Abstract
Background:
Liver dysfunction is a common phenomenon in critically ill patients. Extracorporeal albumin dialysis (ECAD) is established in supporting liver function as a bridge to transplant or recovery. ECAD device OPAL is confirmed in clinical routine and studied in terms of detoxification capabilities, efficacy and efficiency. For the ECAD device ALBUNIQUE, a further developed system requiring less specialised technical equipment, no corresponding literature is available.
Methods:
This retrospective single-center study included patients undergoing ECAD, either OPAL and/or ALBUNIQUE, in a two-year period from 2023 to 2024, with treatment times of 12 h or more. Generalized Estimating Equations were used to compare the effects of ECAD therapy.
Results:
In total, n = 25 patients with ECAD treatments were identified in our institution. Among these patients n = 90 ECAD cycles were eligible for evaluation, thereof n = 58 (64%) treatments with OPAL and n = 32 (36%) with ALBUNIQUE. ECAD treatment resulted in significant reduction of bilirubin, ammonia, creatinine, and urea levels as well as significant increase of negative base excess values. The first ECAD cycle was associated with highest percental changes. There were no significant differences between the parameters gathered by OPAL and ALBUNIQUE.
Conclusion:
Both ECAD systems, OPAL and ALBUNIQUE, were effective in removing bilirubin, reducing ammonia levels, eliminating water-soluble substances and stabilizing metabolic dysfunction without significant differences between the devices. Further, we found no increased bleeding risk during or after application of ECAD treatment in general or for one of the examined devices.
Keywords
Introduction
Liver dysfunction is a frequently seen phenomenon in critical illness and is observed in up to 20% of intensive care patients. 1 Hepatic dysfunction can be divided into acute liver failure, decompensated cirrhosis, acute-on-chronic liver failure, or secondary hepatic failure with a relevant impact on ICU stay and patient’s outcome.1–3
Current therapeutic options in acute or acute-on-chronic liver failure, especially in the case of critical care patients, are scarce. The cornerstones of the treatment are critical care monitoring, standard medical therapy, an early diagnosis, and treatment of underlying causes as well as involving transplant centers to evaluate potential indication for liver transplantation as a treatment option.3,4 Within recent years, several artificial organ support devices have been established for bridging patients to transplantation or recovery. Based on the underlying concepts, they can be divided into systems that use either plasma separation (i.e., plasmapheresis, Prometheus) or albumin dialysis. 5
The following extracorporeal albumin dialysis devices (ECAD) were clinically applied: molecular adsorbent recirculating system (MARS), open albumin dialysis (OPAL), advanced organ support (ADVOS), and single-pass albumin dialysis (SPAD). 6 Most experiences has been gained with the MARS system in cause of ALF and ACLF. Its function is mainly based on albumin-binding effects to eliminate compounds such as bilirubin or bile acids, as well as water-soluble compounds such as ammonia, creatinine, and urea. Thus, it supports excretory liver and kidney function and leads to improvement of various clinical endpoints, such as hepatic encephalopathy (HE), hemodynamic stabilization, benefit to hepatorenal syndrome, and pruritus.5–8
The OPAL system is based on the MARS device, which was introduced in 1995 by Stange et al. 9 The OPAL system consists of two components: the MARS dialyzer (monitor) and a conventional hemodialysis machine. Within the system, three circuits can be defined: (1) patient blood, (2) albumin, and (3) hemodialysis. Recirculating albumin is regenerated by the Hepalbin adsorbant (Hepalbin-Cluster12) that is attached to the MARS monitor. The cluster itself incorporates powdered activated Hepalbin charcoal on a cellulose framework combined with a cationic binding polymer.10,11 Compared to OPAL, the ALBUNIQUE system consists of only two components: conventional dialysis and a simple infusion pump. Within the system, two circuits can be defined: (1) patient blood and (2) albumin-hemodialysis as one circuit. The principles of the two ECAD systems are depicted in Figure 1 and further described in Table 1. For OPAL, a reduction in bilirubin levels, as a surrogate parameter of ECAD efficiency, as well as a decline of water-soluble creatinine and urea, was clinically shown. 6 However, for ALBUNIQUE, no literature is available regarding detoxification capabilities, efficacy, and efficiency. For that, we retrospectively evaluated all critically ill patients receiving either ALBUNIQUE or OPAL in our institution, with a scope on laboratory and clinical variables. We hypothesize that the detoxification and hemodialysis capacities of both ECAD systems are comparable.
