Abstract
Objective
This study aimed to investigate the role of the alpha fetoprotein (AFP) ratio before and after radiofrequency ablation (RFA) in the prognosis of patients with liver cancer.
Methods
A total of 368 patients who underwent RFA for liver cancer in Shenzhen People's Hospital from 2010 to 2020 were randomly divided into the training group and the validation group. Levels of AFP before and after RFA were recorded and their ratios were calculated.
Results
Using the X-tile software, it was found that the optimal cut-off value of the AFP ratio in the training group was 37.9. Both in the training group and the validation group, the relapse-free survival and overall survival of patients with an AFP ratio <37.9 (high-risk group) were significantly shorter than those with an AFP ratio >37.9 (low-risk group) (training group, relapse-free survival, P = 0.0003; overall survival, P = 0.0186; validation group, relapse-free survival, P = 0.0490, overall survival, P = 0.0031). An AFP ratio <37.9 was an independent risk factor for recurrence and survival of liver cancer after RFA.
Conclusion
The AFP ratio can predict the prognosis of patients with liver cancer after RFA. An AFP ratio <37.9 is an independent risk factor for tumor recurrence and survival after RFA.
Introduction
Liver cancer is one of the main causes of tumor-related deaths in China. 1 For liver cancer with limited small lesions, radiofrequency ablation (RFA) is used over hepatectomy because of high repetition, lower trauma, faster recovery, fewer complications, and shorter hospital stay.2,3 Due to the lack of complete tumor tissue to evaluate the therapeutic efficacy, current RFA technology may still be improved to overcome the disadvantage of a low local tumor control rate. 4 To date, no clinical indicator has been made available to evaluate the post-RFA prognosis of liver cancer. Serum alpha-fetoprotein (AFP), which serves as the main tumor marker of liver cancer, has been widely used in the clinical screening, diagnosis, monitoring, and prognostic evaluation of liver cancer.5,6 Previous studies have found that the change in AFP levels before and after hepatectomy for liver cancer can effectively predict liver cancer recurrence. 7 For patients with liver cancer, pre- to postoperative changes in AFP levels are more efficient than simple AFP levels as a prognostic indicator for liver cancer recurrence. 8 However, no studies have reported the range of pre- to post-RFA changes in AFP levels among patients with liver cancer. Therefore, this study retrospectively analyzed 368 patients with liver cancer treated with RFA in our hospital to explore the role of the pre- to postoperative AFP ratio in the post-RFA prognosis of liver cancer, aiming to provide a promising indicator for the therapeutic efficacy of RFA for liver cancer.
Subjects and methodology
Subjects
This study enrolled 368 patients with liver cancer who attended Shenzhen People's Hospital and received RFA as the first line therapy from 2010 to 2020. All patients were clinically diagnosed with liver cancer according to the American Association for the Study of Liver Diseases (AASLD) standard using computed tomography (CT) or magnetic resonance imaging (MRI) and pathological findings and met the indications for RFA. This study was approved by the Ethics Review Committee of Shenzhen People's Hospital.
RFA performance method
The RFA probe was placed directly inside the tumor under the guidance of CT or ultrasound, using either local or general anesthesia. For tumors that were larger than 3 cm, heat was increased throughout the entire lesion by inserting the RFA probe several times or at multiple sites at one time. For these large tumors, ablation was performed repeatedly at multiple sites and at multiple angles. For tumors that were smaller than 3 cm, a single probe was used at a single site. Therapy choices were decided during therapy according to the tumor size. The ablation area should include a 0.5–1 cm margin of apparently normal tissue adjacent to the lesion to prevent tumor metastasis and recurrence.
Indicator recording
AFP levels were measured using the ChemiLuminescence method. Chemiluminescence detection kits for the detection of AFP were purchased from Roche Diagnostics GmbH (Shanghai, China). The ChemiLuminescence method was performed strictly according to the guidelines provided by manufacturer. General clinical indicators and AFP levels were recorded 1 week prior to RFA, and patients’ postoperative AFP levels were recorded at reexamination 1 month after RFA. The pre- to postoperative AFP ratios were calculated. General clinical indicators, included sex, age, treatment history, Eastern Cooperative Oncology Group (ECOG) score, Child–Pugh score, albumin–bilirubin index (ALBI), 9 hepatitis B antigen, tumor size, and number of ablations, were also recorded.
Follow-up
Follow-up after RFA for liver cancer was performed according to the National Comprehensive Cancer Network (NCCN) guidelines, relying on enhanced CT or MRI and medical records to determine recurrence. The primary study endpoint is overall survival (OS), the secondary study endpoint is relapse-free survival (RFS). OS is calculated from the date of diagnosis to the date of death. RFS is calculated from the date of diagnosis to the recurrence after RFA.
Statistical methods
Statistical analysis was performed using SPSS 20.0. Two groups of continuous variables conforming to normal distribution were compared by t-test. Multiple groups of continuous variables were compared through one-way analysis of variance. The optimal cut-off for AFP ratios was obtained using the X-tile software. 10 The survival difference between the two groups was evaluated by Kaplan–Meier curves. Differences with P < 0.05 were deemed to be statistically significant.
