Abstract
Background
The monitoring of anti-p53 auto-antibodies in the peripheral blood has been used in cancer management; however, their clinical significance alone is limited. This pilot study aimed to describe the prevalence of elevated anti-p53 in newly diagnosed or recurrent upper gastrointestinal cancer or colorectal cancer in Chinese subjects. It also evaluated whether the addition of anti-p53 to a set of established tumor markers would allow for the detection of additional cancer cases than when using these markers alone.
Methods
A total of 573 subjects, including 187 healthy individuals, 169 patients with upper gastrointestinal cancer and 217 patients with colorectal cancer were included in this observational, prospective study. All subjects were required to provide up to 10 mL of blood. The following biomarkers were measured: anti-p53, carcinoembryonic antigen, cancer antigen (CA)19-9, and CA72-4.
Results
At the cutoff of 0.02 µg/mL, the sensitivity of anti-p53 in early-stage upper gastrointestinal cancer and colorectal cancer was 8.16% and 26.4%, and in late-stage disease was 7.81 and 28.0%, respectively. The specificity of anti-p53 in the healthy cohort at this cutoff was 98.4%. By adding anti-p53 to other tumor markers, the sensitivities were increased by 8.88%–9.47% in upper gastrointestinal cancer, and by 18.06%–25.00% in colorectal cancer; specificities decreased by 1%–2%.
Conclusion
The addition of anti-p53 to established tumor markers may improve their diagnostic value for patients with colorectal cancer.
Introduction
The p53 gene, located on the distal band of the short arm of chromosome 17p13 1 is a well-known tumor suppressor gene that frequently mutates in human cancers. 2 As mutation of p53 is involved in the early stages of cancer development, the anti-p53 antibody has been investigated as one of the most promising tumor markers for early detection of cancers.3–7 However, the clinical significance of anti-p53 measurement alone in patients with colorectal cancer is limited. 7
Other tumor markers, such as carcinoembryonic antigen (CEA) and cancer antigen 19-9 (CA19-9), have also been used as tumor markers in cancer screening; however, their predictive value varies. For example, the use of CEA in the management of patients with colorectal cancer is well-established.8,9 In patients with colorectal cancers that produce low or no CEA, CA19-9 is suggested to be used for treatment monitoring. 10 Stable or decreasing CEA or CA19-9 have been shown to have good negative predictive values of 96% and 90%, respectively, to exclude progression. 11 However, their positive predictive value to predict subsequent progression on computed tomography imaging was only 57% and 62%, respectively. 11 Elevated CA72-4 levels are associated with tumor stage in gastric cancer. 12 Combining tumor biomarkers may improve their diagnostic value in gastrointestinal (GI) cancers.
In this pilot study, we aimed to explore the prevalence of elevated anti-p53 in early- or late-stage upper GI or colorectal cancer in Chinese subjects and the diagnostic value of anti-p53 in combination with established tumor markers.
Materials and methods
Study design and study participants
In this observational, prospective, case-control study, patients with upper GI or colorectal cancer, and apparently healthy controls, ≥18 years old with written informed consent were recruited at the Fudan University Shanghai Cancer Center, Shanghai, China. All subjects were required to provide up to 10 mL of blood. The following biomarkers were measured for each subject: anti-p53, CA19-9, CA72-4 and CEA using a cobas e 601 instrument (Roche Diagnostics GmbH, Germany).
The study was conducted in full compliance with the principles of the Helsinki Declaration and local regulations. The study protocol was approved by the ethics committee at the research site.
Statistical methods
Elevation of anti-p53 was determined based on a calculated cutoff, derived from the 95th percentile of the anti-p53 value distribution in the healthy cohort. The cutoffs of the other tumor biomarkers were the established cutoffs from their package inserts. The two tumor markers combination was defined as positive if either the other tumor marker or anti-p53 was positive, and negative if both the other tumor marker and anti-p53 were negative.
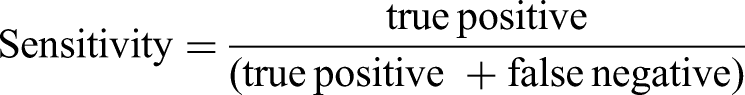
The sensitivity and specificity of anti-p53 and the other tumor biomarkers in each cancer type and the healthy cohort were calculated. All healthy subjects above the cutoff were treated as false positive, and below the cutoff as true negative. All cancer patients above the cutoff were treated as true positive, and below the cutoff as false negative. The sensitivity and specificity were derived using the following formula, and 95% exact confidence intervals (CI) calculated using the Clopper-Pearson method
13
from the binomial distribution.
