Abstract
Proclarix is a new blood-based test to assess the likelihood of clinically significant prostate cancer (csPCa) defined as
Keywords
Introduction
Early detection of clinically significant prostate cancer (csPCa) decreases the specific mortality of PCa. 1 Currently, suspicion of PCa is established from a persistent elevation of serum prostate-specific antigen (PSA) and/or abnormal digital rectal examination (DRE) and then followed up with systematic prostate biopsies; 2 however, this approach has been disapproved due to the high rates of unnecessary biopsies and the overdetection of insignificant PCa (iPCa). 3 Multiparametric magnetic resonance imaging (mpMRI) and guided biopsies have enabled recent improvements in the early detection of csPCa. 2 Nevertheless, the efficacy of this new strategy could be further improved with a more accurate selection of candidates for prostate biopsy, especially when low or moderate likelihood of csPCa is suggested by mpMRI. 2 A Prostate Imaging-Reporting and Data System (PI-RADS) score of <3 indicates a negative mpMRI, and the current negative predictive value of mpMRI is 80%–95%. 4 Additionally, PI-RADS category 3 suggests a moderate risk of csPCa that does not exceed 20%. 5 In these challenging scenarios where the rate of csPCa detection is low, PSA density (PSAD), modern markers, or predictive models can be helpful.6,7
Proclarix is a blood-based marker test that was recently introduced.
8
Proclarix provides a multivariate risk score for csPCa to guide biopsy decision making. This risk score is based on the combination of age and serum measurements of thrombospondin-1 (THBS1), cathepsin D (CTSD), total PSA (tPSA), and free PSA (fPSA).9,10 THBS1 and CTSD were initially identified using a discovery mass spectrometry–based proteomics approach
11
and were subsequently observed in a PTEN knockout mouse model silencing the PI3 K/PTEN cancer pathway that is involved in the carcinogenesis and progression of PCa
12
and in human serum of men with and without PCa.
13
Clinical testing of individual immunoassays for the quantification of several glycoproteins was performed, and THBS1 and CTSD were ultimately selected because their measurement improved the accuracy of the percentage of fPSA in distinguishing men with and without csPCa.
14
This novel diagnostic test has been developed and validated to distinguish men without PCa or iPCa from those with csPCa among men with serum PSA between 2 and 10 ng/mL, prostate volume
Because the performance of Proclarix according to the PI-RADS category has not yet been studied, our objective is to compare the performance of Proclarix and PSAD in the selection of candidates for prostate biopsy after mpMRI.
Materials and methods
Design, setting, participants, and intervention
This was a prospective head-to-head evaluation of Proclarix in a frozen serum collection (https://biobancos.isciii.es/; Reference collection: 0003439) and PSAD in 567 consecutive men with PSA >3 ng/mL and/or abnormal DRE in whom pre-biopsy 3-Tesla mpMRI was performed (Magnetom Trio, Siemens Corp., Germany). From January 2018 to March 2020 at a single academic institution, men with tumors with a score of
Laboratory method for Proclarix evaluation and prostate-specific antigen density assessment
THBS1 and CTSD were measured using the Proclarix kit (Proteomedix, Zürich-Schlieren, Switzerland) as previously described. 8 Serum tPSA and fPSA were re-analyzed for all samples using the Roche Cobas immunoassay system (Roche Diagnostics, Rotkreuz, Switzerland). All measurements were performed in the Proteomedix laboratory in Zürich-Schlieren, Switzerland, with Proteomedix bearing the costs for measurements and reagents. Serum THBS1, CTD, tPSA, percent fPSA, and age were entered into an algorithm that reported a score from 0% to 100%. 10
PSAD was estimated from the MRI-derived prostate volume and the tPSA measured in Proclarix evaluation.
