Abstract
The common marmoset (Callithrix jacchus, C. j.) is an established primate model in biomedical research and for human-related diseases. Monitoring of cardiovascular parameters including blood pressure (BP) is important for the health surveillance of these experimental animals and the quantification of diseases or pharmaceutical substances influencing BP. Measurement guidelines for C. j. do not exist yet; therefore, the present study was carried out to establish a practicable protocol based on recommendations of the American College of Veterinary Internal Medicine (ACVIM). Furthermore, BP data of 49 marmosets (13.8–202.4 months of age) were obtained via high-definition oscillometry to further knowledge of physiological parameters and gender-related differences in this primate. The thighs proved to be the most suitable measurement localization, since systolic values were less variable (left 4.03 ± 2.90%, right 5.96 ± 2.77%) compared with the tail (12.7 ± 6.96%). BP values were similar in the morning and in the afternoon (P > 0.05). Data were highly reproducible within and between several sessions on three consecutive days (P > 0.05) as well as over the course of 20 months (P > 0.05). Furthermore, the measurement time for females was significantly shorter than for males (5:14 ± 1:59 min versus 6:50 ± 1:58 min, P = 0.007). Measurement recommendations for the common marmoset were successfully established. Standardized values enabled a reliable comparison of BP parameters, e.g. for cardiovascular, toxicological or metabolic research.
Blood pressure (BP) represents a relevant marker of the cardiovascular system, especially with regard to heart function and blood vessel condition. It can be influenced by metabolic disorders (e.g. diabetes mellitus, 1 adiposity 2 ) or age-related diseases (e.g. renal failure, 3 cardiac insufficiency4–7). Studies in those fields frequently use animal models. Regarding the medical assessment of the used species, BP is an essential parameter for animal health.
To ensure the acquisition of reliable and comparable values, recording procedures have to be standardized. For pets and rodents appropriate measurement protocols have been developed recently.4,8–10 However, no recommendations exist for small experimental primates.
With regard to biomedical research and human-related diseases the common marmoset is a widely used primate model.11–13 However not all procedures for BP assessment (e.g. restraining techniques or acclimatization periods) seem to be transferable from small animals to these New World Primates due to differences in handling techniques.14,15 Therefore, guidelines for BP measurement in Callithrix jacchus (C. j.) are needed.
The existing gold standard for the assessment of BP is direct intra-arterial BP recording via radiotelemetry, whereby a catheter is implanted in the femoral artery or the abdominal aorta. Continuous and undisturbed measurement of BP is possible using this technique. Nevertheless, it is an invasive method with disadvantages such as technical and financial requirements, potential inflammatory reactions, potential loosening of the detector, as well as limited dwelling time in the vessels for the catheter. 16 A promising alternative is non-invasive measurement of blood pressure (NIBP). It allows fast data acquisition, there is no need for surgical procedures, and it can be used for long-term experiments. Frequently employed NIBP methods are ultrasonic Doppler technology and oscillometry.4,17–19 Doppler technology detects blood flow changes induced by a cuff compression to estimate BP. Oscillometry, on the other hand, is based on the registration of arterial wall oscillations, which are caused by external occlusion. A promising development is high-definition oscillometry (HDO).6,20–22 In contrast to classic oscillometry, this technique allows real-time acquisition of BP values as well as the analysis of pressure waveforms. Therefore, fast measurements and the assessment of cardiac output and arterial elasticity are possible.9,23 Previous studies using HDO in C. j. were performed either on a small number of animals (n = 18) 24 or in very young animals (age ≤ 24 months, n = 30). 25 Furthermore, both studies did not state measurement protocols consistent with the standards of the American College of Veterinary Internal Medicine (ACVIM) by Brown et al. 4 Moreover, studies conducted on other species have indicated that BP is influenced by gender, cuff position and time of day.26,27 None of these parameters have been explored so far for the common marmoset.
Therefore, one aim of the present study was to establish a standardized measurement protocol for NIBP in the common marmoset on the basis of the ACVIM guidelines for small animals. The second aim was to analyse BP data from 49 animals with a broad age range with respect to gender-related variances, in order to increase knowledge of physiological BP values in this species.
