Abstract
Skin tumours are among the cancer types most sensitive to immunotherapy, due to their unique immunogenic features including skin-associated lymphoid tissue, high mutational load, overexpression of tumour antigens, and high frequency of viral antigens. Despite this high immunotherapy response rate, however, ultimately most skin tumours develop similar treatment resistance to most other malignant tumours, which highlights the need for in-depth study of mechanisms of response and resistance to immunotherapy. A bibliographic review of the most recent publications regarding currently in use and emerging biomarkers on skin tumors has been done. Predictive biomarkers of treatment response, biomarkers that warn of possible resistance, and emerging markers, the majority of a systemic nature, are described. Including factors affecting not only genomics, but also the immune system, nervous system, microbiota, tumour microenvironment, metabolism and stress. For accurate diagnosis of tumour type, knowledge of its functional mechanisms and selection of a comprehensive therapeutic protocol, this inclusive view of biology, health and disease is fundamental. This field of study could also become a valuable source of practical information applicable to other areas of oncology and immunotherapy.
Introduction
The skin constitutes the external epithelial barrier of our body, which together with other internal barriers (intestinal, respiratory, and genitourinary), establishes the interface with environmental chemical substances, physical factors (e.g., UV-radiation) and biological threats (e.g., infectious microorganisms). It consists of a multitude of structures (skin appendages, vascular, and nerve plexuses, etc.), a complex immune environment and a specific microbiota, coordinated by sophisticated dermal regulation mechanisms. Skin has an extraordinary neural specialization, endocrine elements, and skin-associated lymphoid tissue (with functional properties and specific nesting), making it a psychoneuroendocrine-immune organ. These characteristics confer the skin with remarkable sensitivity and the ability to respond to physicochemical, infectious, and emotional stress.1,2
Normal immune response of the epidermis is mediated by three types of immune cells: Langerhans cells (LC), γδ T cells, and resident memory CD8+ T cells, which move among keratinocytes. In the dermis, numerous microorganisms live deep within the hair follicles and sebaceous glands surrounded by an immunosuppressive extracellular matrix (ECM), by regulatory T cells (Treg) and dermal dendritic cells (DC), which activate the immune response under pathological conditions. Injury, inflammation, infection, and tumor contribute to the establishment of skin-associated lymphoid tissue (SALT); vascular and nerve stimuli also act directly on immune cells. 3
Under normal conditions, immunogenic tumor clones developing in the skin are eliminated before they can evade the immune system. However, in patients with local or systemic alterations that entail immunosuppression (i.e., decrease in CD4 lymphocytes in HIV-AIDS patients, or decrease in CD8+ lymphocytes in solid organ transplantation patients) a significant increase in cutaneous tumors is observed. These immunosuppressed patients also show lower response to treatment and worse prognosis.
Tumors originating in epithelial elements, such as basal cell carcinoma, have an extraordinarily proliferative index, but can remain localized for long periods of time, showing little tendency to infiltrate and metastasize. On the other hand, tumors originating in melanocytes are recognized as malignant once mitosis appears, showing infiltrative and aggressive clinical behavior. Endocrine tumors, such as Merkel carcinoma, generally exhibit an infiltrative histological pattern and poor evolution, whereas similarly exuberant lymphoid lesions with rich immune responses classified as pseudo lymphomas involve physical, chemical, immunological, or infectious stimulation, but without any tumor. Skin sarcomas, lymphomas, melanomas, and primary cutaneous carcinomas have particular and complex immunogenic characteristics rarely present in other locations in the body. Figure 1 summarizes a range of the topics discussed in the current review, displaying a schematic representation of the skin as an organ, highlighting SALT and listing different factors that may play a role in the development of the main cutaneous tumor types. These particular skin tumor features make dermato-oncology immunopathology a distinctive area of cancer immunotherapy (IT) study.