Results
Clinical relationship between general clinical indicators and liver cancer recurrence in patients with liver cancer
This study included 368 patients with liver cancer, who were categorized into a training group (n = 184) and a validation group (n = 184) in a random manner. These was no significant difference between the two groups in any clinical indicator (Table 1).
Clinical characteristics of the training group and the validation group.
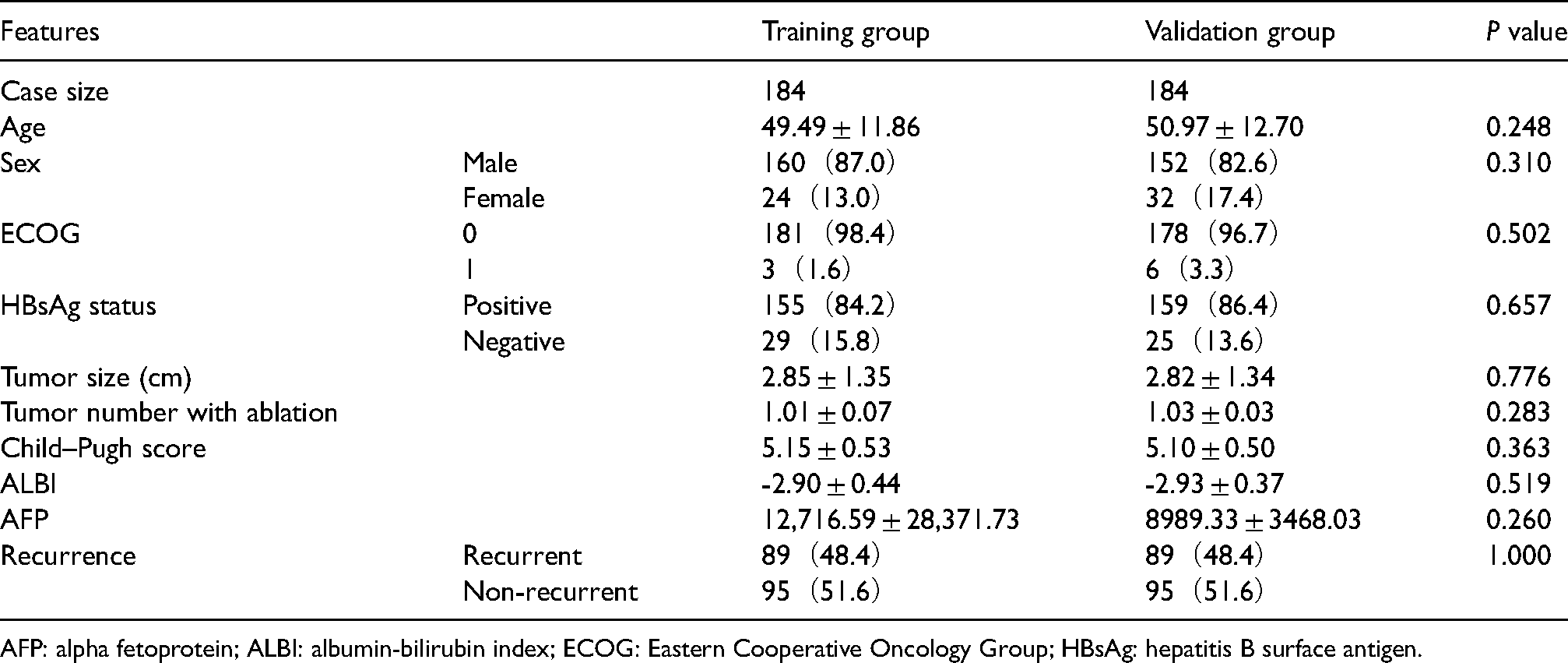
AFP: alpha fetoprotein; ALBI: albumin-bilirubin index; ECOG: Eastern Cooperative Oncology Group; HBsAg: hepatitis B surface antigen.
Relationship between AFP level and general clinical indicators in patients with liver cancer
Using the X-tile software, the optimal cut-off of AFP ratios was determined to be 37.9 in the training group. Comparison between the high- and low-risk subgroups in the relationship of AFP ratio with general clinical indicators (Table 2) indicated that the AFP ratio was associated with postoperative recurrence status and the AFP level.
Relationship between AFP ratio and the clinical characteristics.