Endpoint measurements and definition of clinically significant prostate cancer
The endpoint measurements were csPCa detection rates, rates of avoided prostate biopsies, and rates of overdetection of iPCa. Tumors with an International Society of Uro-Pathology grade group of
Statistical analysis
Comparisons were performed with the Mann–Whitney U test for quantitative variables and with the Chi square and Kruskal–Wallis tests for qualitative variables. Receiver operating characteristic (ROC) curves and areas under the curve (AUCs) were used to analyze efficacies, and the DeLong test for their comparisons. Decision curve analysis (DCA) was used to evaluate net benefits. SPSS v.25 (IBM Corp., Armonk, NY, USA) and R programming language v.3.6.1 (R Foundation for Statistical Computing, Vienna, Austria) were used.
Results
PCa was detected in 298 men (52.6%), of whom 232 (40.9% of all participants) were diagnosed with csPCa and 66 (11.6%) had iPCa. The characteristics of the entire study cohort and a comparison of these characteristics in men without PCa or with iPCa and those with csPCa are presented in Table 1. We note that men with csPCa had significantly higher age, serum PSA, and PSAD; a lower percentage of fPSA; and higher rates of abnormal DRE, PCa family history, and positive mpMRI (PI-RADS
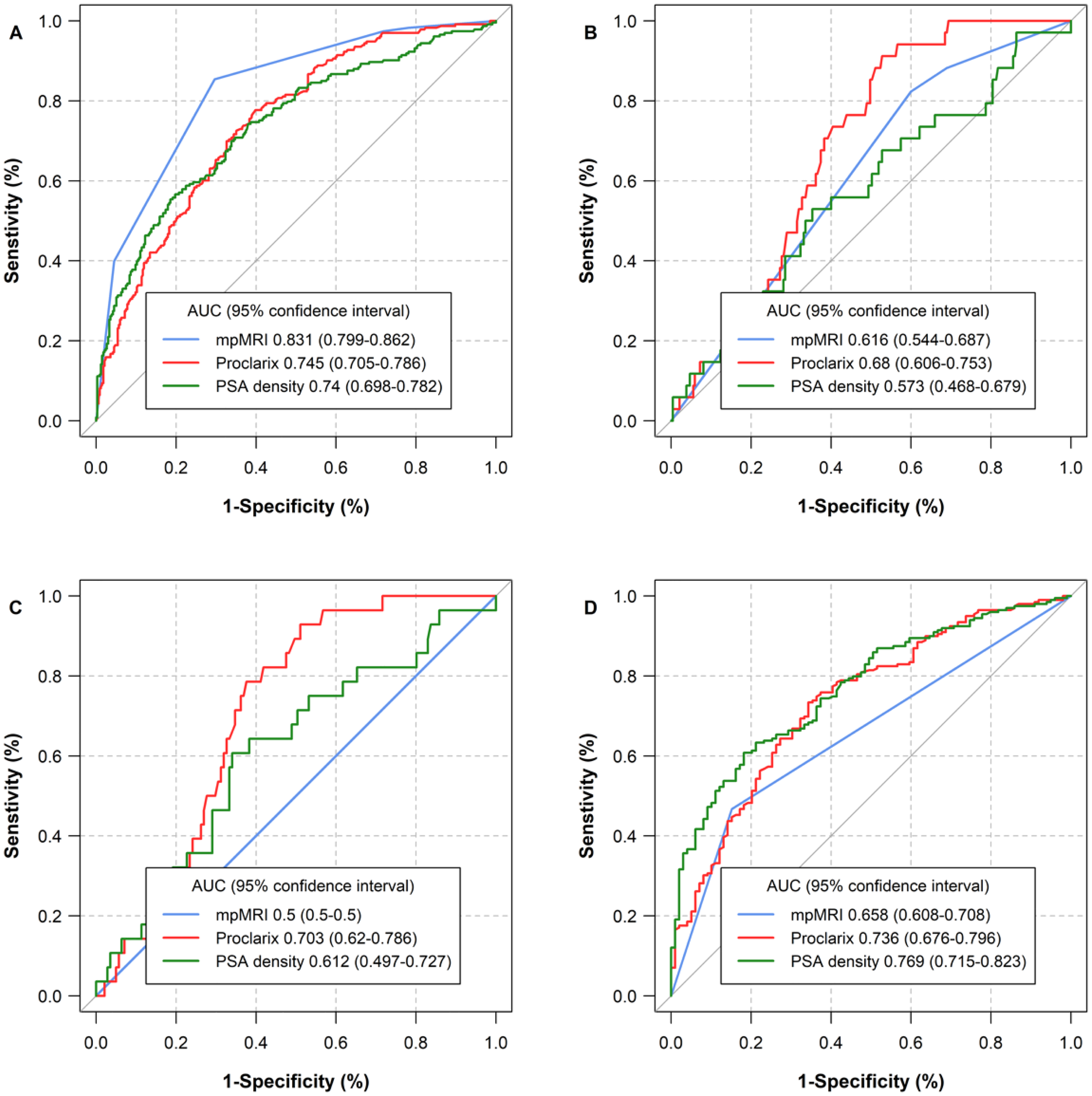
Efficacy of proclarix, PSAD and mpMRI for the detection of csPCa (
Characteristics of the study cohort and comparison between the characteristics of men without PCa or iPCa and that of those with csPCa (>2 grade group).