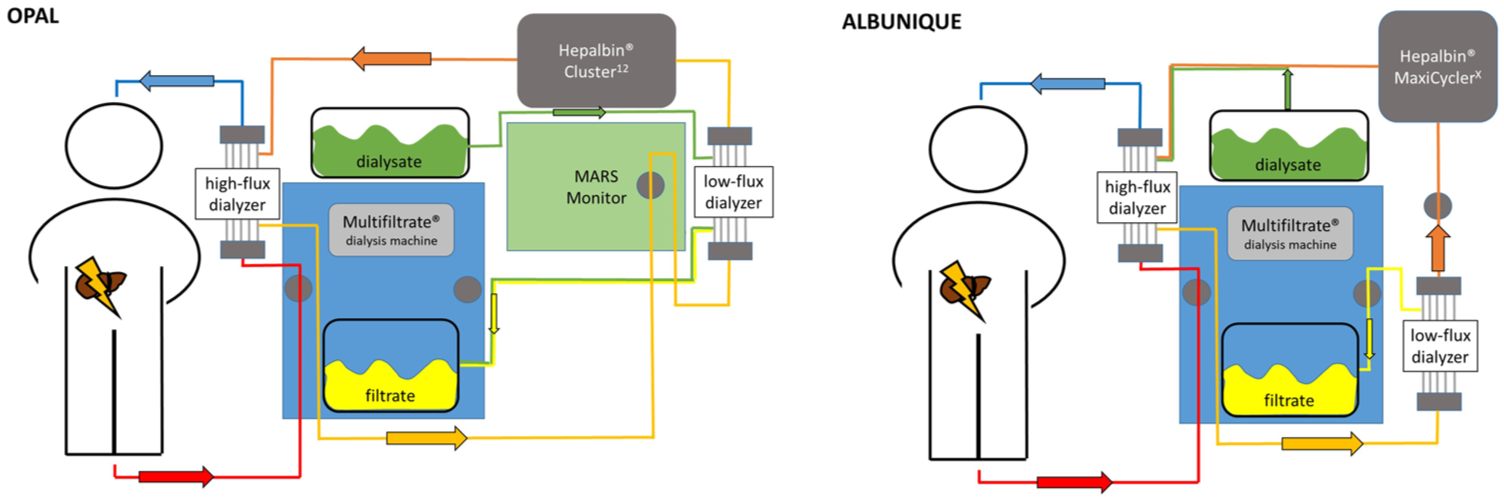
Principles of machine setup among the two different ECAD devices.
Principle mechanism, machine setup, and equipment of the ECAD devices.

Methods
Study design and settings
At the interdisciplinary intensive care unit of our institution the ECAD devices OPAL and ALBUNIQUE were available and frequently in use. Therefore, we performed a retrospective single-centre data analysis of all patients treated with the aforementioned systems between January 2023 and December 2024. ECAD treatments were identified by screening the data management system (SAP, Version 8000.1.8.1161) for Operation and Procedure classification system (OPS) Code 8-858, indicating liver dialysis. All patients (age ⩾ 18 years) with pertinent diagnoses of primary (ALF, ACLF, or transplant failure) or secondary liver failure who underwent ECAD treatment, were included for further evaluation.
The intensivist in charge identified patients with a pertinent diagnosis and scheduled ECAD treatment. According to our local standard operating procedure, ECAD is considered with the following signs and symptoms: (1) liver dysfunction (ALF, ACLF, liver failure after liver surgery) plus (2) plasma bilirubin levels >150–200 μmol/L, and/or (3) symptoms of hepatic encephalopathy (HE) grade II or higher. The selection of using either OPAL or ALBUNIQUE was based on availability and decision of the intensivist.
The study design and evaluation strategy are depicted in Figure 2. The study was approved by the local ethical committee (2025-3666-BO-D).
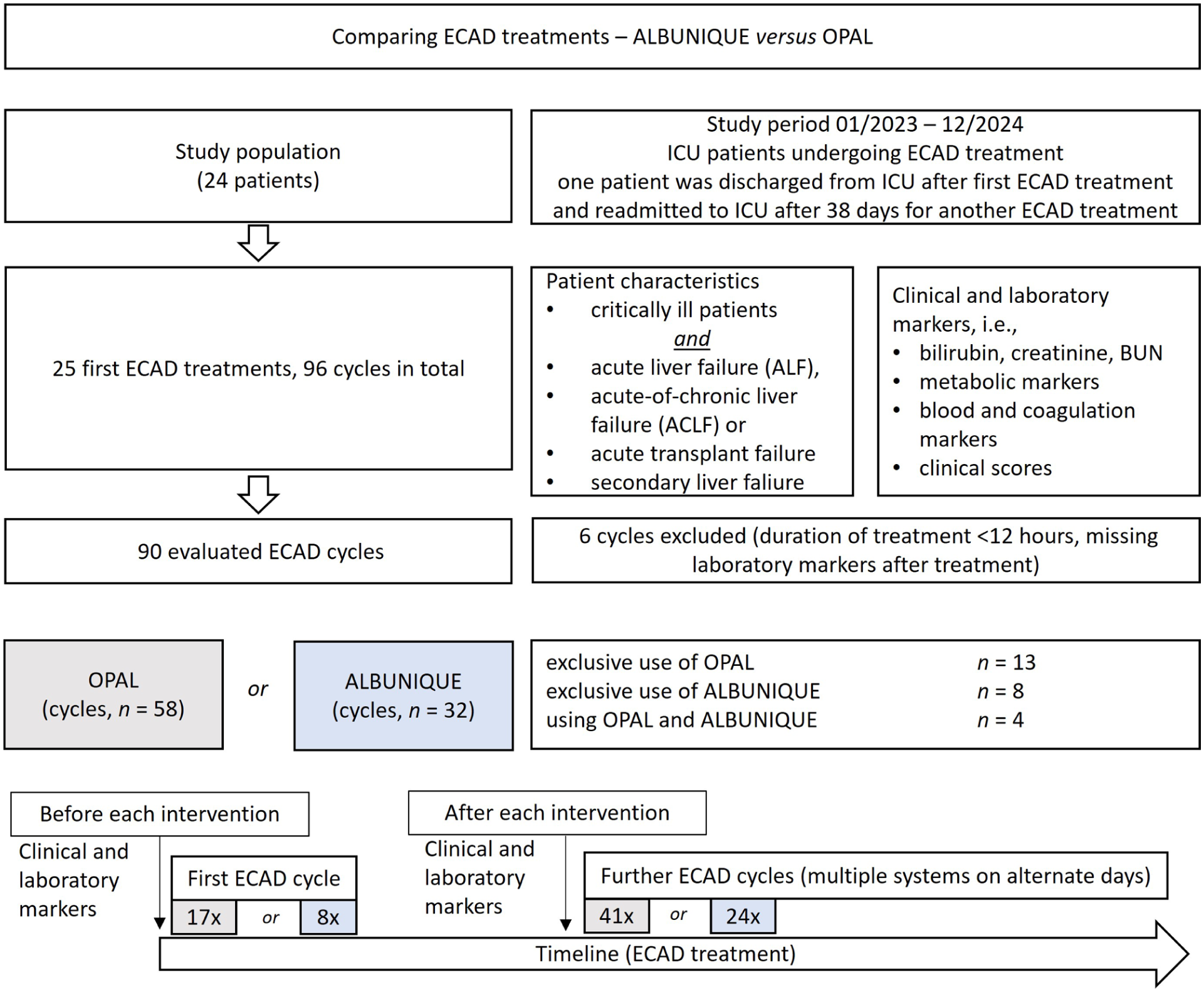
Flowchart depicting the study design and evaluation strategy.