Animals, materials and methods
Animals
In this study 49 common marmosets (23 males, 26 females) were used to acquire BP values over a period of 20 months from May 2012 to December 2013. The majority of the animals were born in the German Primate Centre or the primate house of the University of Leipzig, Germany. Four animals came from private owners and lived in the colony for two years or more prior to this study. All the animals had a median age of 55.33 months (25th percentile [P25] = 45.6 months, 75th percentile [P75] = 98.0 months). Due to the size of the available measuring cuffs and the animals’ leg circumference, the recording of BP values started when the animals were at least one year old. The mean body weight of all animals was 404.9 ± 40.3 g, with no significant gender-related differences.
All animals had a conventional microbiological status and were housed in an open system in pairs or family groups. Housing equipment consisted of steel mesh cages (180 × 80 × 70 cm, University of Berlin, Berlin, Germany) for two animals and a cage of double the size for a maximum of four animals with a steel nest box (17 × 26 × 17 cm), branches, sitting boards, and a cloth hammock. In addition to natural lighting in terms of glass windows in every animal room, an artificial light period was provided between 07:00 and 19:00 h. Room temperature was regulated at 25℃ (24–26℃) with a relative humidity of 40–60% and 10 air changes per hour. Additional red light was given daily from 07:15 to 07:45 h. Changing environmental and food enrichment (e.g. varying presentation of vegetables and fruits, spreading of gum arabic or offering of mealworms, grasshoppers as well as crafted straw containers or plastic balls) was provided weekly to the animals. Feeding took place twice a day. In the morning each animal pair was given a mixture of 20 g of seasonal vegetables and fruits as well as a protein source (e.g. meat, egg). At midday they received commercially available gluten-free pellets (ssniff Spezialdiäten GmbH, Soest, Germany) and gluten-free mash (ssniff Spezialdiäten GmbH). 28 Water was available ad libitum via water bottles. The animals were used to regular handling. After the completion of data acquisition the animals remained in the colony of the University of Leipzig, Germany. During the study, five animals died of causes unrelated to the pathology of the cardiovascular system (e.g. diarrhoea).
Animal husbandry was approved by the regional board of Leipzig, Germany (reference number: 24-9168.11/17/68) and research approval was granted by the Regional Council Leipzig, Germany (TVV 68/12). The non-invasive BP measurements conducted did not require authorization under the German Animal Welfare Act. The study conformed to the German Animal Welfare Act for the care and use of laboratory animals.
Experimental design
This study comprises a preliminary (I) and a main (II) study with the following scopes:
Evaluation of different cuff positions and times of day for recordings. Establishment of a standardized measurement protocol and evaluation of gender-related differences in BP.
