Abstract
Introduction:
Hemodialysis (HD) represents a necessary medical intervention for patients with end stage kidney disease (ESKD) with a high carbon footprint that significantly consumes natural resources (i.e. water). Reduction of dialysate flow rate is one strategy that directly targets water waste during HD.
Methods:
A retrospective multicenter, observational study was conducted over 12 months in patients with AKI-D and ESKD to evaluate treatments with mid-range dialysate flow rates set at Qd 300 mL/min (Green HD) using the Tablo® HD System (Outset Medical) versus flow rates ⩾Qd 500 mL/min (Conventional systems).
Results:
One thousand one hundred ninety-five treatments were performed in 433 patients using Green HD (Qd = 300 mL/min, n = 575) and Conventional systems (Qd ⩾500 mL/min, n = 620). Mean Qd for Green HD was 300.0 ± 0.0 and mean Qd for the Conventional systems was 686.6 ± 88.3 mL/min. Mean blood flow rates were significantly lower among patients treated with Green HD. Reductions in TACurea and post dialysis serum potassium were similar between the two groups. Estimates for resource utilization were lower using Green HD compared to Conventional systems.
Conclusion:
These findings support that higher dialysate flow rates using Conventional systems yield minimal if any benefit in small solute clearance compared to mid-range flow rates. Green HD using the Tablo® HD System results in comparable benefits in ESKD patients with significant conservation of water resources.
Keywords
Introduction
While generally recognized for its positive contributions to modern society, the global healthcare system has a significant negative impact on the environment. High carbon emissions, energy expenditure, excessive water consumption, and the development of medical wastewater containing biomedical toxins and environmental pollutants (e.g. radioactive and pharmacologically active compounds, contaminants, heavy metals, and pathogenic microbes) are key issues that influence the biosphere. 1 Water waste creates a multitude of sustainability challenges including reduced energy reserves, water security, and overall community health. Water conservation efforts aim to reduce upfront water usage and also recycle wastewater for different purposes such as irrigation, laundry, and sanitation. As water scarcity increases worldwide, it is imperative that medical facilities focus on the development of healthcare strategies that reduce water consumption and wastewater generation.2,3
The medical field of nephrology, especially as it relates to hemodialysis (HD), significantly contributes to the high utilization of water and energy resources, and the generation of excessive waste. 4 A call-to-action has been sounded in the field with raised awareness focused on environmentally sustainable practices in dialysis. 5 Patients with end stage kidney disease (ESKD) commonly undergo HD, an interventional treatment that aims to filter and remove waste products and excess fluid from the blood. This purification process consumes a large quantity of water in the creation of the dialysate fluid used to filter waste products from blood. Most devices employ reverse osmosis (RO) for purification and generate a significant amount of wastewater as a byproduct. 4
Water conservation in HD is considered an important responsibility of healthcare practitioners all over the world. Further investigation into the utility of reduced dialysate flux is needed to broaden the understanding of how these techniques can be used in order to significantly reduce water consumption during chronic HD while still ensuring optimum efficacy and efficiency. Multiple reports have demonstrated the ability of mid-range dialysate flow rates (Qd; e.g. Qd300 mL/min) to achieve the recommended URR and Kt/Vurea targets in both the acute and chronic environments.6,7 However, broad-based real-world data on clearance parameters utilizing mid-range rates are lacking. The primary aim of this investigation, the Comparative Outcome Measures of Patients Receiving Acute Renal Therapy (“COMPARE”) study, was to evaluate solute control through reduced water usage in a relatively large population of hospitalized patients with ESKD or acute kidney injury requiring dialysis (AKI-D).
Materials and methods
Study design and participants
A retrospective multicenter, observational study (ClinicalTrials. gov identifier NCT04912024) was conducted at nine facilities in the AdventHealth Orlando (Orlando, FL, USA) and Texas Health Harris Methodist Hospital (Fort Worth, TX, USA) hospital systems over a 12-month period (June 2021 through June 2022). The study was reviewed and approved by the Advarra Institutional Review Board (Pro00046206). In accordance with IRB guidance, a waiver of informed consent was granted, as the research involved no more than minimal risk to participants. Conduct of the study adhered to the Declaration of Helsinki and all study site personnel underwent standard training performed by Outset Medical, Inc. on required study procedures.