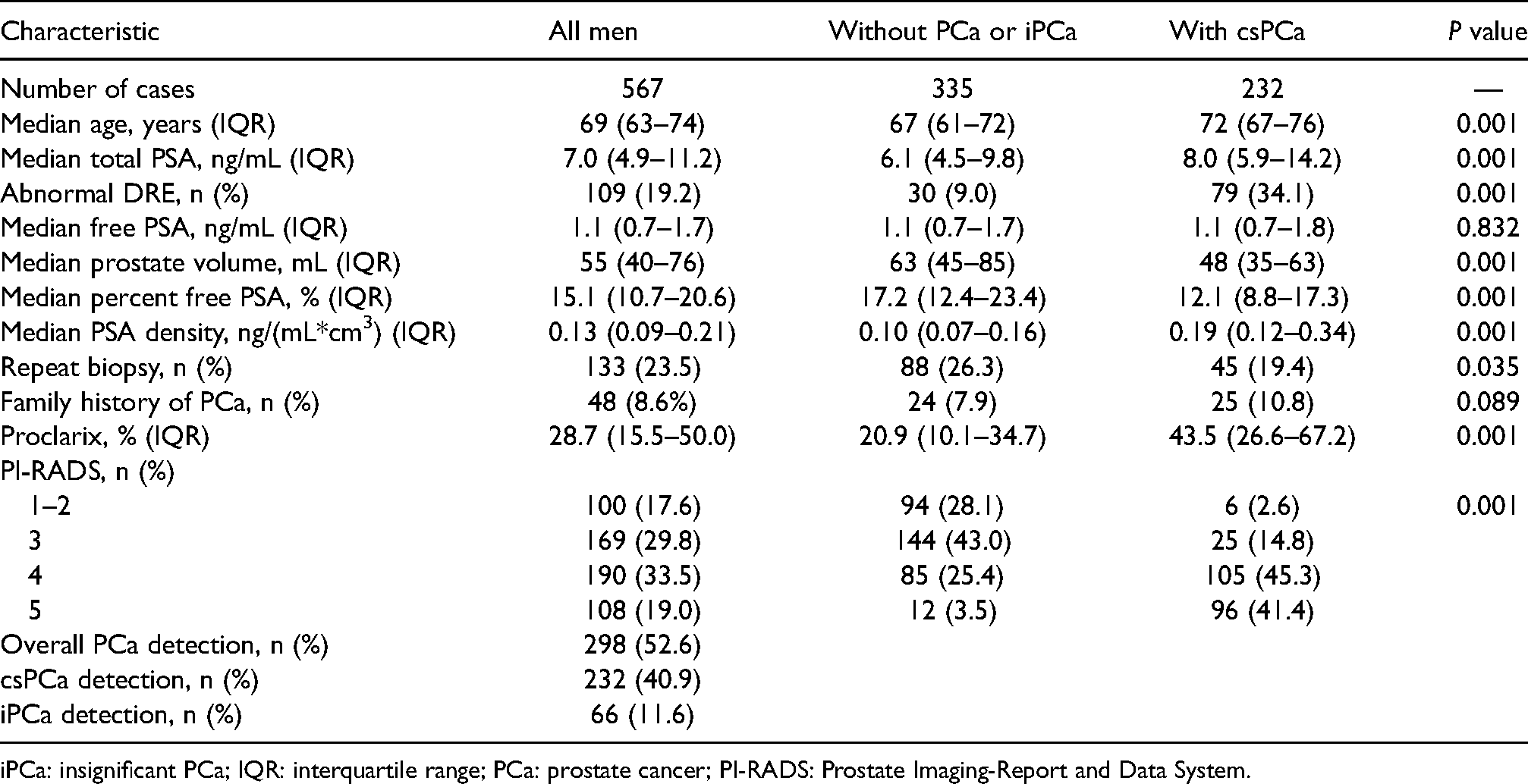
iPCa: insignificant PCa; IQR: interquartile range; PCa: prostate cancer; PI-RADS: Prostate Imaging-Report and Data System.
