Abstract
Background:
Fontan procedure, the standard surgical palliation to treat children with single ventricular defects, causes systemic complications over years due to lack of pumping at cavopulmonary junction. A device developed specifically for cavopulmonary support is thus considered, while current commercial ventricular assist devices (VAD) induce high shear rates to blood, and have issues with paediatric suitability.
Aim:
To demonstrate the feasibility of a small, valveless, non-invasive to blood and pulsatile rotary pump, which integrates impedance and peristaltic effects.
Methods:
A prototype pump was designed and fabricated in-house without any effort to optimise its specification. It was then tested in vitro, in terms of effect of pumping frequency, background pressure differences and pump size on output performance.
Results:
Net flow rate (NFR) and maximum pressure head delivery are both reasonably linearly dependent on pumping frequency within normal physiological range. Positive linearity is also observed between NFR and the extent of asymmetric pumping. The device regulates NFR in favourable pressure head difference and overcomes significant adverse pressure head difference. Additionally, performance is shown to be insensitive to device size.
Conclusions:
The feasibility of the novel rotary pump integrating impedance and peristaltic effects is demonstrated to perform in normal physiological conditions without any optimisation effort. It provides promising results for possible future paediatric cavopulmonary support and warrants further investigation of miniaturisation and possible haemolysis.
Introduction
Functional single ventricular defects are a group of congenital cardiac malformations characterised by a lack of two well-developed ventricles and not amendable via biventricular repair. 1 Single ventricular defects are rare (85 per million live births) but of increasing concern, not least because of the high healthcare utilisation of this patient group. Without intervention in early life, survival to adulthood is extremely rare and death in childhood is typical. 2 Now, the Fontan procedure 3 and its modifications are the standard surgical palliation to treat children with single ventricular defects, although it was originally proposed just to repair tricuspid atresia.
The principle of the Fontan procedure is to bypass the subpulmonary ventricle by routing the systemic venous return directly to the pulmonary circulation, so that the single ventricle is committed to the systemic circulation and the load on the ventricle can be reduced. This neoportal system is often called Fontan circulation. It introduces significant alteration of the normal cardiovascular haemodynamics, due to the reversal of pressure gradient between caval veins and arteries compared to normal biventricular circulations and this causes long term efficiency declination and ultimately leads to fatal systemic complications, such as hepatic failure.
Systemic complications are largely due to upstream venous congestion and downstream decreased cardiac output. In the absence of a pumping chamber to the pulmonary circulation, the single ventricle, which drives the entire circulation has to pull blood through the lungs using a degree of suction which is not physiological. 4 As a result, the systemic venous pressure is markedly elevated, as is the hepatic venous pressure. 5
Therefore the key goal is to reduce systemic venous congestion whilst at the same time lowing pressure in the pulmonary circulation to maintain the pressure gradient. An effective way is to provide subpulmonary assistance, which will permit systemic pressure to be lower than pulmonary pressure and provide sufficient power to overcome pulmonary vascular resistance, the critical contributor to Fontan failure. Nevertheless, all commercial ventricular assist devices (VADs) presently used are primarily designed for systemic arterial support. They are not specifically designed for the widely variable cardiac morphology of Fontan patients. The worldwide experience in supporting the pulmonary circulation in failing Fontan patients is very limited to few successful short-term cases. 6 This motivates us to develop a novel cavopulmonary assist device to provide the 10 mmHg pressure head delivery 7 to overcome the bottleneck in the Fontan circulation.
Integrating the principles of impedance and peristaltic pumping, the proposed device will demonstrate in vitro pulsatile (vs continuous flow in most commercial VADs) and unidirectional flow without the need for a one-way valve. It is impeller-free with no blood contact. Most importantly, the device could be synchronised with the single ventricle via the detection of intracardiac electrocardiograms, hence potentially supporting exercise.
Impedance pumping
This pumping via applied periodic forcing produces controllable and unidirectional flow in pipes without the need of one-way valves. 8 This phenomenon, first discovered by Liebau in 1955, 9 utilises periodic compression of a flexible tube at an asymmetrical position to generate unidirectional and pulsatile flow. The flexible tube is connected to a more rigid tube of differing impedance at either end, and the generated flow rate is non-linearly related to the pinching frequency.