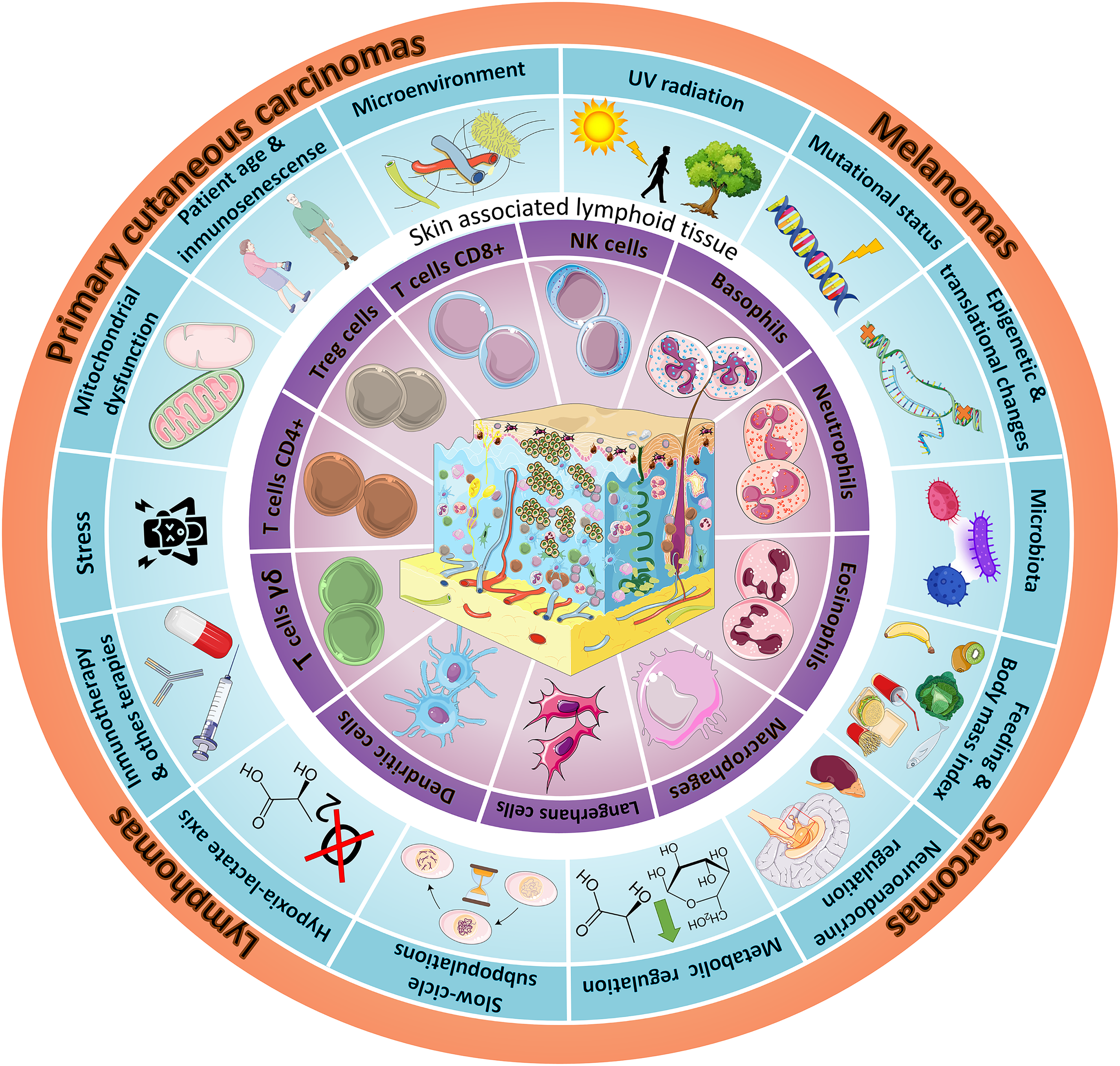
Schematic representation of the complex ecosystem of skin tumors, including the main epidermal, dermal and hypodermal structures (skin appendages, vascular and nerve plexuses, etc.), with particular focus on skin-associated lymphoid tissue (SALT) in the purple circle. The key factors involved in skin tumor development are listed and illustrated in the blue circle. The main types of cutaneous tumors are shown in the orange circle.
