Abstract
Malpositioning of left ventricular assist devices (LVAD) is a risk factor for thrombosis, but its identification from clinical imaging remains challenging. X-rays and CT scans were analyzed and parameters identified that correlated to pump thrombosis. Retrospective imaging data of patients (n = 115) with HeartmateII (HMII) or HVAD were analyzed in two groups (pump-thrombosis PT, n = 15 vs matched control group NT, n = 15) using routine X-rays and CT scans. In CT, directional deviations of the inflow cannula in three-chamber and two-chamber view (α and β angles) were identified. In HVAD PT frontal radiographs showed reduced pump body area and smaller minor axis (PT 41.3 ± 4.8 mm vs NT 34.9 ± 6.0 mm, p = 0.026), and in the lateral radiographs the visibility of the inflow cannula served as a predictive parameter for PT. In HMII patients, no parameters were associated with PT. The angle α differed significantly (NT −1.2 ± 7.5°, PT −22.0 ± 4.7°, p = 0.006) in HVAD patients. Further, correlations of x-ray parameters with CT angles α and β showed that radiographs can be used to identify malpositioned pumps. Well-aligned inflow cannula positions are essential. HVAD patients with a posterior rotation of the inflow cannula have a higher risk of pump thrombosis. This risk can reliably be identified from routine radiographs.
Introduction
In recent years, continuous improvements in mechanical circulatory support have been observed and results are competing with heart transplantation. 1 Nevertheless, adverse events are still limiting factors and impair the long-term success of left ventricular assist devices (LVAD). 2 In particular, thromboembolic complications (pump thrombosis and stroke) are considered one of the most-dreaded complications 3 that might be associated with the pump placement and alignment.
Some studies showed the importance of VAD inflow cannula positioning to reduce specific adverse events. Thromboembolic events were linked to the geometry of the inflow cannula,4,5 the depth of the pump pocket (Abbott HeartMateII (HMII)6–8), pump migration,6,7 the angles of the inflow cannula (pump thrombosis in HMII,8–10 unloading and GI bleeding in Medtronic HeartWare HVAD,11,12 and HMII 13 ) the distance of the cannula tip to the ventricular wall 14 and further to blood stream infections.15–17 Although data exists on positioning of the HMII and pump thrombosis, 7 no data is available on pump thrombosis for the HVAD.
Although magnetically levitated pumps may have a lower risk of pump thrombosis, it is still a relevant pathology with significant morbidity and mortality. 18 Interestingly, the numbers of ischemic strokes that are likely to be caused by thrombi ingested or formed in the pump are in the same range for different pumps (HMII 0.09–0.11, HM3 0.04–0.10, HVAD 0.06–0.17 events/patient-year) with a larger proportion of disabling strokes in the magnetically levitated HM3. 19
Chest radiographs are often performed routinely during outpatient follow-up. Otherwise, although CT imaging provides higher radiation and contrast agent exposure for patients, it offers several advantages of fine spatial resolution and allows the detection of complications (e.g. hematomas, inflow/outflow graft obstruction, aortic thrombi, pump-pocket, or driveline infections).20–24
Although experimental studies have identified possible relationships between flow and thromboembolic complications,11,22,25–28 the mechanistic relationship remains unclear. Nevertheless, the common view of the ideal inflow cannula position seems to be parallel to the septal wall directly facing the mitral valve.22,29–32 It seems clear that an optimized pump position is not optional, but can lead to fewer adverse events, better outcomes and improved patient prognosis. The hypothesis of this study was that there is a difference in the pump position of pump thrombus patients compared to a event free control group. Therefore, we investigated the correlation between pump thrombosis and pump position using radiographs and computed tomography (CT).
Materials and methods
Ethics approval
The local hospital review board (EK1769/2018) approved this study, which includes only retrospectively collected imaging data.