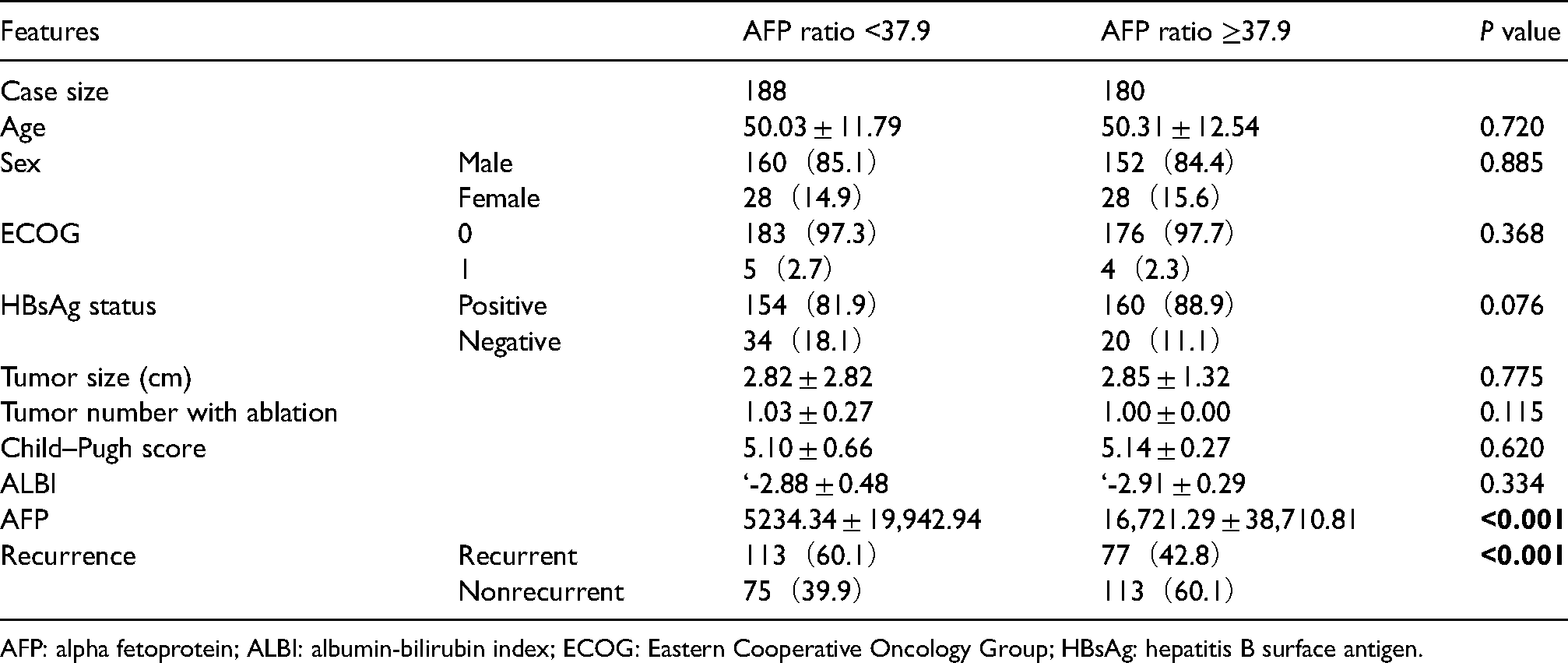
AFP: alpha fetoprotein; ALBI: albumin-bilirubin index; ECOG: Eastern Cooperative Oncology Group; HBsAg: hepatitis B surface antigen.
Relationship between post-RFA prognosis and AFP ratio in patients with liver cancer
The median follow-up time was 22.3 months. In the training group, the median RFS of patients with an AFP ratio >37.9 (low-risk subgroup, n = 90) was 44.47 months, whereas that of patients with an AFP ratio ≤37.9 (high-risk subgroup, n = 94) was 17.07 months, with a significant difference between the groups (P = 0.0003) (Figure 1). OS was also significantly higher in the low-risk subgroup than in the high-risk subgroup (P = 0.0186) (Figure 2). In the validation group, RFS was higher in the low-risk subgroup (n = 90) than in the high-risk subgroup (n = 94) (P = 0.0490) (Figure 3), with the two subgroups having a median RFS of 28.70 and 17.43 months, respectively. Similarly, the OS of patients in the validation group was higher in the low-risk subgroup than in the high-risk subgroup (P = 0.0031) (Figure 4).