Patient characteristics and periprocedural parameters
Clinical data, including age, gender, diagnosis on admission, and laboratory data were recorded from patient data management system COPRA6Live (Version: 213 (03.02.2020 14:09:06)). Based on the extracorporeal liver support system, treatment cycles were divided into two groups: OPAL and ALBUNIQUE. Patients treated with both systems on different days were also included in the analysis. Treatment cycles with less than 12 h treatment time or missing laboratory values after ECAD were excluded from analysis. Laboratory parameters, health scoring values, and vasopressor requirement (i.e., norepinephrine) before and after each treatment were recorded without correction for hemoconcentration at the end of treatment. Transfusion requirements were recorded during each procedure.
Settings of extracorporeal liver support systems (technical description)
OPAL and ALBUNIQUE were built up according to the manufacturer’s instructions and attached to a standard hemodialysis machine (multiFiltrate, Fresenius Medical Care AG, Bad Homburg v.d. Höhe, Germany). In principle, the device contains three parts: a patient blood circuit, an albumin circuit, and a dialysis circuit.
The OPAL device uses the Hepalbin adsorbant (Hepalbin-Cluster12, Albutec GmbH, Rostock, Germany) attached to the MARS-Monitor (Gambro, Lund, Sweden) and is embedded in the albumin circuit.
The ALBUNIQUE device also uses the Hepalbin adsorbant (Hepalbin-MaxiCycler, Albutec GmbH, Rostock, Germany). However, the system runs independent of the MARS-Monitor. The albumin- and the dialysis circuit are embedded as one circuit and run with an infusion pump.
In both systems the albumin circuit was primed with 400 mL of a 20% human albumin solution (Albutein 20 g/100 mL, Grifols Deutschland GmbH, Germany).
Vascular access was obtained through a double-lumen hemodialysis catheter (Gamcath High Flow Double Lumen Catheter Kit, 13 F, Gambro Kathetertechnik Hechingen, Germany), placed in either a femoral or a jugular vein. Blood flow rates were set at between 100 and 150 mL/min according to the patient’s hemodynamics. Albumin flow rates were set at 150 mL/min in case of OPAL or 20 mL/min in case of ALBUNIQUE according to the manufacturer’s recommendations. Both devices were approved to run for up to 24 h as ECAD and furthermore could be used as common hemodialysis for another 48 h.
Blood anti-coagulation regime was maintained using regional citrate application. For that, a citrate solution (4% sodium citrate; Fresenius Kabi, Bad Homburg, Germany) was applied before the hemofilter aiming a final ionized postfilter calcium concentration of 0.25–0.45 mmol/L, followed by calcium reversal by adding a calcium solution (1 N calcium chloride solution; Serumwerk Bernburg AG, Bernburg, Germany).
Statistical analysis
Data are presented as median values (25th to 75th percentile) and categorical data as number and percentage, unless otherwise indicated. To compare the effects of ECAD on several parameters Generalized Estimating Equations (GEE) were used. This method accounts for correlated data due to repeated measurements of several ECAD cycles and devices per patient. We estimated mean differences between the devices with 95% confidence intervals adjusted for the cycle-specific baseline value of the analysed parameter and duration of treatment cycle, respectively. Categorical variables were analysed by χ2 test. With respect to the first treatment cycle group comparisons were evaluated using Wilcoxon rank sum test. A p-value < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS 27 (IBM SPSS statistics). Figures were designed using SigmaPlot version 14.0 (Systat Software, Erkrath, Germany).
Results
Patient characteristics
In total 24 patients underwent ECAD treatment with either OPAL or ALBUNIQUE from January 2023 to December 2024 in our institution. One of the patients was treated twice in this time period and discharged from ICU after the first ECAD treatment period with in total three treatment cycles and readmitted to ICU after 38 days for a second treatment period with further eight treatment cycles. Thus, leading to n = 25 different patient ECAD treatments with 96 cycles collectively.
After excluding six cycles (treatment duration less than 12 h or missing laboratory markers after treatment) n = 90 cycles were eligible for the final evaluation, thereof n = 58 (64%) OPAL and n = 32 (36%) ALBUNIQUE. Exclusive usage of only one of the systems per patient was in n = 13 (52%) OPAL and in n = 8 (32%) ALBUNIQUE. In n = 4 (16%) patients both systems were in use on alternate days (see Figure 2).