The working principle is summarised in Figure 1(a) and (b) which show pressure wave propagation at the start of a single pumping cycle. A single pinching-release motion takes place at an off-central location at time
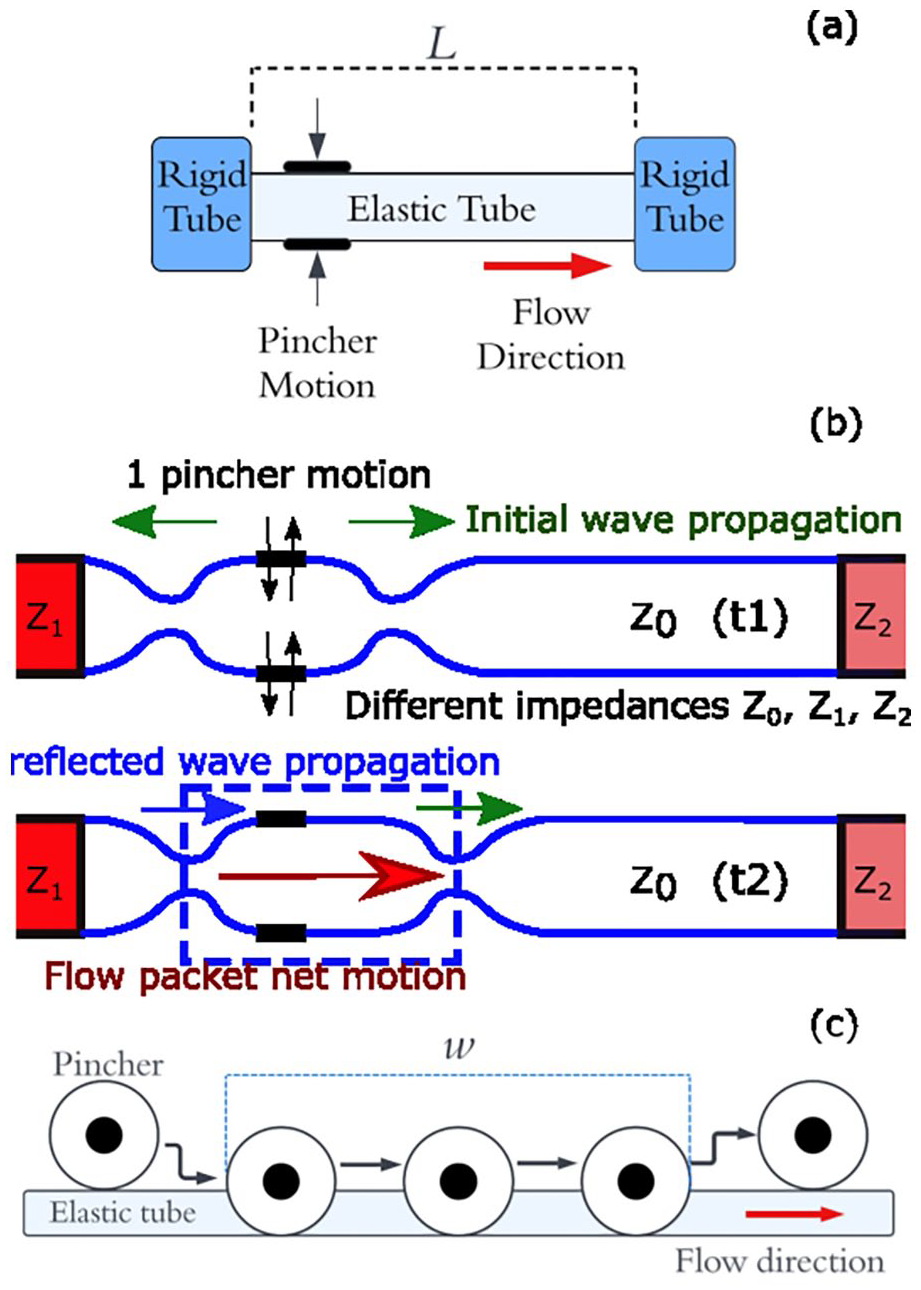
(a) Impedance pumping arrangement. (b) Wave propagation in the pliable elastic tube. (c) Peristaltic pumping mechanism.
