 85 (49.1%) ESCC samples, which were mainly located in the cytoplasm (Figure 1(b)). In contrast, GNA13 was absent or weakly presented in most adjacent normal esophageal tissues (Figure 1(c)). Four groups of different immunostaining intensities are shown in Supplementary Figure 1. Patients were then divided into two cohorts for subsequent survival and prognostic analysis based on the IHC scores.
Association of GNA13 with clinicopathologic characteristics of ESCC
The baseline characteristics of patients were defined as shown in Table 1. All 173 ESCC patients included 134 males (77.5%) and 39 females (22.5%), ranging from 41 to 88 years old (mean, 60 years). The data revealed a strong connection between GNA13 expression level and T stage (P = 0.004), N stage (P < 0.001), TNM stage (P < 0.001), and tumor size (P = 0.018). Notably, the recurrence rate of the high GNA13 expression group was 75.3%, while that of the low GNA13 expression group was 44.3%; patients with GNA13 high expression carried a significantly higher risk of tumor recurrence than those with low GNA13 expression (P < 0.001). Other parameters, which included age, gender, pathological grades, and tumor site did not show any differences between the groups.
Correlation between GNA13 expression level and clinicopathologic indicators in ESCC patients.
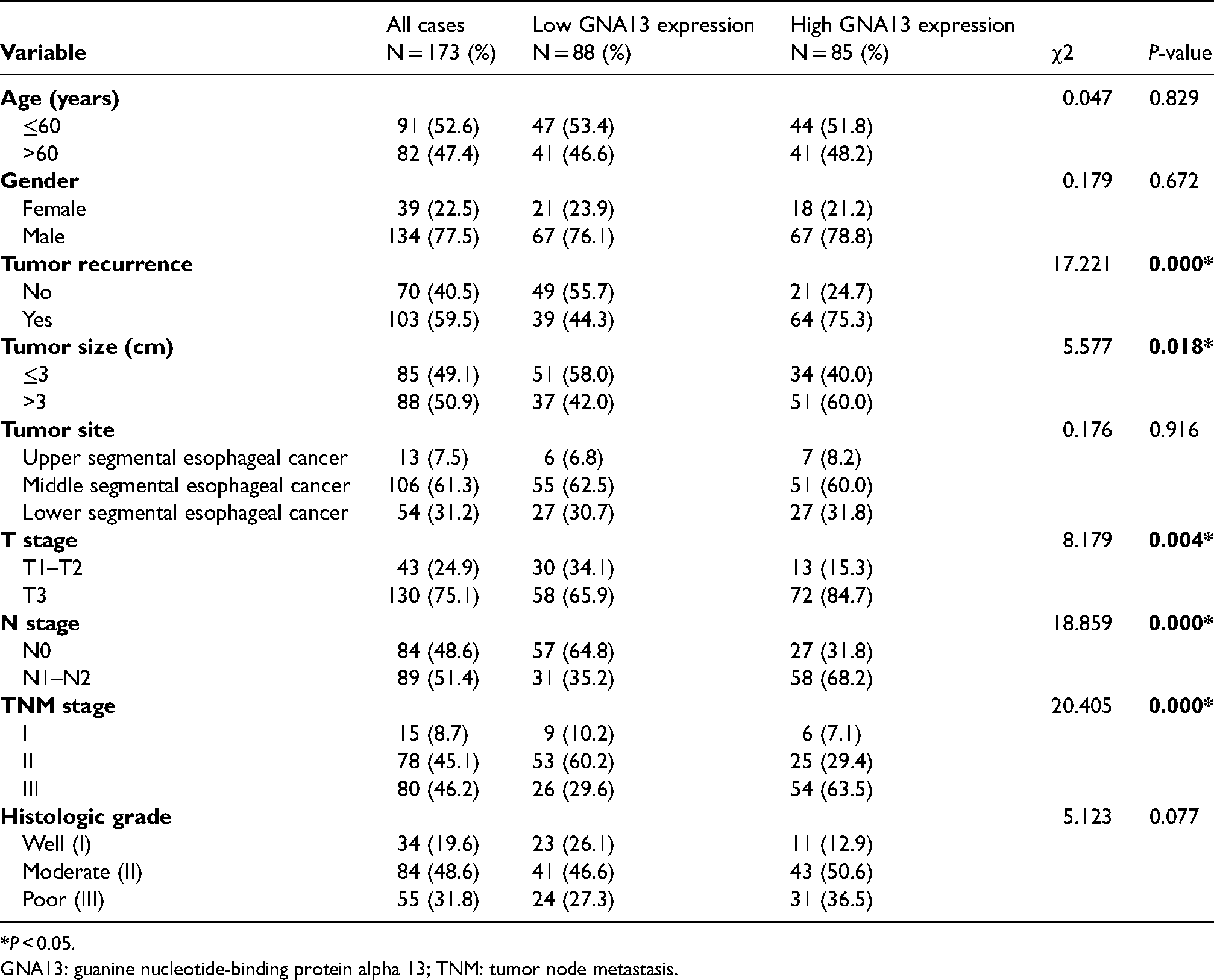
GNA13: guanine nucleotide-binding protein alpha 13; TNM: tumor node metastasis.