Patients received in median three (2.5–5.0) cycles of ECAD, with a minimum of one and a maximum of 11 cycles per patient. Patients had a median age of 58 (38.0–67.0) years, and the majority (n = 16 (64%)) were of male gender. Deterioration of primary liver failure was the leading cause for ECAD in n = 16 (64%) patients, while n = 7 (28%) patients were treated subsequent to acute transplant failure. N = 2 (8%) patients were treated with ECAD following secondary liver failure in context of multiorgan failure. On ICU admission, patients had a median MELD score of 35 (25.7–40.0), an APACHE-II score of 24 (16.5–32.5), SAPS-II score of 47 (37.5–61.5), and median SOFA score of 12 (6.5–15.5). There were no differences between the patient characteristics between both groups. Detailed data on patient characteristics are presented in Table 2. The median run time of OPAL cycle was 22 (17.8–23.0) h, and for ALBUNIQUE cycle 22 (17.0–25.0) h. The majority of patients (n = 17 (68%)) died within ICU stay.
Patient characteristics on ICU admission, total study group and separated by used system.
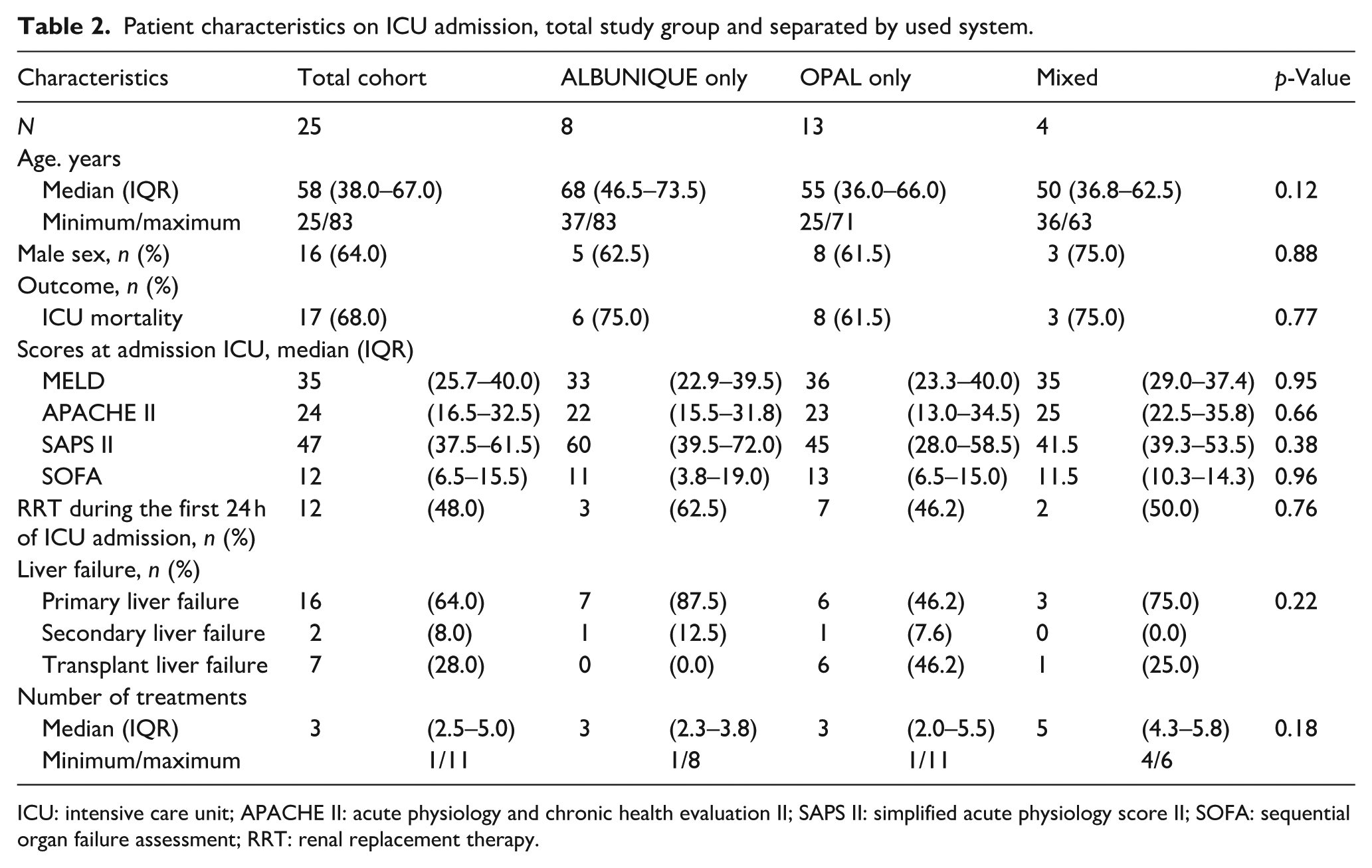
ICU: intensive care unit; APACHE II: acute physiology and chronic health evaluation II; SAPS II: simplified acute physiology score II; SOFA: sequential organ failure assessment; RRT: renal replacement therapy.
Laboratory parameters and clinical scoring
ECAD treatment resulted in a significant decrease of serum bilirubin levels in both devices, with a median bilirubin reduction of −12.0 (−34.4 to −0.3) % (p < 0.01) over all included cycles and −37.3 (−46.3 to −24.5] % (p < 0.01) regarding only the first treatment cycles. For convenience, see Figures 3 and 4. Creatinine levels declined significantly during ECAD treatment, with −16.4 (−31.3 to +2.2) % (p < 0.01) in all cycles and −32.3 (−40.2 to −21.8) % (p < 0.01) after the first application. Urea levels dropped significantly by −10.5 (−24.9 to +2.3) % (p < 0.01) over all cycles and −15.9 (−32.0 to −1.5) % (p < 0.01) after the first run, respectively.