Analysis of the closed loop system was first carried out by Thomann. 12 They demonstrated valveless pumping through a torus made of two elastic tubes of differing elasticity via pinching of the more pliable tube, and modelling of the system using one-dimensional Navier-Stokes equation. This approach was furthered by Moser, 13 demonstrating impedance-defined flow with a closed loop and elastic tube. They suggested that impedance and wave effects are the dominating factors in net pumping, rather than fluid inertia. Ottensen 14 devised a one-dimensional single loop model and compared model performance to qualitative experimental results. Their findings showed a dependence of the net flow rate (NFR) on pinching frequency, location, duty cycle and tube elasticity. The modelling attempt by Borzì and Propst 15 demonstrated the requirements for a net pressure head from open reservoir pumping. The open loop configuration reduces the effect fluid inertia can have on the pumping mechanism, and suggested that wave mechanics within the flexible tube were likely to be a governing factor in impedance pumping. Hickerson et al. 16 investigated pincher position, actuation frequency, materials and systemic resistance using both open and closed loop setups. It was determined that locating the pincher at the end of the flexible tube led to greater maximum NFR, and that NFR went through apparent resonant peaks as frequency increased, which changed with pinching locations.
Interest in biomedical applications of impedance pumping has been growing. Pahlevan and Gharib 17 found that the embryonic human heart functions as a valveless pump due to impedance effects, rather than peristalsis as previously thought. Zislin and Rosenfeld 18 investigated impedance pumping performance in a multi vessel system and concluded that impedance pumping may be a suitable method for flow control of the cardiovascular system. Davtyan and Sarvazyan 19 researched the performance of impedance pumping with biologically relevant vessel properties and actuation frequencies and found that impedance pumping could produce similar NFR to similarly sized peristaltic pumps, a remarkable feat due to the lower energy and smaller tube length compressed by the impedance pump. Building on this, Sarvazyan 20 suggested tissue-based pinching methods with muscle rings, and body sites suitable for biologically implementable Liebau pumps. Recently, Anatol et al. 21 investigated purely asymmetric valveless pumping with biologically relevant vessel sizes and actuation frequencies. The study concluded that the experimental setup could provide suitable NFR and pressures for paediatric extra-vascular support with a mechanically simple device using only asymmetric pumping and neglecting the need for impedance effect. However, the large size apparatus means the system can be operated at its resonant frequency, whilst on a smaller size the resonant frequency would most likely be too high to suit biologically useful frequencies.
Peristaltic pumping
Peristaltic pumping is a form of positive displacement pumping achieved by displacing fluid with a roller inside a compliant tube, forcing fluid forwards and creating net suction behind the roller, resulting in a net positive pumping effect, 20 Figure 1(c). The rollers compress the tube in a similar manner to the impedance pump, but flow direction is controlled by the direction of the rollers motion and the valve behaviour of the rollers, forcing forward flow and restricting backflow. 22 NFR is therefore directly proportional to forcing frequency, the compressed tube length and tube diameter. Flow reversal is only achievable by reversing the direction of the roller compression and, unlike the impedance pump, resonance effects do not affect flow rate. The simplicity and valveless feature make it a useful pump for low flow rates, alongside low fluid shear rates, associated with blood cell damage, due to lack of direct fluid contact. The peristaltic pump has many applications and is particularly useful for accurate dose delivery due to the precision in volume displacement. 23 However it must be large in size to support sufficient flow rate and therefore are not well suited to be used alone as a VAD.