Study population
This retrospective study included 115 LVAD patients, who received either a HeartWare HVAD, (Medtronic Inc.) or a HeartMateII (Abbott Inc.). All implantations were performed with “sew-then-core” technique of the sewing ring at the anatomical apex. After the third POD antiplatelet therapy with acetylsalicylic acid (ASA) was started with device-specific doses from 100 mg (HMII) to 200 mg (HVAD) and INR target range of 2.0–2.5. To best address patient-specific differences, the ASA dose was adjusted individually to remain within the target range. Standard practice for outpatient subtherapeutic INR was done by bridging with enoxaparin (0.5 mg/kg twice daily). 33
For the further analysis 27 patients were not eligible due to early death before the first standing X-ray, non-standard anticoagulation (no ASA), or missing data. Patients with inflow/outflow graft thrombosis were excluded. Of the remaining 88 patients, 15 suffered from intra-pump thrombosis (PT) diagnosed by presence of hemolysis (LDH >3× upper limit of normal, plasma fHb >40 mg/dl), increased LVAD power consumption and/or heart failure not explained by structural heart disease.
To provide a high-quality analysis, the control group (no thrombus—NT) was assigned based on risk factors using propensity score matching. To reduce the selection bias, propensity score matching was applied, which was derived from a non-parsimonous logistic multivariate model. Patients were selected based on one-to-one nearest neighbor matching using a wide range of demographic data, clinical data, and preoperative risk factors 34 to minimize effects of confounding factors. An optimal selection was made among these candidates, resulting in a smaller (15 vs 15 patients) total cohort, but the comparison data sets all have the appearance of a randomized study. 35
Radiologic imaging data
Imaging data were retrospectively collected from the PACS system. The first available standing radiographs (posterior-anterior (PA) and lateral (lat) projections) were exported. Supine images were excluded as they often suffer from poor image quality and poor patient positioning. If available, also routinely performed CT scans were used. All data were analyzed pseudo-anonymized and blinded.