Cell markers for routine diagnostic dermatopathology
Dermatopathologists are routinely required to identify primary skin tumors rather than dermal metastases, at any location, making the use of genetic and immunohistochemical markers essential.4–6 It is crucial to perform a correct and accurate diagnosis of skin tumor typology before starting any treatment, especially IT. The structural and functional complexity of the skin results in greater diversity of neoplastic lesions than in other tissues, therefore requiring epithelial, neural, endocrine, or lymphoid markers to reveal the original linage of the tumoral cells. Identifying immunophenotype, structural elements (e.g., intermediate filaments), stromal cells, and vascular markers, are examples of such pathologist diagnostic tools to identify the precise tumor typology. Once the nature and precise classification of the tumor is defined, the specific management protocols and treatments for the tumor type can be proposed.
Skin tumors as a paradigm of IT
Skin tumors are highly sensitive to IT due to their immunogenic characteristics, which include high mutational load, overexpression of tumor antigens (Ag), and the frequent presence of viral Ag. Although IT response rates reach 40%, most tumors finally develop resistance, reducing the potential clinical advantage of skin tumor immunogenicity. Among multiple resistance mechanisms identified are those related to tumor cellularity, tumor microenvironment (TME), metabolic reprogramming, microbiota, plus neuroendocrine and immune dysfunction. 7 In light of this, research for a single definitive biomarker of IT response/resistance to guide therapeutic decisions seems unrealistic. 8
Here we provide an overview of IT response and resistance to (known and emerging) biomarkers in skin tumors to provide practical aspects for clinical management. Table 1 summarizes the biomarkers in skin tumors related to IT.
Biomarkers in skin tumors related to immunotherapy.

Markers of response to IT in skin tumors
TME and its rich ecosystem
A cutaneous tumor is a functional and interconnected tissue where malignant cells proliferate and migrate depending on their tumor niche or TME. This TME is generated by malignant cells themselves, stromal cells (fibroblasts, macrophages, lymphocytes, DC, vascular and neural cells, among others) and non-cellular elements, or ECM (fibrillar, non-fibrillar protein-glucidic structures and signaling molecules), which can have pro- or anti-tumoral effects. Nevertheless, response to IT is not determined by the specific quality of the TME alone, as tumor burden is also significant. Better results are expected when the tumor burden is low or microscopic, rather than larger (despite the sometimes immunogenic structure of big tumors). For example, in patients with metastatic melanoma treated with anti-programmed cell death protein 1 (PD1), higher tumor burden means lower response rate and worse survival. 9 Lower tumor burden may reduce the probability of mutational burden and production of IT-resistant clones, such as β-2-microglobulin mutated clones. 10 Large tumor volume increases the immunosuppressive characteristics of the TME (inhibiting CD8+ T cells, decreasing oxygenation, producing metabolic alterations, and increasing lactate production), with a subsequent rise in tumor heterogeneity. 11 Additionally, high tumor burden increases the number of fibroblasts and type 2 macrophages, hindering T-cell function. 12
Neuroendocrine regulation of immune cells within the tumor
Dysregulation of the sympathetic nervous system and the hypothalamic-pituitary-adrenal axis modifies the biology of the tumor and its response to IT, by promoting angiogenesis, proliferation and survival of tumor cells, alteration of the immune response and inflammation in the TME. 13