Inclusion criteria were age ⩾18 years and weight ⩾34 kg. All participants were hospitalized with dialysis-dependent ESKD or AKI-D and received at least one HD treatment at Qd300 mL/min or ⩾ Qd500 mL/min using the Tablo® HD system (Tablo) or conventional systems, respectively. Device assignment was dependent on availability at each site. Sites were not selected based on device availability and thus, the devices utilized were those already in practice at their respective facilities. Treatments were excluded if they were for isolated ultrafiltration, had a prescribed treatment duration of <2 h, did not have sufficient lab and prescription data, or were prescribed Qd <300 mL/min on Tablo or Qd <500 mL/min on other systems.
Procedures and data collection
Prescribed and actual treatment parameters were obtained from each patient’s electronic medical records. Data included Qb and Qd, dialysate composition, access type (e.g. tunneled or non-tunneled catheter, arteriovenous graft or fistula), dialyzer type, pre-dialysis weight, and treatment duration. Patients were grouped into those receiving a mid-range dialysate flow rate of Qd300 mL/min via Tablo (Green HD Group) and those treated at a higher dialysate flow rate with a Qd of ⩾500 mL/min on conventional systems (Conventional systems group). Prescribed treatment parameters, including choice of high-flux dialyzer, were based on hospital standard of practice and nephrologist preference.
Blood samples were obtained on all participants pre- and post-treatment. Pre-treatment lab samples were obtained pre-dialysis on the same day as treatment. Post-treatment samples were obtained at least 1 h and no more than 24 h post-treatment. Blood urea nitrogen (BUN), serum bicarbonate (HCO3), and serum potassium (K+) concentrations were measured using standard clinical laboratory methods. Total time-averaged concentration of blood urea nitrogen (TACurea) 8 was calculated to account for differences in body weight, pre-dialysis BUN concentrations, HD session length, and variable time of post-dialysis laboratory measurements.
Tablo® HD System
The Tablo® HD System (Outset Medical, San Jose, CA, USA; “Tablo”) is FDA cleared for the treatment of acute and chronic kidney failure with or without ultrafiltration and generates a maximum Qd of 300 mL/min. Tablo purifies source water and creates dialysate on demand without the need for an external RO from standard concentrates and utilizes novel sensor based, flow balancing technology to deliver highly precise blood flow and mid-range dialysate flow rates to maximize hemodialysis efficacy at these parameters. Through the use of mid-dialysate flow rates and a software controlled, internal dual RO system, the system significantly reduces water consumption during the administration of treatment by approximately 40%–60%. Use of Tablo was selected for the “Green HD” group.
Statistical analysis
The data is presented using continuous variables as mean ± standard deviation, comparing groups with Student’s t-test, and categorical variables as proportions, comparing groups with Fisher’s Exact test. Given the relatively large sample size and multiple comparisons, a p-value of <0.01 was considered to be statistically significant.
Results
Baseline characteristics
A total of 433 hospitalized patients undergoing 1195 treatment sessions were screened by study site personnel and included for analysis. Within this cohort, 408 patients received all treatments within a single category representing 528 treatments at 300 mL/min and 554 treatments at ⩾500 mL/min. Twenty-five patients received at least one treatment in each category, 300 mL/min and ⩾500 mL/min (47 and 66 treatments, respectively).
Baseline demographic characteristics of participants, and general details of the dialysis prescription are presented in Table 1. In addition to the sizeable difference in Qd (300.0 ± 0.0 mL/min vs 686.6 ± 88.3 mL/min, p < 0.01), patients in the Green HD group were on average larger (mean body weight 93.7 ± 29.8 vs 83.8 ± 26.7, p < 0.01), prescribed significantly lower mean Qb (332.5 ± 44.1 mL/min vs 342.7 ± 43.5 mL/min, p < 0.01), and had a generally less favorable distribution of vascular access due to a higher proportion of temporary (non-tunneled) catheters. Access type for the Green HD group showed 31.0% arteriovenous fistula (AVF), 6.8% arteriovenous graft (AVG), 18.0% non-tunneled central venous catheter (CVC) and 44.2% tunneled CVC. Access type for Conventional systems group showed 33.4% AVF, 3.2% AVG, 10.5% for non-tunneled CVC, and 52.9% for tunneled CVC. Dialyzer size was similar among groups. The dialysate sodium, potassium, calcium, base, and fluid temperature of the prescribed dialysate concentrations were similar between both treatment groups.