Radiograph measurements
Radiograph DICOM data were imported with ImageJ (v1.51j8, National Institutes of Health, USA) and geometric measurements were performed (see Figure 1). The pump depth, inflow cannula (IC) length, and the angle of the IC against a horizontal reference line were analyzed (similar to Imamura et al. 11 ). In the PA-radiographs, a possible rotation of the patient was corrected, by checking the spline vertical axis. Furthermore, the distance between the heart contour and the IC-tip was measured. In cases where the IC was not visible and hidden behind the pump body, these values could not be evaluated, but its visibility was noted.
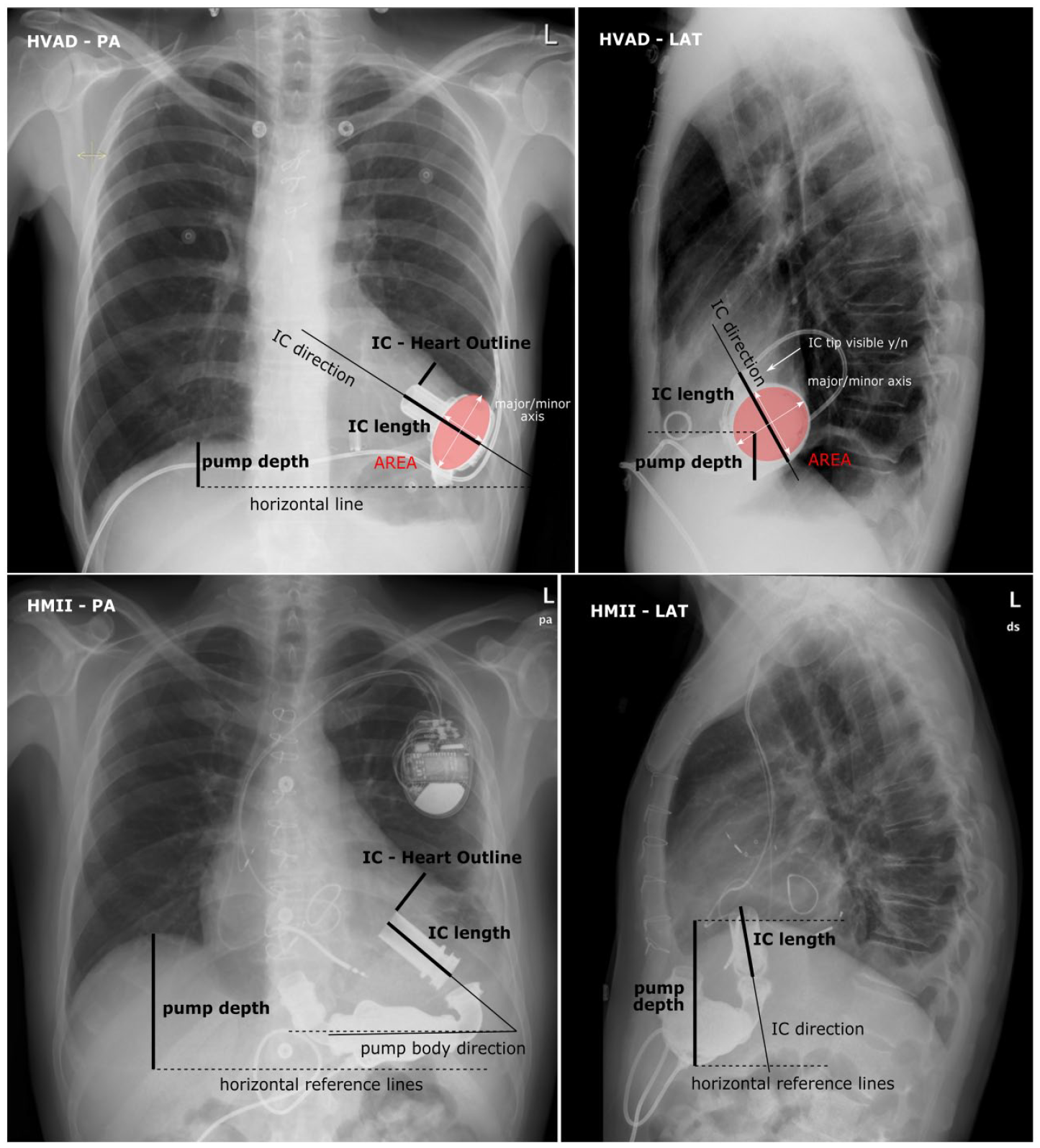
Definition of X-ray parameters: pump depth, IC-horizontal angle, and elliptical approximation for the projected pump body.
Other parameters were defined specifically for the two pump types. The projected HVAD pump body appears in elliptical shape on the radiographs. Elliptical approximations of the pump body and its area, the short and long axis in both frontal and lateral projection were evaluated. For the HMII, the angle between the IC and the pump body was measured.
CT measurements
Multiplanar reconstructions (MPR) from routine CT scans were created in RadiAnt (Medixant, Poznan, Poland) to obtain the three-chamber view (see Figure 2) defined by anatomical landmarks: center of the aortic and mitral valve and the center of the LVAD inflow cannula tip (the apex is usually covered by the IC). Further two angles were defined: Angle α describes the deviation of the IC axis from the axis connecting the cannula tip and the mitral valve. The angle β characterizes the deviation of the IC axis out of the three-chamber view plane. Based on these two angles the deviation from the ideal mitral valve inflow cannula axis was calculated. In addition, at the level of the cannula tip a short axis view was recorded to inspect the IC distances to the endocardial wall.