The effect of stress on the immune system, as a deregulator of the tissue microenvironment and tumor promoter, is pivotal in IT strategies. Recent data suggest that norepinephrine signaling through the beta-2 adrenergic receptor has an immunosuppressive effect on cytotoxic lymphocytes (CTL) by down-regulating cytokines to stimulate its own proliferation. 14 Focusing on ultraviolet-induced squamous cell carcinoma, Dhabhar et al. 15 showed that highly anxious mice had a major tumor burden and significant decrease in (T helper + CTL)/Treg ratio. Likewise, in xenotransplanted B16.F10 melanoma cell line, Kokolus et al. 16 reported that cold stress increases tumor growth and that growth suppression at thermoneutral temperature (30–31°C) is dependent on the presence of CTL cells. The adverse systemic effects of chronic stress on immune cells, together with tumor-associated macrophages (TAM)—which modulate inflammation, immunosuppression, and metastatic potential—limit the benefits of IT. 17 However, recent research conducted by Kokolus et al. 18 found that administration of the beta-blocker propranolol enhances the effect of alphaPD1 + IL2 IT in murine melanoma. This opens the door to the use of such beta-adrenergic blockers as a complement to IT in cutaneous tumors.13,19–21
Pro-tumorigenic response linked to administered therapy
The host-mediated immunological pro-tumorigenic response varies according to treatment used, which could include chemotherapy, radiotherapy, IT, surgery, or molecularly targeted drugs. Beyond TAMs and myeloid suppressor cells (MDSC) which play a pro-tumorigenic role in response to all therapies, each pro-tumorigenic response shows a specificity that must be addressed when identifying specific biomarker therapy. 22 Chemotherapeutic treatments elicit high IL-10 secretion by TAMs, which leads to decreased IL-12 expression by DCs and subsequent suppression of cytotoxic T-cell activity. In locally irradiated tumors, stromal cell-derived factor 1 is induced, driving MDSC recruitment.
Regarding targeted drugs, the proteasome inhibitor bortezomib promotes IL-16 secretion by pro-inflammatory TAMs. Surgical procedures reduce the number of lymphocytes and natural killer cells, leading to immunosuppression, expansion of residual tumor cells, and formation of micro metastases. Additionally, residual tumor cells secrete cytokines that contribute to immunosuppression and recruitment of MDSC and Treg cells. Therefore, some patients may show accelerated tumor progression during IT treatment. 23 To avoid these effects, IT in combination with chemotherapy/radiotherapy has been proposed for different tumors, including melanoma. 24
Patient age and immunosenescence process
Older melanoma patients present lower sentinel lymph node metastases rates, but have higher incidence of visceral metastases and lower survival. 25 Fibroblasts harvested from healthy donors aged >55 induce significantly more invasion of various human melanoma cell lines than fibroblasts harvested from donors aged <35. 26 A similar effect was observed with regard to metastases on aged ECM. 27 Likewise, while BRAF gene mutations are present in 43% of patients with melanoma, these tumors are more numerous and less aggressive in young patients than in the elderly. 28
IT response/resistance is influenced by the enormous diversity in immune cell profiles, infiltrates, and activity in the TME. This variability affects the differential IT response between young and old patients, which may justify adopting age-specific therapy.10,29 Patients aged >60 respond better to anti-PD1, so the probability of anti-PD1 response increases with age. The ratio of CD8+ T cells to Treg cells increases with aging, making tumors more immunogenic. Since the CD25 surface marker is highly expressed on Treg (and is temporarily expressed on T effector cells), anti-CD25 antibodies can be used to influence its presence/function. Analysis of tumor immune cell subtypes in older mice reveals that after anti-CD25 therapy the proportion of CD8+ T cells increases, while Treg cells decrease. The same anti-CD25 therapy in younger mice increases the anti-PD1 response. 30 Depletion of Treg cells in melanoma mouse models does not induce anti-tumor immunity in aged mice, but significantly decreases tumor growth in young mice. 31 Furthermore, despite evidence that immune checkpoint inhibitors are highly effective in older adults, in melanoma patients there are age-mediated intratumoral immune population differences after IT.30,32