GNA13 expression and survival analysis
The difference in disease prognosis between patient groups is presented in Figure 2. The high GNA13 expression group had a median overall survival (OS) of 30.3 months compared to 54.9 months in the low GNA13 group (Figure 2(a)), and a median disease-free survival (DFS) of 16.4 months compared to 53.3 months in the low GNA13 group (Figure 2(b)). As shown, the high GNA13 expression group exhibited a shorter OS and DFS time than the low GNA13 group (OS, DFS: log-rank test, both P < 0.001). In addition, the high expression of GNA13 also can predict worse prognosis of ESCC patients with disease relapse (103/173, OS: P = 0.001; DFS: P = 0.008, Figure 2(c) and (d)), TNM stage III (80/173, OS: P = 0.005; DFS: P = 0.013, Figure 2(e) and (f)) and pathological grade III (55/173, OS: P = 0.017; DFS: P = 0.032, Figure 2(g) and (h)).
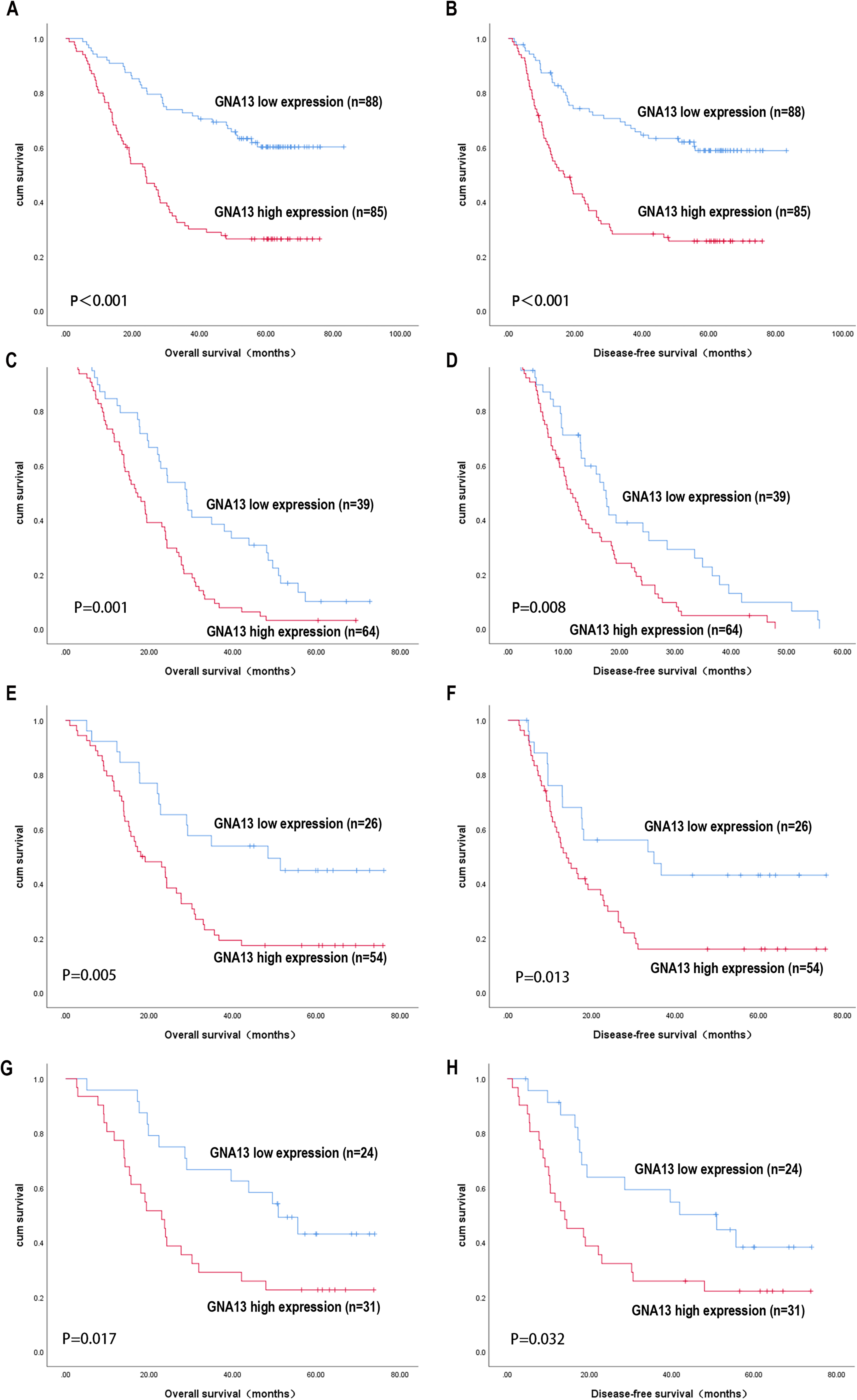