In the preliminary study, the effects of three different cuff placements were analysed in 10 adult common marmosets. The following positions were used: the right and left thighs as well as the tail (approximately 1–2 cm distal to the base of the tail; see Figure 1). To analyse the influence of time of day on BP, six adult animals were selected. Over a period of three consecutive days, the BP of each animal was measured in the morning (08:00 to 11:00 h) and in the afternoon (13:00 to 16:00 h).
Positions of the cuff for blood pressure (BP) measurements in the common marmoset. (a) Left thigh. (b) Tail position.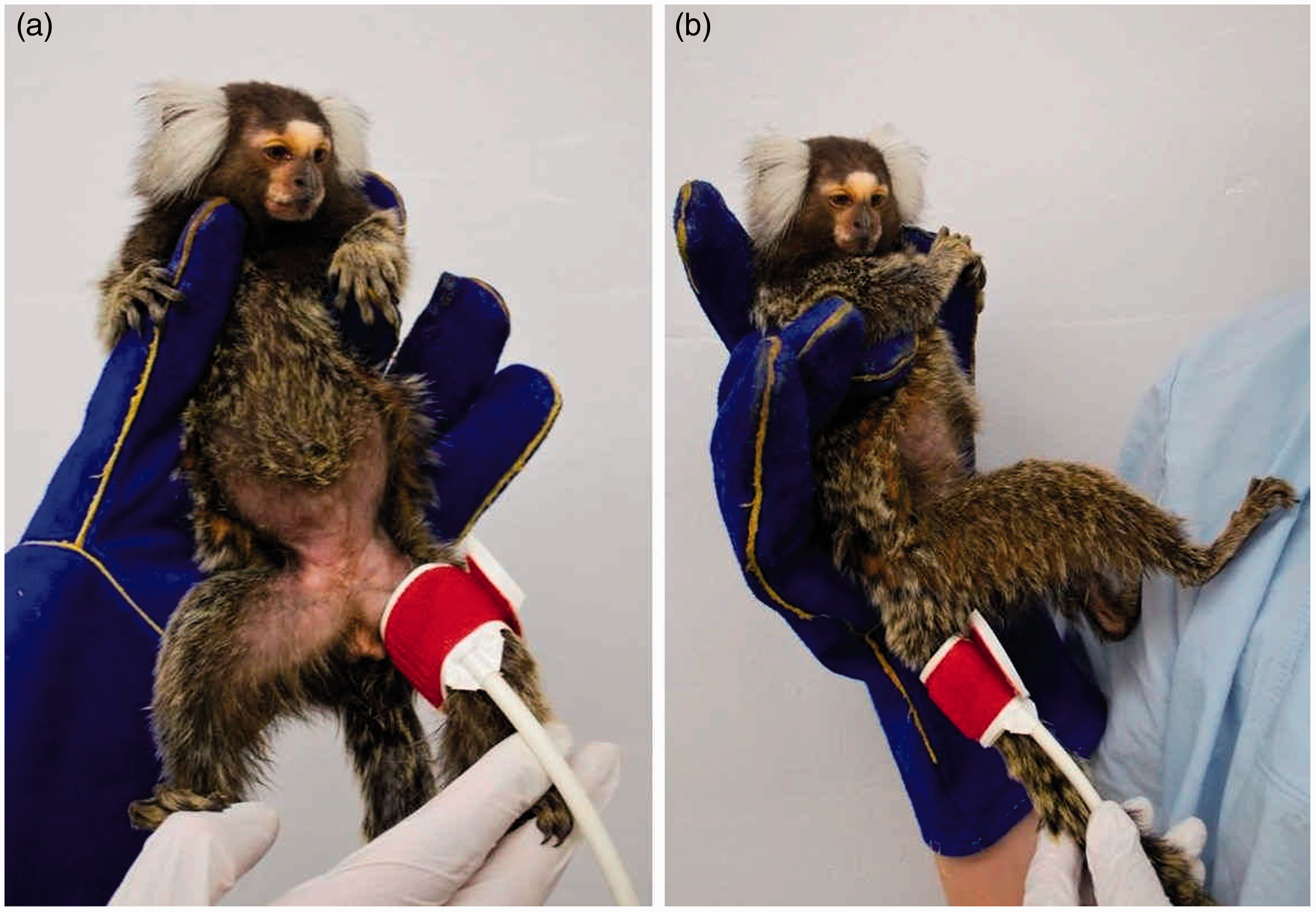
For the main experiment 49 animals were used. Measurements took place throughout the day and were conducted on the left or right thigh.
For both studies the following setting was applied. Animals were accustomed to handling with minimal stress as described elsewhere. 28 For the measurement procedure the animals were placed in a comfortable slightly backward leaning position to ensure that the distance between the heart base and the cuff was maximally 10 cm (see Figure 1). Pulsing of the femoral artery was manually detected and the sensor of the cuff was positioned as described by Egner et al. 9 Data collection started after an acclimatization period of 1–3 min.
Blood pressure measurement
BP data acquisition was carried out with an HDO measurement device (VET HDO Monitor MD PRO Marmoset; S + B medVET GmbH, Babenhausen, Germany) and recorded with the corresponding software (S + B medVET Memodiagnostics MDS Analyse Software Version 2.0.3.0). The analysis software created a computer file with pressure waveforms for every single measurement. The systolic, diastolic and mean arterial BPs (SAP, DAP and MAP) in mmHg as well as the pulse rate (Pulse) in beats per minute (b/min) were recorded.
Air-deflating cuffs with sizes C2 (length 15.2 cm, width 4.4 cm) and V2 (length 12.5 cm, width 3.6 cm) were used. To acquire data about the fitting of the used cuffs, repetitive measurements were conducted at the middle of the thighs and the bases of the tails. The ratios of cuff widths to thigh circumferences were mean 54.5 ± 5.29% (C2) to 45.7 ± 4.43% (V2) and at the tail 102 ± 11.1% for C2 to 85.4 ± 9.28% for V2. The cuff size used (C2 or V2) was chosen depending on the anatomical situation of the legs.
Data were evaluated based on ACVIM statements. The first reading of each session was discarded. At least three readings (comparison of cuff positions) or five readings (other settings, coefficient of variation below 20% in SAP) were conducted as suggested by ACVIM. For small animals these guidelines recommend three to seven measurements per session to obtain representative BP values. Consequently we reviewed the transferability of this statement to the C. j. Overall, 115 sessions were conducted using a total of 36 animals. Each session included seven readings which were analysed and compared by averaging the first three, five or all seven readings.