Experimental method
Pinching mechanism
A prototype of the device is shown in Figure 2. A roller type pincher controlled by a variable speed DC motor was attached to a rigid aluminium frame. The pincher fully occludes the tube at the bottom of its rotation whilst not contacting it at the top. The period of pinching, determined by the arm length
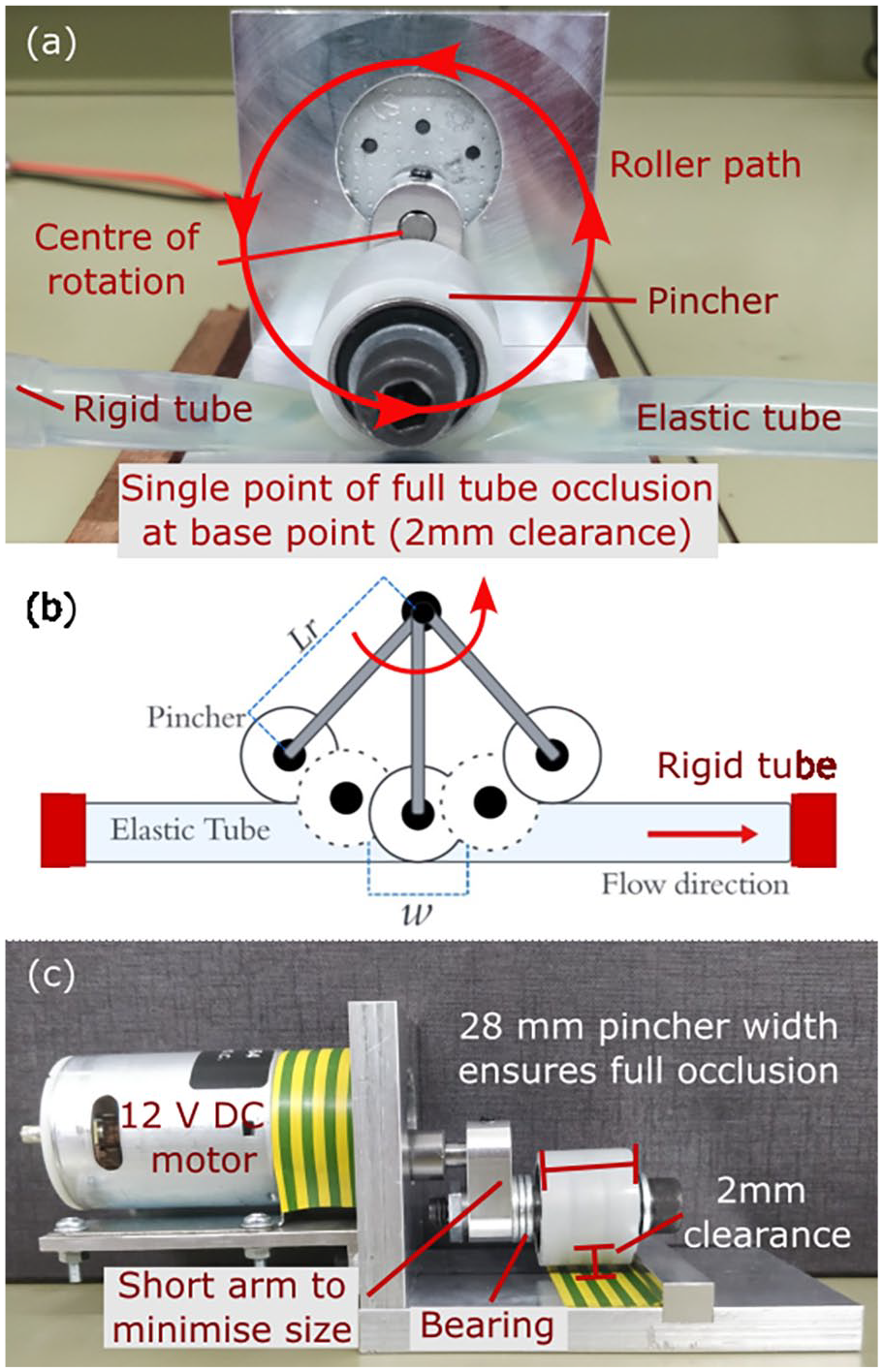
Rotary pumping mechanism: (a) front view, (b) schematics of rotary tube occlusion, and (c) side view. Pincher diameter
Figure 2(b) illustrates the action of the rotary pump, which is an unexplored pinching mechanism for impedance pumping to the best of our knowledge. The extent contributed by the peristaltic effect from the roller ‘pushing’ the fluid alongside impedance effects is investigated. Peristaltic NFR,
where
Experimental testbed
The experimental testbed is displayed in Figure 3. Two plastic water reservoirs, each with base size
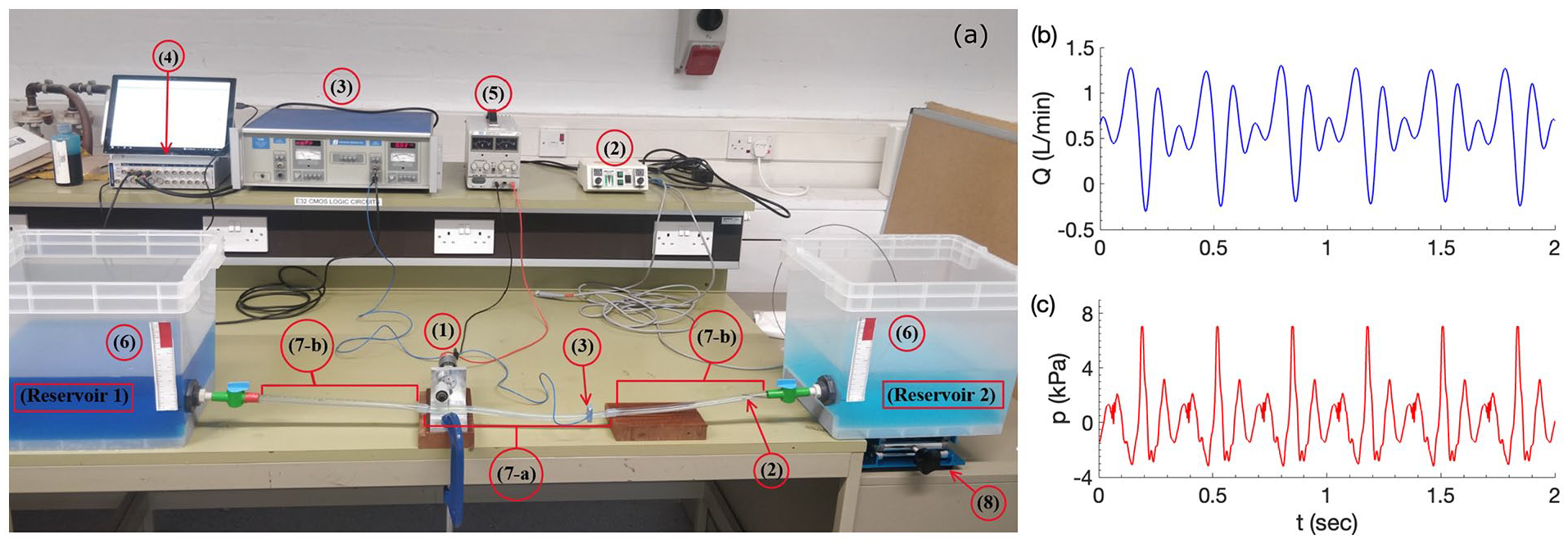
Experimental testbed (a). Typical instantaneous waveforms at pumping frequency
Experimental tube parameters.

The off-the-shelf tubing size used is selected purposely to be within physiological ranges, with the internal diameter of the pulmonary vein, for example, commonly within the range of 9–13 mm. 24 The tubing is slightly smaller than the major cardiac arteries in the adult, 25 but close to that of the paediatric population with mean proximal aorta diameter only about 10 mm for children between 3 and 8 years old. 26
Data collection method
Instantaneous flow rates were measured non-invasively using a Transonic Systems T206 Dual Channel Small Animal Blood Flow Meter with Transonic Flow Probes (3). Pressure was measured by a Millar PCU-2000 Dual Channel Pressure Control Unit with a Mikro-tip catheter pressure transducer (2). A lab stand (8) was used to elevate the reservoir to create background pressure gradients.
Flow rate and pressure waveform data were collected using an ADInstruments PowerLab signal conditioner (4), sampled at 1 kHz, sufficiently high to avoid signal aliasing. NFR was calculated by averaging instantaneous flow for a minimum of 8 s after steady state condition is reached that is, a minimum transient period of of 2 s is allowed before measurement is taken. Figure 3(b) shows typical instantaneous flow and pressure waveforms, where momentary flow reversal and pressure wave reflection can be clearly observed.
