Abstract
We report sterile peri-graft abscess formation following an ascending aortic and hemiarch replacement for acute type A aortic dissection, possibly caused as a reaction to BioGlue®. The patient was successfully treated by drainage, cleaning, removal of BioGlue® remnants, three sessions of negative-pressure wound dressing, and secondary chest closure.
Introduction
Many different tissue sealants are available on the market today. Indications for using these products include supporting suture lines to achieve better hemostasis, strengthening of the fragile tissues, and inducing adherence between tissues for faster healing. 1 BioGlue® (CryoLife International Inc, Kennesaw, GA) is one of the most widely used surgical adhesive agent. It is composed of purified bovine serum albumin and glutaraldehyde, which, combined, form a covalent bond between their molecules and the tissue proteins at the surgical site, resulting in a mechanical seal. 2 We report a sterile peri-graft abscess formation following an ascending aortic and hemiarch replacement for acute type A aortic dissection, possibly caused as a reaction to BioGlue®.
Clinical summary
A 54-year-old male patient underwent an emergency ascending and hemi-arch aortic replacement for acute type A dissection (Figure 1(a)). The anastomotic suture line was reinforced by application of BioGlue® tissue sealant due to the difficulty in achieving satisfactory hemostasis, which is not routinely used in our local protocol. The initial post-operative course was uneventful. On postoperative day (POD) 7, the patient developed fever and an inflammatory syndrome. Blood culture was negative. A broad-spectrum antibiotic therapy was started due to a suspicion of pneumonia. Chest CT scan was performed on POD 11 because of a persistent fever and an inflammatory syndrome showed a suspicious peri-graft fluid collection (Figure 1(b)). The patient underwent a surgical revision for a suspicion of acute mediastinitis. During the procedure, a whitish milky liquid was drained (Figure 1(c)). The surgical site was drained, mechanically cleaned with water jet (OrthoLav®) to remove all remnants of BioGlue®. The chest was left open with a negative-pressure wound dressing. The patient became rapidly apyretic and the inflammatory markers normalized. The histopathologic analysis of the peri-graft process didn’t seem related to an infectious abscess. Indeed, a fibrin-inflammatory reaction without microorganisms in direct microscopic observation and dedicated staining was found. After a 10 days follow-up with three consecutive vacuum dressing changes, the chest was closed and patient’s recovery was uneventful without fever or any inflammatory response. He was discharged to another hospital at POD seven following final sternal closure. All samples which were analyzed by broad spectrum microbiological cultures remained negative throughout. Three months after surgery, a chest CT control showed regular post-operative images without having any residual collection (Figure 1(d)).