Criteria for artefact assessment.

In the cases of low-grade artefacts, corresponding values were included in measurements, whereas artefacts classified as high were discarded and repeated. MO: movements; VE: vocal expressions, MT: muscle tremor; NI: nibbling; MF: measurement fault; EX: excitement; BP: blood pressure.
Statistics
Statistical analysis was performed with IBM SPSS Statistics Version 22 (IBM, Chicago, IL, USA). All data were tested for normal distribution by means of the Kolmogorov–Smirnov test. In the case of normally distributed data (presented as mean ± standard deviation [SD]), t-tests for independent samples were conducted to examine the values for significant differences between the groups. Comparisons of localizations or repetitive measurements within or between sessions were analysed using a repeated measurements analysis of variance (ANOVA) with Bonferroni adjustment. In the case of not normally distributed data (analysis of the artefacts) Wilcoxon tests were conducted. Due to the limited sample size, data of the preliminary experiment (time of day comparison) were analysed as not normally distributed. Their values are given as median, P25 and P75. The correlations of BP values and artefact occurrence were explored and determined via linear regression analysis. Comparison of artefact values with regular BP data was done by averaging all respective artefact data during one measurement session and relating it to the mean of all the remaining values. The level of significance was defined at P < 0.05, with P < 0.01 being highly significant.
Results
(I) Preliminary experiment
Comparison of localization during the preliminary experiment.
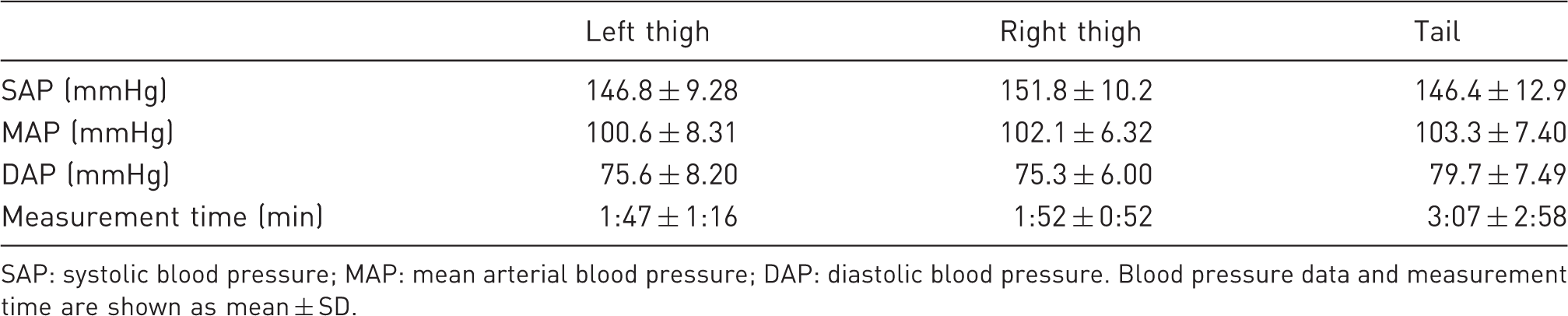
SAP: systolic blood pressure; MAP: mean arterial blood pressure; DAP: diastolic blood pressure. Blood pressure data and measurement time are shown as mean ± SD.
The variances of systolic values (cvSAP) at the left and right thighs were significantly lower than at the tails (4.03 ± 2.90%, 5.96 ± 2.77%, 12.7 ± 6.96%, respectively; P ≤ 0.01). cvSAP results ranged between 2.43% and 22.3% for the tail whereas the maximum values for the left and right thighs were markedly lower (9.49% and 10.5%, respectively).
Values for SAP, MAP, DAP and Pulse on three consecutive days for comparison of measurements in the morning (a.m.) and afternoon (p.m.).