Results
Effect of pumping frequency
Experimental setup to investigate the effect of pumping frequency on NFR is illustrated in Figure 4(a). Note that the rotary pincher is located at an off-central location. In order to examine the effect of tube size, two sets of elastic-rigid tube pairs, as described in Table 1, are tested. Owing to the push fit connection method, the size of the rigid tube needs to vary with the elastic tube size. The results are shown in Figure 4(b), which suggests an approximately linear NFR increment with
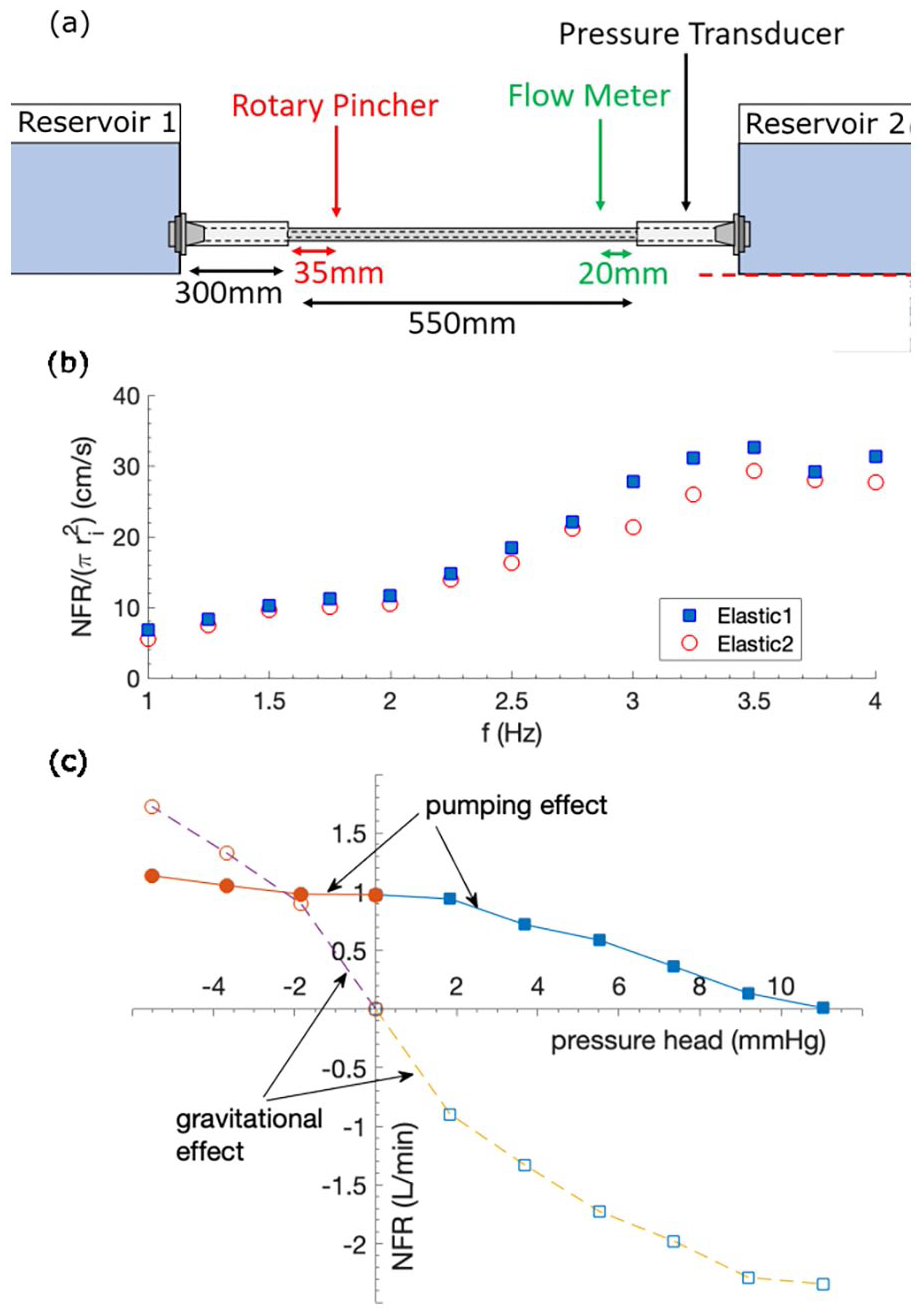
(a) Experimental setup to test the effect of pumping frequency; not to scale. (b) Effect of
It is also evident from Figure 4(b) that NFR is proportional to the elastic tube cross-sectional area as the data points reflect good degree of collapse over the tested
Effect of pressure head
The ability for the pump to deliver pressure head, that is, produce flow against an adverse (positive) pressure, is essential for a VAD. That is, pressure head delivery of 110 mmHg for the full systemic support and 10 mmHg for cavopulmonary support; see
The results are shown in Figure 4(c). Also included for comparison is the gravitational driven NFR, when the pump does not operate.