Table 2 shows the performance of Proclarix and PSAD both in all men and according to the PI-RADS categories. We note that Proclarix was able to detect all the 6% of csPCa detected in the systematic biopsies performed in the 100 men with negative mpMRI, although prostate biopsy was required in 70% of them. In contrast, PSAD detected 50% of csPCa and required 71% of systematic biopsies. In the subset of 169 men with a PI-RADS of 3, Proclarix avoided 21.3% of biopsies and detected all 25 cases of csPCa, while PSAD avoided 26.2% of biopsies, but missed 16.0% of csPCa. In men with PI-RADS of 4 and 5, Proclarix avoided 12.1% and 5.6% of biopsies, but misdiagnosed 4.8% and 1.0% of csPCa, respectively. PSAD avoided 18.4% and 9.3% of biopsies and misdiagnosed 11.4% and 4.2% of csPCa, respectively. The net benefit of Proclarix and PSAD on the biopsy of all men is presented in DCAs of Figure 2(a) and according to the PI-RADS categories in Figure 2(b) to (e).
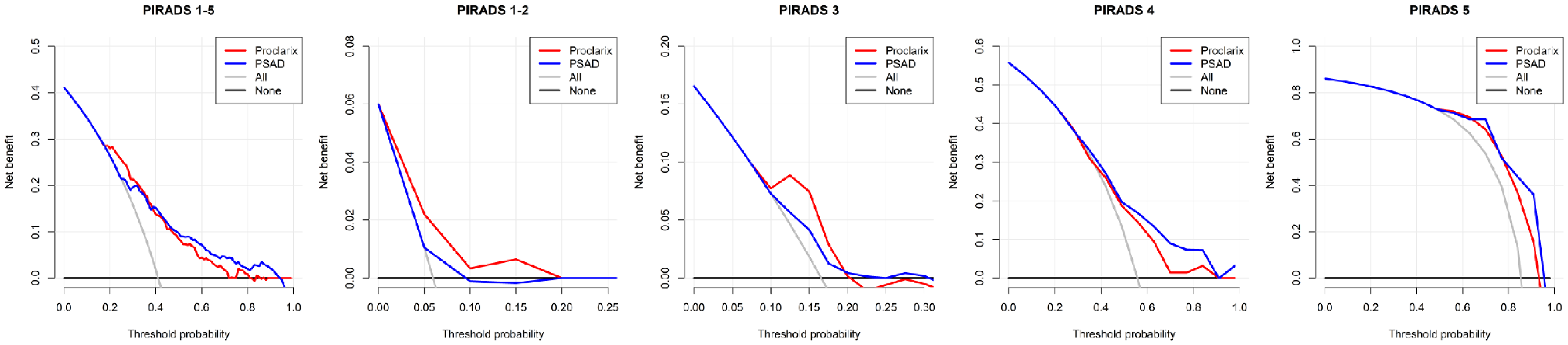
Net benefit of proclarix and PSAD instead of biopsy all men in the entire study cohort (PI-RADS 1–5), and according to the PI-RADS categories.