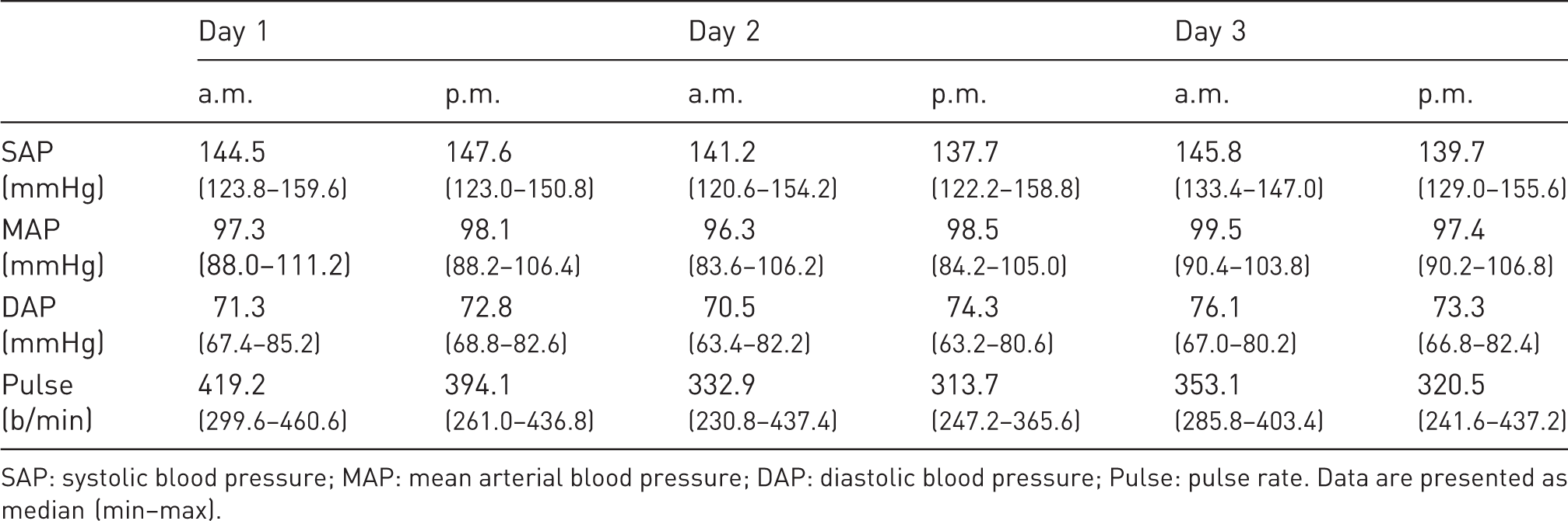
SAP: systolic blood pressure; MAP: mean arterial blood pressure; DAP: diastolic blood pressure; Pulse: pulse rate. Data are presented as median (min–max).
In general, the preliminary experiments demonstrated that thighs should be used as the measurement localization. Furthermore, readings could be conducted either in the morning or in the afternoon. These principles were applied in the main experiment following.
(II) Main experiment
In the main experiment, the BP of 49 C. j. was measured. The mean BP of all animals amounted up to 145.7 ± 9.0 mmHg (SAP), 98.3 ± 6.46 mmHg (MAP) and 72.6 ± 6.44 mmHg (DAP). The pulse rate averaged 406.5 ± 32 b/min. All BP parameters were highly correlated with each other (SAP–MAP: r = 0.834; SAP–DAP: r = 0.56; MAP–DAP: r = 0.924). BP and Pulse did not differ between males and females (SAP: 144.8 ± 8.40 mmHg, 146.5 ± 9.56 mmHg, P = 0.53; MAP: 96.8 ± 5.01 mmHg, 99.6 ± 7.34 mmHg, P = 0.12; DAP: 70.7 ± 4.93 mmHg, 74.2 ± 7.25 mmHg, P = 0.06; Pulse: 403.7 ± 31.2 b/min, 409.0 ± 33.1 b/min, P = 0.56).
The mean measurement time was 5:59 ± 2:07 min. Analysis of the data for gender-related differences showed that measurement took significantly longer in males (6:50 ± 1:58 min) than females (5:14 ± 1:59 min); P = 0.007. To exclude possible seasonal effects on BP, a median of 6 (P25: 4, P75: 9.5) sessions per animal were conducted, evenly distributed throughout the year. No statistically significant differences between these sessions or seasons were observed (repeated measurements ANOVA, α > 0.05).
Artefact values and differences to the normal values from the same session (behind the slash) .
