Abstract
Introduction:
Indications for treating Benign Prostatic Hyperplasia include reversing signs and symptoms or preventing the progression of the disease. Alpha-blockers are the most effective, least costly, and best tolerated of the drugs for relieving LUTS. The aim of the study is to investigate the immediate impact of alpha-blocker medications on lower urinary tract symptoms (LUTS).
Materials and methodology:
About 100 patients were included in the study—50 patients in each of the groups A (tamsulosin) and B (silodosin). The first visit was the baseline examination before starting alpha-blockers and included history, DRE, UFM, USG KUBP with PVR, IPSS, serum PSA, serum creatinine, urine analysis, urine culture, and sensitivity. All above parameters were also at 1 week, 1 month, and 3 months following starting of alpha-blockers respectively, and compared with baseline.
Result:
As of the first, second, third, and fourth visits, the mean Qmax in group A was 10.3 ± 3.3 s, 15.08 ± 2.80 s, 15.66 ± 3.18 s, and 15.12 ± 3.24 s, respectively, while in group B it was 10.1 ± 3.1 s, 14.88 ± 2.80 s, 15.18 ± 3.18 s, and 15.08 ± 3.24 s, respectively (p < 0.001). The mean voiding time was 40.87 ± 23.91 s, 36.41 ± 20.73 s, 34.85 ± 21.37 s, and 32.07 ± 21.81 s, respectively in group A, and 41.27 ± 15.49 s, 37.23 ± 21.34 s, 38.59 ± 20.83 s, and 33.10 ±22.08. In group A, the mean PVR and IPSS scores were improved and also improved in group B.
Conclusion:
The first dose of tamsulosin and silodosin improves UFM and predicts the mid-term change in UFM as well as IPSS indices in the treatment of BPH-related LUTS.
Introduction
The most frequent cause of lower urinary tract symptoms (LUTS) in males is benign prostatic hyperplasia (BPH), which has a strong correlation with aging. 1 Although the pathophysiology of LUTS/BPH is not fully understood, a growing body of research indicates that chronic prostatic inflammation plays a significant role in the development of BPH in general and is directly related to the severity of the condition.2–4 Twenty-three percent of men over the age of 50 experience lower urinary tract symptoms (LUTSs) brought on by benign prostatic hyperplasia (BPH), with moderate to severe LUTS occurring in up to 50% of BPH patients.5,6
When treating males with moderate to severe lower urinary tract symptoms with medication, the first line of treatment is alpha-blockers.7,8 The effectiveness of alpha-blockers as measured by symptom ratings in randomized controlled studies is the basis for current guideline recommendations.1,8,9 However, in everyday practice, patients and physicians are more concerned with symptom improvements that reflect shifts in the subjective evaluation of symptoms, which may not be reflected in these symptom scores. The randomized controlled studies on which doctors rely for guidance typically report mean changes in symptom ratings rather than the number of patients who experience improvement, making it challenging for clinicians to predict the likelihood of improvement. Furthermore, the multifaceted etiology of LUTS in the male population makes it difficult to pinpoint who will benefit from alpha-blocker medication. Therefore, when a prescription for therapy is requested, doctors must depend on the general recommendation to begin alpha-blockers.1,6,9 Clinically useful information would aid physicians in determining which patients are most likely to improve after using an alpha-blocker.
The study aims to investigate the immediate impact of alpha-blocker medications on lower urinary tract symptoms (LUTS) and to determine whether the initial response to alpha-blocker treatment can be used as a predictor for long-term improvement in patients with benign prostatic hyperplasia (BPH).
Materials and methodology
The ethics committee at SMS Medical College and Hospital in Jaipur, Rajasthan, India approved the design of prospective research, which was conducted from July 2021 to December 2022. Following approval, written informed permission was collected from each participant. The ICH-GCP guideline was followed in the conduct of the study. Every patient over 40 who came with BPH-related symptoms to the urology outpatient clinic at SMS Medical College and Hospital Jaipur, Rajasthan, India was enrolled. Patients with a history of prostate-related medical or surgical treatment, post-void residual urine volume (PVR) greater than 150 ml, symptoms of neurogenic bladder, urinary tract stones, suspected prostate cancer (prostate-specific antigen [PSA] >4 ng/dl), and/or abnormal digital rectal examination (DRE) were excluded.