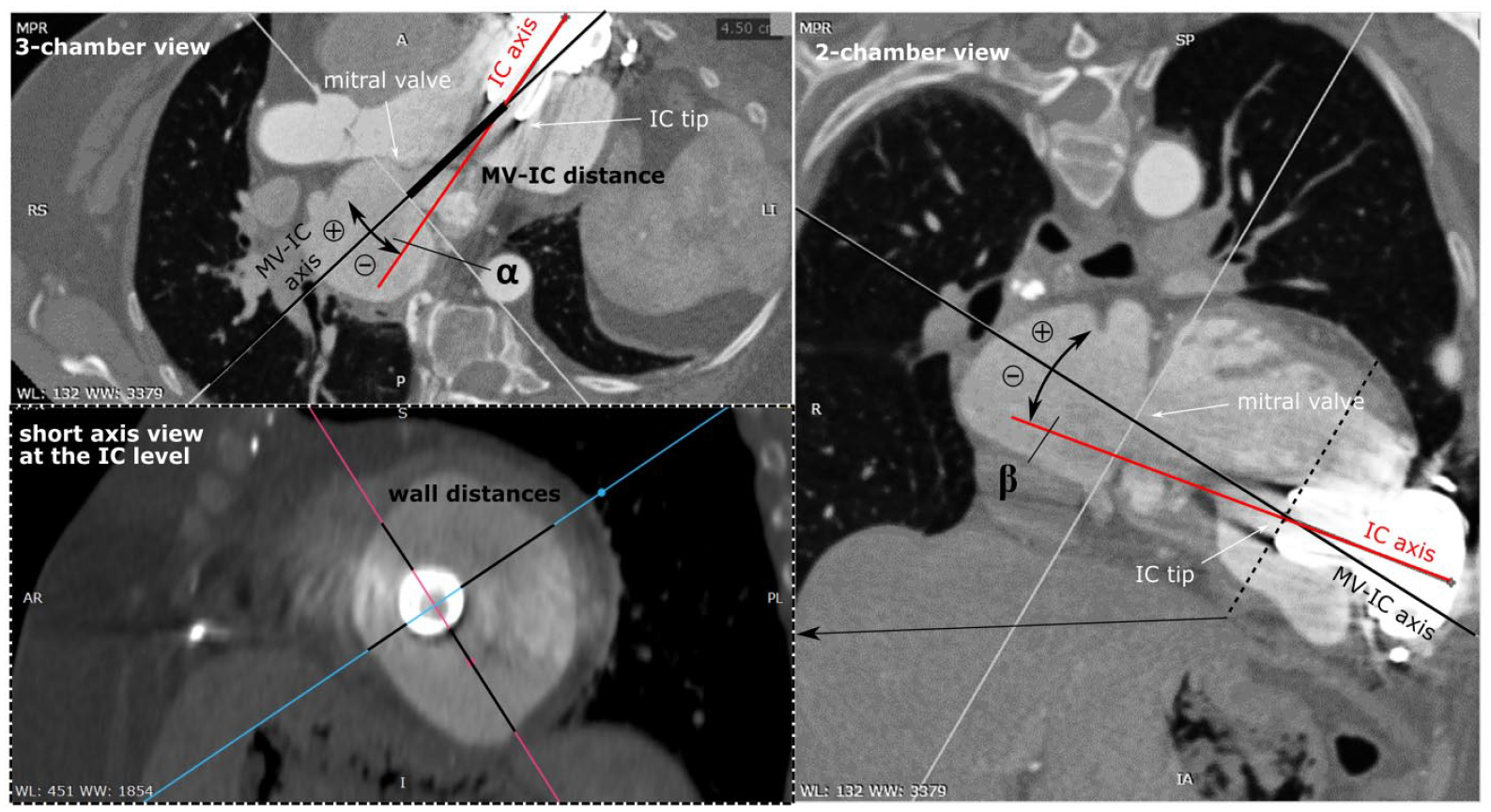
Multiplanar reconstruction of the CT-scans: definition of the angle α in the three-chamber view, the angle β in the two-chamber view and the wall distance measurement of the inflow cannula in the short-axis view.