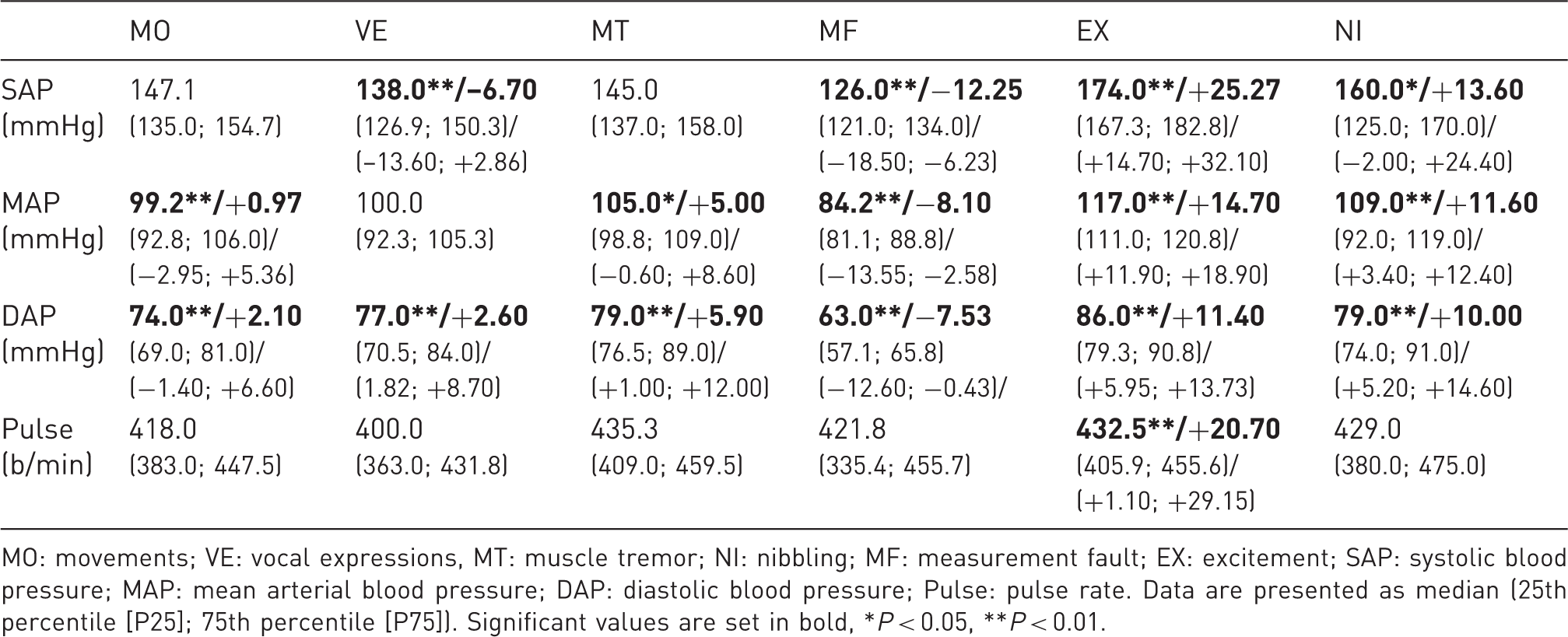
MO: movements; VE: vocal expressions, MT: muscle tremor; NI: nibbling; MF: measurement fault; EX: excitement; SAP: systolic blood pressure; MAP: mean arterial blood pressure; DAP: diastolic blood pressure; Pulse: pulse rate. Data are presented as median (25th percentile [P25]; 75th percentile [P75]). Significant values are set in bold, *P < 0.05, **P < 0.01.
The values of the second readings for SAP and MAP were significantly different from the other readings (P < 0.05). They varied by 2.24 ± 3.43 mmHg and 1.12 ± 1.69 mmHg from the mean values of each session (P < 0.01). DAP and Pulse showed no significant changes throughout a session.
Comparison of averaged blood pressure data of three, five or seven consecutive readings.
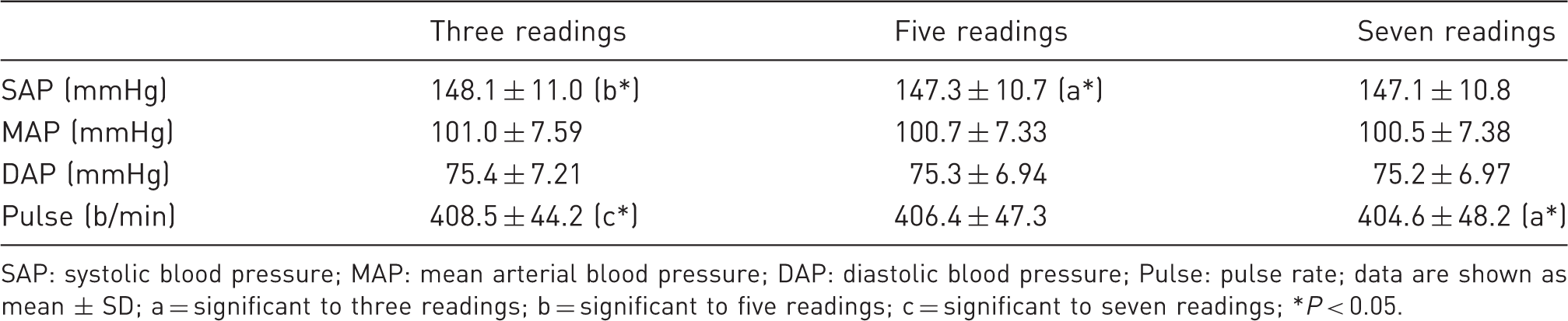
SAP: systolic blood pressure; MAP: mean arterial blood pressure; DAP: diastolic blood pressure; Pulse: pulse rate; data are shown as mean ± SD; a = significant to three readings; b = significant to five readings; c = significant to seven readings; *P < 0.05.