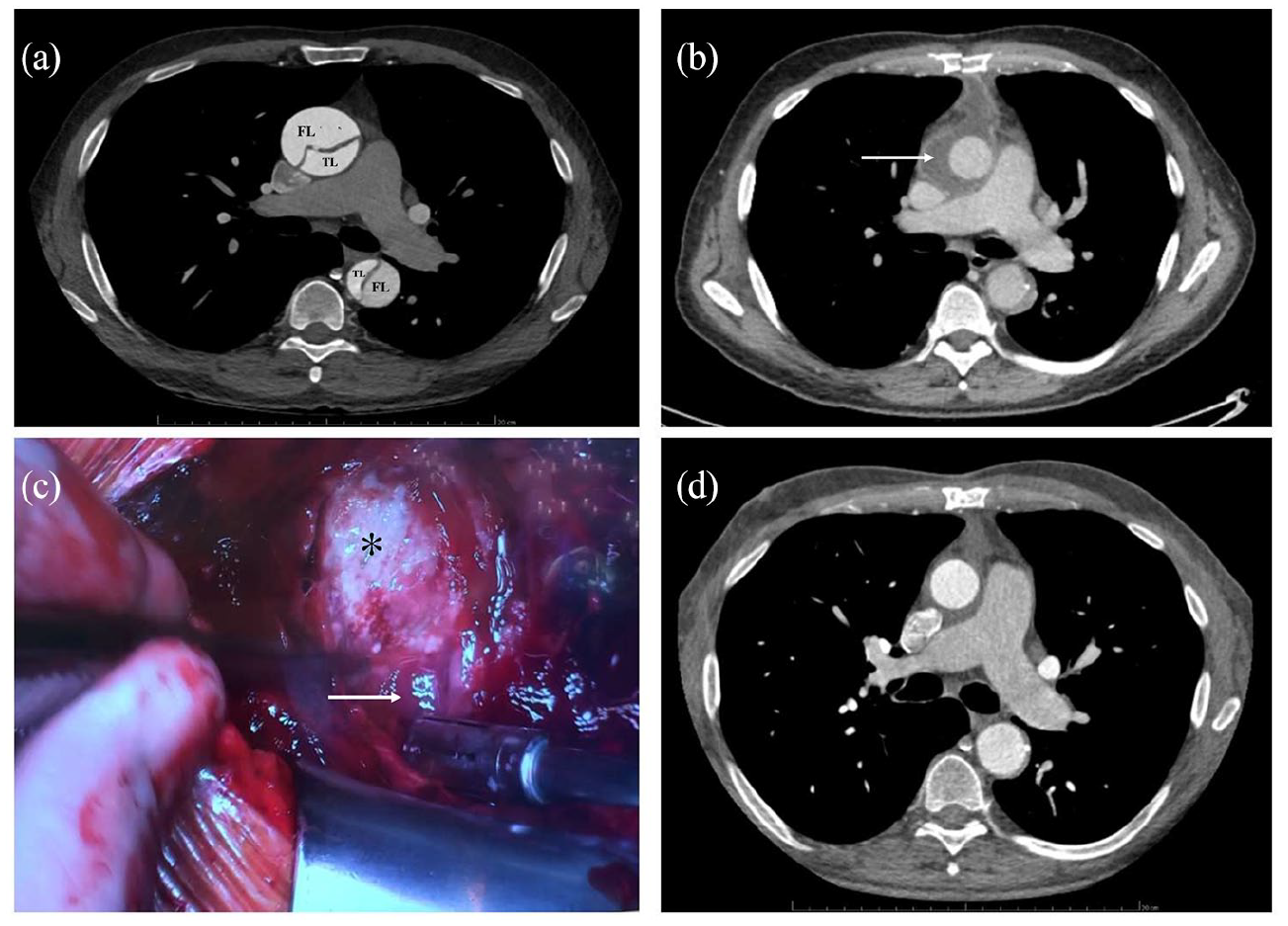
(a) Contrast-enhanced chest computed tomography: acute type A dissection with true lumen (TL) and false lumen (FL), (b) contrast-enhanced chest computed tomography: peri-graft fluid collection (arrow), (c) operative view. Peri-graft (asterix) fluid collection (arrow), and (d) contrast-enhanced chest computed tomography: no residual collection.
Discussion
Several studies reported complications potentially attributed to use of BioGlue® like toxicity to nerves and cardiac conduction tissue, re-dissection, pseudo-aneurysm formation, and adhesive embolization. Valve dysfunction due to deposits of the glue on prosthetic valve leaflets after leakage through aortic suture lines was also reported. Some reports have described tissue damages (necrotic, fibrosed, and excessively thinned aortic tissue) which were observed during reoperations at the site of BioGlue® application. 2 The formation of a sterile abscess following this product is a very rare complication as evidenced by some case reports in the literature.2,3 Although the mechanism of the sterile abscess formation is not clearly associated to BioGlue®, the localized collection of the liquid next to this product remnants reminds us of the need to use this product with caution. The mechanism behind this complication could be secondary to a massive foreign-body reaction with, at histological examination, numerous multinucleated giant cells.4,5 Surgical drainage, mechanical cleaning, removal of the tissue sealant remnants can be applied as an effective treatment on the spot of sterile abscess collection. 3 However, in our experience, a suspicion of mediastinitis because of the presence of an infective-like liquid collection, leaving the wound open with a negative pressure dressing until a sterile abscess is proven seems to be more careful.
Conclusion
Despite the usefulness of all tissue sealants including BioGlue® in cardiovascular surgery, serious complications secondary to them are highlighted in the literature. The formation of a sterile abscess remains a rare complication but have a serious repercussion on a patient with the necessity of surgical revision. In this context, we recommend to limit the use of BioGlue® only to indicated cases with a word of caution.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