Effect of elastic tube length
To explore the feasibility for miniaturisation of the pump system, the effect of length
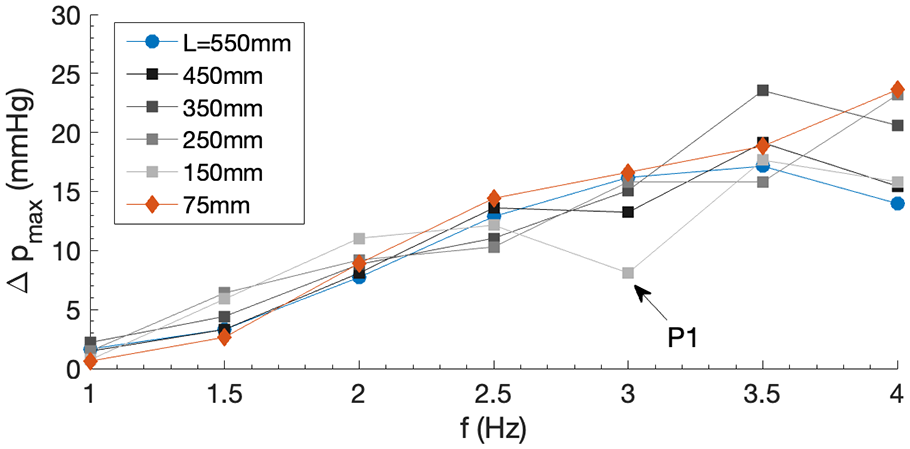
Dependence of maximum pressure head delivery
Effect of impedance versus peristalsis
As an attempt to separate the effects of impedance and peristalsis on NFR, the pumping position was varied off-centrally, assuming peristaltic pumping does not strongly depend on the pumping position. The experimental design is illustrated in Figure 6(a). At the centre of the elastic tube,
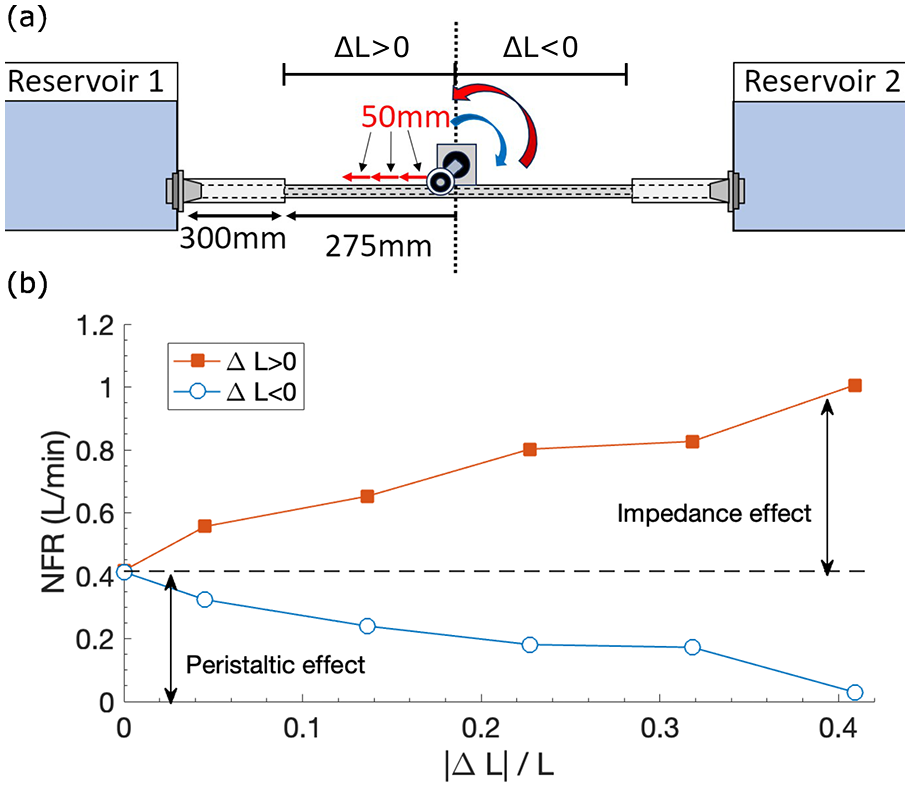
(a) Experimental setup to separate the impedance and peristaltic effect; not to scale. Pincher frequency
Discussion
In healthy adults, heart beats at
The linear behaviour of NFR against pumping frequency shown in Figure 4(a) exhibits some similarity to impedance pumping at low frequency,10,11 but with improved linearity attributed to the peristaltic pumping effect, equation (1). This improved linear behaviour therefore is arguably better suited for supporting cardiac output compared to pure impedance pumping because of this improved predictability for motor control.