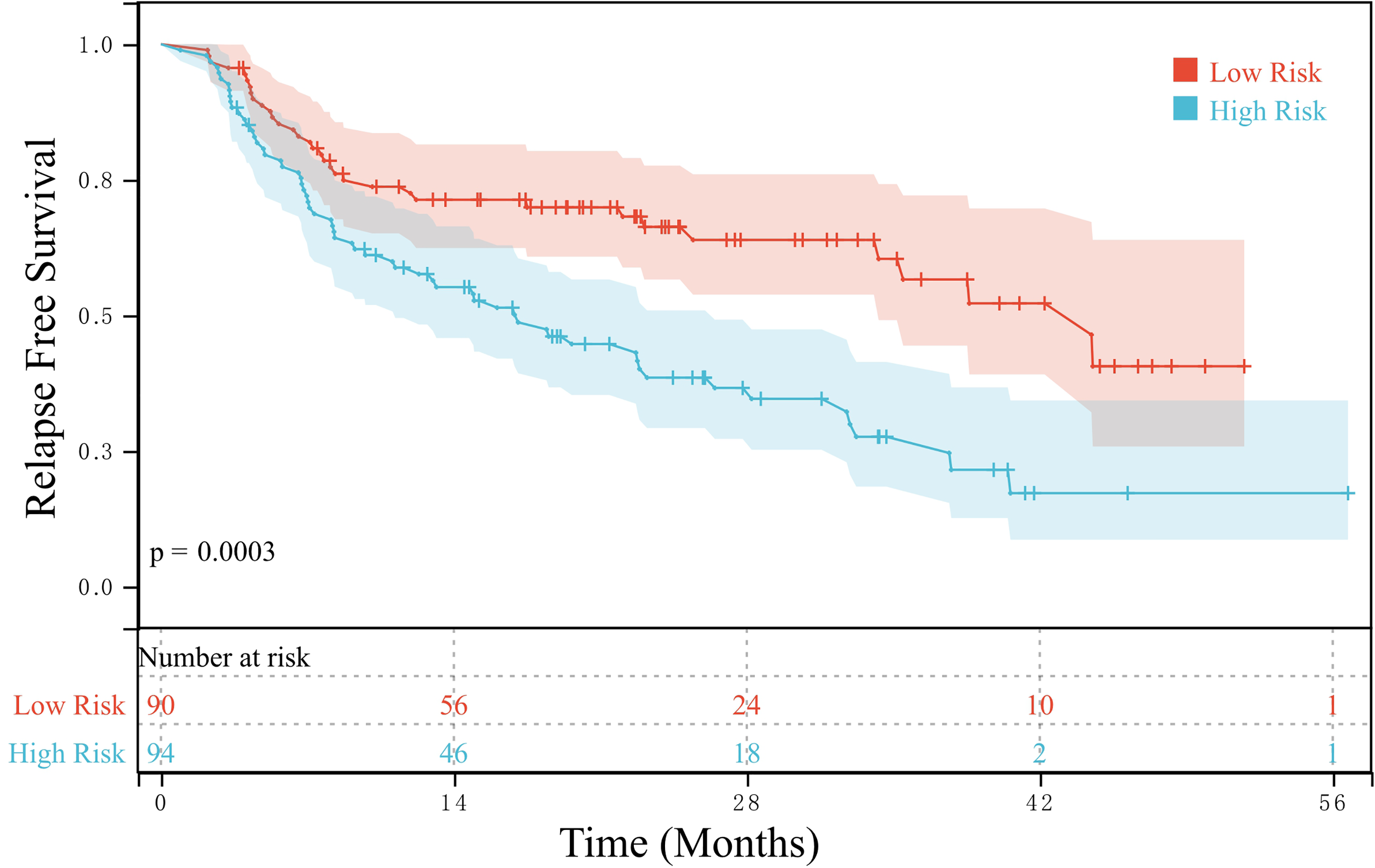
RFS of the high- and low-risk subgroups in the training group.
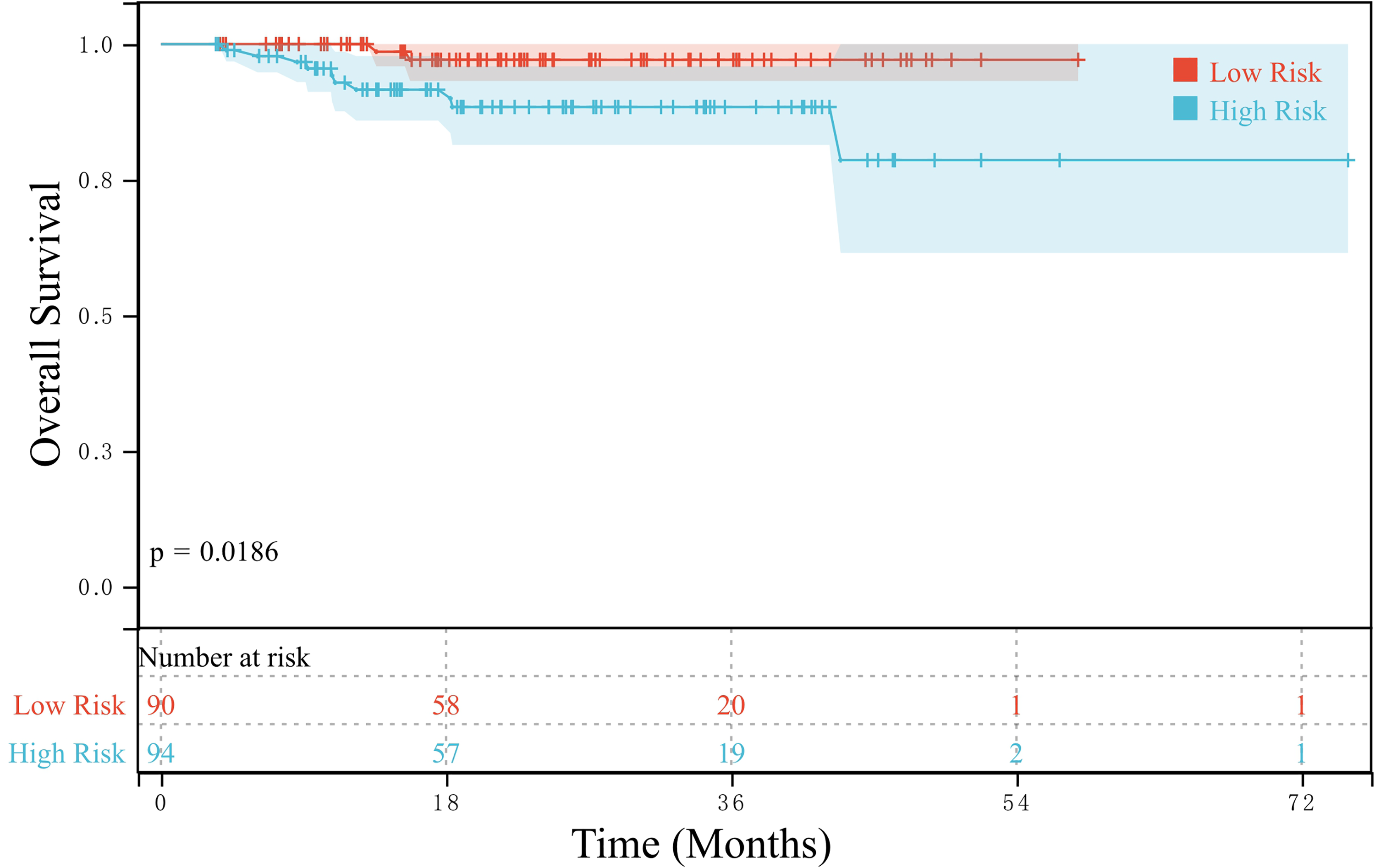
OS of the high- and low-risk subgroups in the training group.