Following the screening of 110 individuals, 100 patients were included in the study—50 patients in each of the groups A (tamsulosin) and B (silodosin). The patients were randomized by computer-generated random numbers and allocated by opaque envelop method. The first visit was the baseline examination before starting alpha-blockers and included history, DRE, UFM, USG KUBP with PVR, IPSS, serum PSA, serum creatinine, urine analysis, urine culture, and sensitivity. Patients in group A were directed to take 0.4 mg of tamsulosin at bedtime and group B patients took 8 mg silodosin. UFM, PVR, and IPSS were also assessed at the second, third, and fourth visits which took place 1 week, 1 month, and 3 months following starting of alpha-blockers respectively, and compared with baseline.
Statistical analysis
The Shapiro-Wilk test was used to determine if the data were normal. Parametric tests were used to examine data having a normal distribution; nonparametric tests were used to study data lacking a normal distribution. Both Kruskal-Walli’s variance analysis and one-way analysis of variance (ANOVA) were employed to assess the data from more than two independent groups. Wilcoxon signed ranks test and the paired t-test was used to evaluate data from two separate periods in the same group. After one-way ANOVA and Kruskal-Walli’s variance analysis, multiple comparisons were made using the GraphPad software version 3.0, Tukey HSD test. For repeated measurements at various follow-up points and periods, repeated measures ANOVA with a Greenhouse-Geisser correction was utilized. Multivariate analysis for prediction of improvement with prostate size, PSA, age, PVR, and baseline IPSS. The level of statistical significance was 0.05. the sample size was calculated by the incidence of 8%, with a 95% confidence interval, 5% of margin error, with 2% dropout rate, and 80% of the power. we got 100 sample sizes that were divided into 50 in each groups.
Result
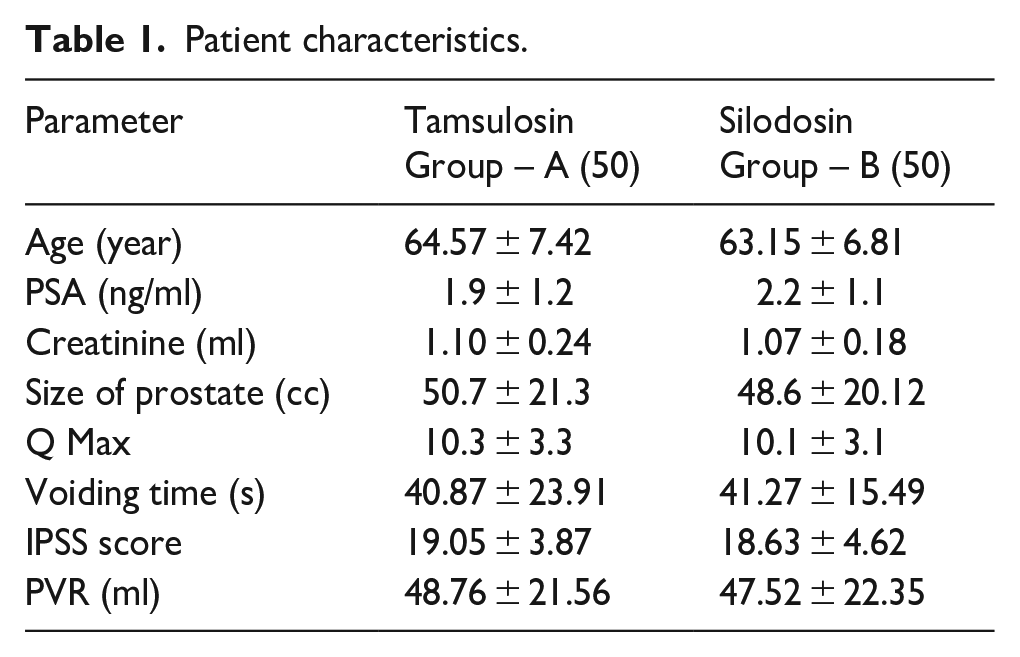
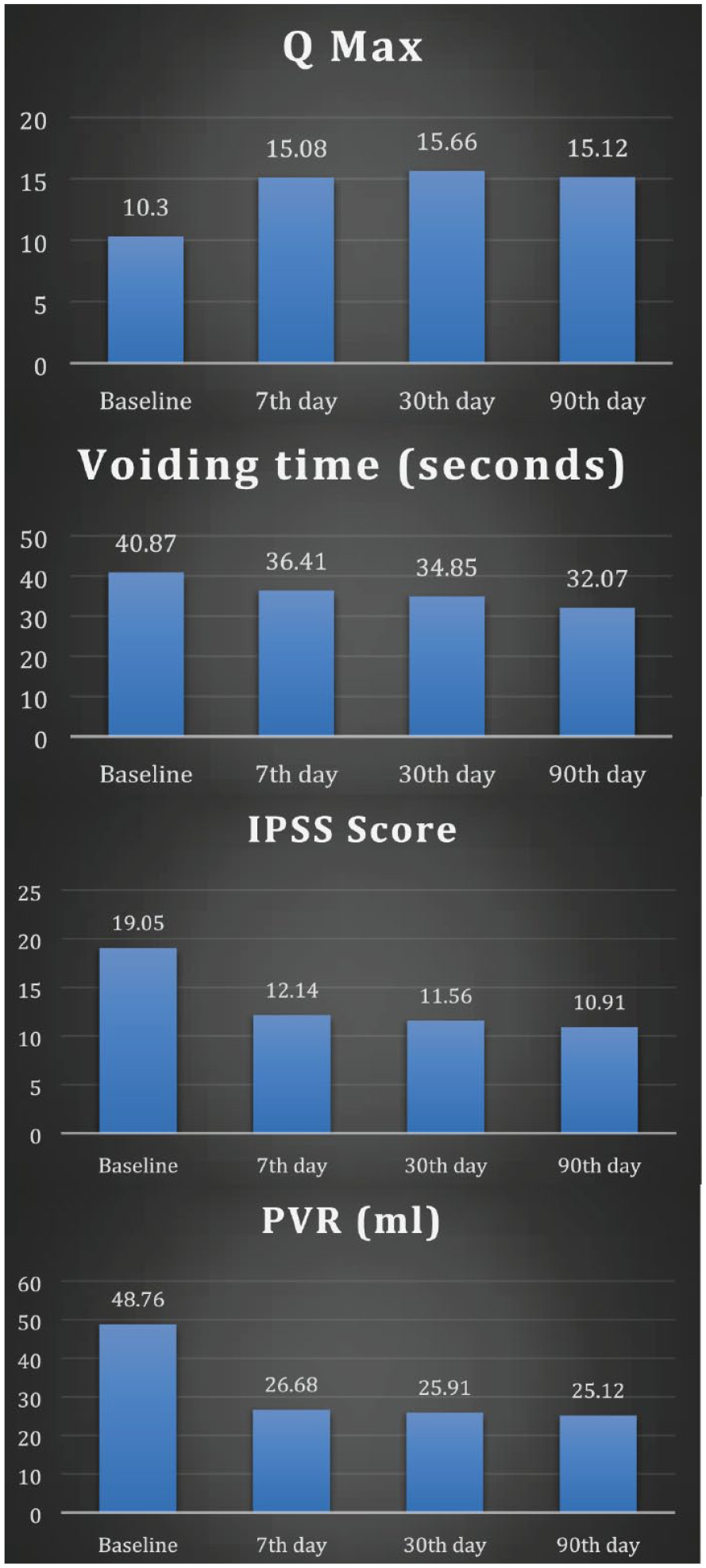
The study comprised 100 patients (50 in each group A and group B) with mean PSA levels of 1.9 ng/dl in group A and 2.2 ng/dl in group B, mean prostate volumes of 50.7 cc in group A and 48.6 cc in group B, and mean age of 64.57 ± 7.42 years and 63.15 ± 6.81 years, respectively (Table 1). As of the first, second, third, and fourth visits, the mean Qmax in group A was 10.3 ± 3.3 s, 15.08 ± 2.80 s, 15.66 ± 3.18 s, and 15.12 ± 3.24 s, respectively (p < 0.001), while in group B it was 10.1 ± 3.1 s, 14.88 ± 2.80 s, 15.18 ± 3.18 s, and 15.08 ± 3.24 s, respectively (p < 0.001) (Table 2; Figures 1 and 2). At the first, second, third, and fourth visits, the mean voiding time was 40.87 ± 23.91 s, 36.41 ± 20.73 s, 34.85 ± 21.37 s, and 32.07 ± 21.81 s, respectively (p < 0.001) in group A, and 41.27 ± 15.49 s, 37.23 ± 21.34 s, 38.59 ± 20.83 s, and 33.10 ± 22.08 (Figures 1 and 2; Table 2).