Although BP values did not show gender-related variances, it was generally quicker to obtain readings in female C. j. The second measurements differed significantly from the mean values in each session; nevertheless the amount was only by about 2 mmHg or less. Moreover, the averaged values of each session differed just slightly with an increase in the number of readings, and artefacts influenced BP values in varying amounts.
Discussion
As the importance of common marmosets as a primate model increases, reliable and standardized BP values become important. For dogs and cats several guidelines exist.4,8,9 Nevertheless, it is questionable whether recommended standardized procedures like the ACVIM guidelines are easily transferable to the common marmoset. Therefore, one aim of this study was to develop a measurement protocol based on the recommendations for dogs and cats (Table 4). Furthermore, published NIBP data for C. j. are limited to few or very young animals. But due to the increasing lifespan of marmosets under human care, further knowledge of physiological values in this laboratory species is necessary. Another aim of the present study was therefore to obtain BP data from a larger number of animals of both sexes and with a broader age range. These recommendations and data are described below.
Different localizations for BP measurements are established for dogs and cats (thighs and tail)4,26,29 as well as for rodents (tail). 10 In the present study these localizations were tested in the common marmoset. Both the thighs and the tail showed comparable data with regard to BP values and times of acquisition. However, when using the commercially available cuffs, tail measurements proved to be less accurate (cvSAP up to > 20%); therefore we do not recommend this localization for BP acquisition in common marmosets.
Suitable cuff sizes should have a cuff width/leg circumference ratio of 30–40% as recommended by ACVIM for BP measurements in dogs and cats. Nevertheless, with a ratio of 45.7% (V2) and 54.5% (C2) the available cuff sizes exceeded these reference values considerably in the common marmoset. Younger (<1 year) and older (accelerating after 8 years) C. j. have substantially less body musculature compared with adult animals,13,30 therefore the available cuffs are not applicable in these cases.
With regard to the time of day (morning versus afternoon), BP values and acquisition times proved to be similar in this study. These findings are in line with NIBP readings in cynomolgus monkeys, where no daytime dependency of BP was observed. 27 In contrast to these findings, BP values obtained with telemetry show curve progressions which mirror the daytime dependent multimodal course of activity that is exhibited by marmosets in the natural habitat as well as in human care.31,32 The divergent results are most likely caused by the differences in measurement techniques. Whereas telemetry allows an undisturbed daily routine with periods of activity and recovery, the regular circadian cycle of the animals has to be interrupted for NIBP measurements. It is probable that due to the essential handling of the animals, parameters like stress, excitement or habituation mask the naturally occurring variations in BP values.
According to the ACVIM guidelines for dogs and cats, the first reading of each session was excluded from BP analysis. 4 As previously reported by Bodey et al., the mentioned reading is significantly higher than the following measurements and does not represent the average BP. The increase in BP is probably triggered by the sound of the measurement device or the pressure on the thigh, and these effects disappear once the animal is familiar with the situation 18 .
The evaluation of the analysed readings showed that the second reading was significantly higher than the mean of a session. Nevertheless, the increase was only 2 mmHg; therefore the impact on the overall BP is negligible and this reading can be included in recordings.
In the main study, various numbers of consecutive readings were compared with each other. The values differed only by about 1 mmHg for SAP and even less for all other BP parameters. Bodey et al. 18 , Schmelting et al. 22 and Vincent et al. 33 observed similar results when measuring cynomolgus monkeys and dogs. Little or no changes in BP were detected throughout a measurement session. These results demonstrate that the acclimatization period of 1–3 min used in this study is adequate.