Patient baseline parameters.
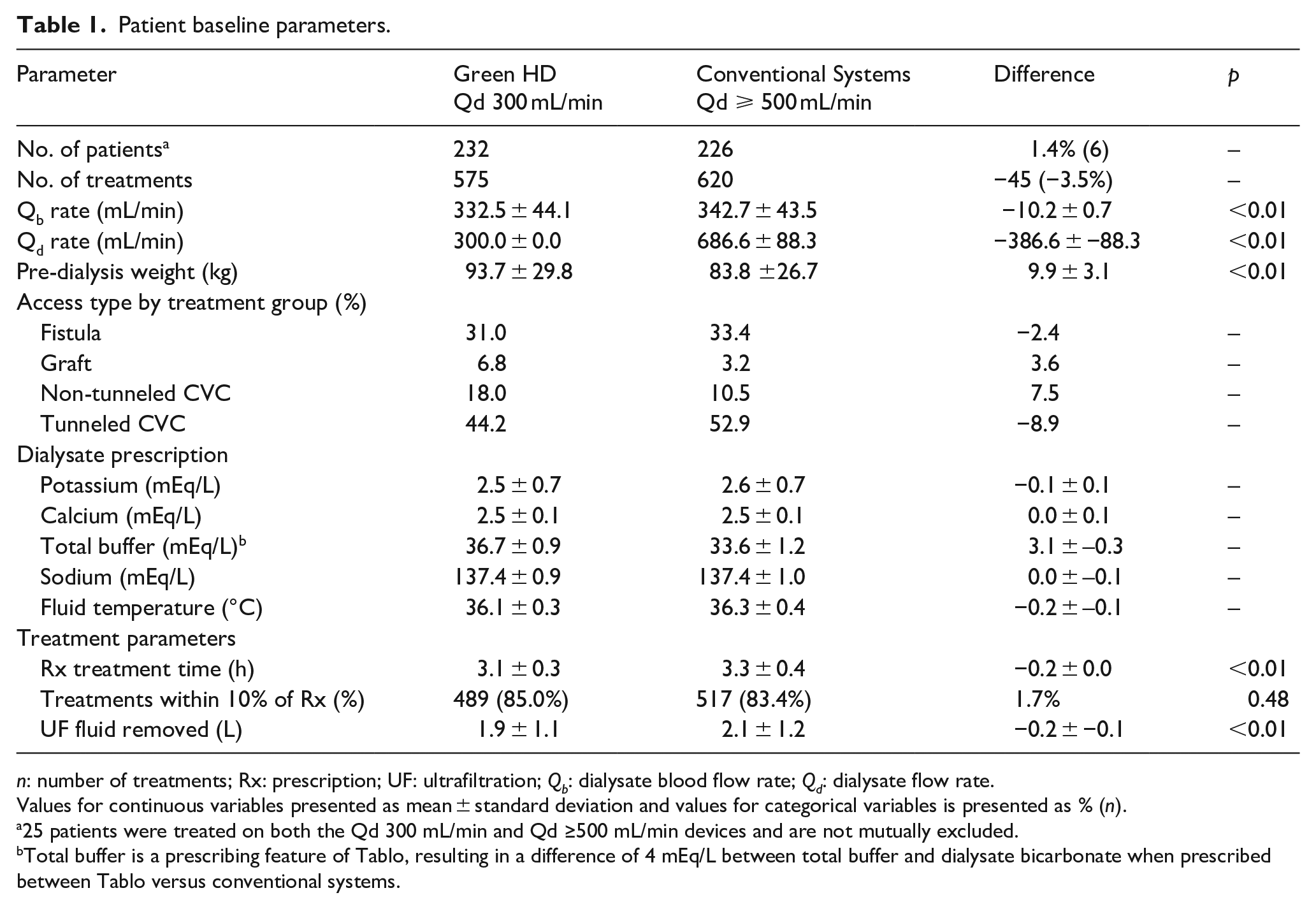
n: number of treatments; Rx: prescription; UF: ultrafiltration; Qb: dialysate blood flow rate; Qd: dialysate flow rate.