Kaplan–Meier survival analysis of overall survival and disease-free survival in ESCC patients with high vs. low GNA13 expression. Survival analysis in total ESCC patients (a) and (b); disease relapsed (c) and (d); TNM stage III (e) and (f); and pathological grade III (g) and (h) ESCC patients.
Univariate and multivariate analysis of OS and DFS
The prognostic prediction value of GNA13 is shown in Table 2 and Table 3. Our results showed that increased GNA13 expression, advanced T stage (III), TNM stage (III), pathological grade (III), and lymph node metastasis at baseline result in a worse prognosis. However, only high GNA13 expression (OS, DFS: P < 0.001) and lymph node metastasis (OS: P = 0.032; DFS: P = 0.006) were independent predictors of OS and DFS. On the other hand, the age, gender, tumor size, and tumor site did not exert a significant effect on prognosis by the univariate logistic regression model.
Univariate and multivariate Cox regression analysis of clinicopathologic variables in 173 ESCC patients for overall survival. .
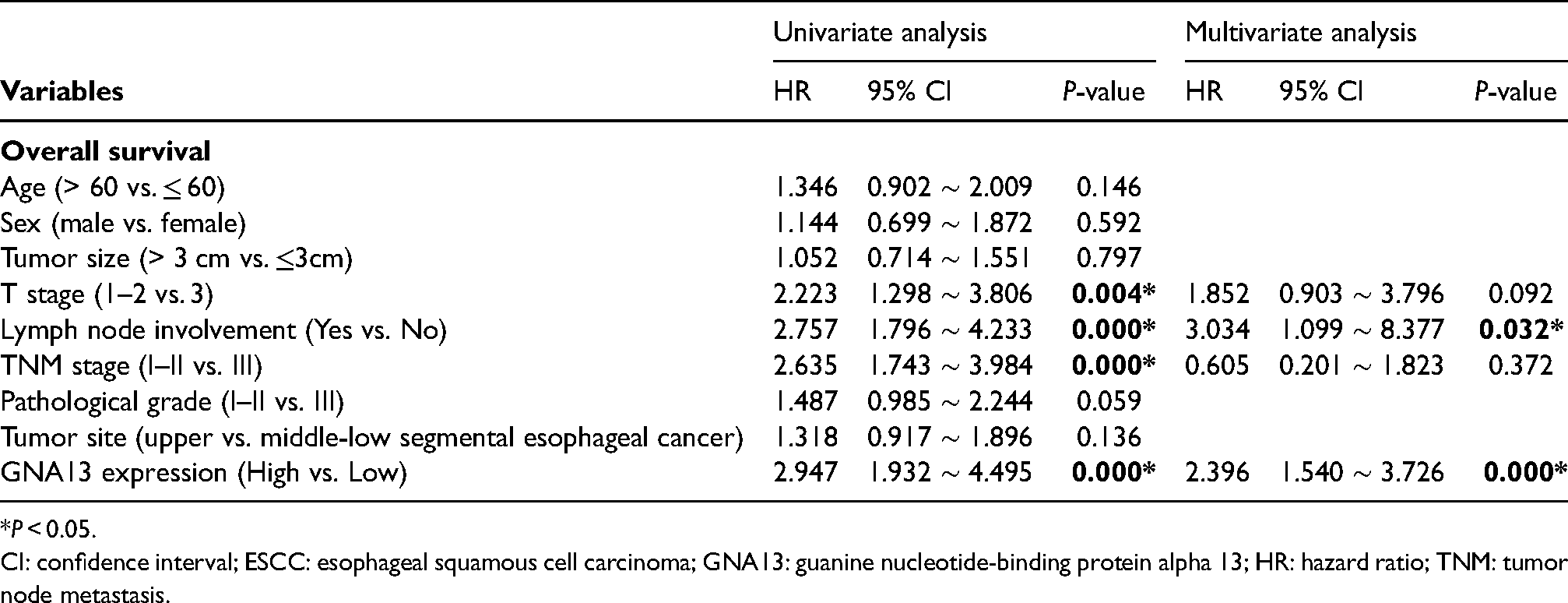
*P < 0.05.