Body mass index and metabolism
Obesity and body mass index (BMI) usually play an adverse role in most tumors, but they seem to have an inverse effect in metastatic melanoma.33–35 Repeated observations in (mainly male) patients with metastatic melanoma treated with targeted therapy and IT (without chemotherapy) have shown that obesity (taking BMI = 25 kg/m2 as a cut-off point) is associated with better survival, regardless of other traditional prognostic factors. This so-called “obesity paradox” in cancer paves the way for investigations exploring connections between the amount of adipose tissue and central or peripheral distribution, the type of oncological therapy used, and the possible alteration of specific pathways, and the associated metabolic dysfunction. 36 Obesity-induced low chronic inflammation is known to elevate levels of cytokines IL-1, IL-6, and TNF-α. 37 Furthermore, increased leptin has been found to trigger PD1 expression in T cells. This increased PD1 may mediate a mechanism of immune suppression of T cell responses induced by obesity-associated inflammation. However, a recent study showed that obesity promotes the T-cell depletion phenotype facilitating tumor growth, while at the same time conferring greater sensitivity to anti-PD1 IT. 38 Other studies suggest that the obesity paradox is the result of analysis biases and confusion. 36 More research is needed to understand the effect of obesity on IT treatment in skin tumors.
Regarding lipid metabolism, variations in fatty acid uptake, oxidation, and storage seem to play an essential role in tumor cell migration. It has been observed that melanoma cells can reprogram the TME using a variety of nutrients that allow tumor growth and progression. Furthermore, lipids such as glycerophospholipids, sphingolipids, sterols, and eicosanoids can contribute to the plasticity and aggressiveness of melanoma. A recent review of this topic and its potential therapeutic strategies in melanoma can be found in Pellerin et al. 37
Markers of resistance to IT in skin tumors
Mutational genetic status
Malignant melanoma is characterized by genetic and epigenetic alterations and has marked resistance to most current therapies. In cutaneous melanoma, genetic alterations have been linked to drug resistance. However, this resistance seems to be related to changes in transcriptional programs within cell subpopulations. Studies in melanoma patients have observed different responses to IT depending on tumor burden mutational status, but not on tumor mutational heterogeneity. Different types of mutational heterogeneity can be seen within the same tumor type: intertumoral heterogeneity (between different patients), intratumoral heterogeneity (between different areas of the same tumor) and spatial/temporal heterogeneity (between primary tumor and metastases), and these must be taken into account when evaluating biomarker study results and resistance to IT. The BRAF (V600E) mutation does not show specificity, due to its heterogeneous presence in the tumor 39 ; neither does it necessarily imply malignancy, since there are benign nevi with this oncogenic mutation. 40 Other interesting heterogeneous point mutations in melanomas are in BRAF (V600 K, V600Q, and V600R), NRAS (Q61 K, Q61L, Q61H), and C-KIT (especially associated with older age at diagnosis). 41 Patients with NRAS mutated melanoma have a better response to IT than those with BRAF mutated ones.42,43 In BRAF mutated melanomas, patients with BRAF V600 K tumors (approximately 20–30% of melanomas) have shown better outcomes and overall survival than patients with BRAF V600E tumors (approximately 70–80% of melanomas) after PD1 treatment. 44 More studies are needed to confirm these relationships and to identify the IT response in patients with intratumoral and spatial/temporal heterogeneous point mutated tumors or with other mutations such as C-KIT or MTS1. The p16/CDKN2 (MTS1) gene encoding for the p16 inhibitor of cyclin D/CDK4 complexes is frequently heterogeneously mutated and deleted in a large proportion of melanoma cell lines.