Values for continuous variables presented as mean ± standard deviation and values for categorical variables is presented as % (n).
25 patients were treated on both the Qd 300 mL/min and Qd ≥500 mL/min devices and are not mutually excluded.
Total buffer is a prescribing feature of Tablo, resulting in a difference of 4 mEq/L between total buffer and dialysate bicarbonate when prescribed between Tablo versus conventional systems.
Treatment and lab parameters
The prescribed dialysis session length (3.1 ± 0.3 h vs 3.3 ± 0.4 h) and ultrafiltration fluid removal were significantly lower (1.9 ± 1.1 vs 2.1 ± 1.2, p < 0.01) for treatments in the Green HD group (p < 0.001; Table 1). Individual treatments achieving at least 90% of their prescribed session length were similar (85% vs 83.4%; p = 0.48) between groups. When adjusted for body weight, rates of fluid removal were slightly higher in patients using Conventional systems (0.127 mL/min/kg or 7.59 mL/kg/h/kg) compared to those in the Green HD group (0.110 mL/min/kg or 6.54 mL/kg/h) due to the higher body weight within the Green HD group. Green HD patients had significantly higher BUN concentrations pre- and post-treatment and were more likely to present with hyperkalemia (⩾5.5 mEq/L). Both groups had similar post-dialysis K+ and HCO3 concentrations, and similar percent reductions in BUN (Table 2). To adjust for variability in post-treatment blood sampling (>1–>24 h), laboratory parameters were stratified by timing of post-session blood sampling. Treatments associated with post-dialysis laboratory determinations obtained fewer than 12 h post-dialysis were analyzed separately (Green HD, n = 47; Conventional systems, n = 36). Similar to total cohort findings, the Green HD group was significantly heavier, spent less time on treatment, and had higher BUN levels pre (p = 0.007) and post-treatment (p = 0.002, Table 2). Similar post-K+ levels, post-HCO3 concentrations, and comparable percent reductions in BUN levels were observed (Table 2). A sub analysis was performed on the 25 patients who received at least one treatment at Qd 300 mL/min and at least one treatment at Qd 500 mL/min. Despite significant differences in mean Qd (300.0 ± 0.0 mL/min vs 691.1 ± 84.7 mL/min; p < 0.01) and treatment duration (3.0 ± 0.3 h vs 3.3 ± 0.3 h; p < 0.01), there were no differences in post-treatment K+, HCO3, or TACurea.
Lab parameters: pre- and post-treatment and by post-treatment lab timing.