Statistical evaluation
All collected data were entered into a spreadsheet and statistically analyzed in SPSS 26 (SPSS Inc., Chicago, IL, US). Metric data are presented as mean ± standard deviation for normally distributed data or as median with the interquartile range for non-normally distributed data. The normality of the data was verified using the Shapiro-Wilk test. Statistical comparisons of continuous variables between the PT and NT groups were performed using the Students t-test for normally distributed data and the Mann-Whitney U-test for non-normally distributed data. Fisher’s exact test was used to assess for statistical significance of categorical variables.
Correlations of CT and radiographic data were calculated using Pearson correlation coefficients for normally distributed data without outliers or using Spearman correlation coefficients for the data that were not normally distributed or contained outliers. A Receiver Operating Characteristic (ROC) curve to assess the visibility of the IC in lateral X-rays, as a categorical variable, to predict the occurrence of pump thrombosis was generated along with the area under the curve (AUC). The statistical significance level was set at a p-value of 0.05.
Results
Baseline characteristics
Of the 88 patients 15 (17% of the total cohort) experienced pump thrombosis at a median time of POD 343 (IQR 165–617) of which 8 patients were HVAD (14% of all HVAD recipients, median POD 666 (IQR 339–964)) and 7 patients were HMII patients (22% of all HMII recipients, median POD 808 (IQR 351–1120)). The baseline characteristics of the event group suffering from PT (n = 15) and the whole control cohort (n = 73) including propensity score-matched group (n = 15) control group are summarized in Table 1.
Baseline characteristics of the pump thrombus group (n = 15) and the propensity score matched control group (n = 15).
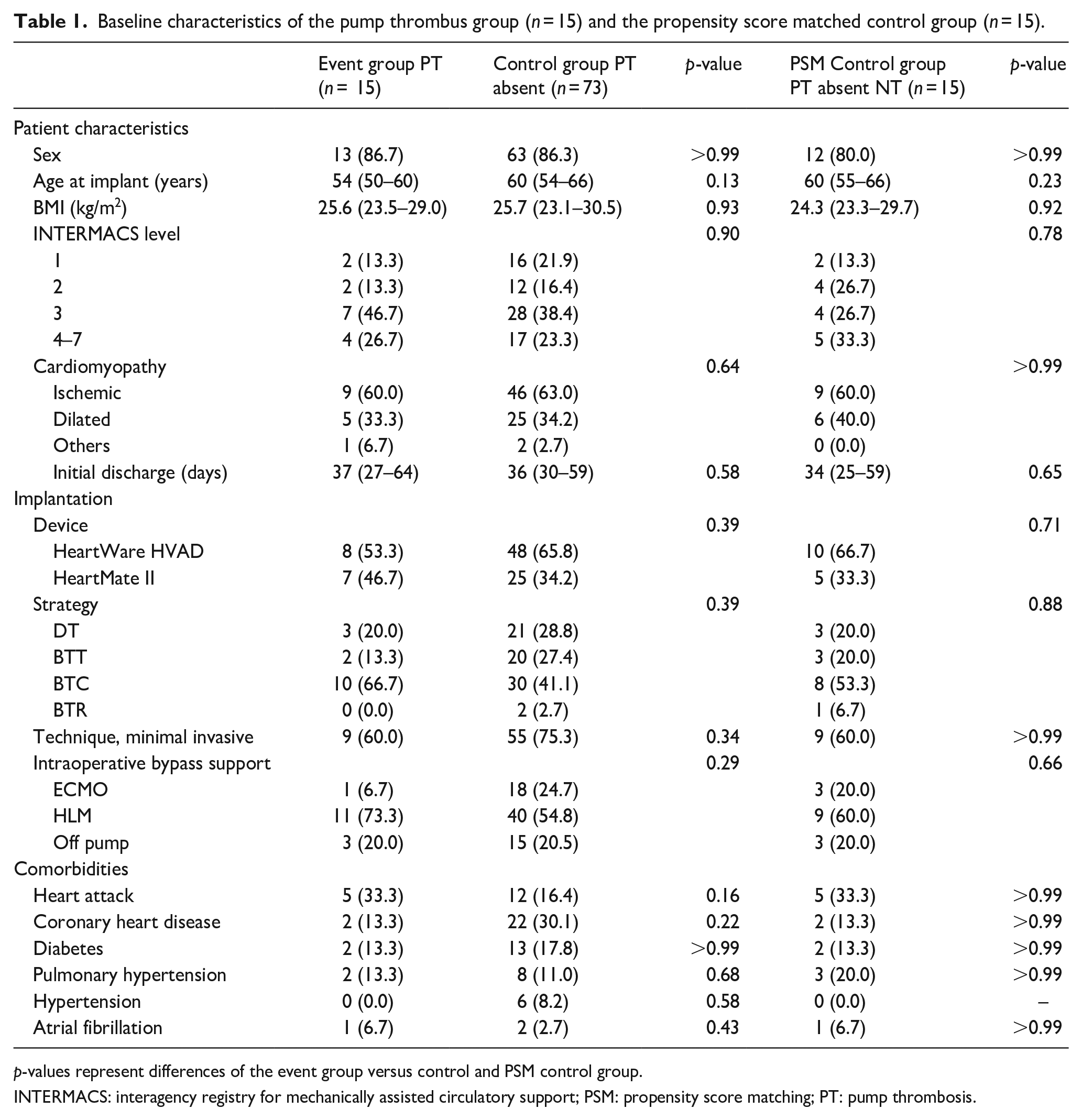
p-values represent differences of the event group versus control and PSM control group.
INTERMACS: interagency registry for mechanically assisted circulatory support; PSM: propensity score matching; PT: pump thrombosis.
Radiograph imaging data
Depending on the postoperative course, the POD of the first standing radiograph was 24 days (IQR 14–67 days). If no lateral radiograph was included in the defined first standing radiograph, the next routine radiograph within 2 months was used.