Patient characteristics.
Parameters at baseline, 7th day, 30th day, and 90th day in Silodosin group B.

Parameters at baseline, 7th day, 30th day, and 90th day in tamsulosin group A.
Parameters at baseline, 7th day, 30th day, and 90th day in Silodosin group B.
In group A, the mean PVR at the first, second, third, and fourth visits was 48.76 ± 21.56 ml, 26.68 ± 24.85 ml, 25.91 ± 24.72 ml, and 25.12 ± 25.96 ml, respectively (p < 0.001); in group B, it was 47.52 ± 22.35 ml, 25.71 ± 23.29 ml, 24.58 ± 22 ml (Figures 1 and 2; Table 2). After the seventh day, the effects of the first dosage of tamsulosin and silodosin significantly increased Qmax, decreased voiding time, and preserved residual urine (PVR), whereas there was no statistically significant difference between the seventh day, the first month, and the third month (Table 3).
Parameters at baseline, 7th day, 30th day, and 90th day in group A versus group B.
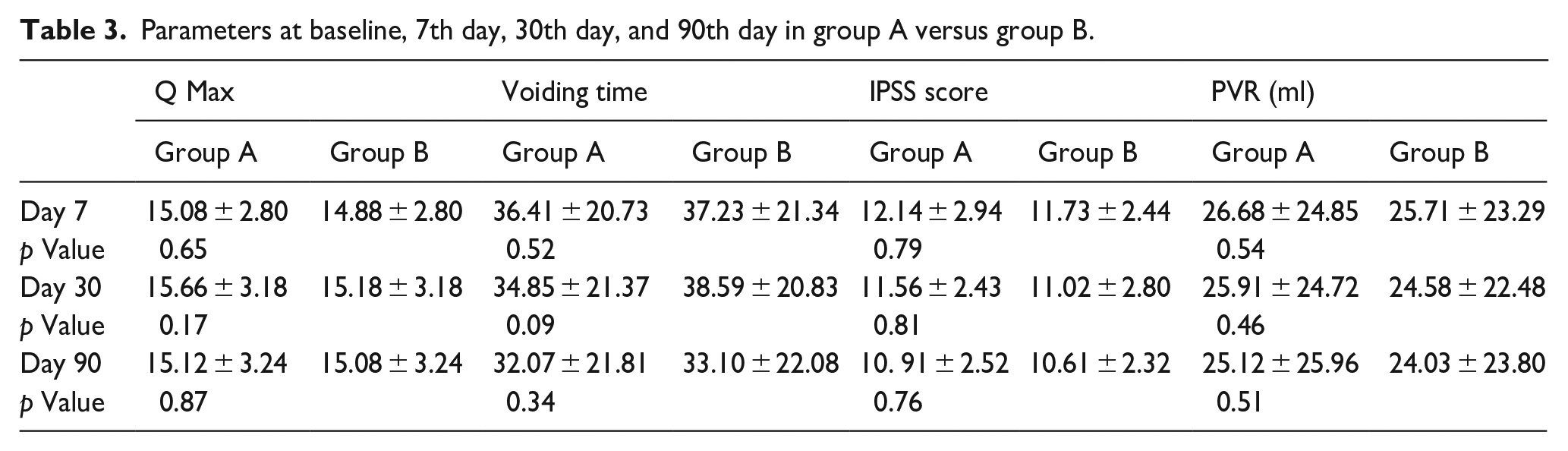
p < 0.001 indicates that the IPSS considerably improved after 1 and 3 months. Between 1 and 3 months, there was no statistically significant difference in the IPSS (Table 4). There is no correlation between prostate size, PSA, age, PVR, baseline IPSS, and response to tamsulosin and silodosin, according to multivariate analysis between improvement of IPSS and other parameters in the study group (p > 0.05).