Performance of proclarix at 10% threshold and PSAD at 0.07 ng/(mL*cm3) threshold for csPCa (
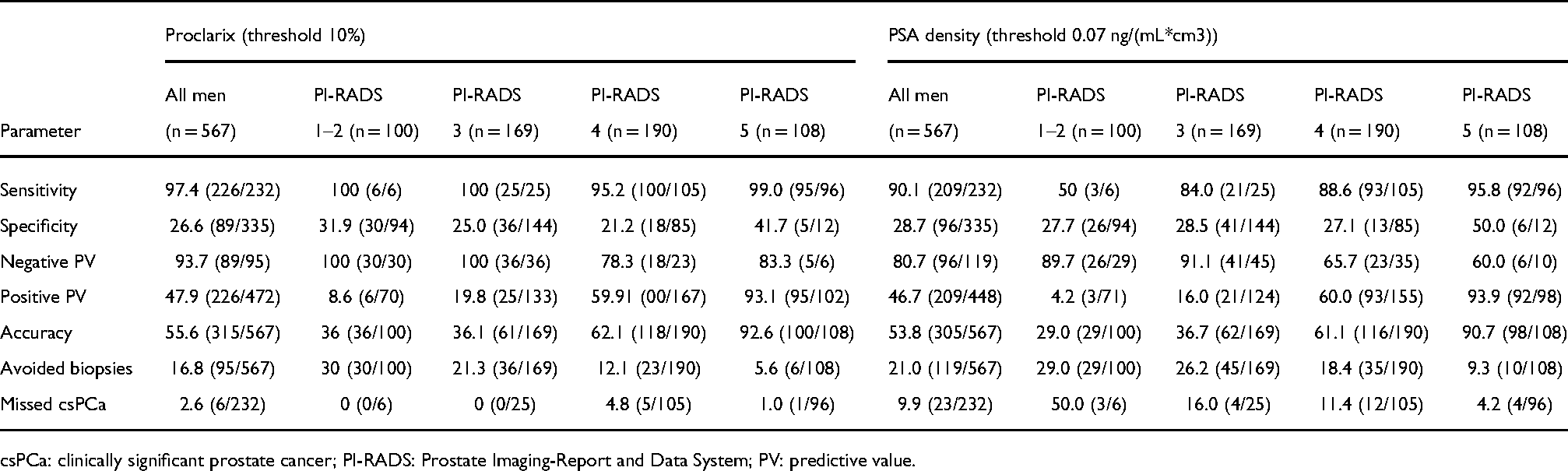
csPCa: clinically significant prostate cancer; PI-RADS: Prostate Imaging-Report and Data System; PV: predictive value.
Discussion
The present study confirms that Proclarix is a very sensitive marker of csPCa (grade group of
PROPOSe was the first study analyzing Proclarix in men with pre-biopsy mpMRI. The authors reported results from 108 men with positive mpMRI in whom guided and systematic biopsies were performed.
14
After fixing a sensitivity for
Comparisons between Proclarix and other markers are needed.18–20 In our opinion, a major strength of Proclarix is its high sensitivity for tumors with a grade group of
The limitations of our study include its partially retrospective design and the lack of external validation. Prospective and multicenter studies mimicking real clinical practice are needed, especially studies comparing the existing markers. However, a common limitation of these studies is measuring the rate of csPCa in prostate biopsies, which does not represent the true pathology observed in the whole prostate gland. A strength of our study was to perform systematic biopsies in men with negative mpMRI (PI-RADS <3) which allowed the possibility to know that Proclarix increased the negative predictive value of mpMRI from 94% to 100%.
Finally, we note that Proclarix can improve the selection of candidates for prostate biopsy after mpMRI, especially in men with low or moderate risk of csPCa defined by a PI-RADS score
Footnotes
Acknowledgments
Proteomedix provided Proclarix evaluations. This study was partially supported by the Instituto de Salut Carlos III (ES) PI20/01666.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Instituto de Salud Carlos III, (grant number PI20/01666).