Impact of epigenetic markers
Chromatin epigenetic changes are reversible. Clinically relevant observations have indicated the benefit of “drug holidays,” consisting of treatment interruption or intermittent treatment, which delays epigenetic resistance (not effective if drug resistance is genetically regulated). For example, resistance to selective BRAF inhibitors can be reversed significantly after discontinuation of treatment. 45 Slow-cycling cancer cell subpopulations, including those present in melanoma, have been implicated in drug tolerance reversal processes. A small fraction of cells has repeatedly been found to survive treatment at high concentrations and can regain sensitivity after a drug holiday. This sensitization is done by eliminating (by RNA interference) KDM5A (Lysine Demethylase 5A), confirming that this histone demethylase is important in creating the reversible drug tolerance state. 46 Chronic exposure to external stressors such as hypoxia, lack of nutrients, and other drug treatments can also lead to tolerant tumor cells.
Dysregulation of histone demethylases results in aberrant histone methylation patterns, which have been linked to melanoma pathogenesis. Using the H3K4me3 JARID1B/KDM5B demethylase as a biomarker, a subpopulation of slow-cycling cells (critical for continued tumor growth) has been characterized among the main proliferating population. 47 This dynamic subpopulation depends on epigenetic alterations of melanoma and is critical for the melanoma maintenance since most of the therapies target proliferating cells. Furthermore, characterization of the slow-cycling JARID1Bhigh phenotype reveals high expression of mitochondrial bioenergetic enzymes and mitochondrial respiratory chain blockage, related to intrinsic multidrug resistance in melanoma. 47
Due to their reversible nature, epigenetic and post-translational changes are a growing focus in cancer research with the goal of preventing or reversing resistance to current therapies. 48 The combination of epigenetic targeting drugs with other anticancer agents, including IT, is a promising way to improve the efficacy of skin cancer treatments.
TME strategies
The TME works as the modulator of this dynamic ecosystem, and provides the tools through the intrinsic and extrinsic factors capable of inducing TME reprogramming. 7 An emerging strategy for cancer treatment is to revert the malignant phenotype targeting the TME instead of (or in addition to) cancer cells.
Different strategies can be added to TME reprogramming to avoid the risk of IT resistance mechanisms, such as metabolic readjustment, vitamin D for its effect on tumor stroma or intratumoral immune response reprogramming with the aim of increasing IT response.49–53 In this regard, several local factors that activate the immune response, ECM bio tensegrity, and the adaptive process of angiogenesis all contribute to TME reprogramming. One promising strategy concerning ECM bio tensegrity lies in selective bromodomain-7/9 inhibitor TP-472. A recent study demonstrated that this inhibitor downregulates genes encoding for integrins, collagens, fibronectins, and metalloproteins in melanoma cell lines. 54 Regarding angiogenesis, in melanoma mice models the combination of axitinib (a VEGFR1, 2, and 3 inhibitor) with an anti CTLA-4 treatment has been shown to improve animal survival. Another emerging angiogenesis-related strategy is generation chimeric antigen receptor T cells (CAR-T) anti-VEGFR, which has been shown to act synergistically with CAR-T against melanoma, eradicating B16-induced melanoma. 55
Several methods have recently been developed for systematized evaluation of the host immune response regarding TME reprogramming. The Immunoscore assay quantifies immune cell density to predict patient prognosis and suggest clinical treatment approaching from a spatial perspective that includes the core of the tumor and its invasive margin, with the contribution of T-cell subpopulations (CD3, CD8, and CD45RO).56,57 The TIME (Tumor Immune MicroEnvironment) classification based on the presence or absence of programmed death ligand 1 (PD-L1) and tumor-infiltrating lymphocytes (TIL) determines four subtypes of suppressive tumor immune microenvironment: T1 (PD-L1−, TIL−), T2 (PD-L1+, TIL+), T3 (PD-L1−, TIL+), and T4 (PD-L1+, TIL−). 58 T2 tumors correlate with better response to anti-PD-1 therapy. 59 Although T3 tumors have TIL, they do not express PD-L1; nonetheless, co-stimulation of T3 tumors with OX-40 or 4-1BB agonists could disrupt T-cell tolerance and reprogram the TME into a more treatable tumor. 60 The TIME that shows abundant immune cells in the periphery, but is empty of CTL in the tumor core, is called TIME infiltrated-excluded, meaning that adaptive immunity is unable to recognize malignant cells. On the other hand, infiltrated-inflamed TIME is characterized by abundant CTLs, and therefore were considered highly immunogenic tumors. Nanoparticles represent an interesting tool for immune TME reprogramming to improve IT. Some nanoparticles enable us to control cytokine expression, reprogram TAMs, suppress Treg, and reduce MDSC populations. 61 The benefits of all these TME reprograming strategies on IT need further exploration.