Additionally, anti-p53 results (above cutoff or below/equal to cutoff) were correlated to the patient characteristics (i.e., age, gender, smoking status, body mass index (BMI), cancer histology, and stage) in colorectal cancer and upper GI cancer. The Wilcoxon test was used for age and BMI, and the Fisher’s exact test was used for all other variables. Correction for multiple testing was performed with the Benjamini–Hochberg false discovery rate (FDR) correction for each cancer type individually. The significance level was set as an adjusted P value < 0.05.
Results
In total, 607 subjects including 190 healthy controls, 233 patients with colorectal cancer, and 184 patients with upper GI cancer were enrolled in the study. Of these, 3 (1.58%) healthy controls, 16 (6.87%) patients with colorectal cancer and 15 (8.15%) with upper GI cancer were excluded as they failed the eligibility criteria. A total of 573 subjects, including 187 healthy controls, 217 patients with colorectal cancer, and 169 patients with upper GI cancer were included and analyzed. One anti-p53 measurement was missing for one patient with colorectal cancer. Patients with colorectal cancer included 88 (40.6%) females, with a mean age of 61.4 years; patients with upper GI cancer included 41 (24.3%) females, with a mean age of 60.9 years. Demographics and baseline cancer characteristics are summarized in Table 1.
Demographic and baseline tumor characteristics.
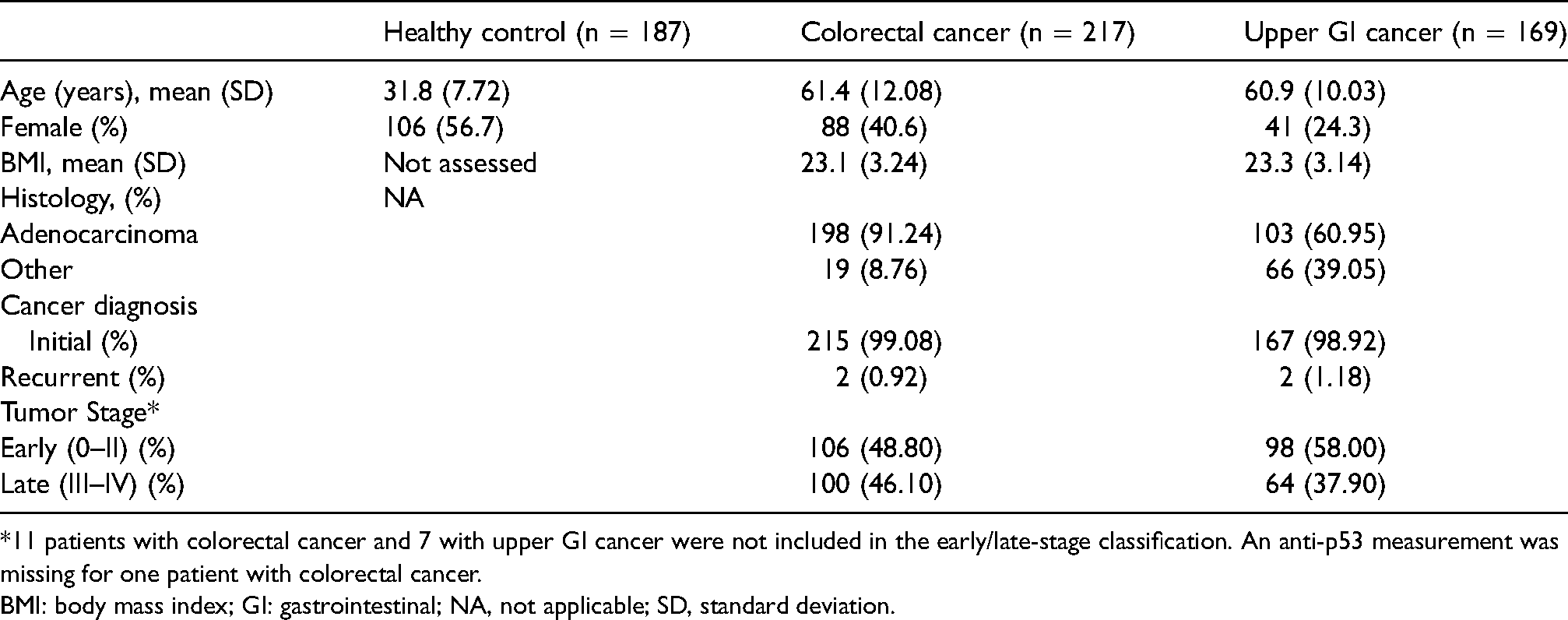
*11 patients with colorectal cancer and 7 with upper GI cancer were not included in the early/late-stage classification. An anti-p53 measurement was missing for one patient with colorectal cancer.