The frequency range investigated,

where
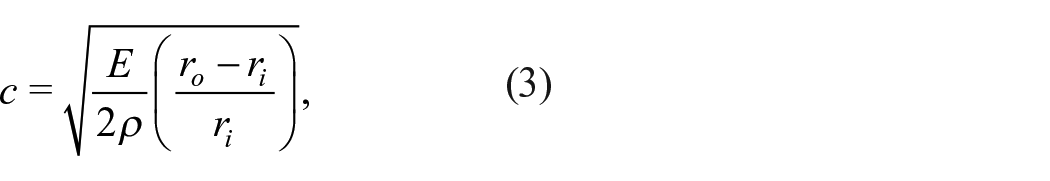
where

with
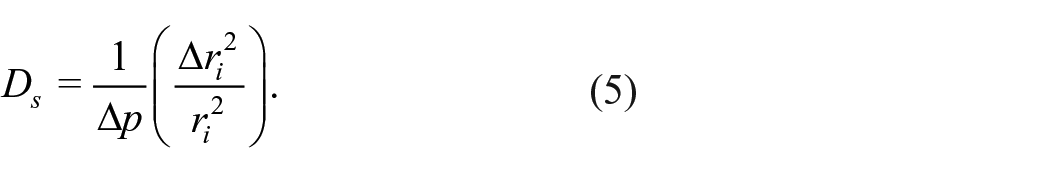
It represents the fractional change of cross-sectional area upon change of fluid pressure. This equation is a slightly simplified model, and may be of use when
For the 2 elastic tubes used in this study,
As expected, Figure 4(c) demonstrates that NFR drops in a reasonably linear manner as the adverse pressure head increases (positive range), but it delivers significant flow against the increasing gravitational effect. For favourable pressure head (negative range), it is interesting to see that the pump is able to regulate NFR in the system, as NFR changing upon pressure head gets flatter compared to that of the pure gravitational effect. This shows that the pump has an active control of system NFR.
The effect of
Results shown in Figure 6(b) illustrate that at
Limitation
In this feasibility study with in vitro tests, majority of the components used to build the device are off-the-shelf and of low cost. No special effort has been made to optimise any of its specifications, for example, elastic material property or design of motor pumping programmes. Future works include miniaturisation of the whole device and identification of optimal operational conditions, from which greater potential may be expected.
Conclusion
This study demonstrates a ‘first version’ prototype of a pump, which integrates positive characteristics of impedance and peristaltic effect, and compromises weaknesses of the two. It is non-invasive (no contact with blood), pulsatile, valveless and potentially can be made of small size, features that are not commonly seen in commercial VADs available in the market. Net flow rate and maximum pressure delivery are both reasonably linearly dependent on pumping frequency within normal physiological range. Positive linearity is also observed between net flow rate and the extent of off-central pumping position. It regulates flow rate in favourable pressure head against gravitational effect and linearly overcomes adverse pressure head. Additionally, the length of the elastic tube is shown to have insensitive impact on the pump performance. These features are potentially advantageous for control strategy design and miniaturisation of a cavopulmonary supporting device, which are left for the future study.
Footnotes
Acknowledgements
The authors would like to thank Z. Woodward for their initial contribution to this work, especially to the experimental testbed and instrumentation setup.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