Emerging IT-related markers in skin tumors
Normal and tumor microbiota
The microbiota has important physio- and oncopathogenic roles in areas including immune response activation rate, metabolic diseases, and TME effects. 62 Under normal circumstances, the skin microbiome itself is as diverse as the gut microbiome. Like the gut microbiome, it affects the risk of several diseases including cancer. Additionally, the gut microbiome may directly influence the risk of cancer of skin and other organs by promoting systemic inflammation. 63 A resident microbiome has currently been identified in common cancers such as breast, brain, or lung, and others such as melanoma. 64 Gram-negative lipopolysaccharides (LPS) are present in most tumor types, whereas the lipoteichoic acid of gram-positive bacteria (found inside macrophages) is far more specific to melanoma. Studies have shown that bacterial composition and diversity differ significantly between skin and melanoma microbiomes, suggesting a strong relationship between melanoma development and skin microbiome changes. 65
Two independent studies have shown that PD1 IT responders in melanoma patients had different gut microbiome profiles to non-responders.66,67 Furthermore, one of these studies demonstrated that transplantation of PD1 responders’ fecal microbiota into germ-free mice lead to reduced melanoma tumor size and improved response to anti-PD-L1 IT. 66 A conserved microbiota that promotes skin immune function, proper metabolism, and effective immunity can monitor, detect, and eliminate potential immunogenic tumor clones developing on the skin. In the mutual reconfiguration between the immune system and the tumor, local and systemic microbiota, especially intestinal ones, are related both to the etiopathogenesis of the tumor and to treatment response. Compared with normal skin, changes in tumor microbiota involve a series of metabolic and immune modifications that alter the TME, allowing tumor onset and progression, and determining its evolution and prognosis. For these reasons, the microbiota and the microbiome constitute important specific biomarkers which must be included in an expanded vision of skin cancer.
Metabolic regulation of the immune response
The emerging field of immunometabolism addresses changes in the intracellular metabolic pathways of immune cells. 68 Metabolic alterations of TME deprive T cells of the necessary energy to carry out their cytotoxic function (which is recovered by anti-PD-L1 treatment) by modifying the common metabolic pathways between tumor cells and immune cells.
Fatty acid metabolism represents an interesting field in this context. While fatty acid oxidation seems to promote non-inflammatory immune cell development and activity, fatty acid synthesis positively regulates the pro-inflammatory function of immune cells of the innate and adaptive immune systems. Inflammatory stimuli such as LPS and cytosines induce higher synthesis of fatty acids in macrophages, which in turn can form the basis for producing different bioactive molecules with a protective role in TME. 69
The role of tryptophan metabolism in immune function is associated with the propensity of tumor cells to evade immune cell responses. The immunoregulatory role of tryptophan metabolism in the immune system has focused on the role of indoleamine-2,3-dioxygenase (IDO), which is responsible for the rate-limiting step in tryptophan catabolism. IDO is expressed in both tumor cells and TME, and its levels have been correlated with poor prognosis in melanoma and other tumors. 70
Mitochondrial dysfunction and the hypoxia-lactate axis
The role of mitochondria is closely linked to the metabolic function of immune and tumor cellularity, glucose availability and the Warburg effect. 71 In addition, mitochondria status is linked to the mutagenic role of free radicals, the apoptosis process, and the availability of cellular energy. Due the nature of the processes in which it is involved, mitochondria could be the cornerstone of the entire oncogenic process, and has been especially studied in the melanoma framework. 72
Cellular responses regulated by oxygen include chromatin remodeling in hypoxic conditions. Lactate (tumor immune regulator) acts as a key mediator of the effects of hypoxia and increased anaerobic or aerobic glycolysis on the macrophage phenotype. The general role of lactate seems to be promoting the shift from an inflammatory to reparative macrophage phenotype. In addition, lactate can act on organelles (lysosomes, mitochondria) and nuclei to modulate signaling mechanisms and gene expression. Hence, lactate acts as a link in the chain of mechanisms by which various pathways are activated by hypoxia (including hypoxia-inducible factor (HIF), acidosis, and chromatin remodeling) and cooperate with inflammatory pathways. Lactate also acts epigenetically by modifying histones and promoting the transcription of homeostatic genes. 73 Therefore, strategies to modulate inflammation focus on hypoxia, glycolysis-induced lactate, and acidosis.