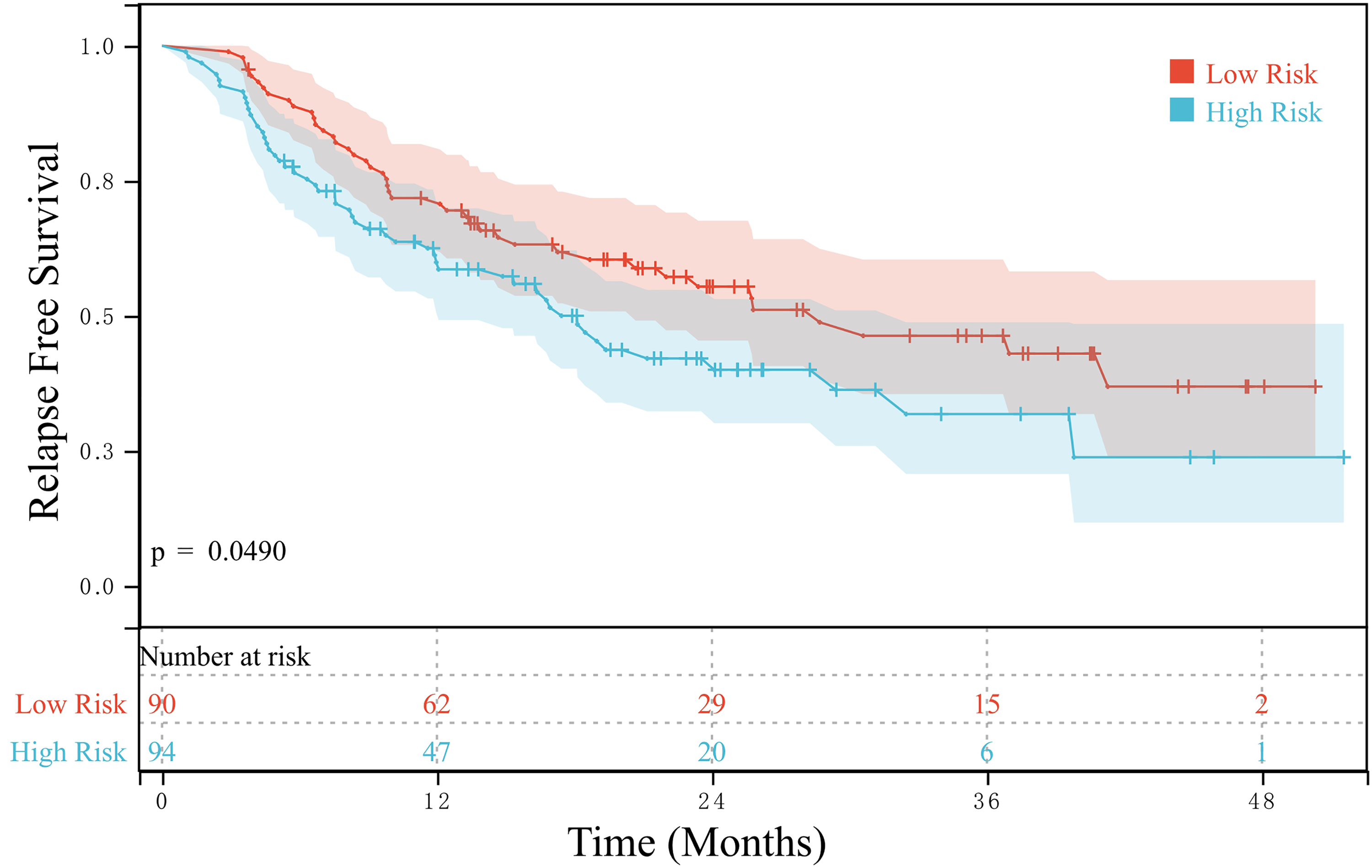
RFS of the high- and low-risk subgroups in the validation group.