BMI: body mass index; GI: gastrointestinal; NA, not applicable; SD, standard deviation.
Clinical performance of anti-p53
The cutoff for anti-p53 derived from the healthy controls was 0.02 µg/mL and the specificity was 98.40%. The prevalence of anti-p53 in patients with early-stage upper GI and colorectal cancers was 8.16% and 26.40%, respectively (Table 2). For patients with late-stage upper GI and colorectal cancers, the prevalence of anti-p53 was 7.81% and 28.00%, respectively. No stage-dependent difference was observed for anti-p53 in either colorectal cancer or upper GI cancer. The anti-p53 distribution for patients by stage is shown in Figure 1(a) and Table 2, and for all patients in Figure 1(b) and Table 2.
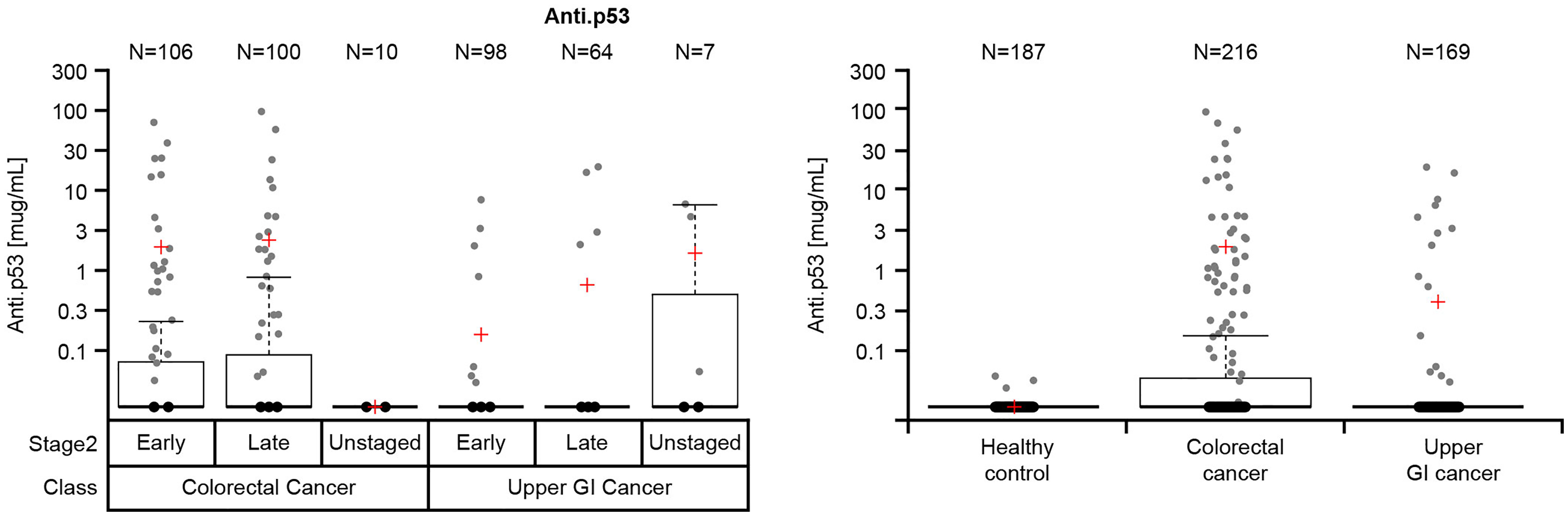
Anti-p53 distribution in (a) patients with early- late-stage and unstaged colorectal and upper gastrointestinal cancers and (b) healthy controls and all patients with colorectal and upper gastrointestinal cancers*. *An anti-p53 measurement was missing for one patient with colorectal cancer.