Parameters at baseline, 7th day, 30th day, and 90th day in tamsulosin group A.

Discussion
Our goal was to determine whether the short course of oral tamsulosin 0.4 mg and silodosin 8 mg is effective in terms of UFM parameters and whether the first change of UFM parameters could predict the mid-term results in terms of UFM parameters and IPSS indices. We used standard doses of 0.4 mg tamsulosin and silodosin 8 mg, once daily orally administered after at bedtime for 50 patients in each group, respectively. In our study, we employed a pre-selected group with “moderate-to-severe LUTS” and patients with a mean age of over 60. This group of people was a good candidate for alpha blockade therapy. 10
Since the tissue level occurs at the 7.6–10.9 days mark with continuous therapy, we opted to analyze the UFM on the seventh day after the first dosage. We used 0.4 mg of tamsulosin in our study because a study by Chung et al., 11 which included 116 patients from three urology centers, showed that this dosage was more effective than tamsulosin 0.2 mg, and we also used 8 mg of silodosin because Boeri et al. 12 show that this dosage effectively relieves symptoms. We employed a pre-selected group with BPH “moderate to severe LUTS” and a greater prostate volume for our series. Akin et al. chose 40 patients with moderate symptoms for a study that was similar to ours; the mean IPSS was 16.46 5.77 and the mean prostate volume was 35.77 3.86 cc in TRUS; we chose additional patients with larger prostate volumes (50.7 21.3 and 48.6 20.12) and higher mean IPSS (19.05 3.87 and 18.63 4.62) to confirm the findings of Akin et al. 13
At the first week of tamsulosin and silodosin, respectively, as well as the first and third months of treatment from baseline, there was a statistically significant increase in Qmax and decrease in PVR for approximately 50/50 patients; however, there was no statistically significant difference in Qmax and PVR between the first week of tamsulosin and silodosin as compared to the first and third months of treatment. This indicates that 1 week of treatment may be able to predict LUTS improvement shortly and if any other intervention is required or not. Furthermore, according to our findings, baseline UFM measures including Qmax, voiding duration, PVR, baseline IPSS, prostate volume, age, and PSA are not indicators of how well a patient may respond to tamsulosin and silodosin. These results concur with those of Kang et al. 14 and Akin et al. 13
Both Group A and Group B saw a statistically significant decline in IPSS scores from baseline at the first and third months of therapy. These findings concur with those of Djavan et al. 15 and Akin et al. 13 Insignificant differences were between the IPSS values in the first and third months.
Limitation of the study
Conclusion
In the treatment of BPH-related LUTS, our prospective study shows that 1 week of tamsulosin 0.4 mg and silodosin 8 mg seems to be effective in improving UFM parameters and can predict the mid-term change in UFM parameters as well as IPSS indices. This allows us to determine whether this treatment will be sufficient after the first week or whether we will need to pursue additional treatment options.
Footnotes
Acknowledgements
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