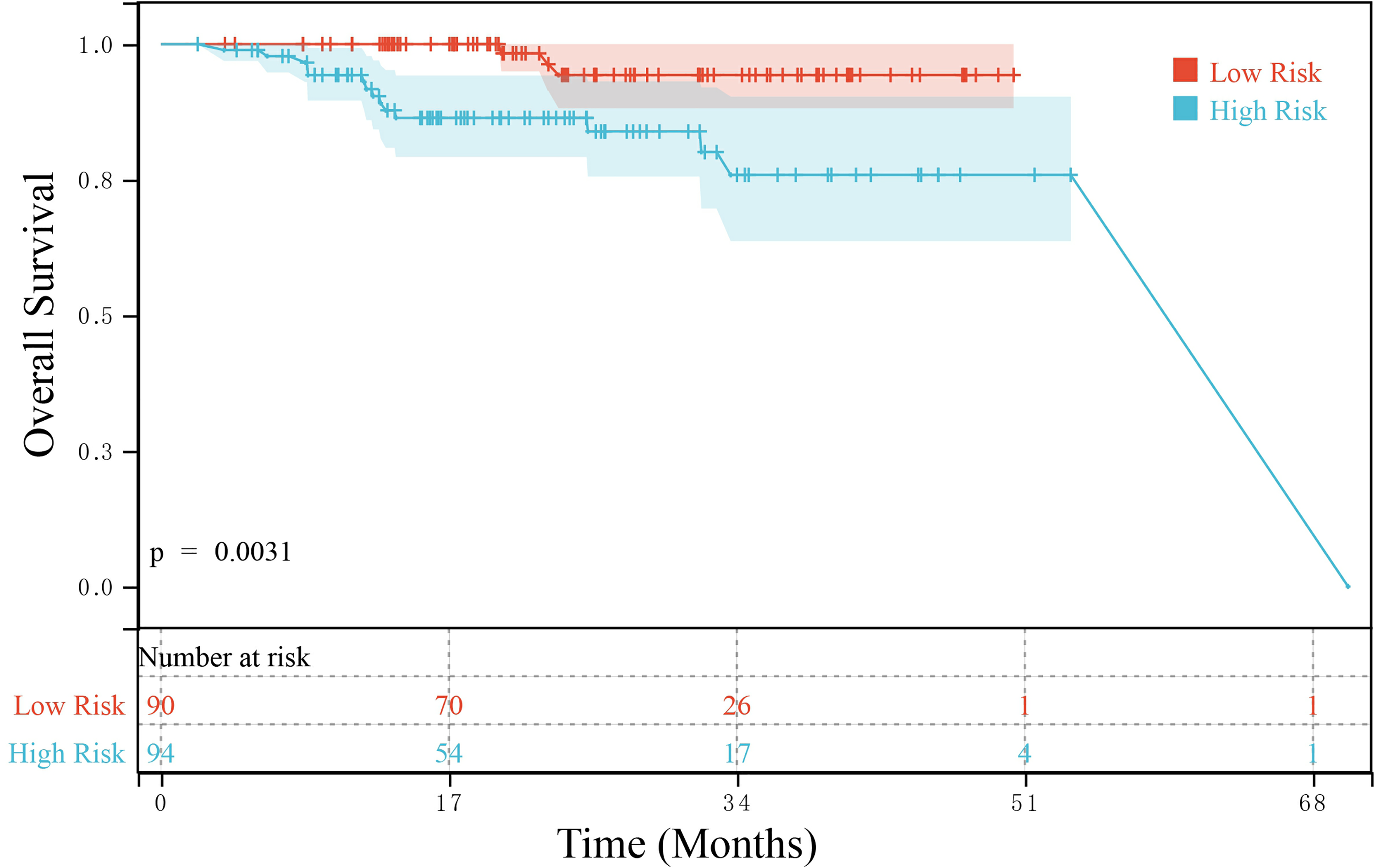
OS of the high- and low-risk subgroups in the validation group.
Independent factor analysis of post-RFA recurrence of liver cancer
The Cox proportional hazard regression model was adopted to analyze clinical indicators and AFP ratios of patients with liver cancer, wherein the significant factors identified on univariate analysis were included in the multivariate analysis. In terms of RFS, AFP ratio <37.9 (hazard ratio (HR) = 1.811 (95% confidence interval (CI): 1.289–2.545)) were identified as independent risk factors for post-RFA recurrence of liver cancer (Table 3). In terms of OS, AFP ratio <37.9 (HR = 4.421 (95% CI: 1.659–11.781)) was identified as an independent risk factor for post-RFA survival of patients with liver cancer (Table 4).
Risk factors for post-RFA recurrence of liver cancer.