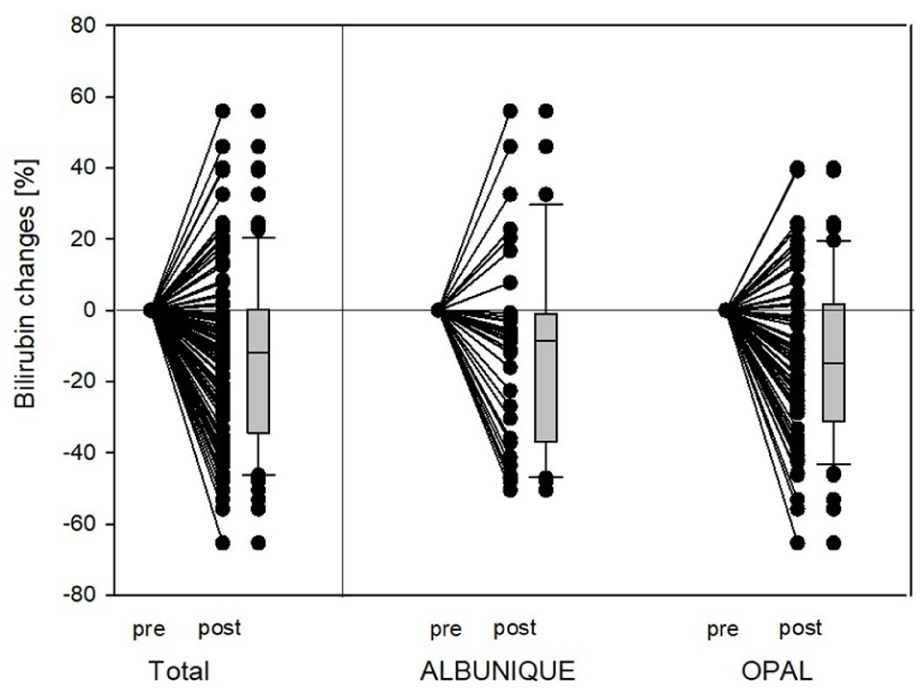
Percental changes of bilirubin levels during ECAD with either OPAL or ALBUNIQUE. Dots represent changes of bilirubin levels after treatment in comparison to initial values (all cycles). Boxplots summarize the entire treatment sessions.
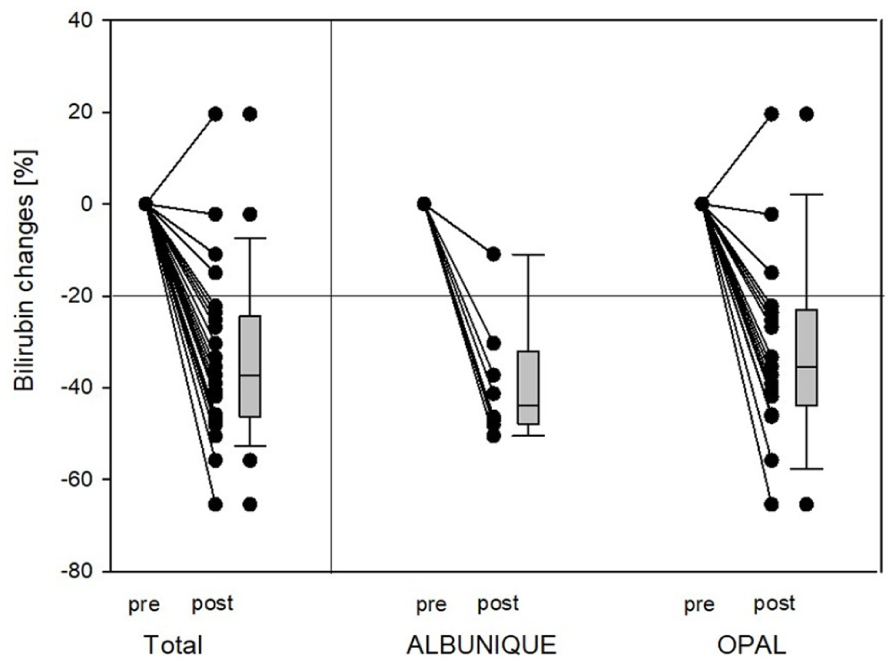
Percental changes of bilirubin levels during ECAD with either OPAL or ALBUNIQUE. Dots represent changes of bilirubin levels after treatment in comparison to initial values (first cycles only). Boxplots summarize the entire treatment session.
Ammonia levels were significantly reduced by −4.8 (−23.5 to +6.4) % (p = 0.015) over all cycles and −5.1 (−31.7 to 8.9) % (p = 0.048) in the first treatment cycle. Base excess (BE) significantly changed during ECAD with a median of −32.8 (−88.7 to +38.2) % (p = 0.049) over all cycles. However, due to high variability, BE levels were not found to be significantly different (−57.1 (−116.1 to +27.1) % (p = 0.44)) after the first treatment cycle. Comparing both ECAD devices, no significant differences were found between the obtained clinical parameters.