Sensitivity of anti-p53 at a cutoff of 0.02 µg/mL in patients with colorectal and upper GI cancers, by cancer stage and in healthy controls.*
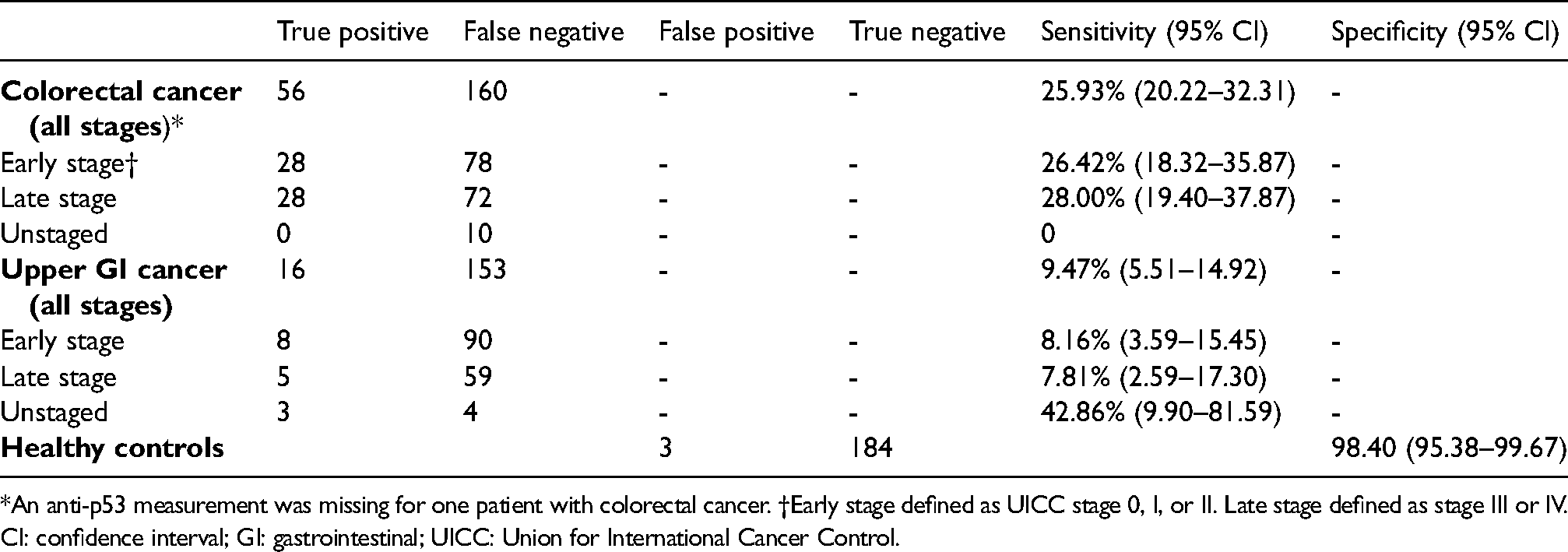
*An anti-p53 measurement was missing for one patient with colorectal cancer. †Early stage defined as UICC stage 0, I, or II. Late stage defined as stage III or IV.
CI: confidence interval; GI: gastrointestinal; UICC: Union for International Cancer Control.
Clinical performance of tumor markers plus anti-p53
Compared with the tumor marker alone, the combined marker sensitivity increased by 18.06%–25.00% in colorectal cancer and by 8.88%–9.47% in upper GI cancer (Table 3); the specificity decreased by 1%–2%.
Clinical performance of the tumor markers in combination with anti-p53 in all patients.