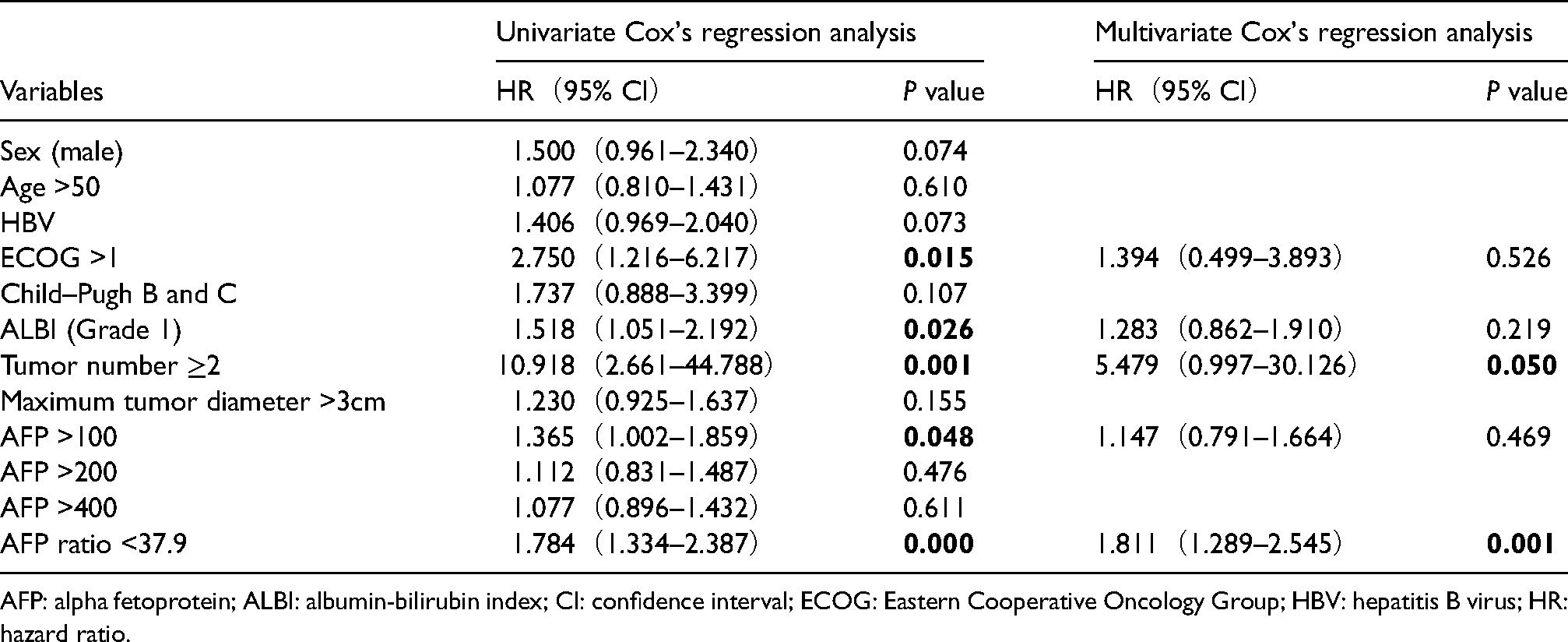
AFP: alpha fetoprotein; ALBI: albumin-bilirubin index; CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; HBV: hepatitis B virus; HR: hazard ratio.
Risk factors for post-RFA survival of patients with liver cancer.
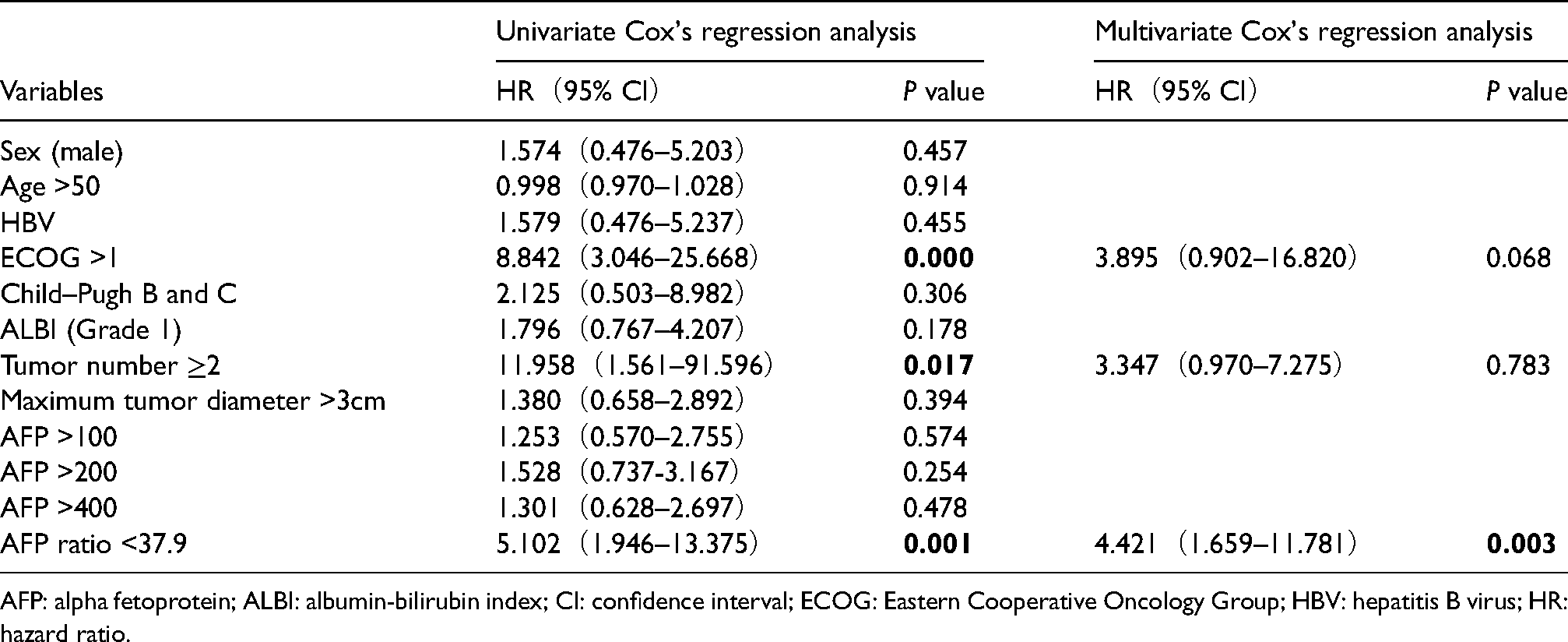
AFP: alpha fetoprotein; ALBI: albumin-bilirubin index; CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; HBV: hepatitis B virus; HR: hazard ratio.