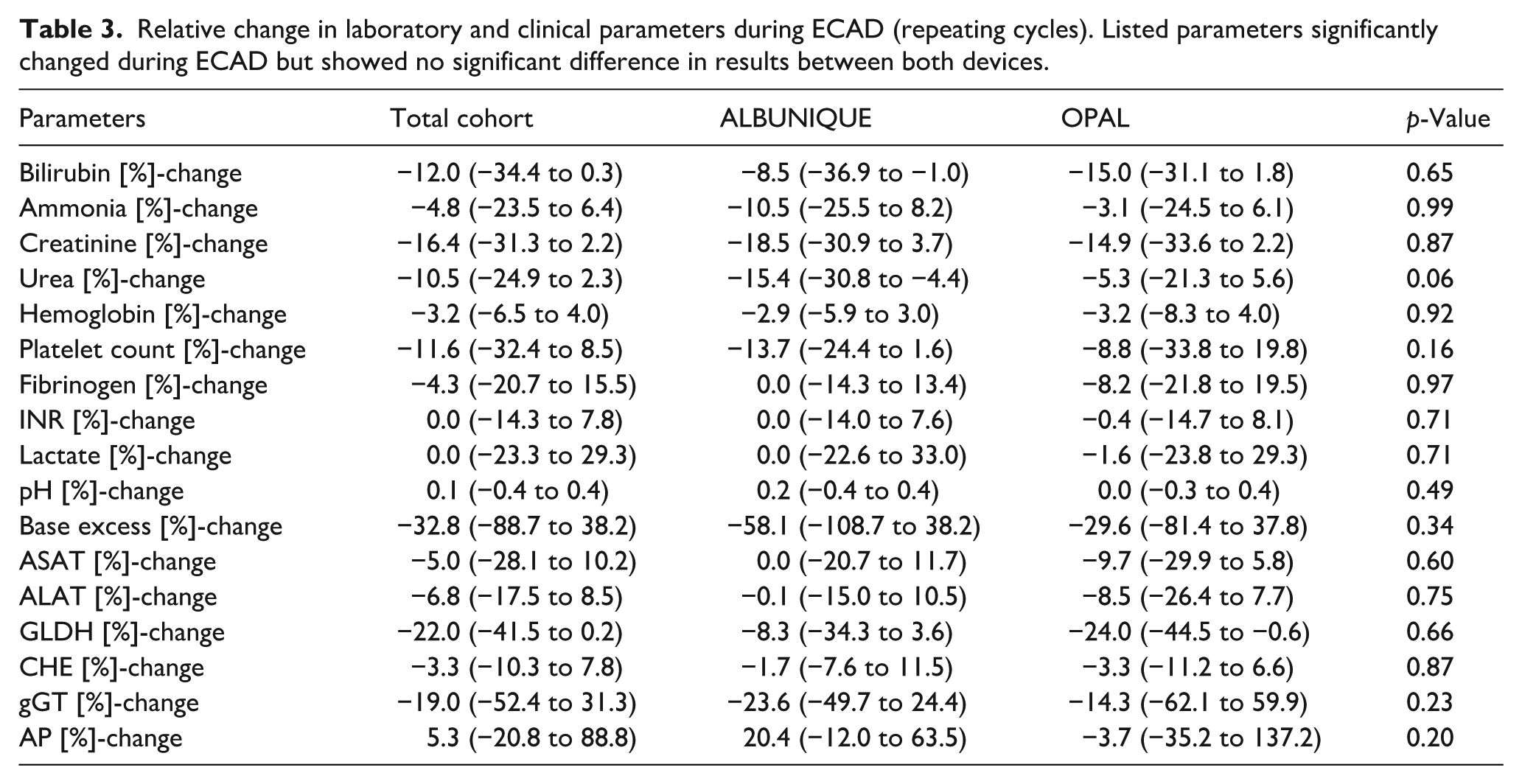
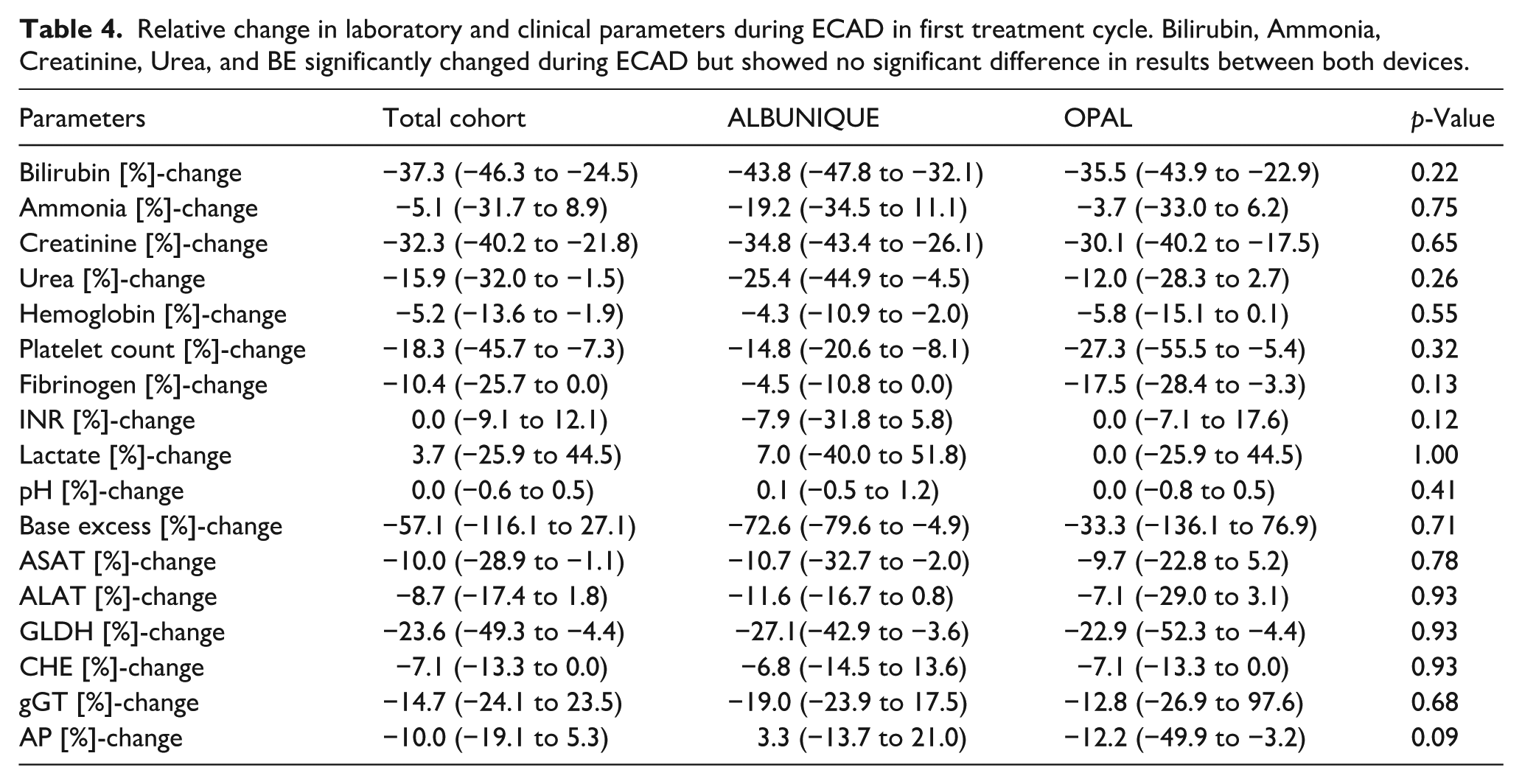
Besides the aforementioned parameters, we found no significant changes in hemoglobin, hematocrit, platelets, fibrinogen, pH, or lactate values, neither across all repeating cycles nor only during the first treatment cycles. Percentage changes of parameters of all repeating ECAD cycles are shown in Table 3, percentage changes of parameters of the first ECAD cycle are shown in Table 4, and in more detail in Supplemental Tables S1 and S2.
Relative change in laboratory and clinical parameters during ECAD (repeating cycles). Listed parameters significantly changed during ECAD but showed no significant difference in results between both devices.
Relative change in laboratory and clinical parameters during ECAD in first treatment cycle. Bilirubin, Ammonia, Creatinine, Urea, and BE significantly changed during ECAD but showed no significant difference in results between both devices.
Bleeding tendency, transfusion requirement, adverse events
Out of all patients, n = 5 (20.0%) showed spontaneous bleeding tendency on ICU admission. Bleeding tendency was also seen before and during n = 5 (20.0%) and after (n = 6 (24.0%) ECAD treatment. In relation to all included treatment cycles (n = 90 (100%)), bleeding tendency was reported whilst admission to ICU (n = 16 (17.8%)), before, (n = 22 (24.4%)), during (n = 21 (23.3%)), and after (n = 19 (21.1%)) application of ECAD. Nevertheless, transfusion during ECAD was commonly rare. Red packed blood was substituted in 0 (0–4) cases, while platelets, coagulation factor I, prothrombin complex (factor II, VII, IX, and X) or fresh frozen plasma were not substituted while on ECAD. Three treatment sessions of ECAD (one ALBUNIQUE and two OPAL) were prematurely terminated due to filter clotting. In all other cycles, no ECAD-related adverse events were reported.
Discussion
In the current literature, only a few retrospective studies or case reports are available comparing different ECAD systems with focus on detoxification capacity, processing times, anticoagulation requirements, periprocedural bleeding or reduction of pruritus. For all investigated devices, removal of albumin-bound as well as water-soluble substances was shown. Moreover, most systems were able to stabilize metabolic (dys-)function.6,10 As one of the newest advances in the development of ECAD devices, the ALBUNIQUE