CI: confidence interval; ESCC: esophageal squamous cell carcinoma; GNA13: guanine nucleotide-binding protein alpha 13; HR: hazard ratio; TNM: tumor node metastasis.
Univariate and multivariate Cox regression analysis of clinicopathologic variables in 173 ESCC patients for disease-free survival.
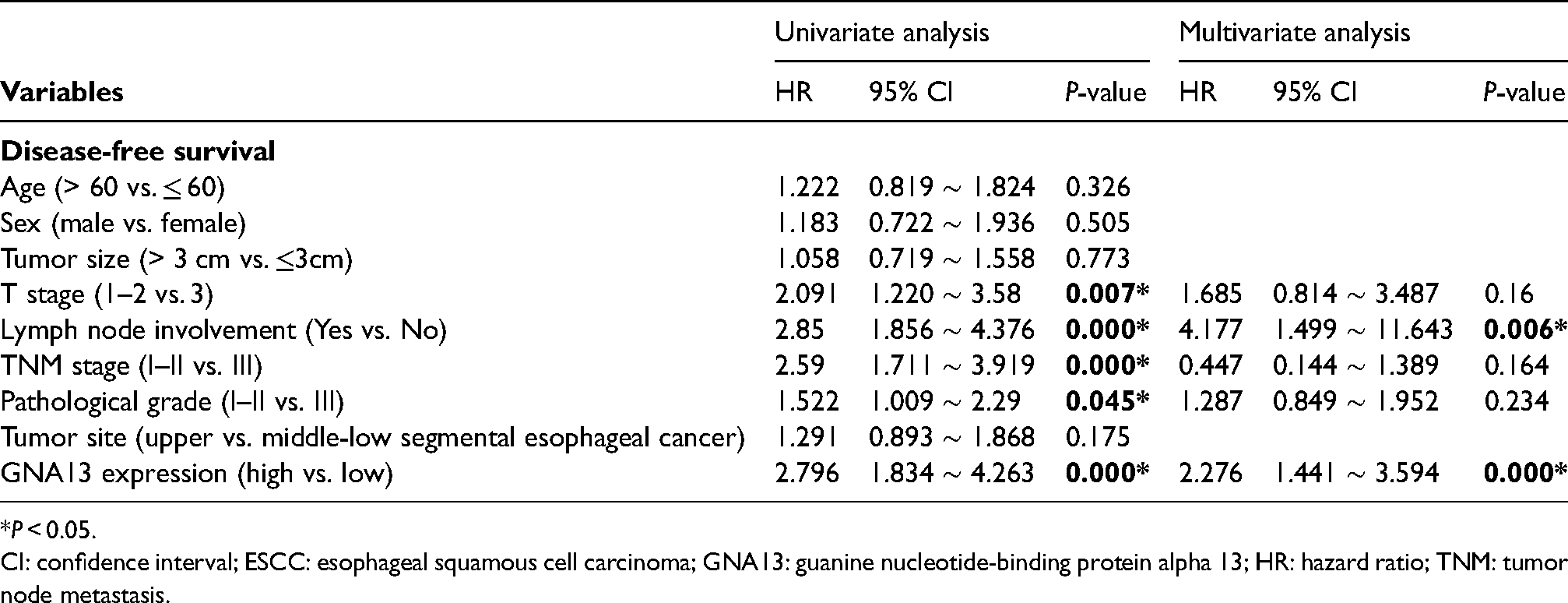
*P < 0.05.