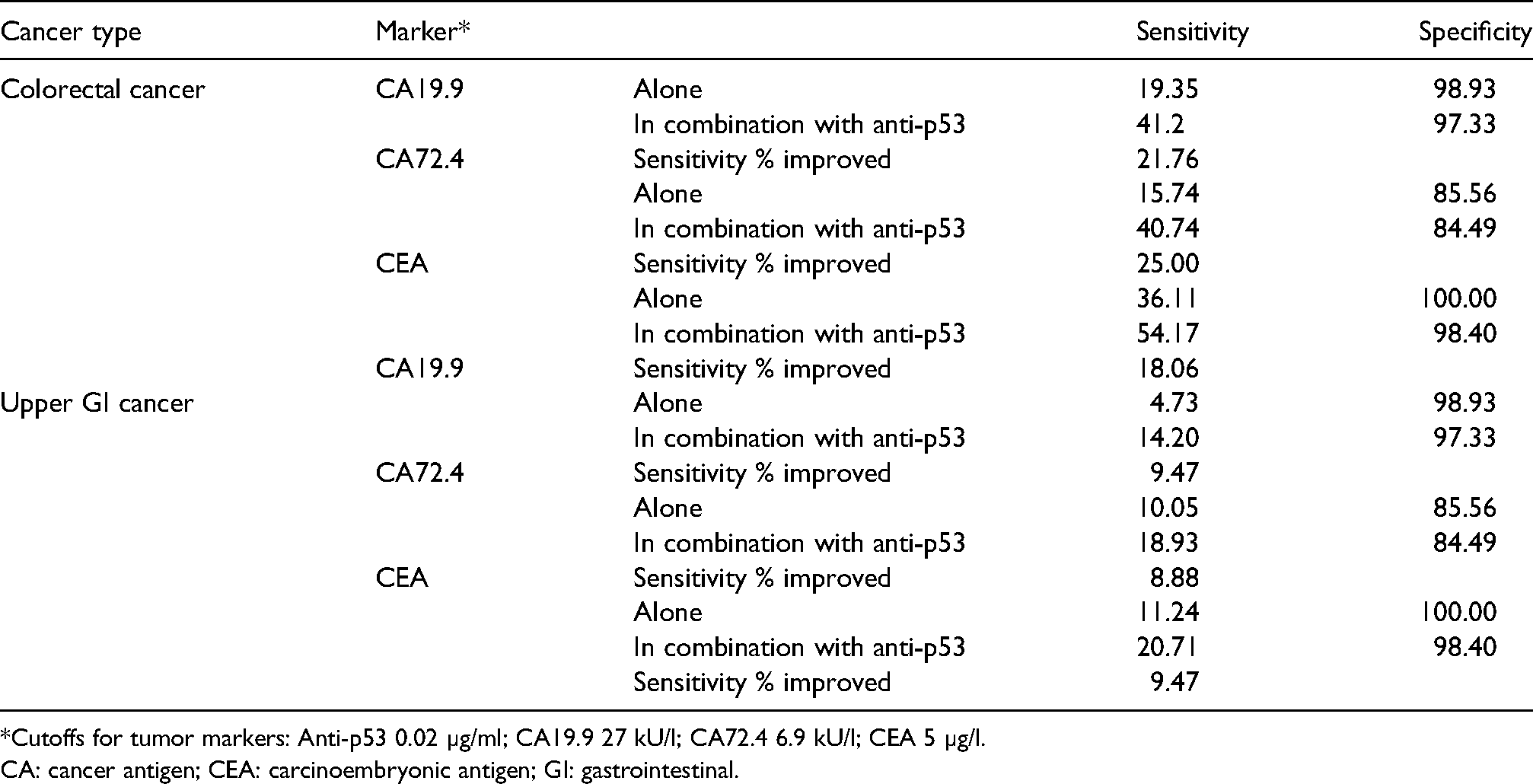
*Cutoffs for tumor markers: Anti-p53 0.02 µg/ml; CA19.9 27 kU/l; CA72.4 6.9 kU/l; CEA 5 µg/l.
CA: cancer antigen; CEA: carcinoembryonic antigen; GI: gastrointestinal.
In all patients with colorectal cancer, the biggest sensitivity improvements were seen with the combination of CA72.4 and anti-p53 (25.00%) and CA19.9 with anti-p53 (21.76%) (Table 3). The combination of CEA with anti-p53 showed the highest sensitivity and specificity in all patients with colorectal cancer (54.17% and 98.4%, respectively) and upper GI cancer (20.71% and 98.4%, respectively) (Table 3). Data by early- and late-stage disease are presented in Supplementary Table 1.