system was introduced into clinical practice. Therefore, studies investigating the effects of ALBUNIQUE are of utmost importance. Our study is the first to compare ALBUNIQUE with other ECAD systems. As OPAL and ALBUNIQUE were based on the same mode of albumin regeneration via a novel type of charcoal adsorber, this evaluation lend focus on detoxification capabilities between the both devices.
The results of our retrospective single-centre study can be summarized as follows:
Both devices were effective in removing bilirubin, without differences between the devices.
Both devices were able to remove water-soluble substances, i.e., creatinine and urea, and were able to stabilize metabolic dysfunction by negative base excess, without significant differences between the devices.
Ammonia levels were significantly reduced during OPAL and ALBUNIQUE treatments.
Periprocedural bleeding was a common finding in our ECAD patient cohort. However, our data found no increased bleeding risk during or after ECAD treatment in general or for each of the devices. Moreover, application of blood products were rare.
ECAD devices were established to remove hydrophobic substances accumulating under compromised liver function and represent a valuable therapeutic tool treating various types of liver failure. 7 MARS, developed by Stange and Mitzner in the 1990s, is a widely known and well-studied albumin-dialysis method since its broad clinical use in 1998.8,12
OPAL represents a further development of the MARS system. The classical OPAL setup requires the same technical configuration, employing a MARS monitor and a continous venovenous hemodialysis (CVVH), but a novel charcoal adsorber replaces the former adsorber columns. In a randomized multicentre cross-over study comparing the efficiency of OPAL and MARS, similar reduction rates of bilirubin in both systems were described. However, only OPAL was able to improve albumin binding capacity and to reduce significant higher bile acid levels. Moreover, OPAL provided a longer run time of up to 24 h in comparison to MARS, which was usually terminated after 8 h of application. 13 Within the ALBUNIQUE concept, the novel type of charcoal adsorber is implemented within a conventional hemodialysis (CVVH) circuit, without the need for the MARS monitor. Therefore, ALBUNIQUE might be easier to set up as OPAL, and may provide longer application times compared to MARS.