CI: confidence interval; ESCC: esophageal squamous cell carcinoma; GNA13: guanine nucleotide-binding protein alpha 13; HR: hazard ratio; TNM: tumor node metastasis.
Discussion
ESCC is one of the most common malignancies seen in clinical practice. Radical surgery remains the standard treatment for patients with operable ESCC. In the case of lymph node metastases, a combination of chemotherapy or chemoradiotherapy is now recommended as postoperative adjuvant therapy or preoperative neoadjuvant therapy.
12
Also, immunotherapy as an adjuvant therapy can reduce the risk of disease relapse.
13
In general, a major cause of poor prognosis for ESCC is tumor recurrence and metastasis
GNA13 proteins belong to a subfamily of the G12 protein family.14,15 Like the GNA12, GNA13 has been proved to play a potential carcinogenic role in multiple human malignancies.16–18 In our previous study, we showed increased GNA13 expression in both gastric and hepatocellular cancer with a worse prognosis.9,10 Rasheed et al.19,20 proved that the expression level of GNA13 in prostate cancer and breast cancer is positively correlated with the aggressiveness of cancer cells. They also found that high expression of GNA13 was associated with more malignant biological manifestations in head and neck squamous cell carcinoma (HNSCC) and could induce the drug resistance to cytotoxic drugs. In addition, the downregulation of GNA13 or its downstream effectors could reverse this phenomenon. 21 These results provide potential strategies for drug resistance and treatment of squamous cell carcinoma. In contrast, in glioma, Liu et al. 22 found that overexpression of GNA13 could induce temozolomide sensitization, to improve prognosis. GNA13 mutations are less common than upregulation in solid tumors. However, GNA13 mutations have been reported in bladder cancer and hematological malignancies; especially in B-cell origin hematological malignancies GNA13 has presented a potential tumor-suppressive role.23,24 Thus, the expression patterns and effects of GNA13 vary in different types of tumors. In this study, we first found that GNA13 protein expression was prominently higher in the cytoplasm of ESCC tissue compared with the normal esophageal tissue. Also, GNA13 expression level was closely related to several indicators of poor prognosis including the depth of invasion, lymph node metastasis, TNM stage, and tumor size. Follow-up data suggested that high GNA13 expression patients were at a higher risk of tumor recurrence than those with low expression in ESCC. Moreover, we found that the TNM stage, and pathological grading were factors that affect the prognosis of ESCC patients after esophagectomy, but were not independent predictors. Instead, high GNA13 expression and lymph node metastasis were independent predictors for shorter OS and DFS. Also, subgroup analysis showed that overexpression of GNA13 could also predict poor prognosis in patients with advanced tumor stages, pathological grades, and in those with disease relapse. Due to the small sample size with single center data and the limited number of TNM I patients, residual confounding cannot be excluded. In subsequent experiments, the sample size should be expanded for further verification and analysis. Also, mechanism experiments are needed to investigate the role of GNA13 in ESCC occurrence, development, and metastasis, which will be solved in our future work.
In conclusion, this study demonstrated that GNA13 expression was overexpressed in ESCC tissues. Furthermore, GNA13 overexpression was an independent predictor of tumor recurrence and worse survival for ESCC patients who had undergone esophagectomy. Therefore, GNA13 could be utilized in conjunction with clinicopathological parameters to help distinguish different prognostic subgroups. More attention and closer follow-up are needed for ESCC patients with high GNA13 expression after surgery, and it may be appropriate to provide more aggressive adjuvant therapy for these patients.
Supplemental Material
sj-jpg-1-jbm-10.1177_03936155221106799 - Supplemental material for Overexpression of GNA13 correlates with poor prognosis in esophageal squamous cell carcinoma after esophagectomy
Supplemental material, sj-jpg-1-jbm-10.1177_03936155221106799 for Overexpression of GNA13 correlates with poor prognosis in esophageal squamous cell carcinoma after esophagectomy by Zichun Pan, Zhousan Zheng, Wen Ye, Cui Chen and Sheng Ye in The International Journal of Biological Markers
Footnotes
Acknowledgments
This study was supported by the Nature Science Foundation of China (No. 81772513).
Author contributions
Sheng Ye and Wen Ye contributed to the design of the experiment. Zichun Pan and Zhousan Zheng were involved in experiments and paper writing. Cui Chen and Wen Ye performed data acquisition and analyses. Zichun Pan and Zhousan Zheng contributed equally to this work and are co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nature Science Foundation of China, (grant number 81772513).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