Analysis of CEA combined with anti-p53 showed 39 (18.06%) colorectal patients who were not detected by CEA alone but were detected by adding anti-p53 (Supplementary Table 2).
AUC of the multi-markers plus anti-p53
Adding anti-p53 to CEA improved the AUC for discriminating patients with colorectal cancer from healthy individuals: the AUC of CEA alone for detecting colorectal cancer was 86.4%, and improved by 3.2% to 89.6% by adding anti-p53 (Figure 2). The AUCs for the other biomarkers (CA19.9, CA72.4, CEA) alone and combined with anti-p53 are presented in Supplementary Table 3.
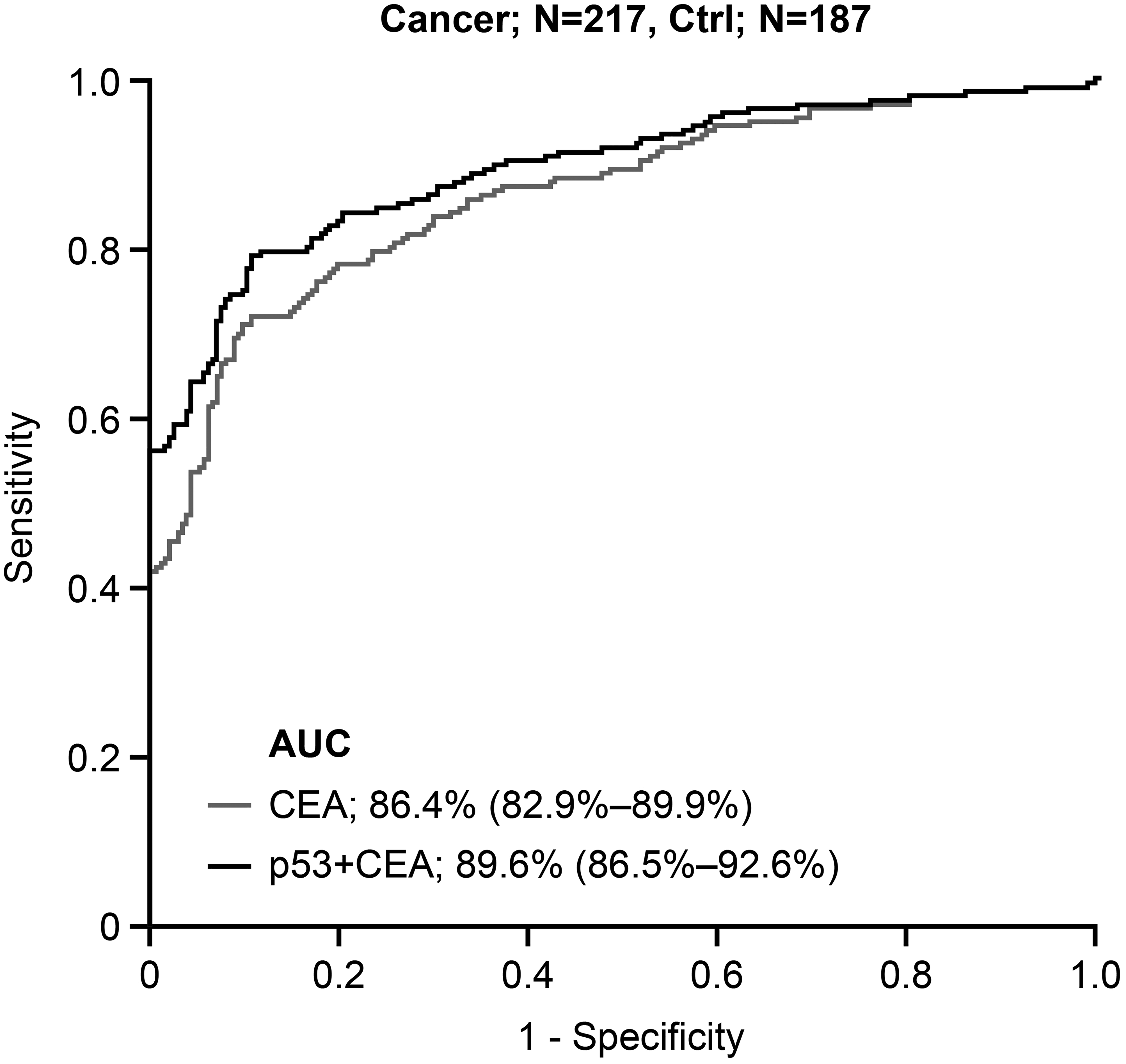
Receiver operating characteristic curve analysis in colorectal cancer (CEA vs. CEA + anti-p53).