In the frontal radiographs, the differences in the evaluated angles and distances did not reach statistical significance for both pump types (see Table 2). However, in the HVAD PT patients the short axis of the elliptical projection was larger (PT 41.3 ± 4.8 mm vs NT 34.9 ± 6.0 mm, p = 0.026). No differences were found in HMII patients.
PA radiograph measurements.
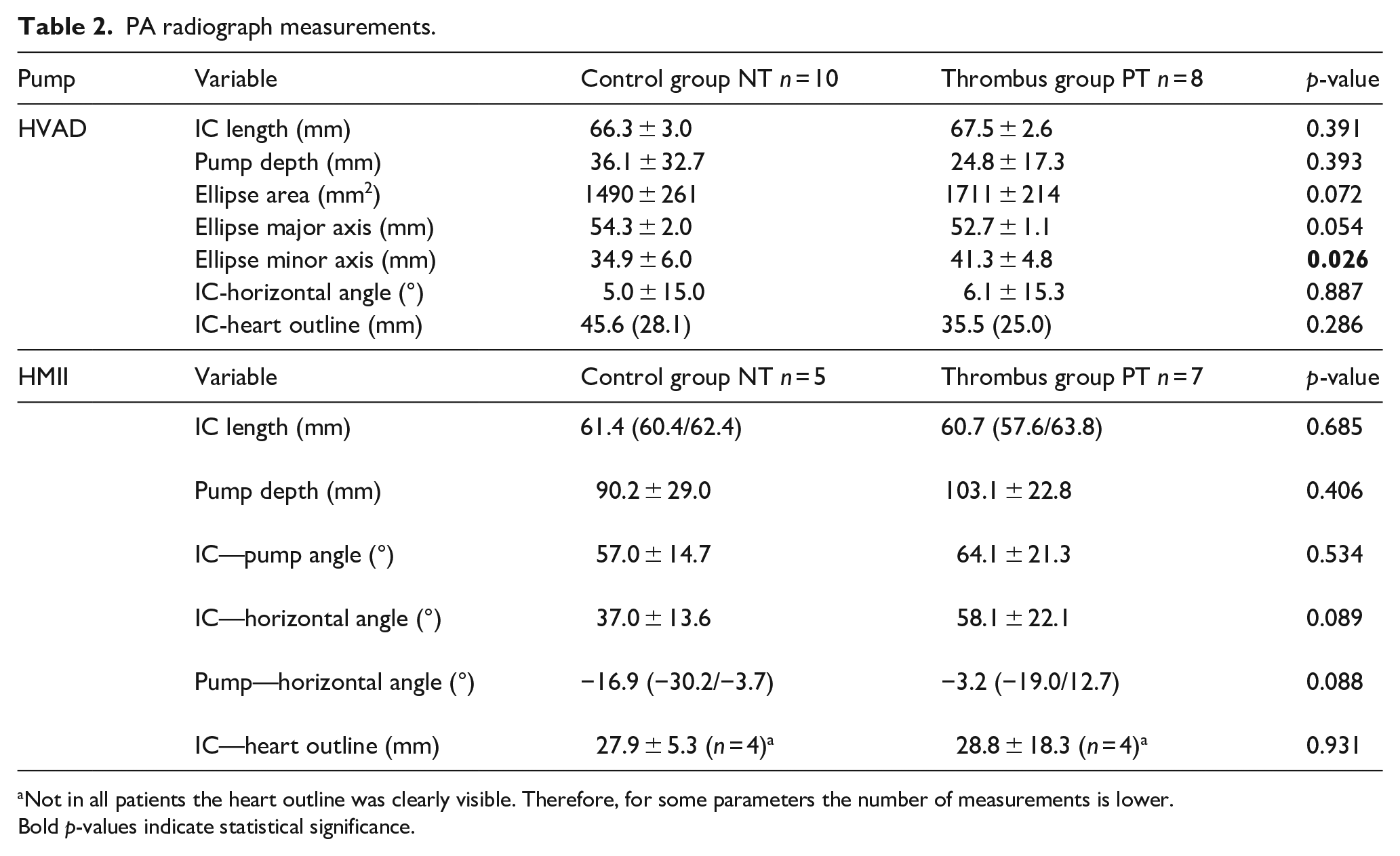
Not in all patients the heart outline was clearly visible. Therefore, for some parameters the number of measurements is lower. Bold p-values indicate statistical significance.
In the lateral radiographs, the differences in the evaluated angles and lengths did not reach statistically significant differences for both pump types (see Table 3). Trends in HVAD patients were identified: PT patients showed a smaller pump depth and a smaller minor axis of the projected ellipse. Further, the visibility of the IC cannula was higher in PT patients. Using the Receiver Operating Characteristic (ROC) curve, a visible IC in the lateral X-ray image provided a sensitivity of 80% and a specificity of 75% (C-statistic = 0.78) for the prediction of PT. For the HMII no differences in the evaluated parameters were found.
Lateral radiograph measurements.