Conclusions
The skin plays an important role in adapting whole-body physiology, including the capacity to produce a wide variety of hormones, neurotransmitters, and cytokines; also skin microbiota plays an integral role in the maturation and homeostatic regulation of host immune networks with systemic implications. 74 The particular characteristics of skin tumors make oncodermatopathology a unique field of observation, research, and learning from an immunological perspective.
Cutaneous tumors represent the most immunogenic group among solid neoplasms and are the most sensitive to immunotherapeutic approaches. The enriched cellular composition, extraordinary neural specialization, presence of endocrine elements and a SALT with functional properties and specific nesting, altogether endow the skin with remarkable sensitivity and responsiveness to physicochemical, infectious, and emotional stimuli. Melanoma and other skin tumors are among the first cancer types whose treatment aims to restore and enhance immune response to regain control of the tumor. 65 In spite of their high sensitivity to immune checkpoint inhibitors, a sizeable proportion of skin tumors (mainly melanoma, squamous cell carcinoma of the skin, and Merkel carcinoma) are primarily resistant to immunotherapeutic approaches. Furthermore, most patients who initially show some degree of response finally relapse, need subsequent treatments, and ultimately die as a consequence of disease progression. A huge number of clinical trials are currently addressing how to improve these clinical outcomes, so combination strategies using—among others—anti-CTLA and/or anti-PD1/PD-L1 monoclonal antibodies plus other immunomodulatory agonists or antagonists mAbs, antiangiogenic-targeted therapies, and intralesional agents, have raised great expectations. However, these new therapies should be developed based on a robust biological background, which is often lacking. At this stage, a comprehensive and thorough overview of the putative mechanisms that intervene in skin oncological processes with their respective study and follow-up biomarkers would shed light on this complex field, and ideally contribute to personalized treatments.
Expanding the knowledge and research base of skin tumors and their therapies, especially IT, puts the spotlight on three broad groups of biomarkers that help guide therapeutic decisions. These are predictive biomarkers of treatment response, biomarkers that warn of possible resistance, and emerging markers—the majority of a systemic nature. Among them, it is worth highlighting elements that focus not only on tumor cellularity itself and on its genetic alterations, but also especially on the TME and its rich ecosystem, neuroendocrine and systemic regulation of immunity. In summary, factors such as age, tumor burden or the immunosenescence process, the impact of epigenetic markers, microbiota, mitochondrial dysfunction and tumor and immune metabolism should be underlined as emerging biomarkers in IT of skin tumors. This comprehensive view of biology, health, and disease is pivotal in order to accurately diagnose the tumor type, understand its functional mechanisms and select a comprehensive therapeutic protocol.
Footnotes
Author contributions
I. Vieco-Martí and A. López-Carrasco contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the ISCIII, ERDF, Centro de Investigación Biomédica en Red de Cáncer (PI20/01107 and CIBERONC contract CB16/12/00484).