Discussion
As part of precision surgery, RFA plays an important role in the personalized treatment of liver cancer. However, postoperative recurrence remains the main factor limiting its therapeutic efficacy. 11 The 3-year cumulative recurrence rate of liver cancer after RFA is as high as 53.4%. 2 The median OS of hepatocarcinoma (HCC) patients treated with RFA is significantly different than the median postoperative recurrence survival (62 month vs. 22 month). 12 There are many drugs, including sorafenib and angiotensin receptor inhibitors, that have been developed to significantly improve the prognosis of HCC after resection.13,14 However, the method to select the most suitable drug for patients as an adjuvant therapy, especially after RFA, remains short of study. Therefore, discovering risk factors and predictors of liver cancer recurrence after RFA is of great significance for the precise and personalized post-RFA management of liver cancer. Several factors have been reported to affect the post-RFA recurrence of liver cancer, including tumor size and preoperative AFP level. 15 In this study, patients who received RFA were randomly assigned into the training and validation groups. The pre- to postoperative AFP ratio could be used as an independent predictor of liver cancer recurrence after RFA in both groups.
A previous study has shown that tumor number, tumor size, Child–Pugh score, PS grade and lymphocyte-to-monocyte ratio are independent risk factors of HCC recurrence after RFA.12,16–18 However, in this study, tumor ablation number, tumor size >3 cm, Child–Pugh score, and ECOG grade were not found as independent risk predictors. Compared to previous studies, our study has a large sample size and high verification ability.
The present study revealed that AFP level fluctuations, especially pre- to postoperative changes, can serve as a predictor of post-RFA recurrence and survival in patients with liver cancer. Currently, no study has reported correlations between changes in AFP levels and post-RFA recurrence of liver cancer. However, previous studies have shown that postoperative AFP level fluctuations in patients with liver cancer can serve as a monitoring indicator of postoperative recurrence. 8 Nobuoka et al. 7 showed that a shift from positive AFP levels before hepatectomy to negative AFP levels after hepatectomy can be used to predict the postoperative recurrence of liver cancer. However, the feasibility of using pre- to postoperative changes in AFP levels as a predictor of post-RFA recurrence of liver cancer has not yet been explored. In the present study, the pre- to postoperative AFP ratio was used to present the pre- to postoperative changes in AFP levels. The higher the AFP ratio, the larger the postoperative decrease in AFP, and vice versa. Nobuoka et al. 7 have reported that regardless of whether the preoperative AFP level is normal, a pre- to postoperative increase in AFP levels is a predictor of poor prognosis. The present study also showed that pre- to postoperative changes in AFP levels can be used to assess post-RFA recurrence and survival and that the pre- to postoperative AFP ratio serves as a potential evaluation index. Univariate and multivariate analyses also revealed that the pre- to postoperative AFP ratio is an independent predictor of post-RFA recurrence and survival in patients with liver cancer. For the patients with a high AFP ratio, follow up frequency must be increased to identify early recurrences or metastases of the lesion, and give targeted therapy to increase patient survival.
Although the present study verified the usefulness of the pre- to postoperative AFP ratio as a predictor of post-RFA recurrence and survival in patients with liver cancer based on the randomization of patients into a training group and a validation group, it still had some limitations. First, this was a single-center retrospective study with some clinical symptoms missing and may also be subject to data bias. 19 Second, given the differences in AFP testing among different hospitals, more studies are required to verify whether the pre- to postoperative AFP ratio varies between differing reagents or equipment. As more clinical studies are conducted, the pre- to postoperative AFP ratio may be identified to play a greater role in the comprehensive treatment of liver cancer.
Footnotes
Ethical approval
This study was approved by the Ethics Review Committee of Shenzhen People's Hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Medical Science and Technology Research Fund, Shenzhen Key Medical Discipline Construction Fund, National Natural Science Foundation of China, (grant number No. A2021230, No.SZXK015, No. 52077216).