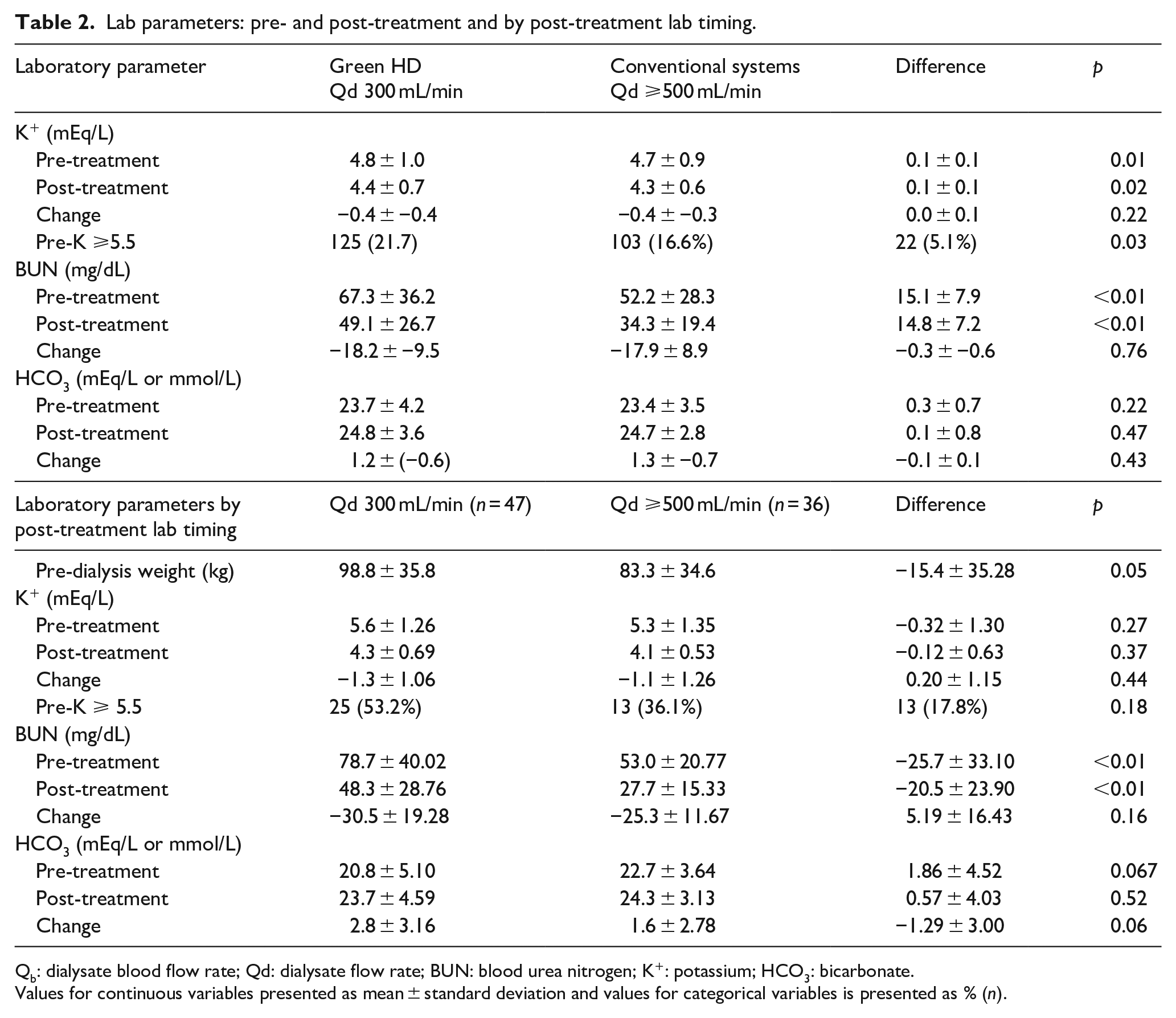
Qb: dialysate blood flow rate; Qd: dialysate flow rate; BUN: blood urea nitrogen; K+: potassium; HCO3: bicarbonate.
Values for continuous variables presented as mean ± standard deviation and values for categorical variables is presented as % (n).
A companion analysis stratified by body size comparing treatment and laboratory data for each group based on pre-dialysis patient weight (<90 and ⩾90 kg) demonstrated a higher proportion of sessions in the Green HD group were performed on heavier patients (50.1% versus 32.3%, p < 0.01). In addition to the expected difference in prescribed dialysate flow rate, it was noted that treatment time was significantly shorter for the Green HD group across both weight strata (3.0 ± 0.3 h vs 3.3 ± 0.3 h, p < 0.01 and 3.1 ± 0.3 h vs 3.3 ± 0.4 h, p < 0.01; Supplemental Table 1).
Resource utilization
Compared to conventional systems, Green HD reduced water volume use by 2.3× per patient and would decrease usage by ~25,000 L per patient annually. Water usage calculations assume a 50% RO efficiency based on prescribed dialysate flow rate. Patients in the Conventional systems group receiving three treatments weekly at 3.5 h per treatment at an average Qd of 686 mL/min would utilize approximately 45,000 L of water per year compared to less than 20,000 L used by patients in the Green HD group (56.3% reduction; Table 3).