For the intended clinical application of NIBP in C. j. a short restraint time is preferable as it is more practicable and reduces the stress on the animal from handling. Furthermore, research conducted with other primate species indicates that longer measurement sessions lead to a continuous rise in heart rate as well as restlessness. 34 Our results show that three to five readings per session are sufficient for obtaining reliable BP data from common marmosets. Since cognitive or behavioural studies generally use a testing period of 10–15 min without having negative effects on experimental results, this time frame can be used as a maximum range for BP recordings.
With regard to the analysed artefacts, the respective influences on BP parameters differed considerably. Whereas MO, VE and MT had only minimal effects, the occurrence of EX, NI and MF resulted in higher deviations from the mean values. Interestingly, most artefacts influenced the pulse rate only marginally. Concerning the practical implementation of NIBP measurement, this suggests that with regard to MO, VE and MT readings can be included while taking the animals’ behaviour into account. Data should be discarded and repeated in cases of high-grade artefacts, EX, NI or MF. EX has a particularly strong effect as the resulting stress or excitement has great influence on BP, which therefore emphasizes the importance of a quiet measurement location, experienced staff and sufficient adaptation time before the beginning of NIBP readings.
In the preliminary study, BP was recorded on three consecutive days. BP parameters did not change between sessions. This is in line with previous findings in cynomolgus monkeys,22,27 where BP values did not differ in or between several sessions. As stress is a major parameter for a temporarily increased BP, one reason for the minimal variation of the BP patterns might be that the animals had become habituated to handling as well as clinical routines. Nevertheless, the pulse rate tended to decrease from day 1 to day 3. Although the differences were not significant, these results have to be taken into account when planning experimental set-ups. Whereas single measurement sessions are sufficient for the acquisition of reliable BP data in trained animals, pulse rates should be analysed over a period of consecutive days to generate conclusive results.
Concerning the assessed BP values in this study in general, our results are partly in line with previously published data for NIBP readings in common marmosets. Compared with telemetrically measured values from undisturbed animals, restraining procedures cause markedly higher BP values in animals. 25 Nevertheless, handling is necessary in this non-invasive measuring method. Therefore, the induced rise in BP has to be mentioned when evaluating the obtained values. Regardless of the higher values, NIBP recording has some important advantages compared with invasive data acquisition (e.g. no operations are required, it is suitable for long-term investigations, and has lower acquisition costs). 16 In addition HDO data are reproducible and devices can detect changes in BP similar to telemetry.20,35 Thus HDO is applicable in clinical settings despite the comparatively higher values.
Studies using NIBP assessment found values ranging from 120.0–153.5 mmHg (SAP), 91.6–96 mmHg (MAP), 64.3–71.1 mmHg (DAP) and 312–390 b/min (Pulse).24,25,32,36,37 This wide range of values may be due to different measurement devices but may also be caused by varying BP protocols. This indicates the importance of standardized procedures for the acquisition of comparable values. Whereas the SAP values and pulse rate observed in our study confirm the mentioned publications, our data for MAP and DAP were slightly higher than those previously reported. Besides the above-mentioned differences in devices, these variances might be due to the wider range in age and body weight of the animals used in our study, which could therefore indicate that these parameters influence BP in the common marmoset. Age and body weight should therefore be taken into account when conducting future studies in this laboratory primate.
No seasonal variations in BP values were observed during the course of our study. This is most likely due to the housing of the animals. They were held in a standardized environment with regulated day–night cycles and a constant temperature, so season-dependent effects are unlikely.
In this study, female animals were significantly easier to measure than males because less time was required to achieve analysable values from female animals. This is consistent with females performing better in tasks of food retrieval or in trainings to enter transport boxes.38,39 Our findings support the hypothesis that male animals require more attention and care during NIBP measurements. BP values did not differ with regard to gender. This is in contrast to results in dogs and humans,26,40 where males have higher BP. By contrast, marmosets do not show gender-related body weight differences, 41 which could contribute to BP differences in the species mentioned. Nevertheless, potential gender-specific variations in BP might be masked by the observed influence of restraint on BP.
Recommendations for non-invasive blood pressure (NIBP) measurements in the common marmoset (according to the ACVIM guidelines).

Footnotes
Acknowledgements
The authors would like to thank Dr Anne Weißmann, Vicki Bachner, Christin Meinhardt and Robert Nestler for their contribution to this work. In addition, we would like to thank Dr Beate Egner for her technical support during the study.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