Correlation of demographic and clinical variables with anti-p53
We further explored the clinical and demographic variables that might correlate with anti-p53, including gender, age, smoking status, BMI, and sub-histology. However, no significant results were found after adjusting the P-value with the Benjamini–Hochberg FDR correction for multiple testing.
In colorectal cancer, there tended to be more subjects with anti-p53 above 0.02 µg/mL in the current smoker group versus ex- and non-smokers (P = 0.0564). The BMI in the anti-p53 positive group was slightly higher than in the anti-p53 negative group (P = 0.0564) in patients with colorectal cancer. In upper GI cancer, no significant results were found to correlate with anti-p53.
Discussion
This pilot study investigated the diagnostic potential of serum anti-p53 antibodies in GI cancer. Germline mutations of the p53 gene commonly occur in the early phases of cancer development, and the resulting accumulation of mutant p53 proteins is often accompanied by the production of anti-p53 antibodies. 14 Thus, it can be hypothesized that anti-p53 antibodies may be detectable in early-stage cancers. In this study, we saw a similar anti-p53 distribution in early- and late-stage GI cancer. The results indicate that the prevalence of anti-p53 antibodies in upper GI and colorectal cancer was low, but their specificity in healthy controls was high (98.40%, [95% CI: 95.38–99.67]).
In current clinical practice, tumor markers are widely used in cancer diagnosis and monitoring; however, greater sensitivity is urgently needed. The combination of tumor markers is proven to increase sensitivity but often at the expense of specificity. In our study, the addition of anti-p53 to the CA19.9, CA72.4, and CEA tumor markers improved the sensitivity to detect cancer with a minimal loss of specificity, based on the healthy controls. The greatest improvement in sensitivity was seen in patients with colorectal cancer, while the benefit of adding anti-p53 to tumor markers in upper GI cancer was relatively small.
No correlation between demographics and clinical variables with anti-p53 was observed. The close-to-significant results may have been caused by unknown confounding factors, and further analysis with a larger sample size is recommended.
Several limitations of this study warrant mention. First, the cutoff value used in the study needs to be better defined to improve sensitivity. Second, the total number of patients in each group was relatively small, and the statistical power was therefore limited. Third, a more specific type of cancer within upper GI cancer should be investigated.
In conclusion, multi-marker analysis using anti-p53 may improve the diagnostic value for colorectal cancer patients. However, it is necessary to conduct further studies to validate anti-p53 autoantibodies for clinical use in China. These studies should evaluate anti-p53 and colorectal cancer markers in a multi-center setting with appropriate benign disease controls.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155221078602 - Supplemental material for Prevalence of elevated Anti-p53 in Chinese patients with upper gastrointestinal or colorectal cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155221078602 for Prevalence of elevated Anti-p53 in Chinese patients with upper gastrointestinal or colorectal cancer by Min Wang, Suhong Xie, Xiang Gao, Jingjing Feng, Minjie Deng, Jiajun Sun, Ying He, Horst Donner and Renqun Lu, Lin Guo in The International Journal of Biological Markers
Footnotes
Acknowledgments
The authors thank the patients and their families and the participating study teams for making this study possible; David Morgenstern (Roche Diagnostics) for scientific consulting support. Editorial support was provided by Jade Drummond of inScience Communications, Springer Healthcare (UK) and was funded by Roche Diagnostics.
COBAS and COBAS E are trademarks of Roche.
Data sharing statement
Qualified researchers may request access to individual anonymized patient-level data through the clinical study data request platform (https://vivli.org/). Further details on Roche's criteria for eligible studies are available here: https://vivli.org/members/ourmembers/. For further details on Roche's Global Policy on the Sharing of Clinical Information and how to request access to related clinical study documents, see here: ![]() .
.
Funding
The study was sponsored by Roche Diagnostics.
Declaration conflicting of interest
Ying He and Donner Horst are employees of Roche diagnostics. The authors declare no other conflict of interests.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.