Resource utilization estimations in mid-range vs. higher dialysate flow rates.
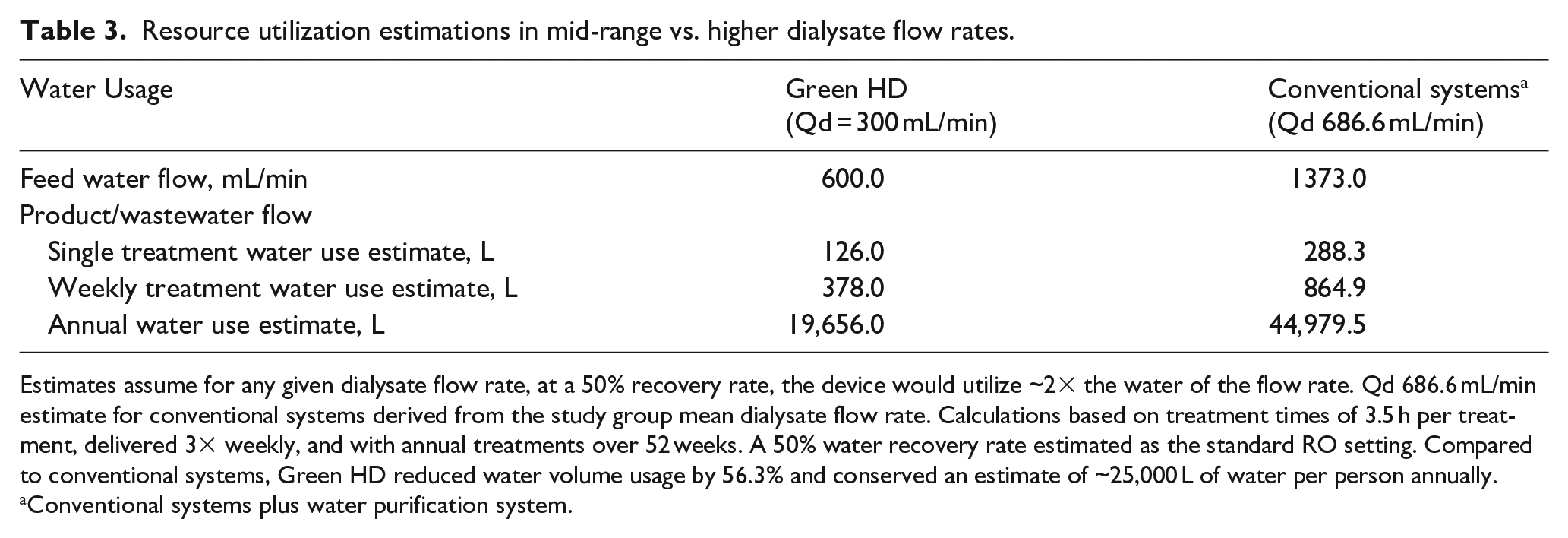
Estimates assume for any given dialysate flow rate, at a 50% recovery rate, the device would utilize ~2× the water of the flow rate. Qd 686.6 mL/min estimate for conventional systems derived from the study group mean dialysate flow rate. Calculations based on treatment times of 3.5 h per treatment, delivered 3× weekly, and with annual treatments over 52 weeks. A 50% water recovery rate estimated as the standard RO setting. Compared to conventional systems, Green HD reduced water volume usage by 56.3% and conserved an estimate of ~25,000 L of water per person annually.
Conventional systems plus water purification system.