Decreasing serum bilirubin levels during ECAD reflect the surrogate to describe and monitor the removal of albumin-bound toxins in clinical practice. The ability of OPAL to reduce bilirubin was shown within the literature compared to MARS or other ECAD devices.6,10,14 In the present study, the OPAL device was able to reduce patient’s bilirubin levels significantly. The reduction was more pronounced within the first treatment cycle, but also found to be consistent over all treatment cycles. The same pattern was seen in the patient group with ALBUNIQUE treatment. Therefore, we found no significant differences between both devices. However, based on the retrospective study design, we were not able to provide data regarding bile acid removal or improvement of albumin binding capacity, as these parameters do not represent daily clinical routine. Within the literature bile acid reduction 15 as well as improvement of albumin binding capacity 16 were shown to be related to the degree of liver dysfunction.
As both ECAD systems were combined with continuous venovenous hemodialysis (CVVH), creatinine and urea levels significantly declined during the treatment period. Sufficient reduction of water-soluble substances were already shown for OPAL compared to MARS and other ECAD devices.6,10,14 However, both devices OPAL and ALBUNIQUE offer a longer cycle run time compared to MARS of up to 24 h without interruption of RRT treatment with consequently more effective reduction of water-soluble substances. Currently, little is known about renal recovery after ECAD treatment. Improving renal function and hepatorenal syndrome was only shown in patients treated with MARS so far. 17 In the light of modern biomarkers (e.g., NGAL 18 or Cystatin-C 19 ) for the prediction and monitoring of renal function, future research could implement the role of ECAD application in monitoring hepatorenal syndrome in this field.
Liver dysfunction and subsequent multiorgan failure are often associated with metabolic derangement, including pH and base excess values. 20 Metabolic derangement itself has tremendous impact on hemodynamic stability and catecholamine dependency. Therefore, ECAD application aims to stabilize patient’s metabolic function by (1) improving liver function and (2) resolving acidosis by alkaline dialysis. 21 In this respect, MARS was shown to reduce the requirement of vasoactive substances and stabilization of patient hemodynamics after several treatment cycles. 7 Moreover, ECAD—including OPAL—was shown to significantly increase metabolic derangements—defined by lowered pH and base excess levels. 6 In the current study both devices were shown to increase negative base excess and low pH levels. However, we found no impact on catecholamine requirements, probably related to limited application of ECAD and pronounced critical illness in our patient cohort.
Encephalopathy represents a key finding in hepatic dysfunction and is one of the diagnostic elements in defining hepatic failure. 22 Among others, increased ammonia levels were one of the pathophysiological aspects resulting in hepatic encephalopathy. 23 As ammonia measurement is implemented in clinical routine, the role of ECAD improving HE by reducing ammonia levels seem reasonable. In this respect, OPAL was able to reduce high ammonia levels in critically ill patients with liver dysfunction 6 and both, MARS and OPAL, showed improvement of hepatic encephalopathy. 13
Coagulation disorder is a common finding among patients with severe liver dysfunction and carries the risk of spontaneous bleeding and coagulation.24,25 Within the literature, disturbances in coagulation were not correlated to bleeding risk and therefore, correction is not recommended prior to ICU stay related invasive procedures, except for disturbances in platelet count (<20 Gpt/l). 26 Bleeding tendency was also a common finding within the current study population, most notably from mucosal tissue, gastrointestinal tract (GIT) or indwelling catheter sites. ECAD did not enhance bleeding tendency. Therefore, transfusion of blood products was rare among the patient population. ECAD was reported a safe procedure in the light of periprocedural bleeding and application of blood products6,27–29 and in line with previous reports.30,31 In the light of ECAD, a Delphi consensus of international experts recommended initiation of ECAD only with platelet counts of >50 Gpt/l and fibrinogen levels of >1 g/L. 5
Limitations of the current study include the retrospective character, small sample size and the monocentric study design. Moreover, the allocation of OPAL versus ALBUNIQUE treatments was not undergoing a randomization process and may therefore have an effect towards selection bias. Due to the limited data on beneficial indications for ECAD a relatively small sample size was recruited in the current analysis, especially regarding the ALBUNIQUE group. Thus, the results of the study may be interpreted in terms of considerations rather than generalization. However, our study truly reflects clinical routine of a special ICU patient cohort.
Conclusion
Both ECAD systems OPAL and ALBUNIQUE showed to be effective in removing bilirubin, reducing ammonia levels, eliminating water-soluble substances and stabilizing metabolic dysfunction without significant differences between the devices. Our data found no increased bleeding risk during or after application of ECAD treatment in general or for one of the examined devices. Further studies are needed focusing on bile acid reduction or enhancement of albumin binding capacities and with respect of ECAD treatment to organ recovery and survival benefit in this crucial patient cohort.
Supplemental Material
sj-docx-1-jao-10.1177_03913988261429916 – Supplemental material for Comparison of the liver support systems OPAL and ALBUNIQUE in critically ill patients with liver dysfunction
Supplemental material, sj-docx-1-jao-10.1177_03913988261429916 for Comparison of the liver support systems OPAL and ALBUNIQUE in critically ill patients with liver dysfunction by Aron Krause, Julia Leonhardt, Christian von Loeffelholz, Christoph Sponholz and Oliver Sommerfeld in The International Journal of Artificial Organs
Footnotes
Author contributions
OS, CS, and AK: Concept/design; Data collection/analysis/interpretation; Statistics; Drafting article. CvL and JL: Data interpretation; Critical revision of article, Approval of article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AK, JL, CS and OS declares that there is no conflict of interest. CvL received honoraria for lectures, presentations or educational events from Fresenius Kabi, not related to this work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