Discussion
The current study demonstrated that dialysate flow rates of 300 mL/min yielded no decrement in small solute clearance relative to Conventional systems utilizing dialysate flow rates between 67%, and on average, more than 100% higher. In this study population, patients treated with mid-range dialysate flow rates were larger and treated at lower blood flow rates—both of which would have predicted poorer efficiency of solute clearance. While allocation to the two systems was not randomized, these findings suggest that among hospitalized patients receiving HD, a dialysate flow rate substantially higher than 300 mL/min does not provide patients with a meaningfully “better” treatment. These findings expand on previous reports that higher Qd has a limited effect on in vivo urea clearance and again challenges the assumption that increased urea mass transfer observed at higher dialysate flow rates with various high flux dialyzers in vitro can be extrapolated in vivo.9 –11
Estimations for water resource utilization in the Green HD group showed a significant reduction in water expenditure compared to Conventional systems. According to the 2023 Centers for Disease Control and Prevention’s annual report on CKD in the United States (US), more than 808,000 individuals in the US are living with ESKD and 68% of them are on dialysis (549,440 patients). 12 Assuming the common chronic dialysis schedule of 3×/week, this equates to over 70 million dialysis treatments annually in the US. With the additional acute HD treatments for hospitalized patients with ESKD or AKI-D delivered annually, there exists a monumental opportunity for water conservation with a standardized reduction to mid-range dialysate flow rates.
Globally, it is estimated that more than 5 million people will require chronic dialysis treatment by 2030. 13 The field of nephrology must explore environmentally sustainable care, especially as HD is recognized for its disproportionately high and repetitive utilization of water resources. 14 Education efforts for nephrologists and all health care professionals regarding the environmental cost of medical therapy must continue and timely deployment of strategies to reduce this impact are critical.
Limitations of this study include the lack of a control arm and fixed parameters other than Qd. Given variability of post-dialysis blood sampling, URR could not be calculated on the majority of sessions. The current study focused on regulation of smaller molecular weight substances, but did not explore the control of middle molecules which may also contribute to uremic symptoms and adverse health events related to kidney failure. Larger RCTs including evaluation of middle and large weight molecules would be necessary to fully evaluate the impact of mid-range dialysate flow rates in ESKD and AKI-D.
Conclusions
This multi-center study comparing a Green HD arm using mid-range Qd 300 mL/min with the Tablo HD System to Conventional systems delivering higher Qd (686.6 mL/min) demonstrates no change in the control of small solutes achieved at similar treatment times across a wide range of patient weights and volumes of distribution. The Tablo HD System achieves similar clearance parameters using less than half of the water of Conventional systems with typical dialysate flow rates. Expansion of evidence supporting use of mid-range Qd rates provides a path to reducing the environmental burden associated with the HD for the treatment of acute and ESKD.
Supplemental Material
sj-pdf-1-jao-10.1177_03913988251337228 – Supplemental material for Utility of a water-sparing approach for reducing dialysate flow rates in hospitalized patients with kidney failure
Supplemental material, sj-pdf-1-jao-10.1177_03913988251337228 for Utility of a water-sparing approach for reducing dialysate flow rates in hospitalized patients with kidney failure by Amay Parikh, Kirsten Drake, Craig Woda, Cynthia D’Alessandri-Silva and Michael Aragon in The International Journal of Artificial Organs
Footnotes
Acknowledgements
The authors would like to recognize and thank Ms. Yaadveer Chahal (Outset Medical, Inc.) for her overall management of the study; and the patients, clinicians, and research teams at the participating research sites. We thank Stephanie E. Tedford, PhD, of Pharmacologics, Inc, who, on the behalf of Outset, Inc. assisted in the implementation of author revisions.
Author contributions
The study was conceptualized by Dr. Aragon and Mr. Schumacher. Data was gathered by Dr. Parikh and Ms. Drake and analyzed by Mr. Schumacher and Mr. Gunter. The manuscript was drafted by Mr. Schumacher, Dr. D’Alessandri-Silva, Dr. Woda, and Dr. Parikh, and was edited by Dr. Aragon, Dr. D’Alessandri-Silva, Dr. Woda, and Ms. Drake.
Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available due to the study sponsor’s liability of competitive exposure. They are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Aragon and D’Alessandri-Silva are employees of Outset Medical, Inc. The rest of the authors have no Conflict of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by Outset Medical, Inc.
Ethics and consent statement
The study was reviewed and approved by the Advarra Institutional Review Board (Pro00046206). In accordance with IRB guidance, a waiver of informed consent was granted, as the research involved no more than minimal risk to participants.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