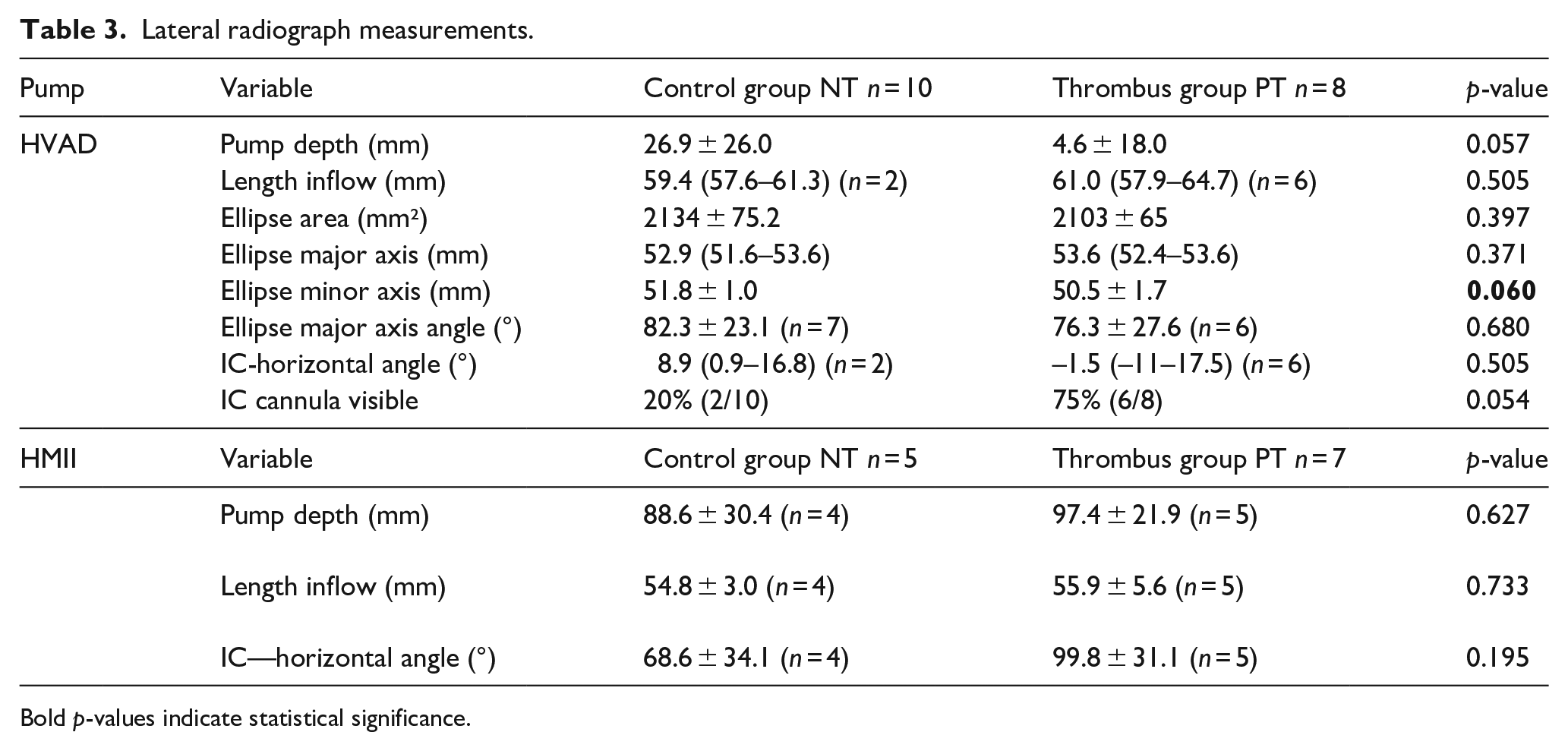
Bold p-values indicate statistical significance.
CT data
A total of 18 routine CT scans were available in the hospital’s PACS system (PT 11, NT 7; HVAD: 8 PT 5, NT 3, HMII: 10 PT 6, NT 4) with a mean POD of 273 days (72-404 days). One dataset had to be excluded due to insufficient image quality.
In HVAD-PT patients, the deviation from the mitral-apical axis (angle α) was significantly greater compared to the NT group (see Table 4 and Figure 3). On the other hand, the HMII inflow cannulas were not aligned with this axis either, but again no differences were found. Differences were also visible in other parameters, but below the statistical significance level considered. Overall, the distance of the IC to the endocardial wall was smaller in the PT group (7.5 ± 8.2 mm, n = 10) than in the NT group (11.6 ± 5.0 mm; n = 7; p = 0.030) for all pumps (see Figure 3).
CT Measurements of the two pump types with and without pump thrombosis.
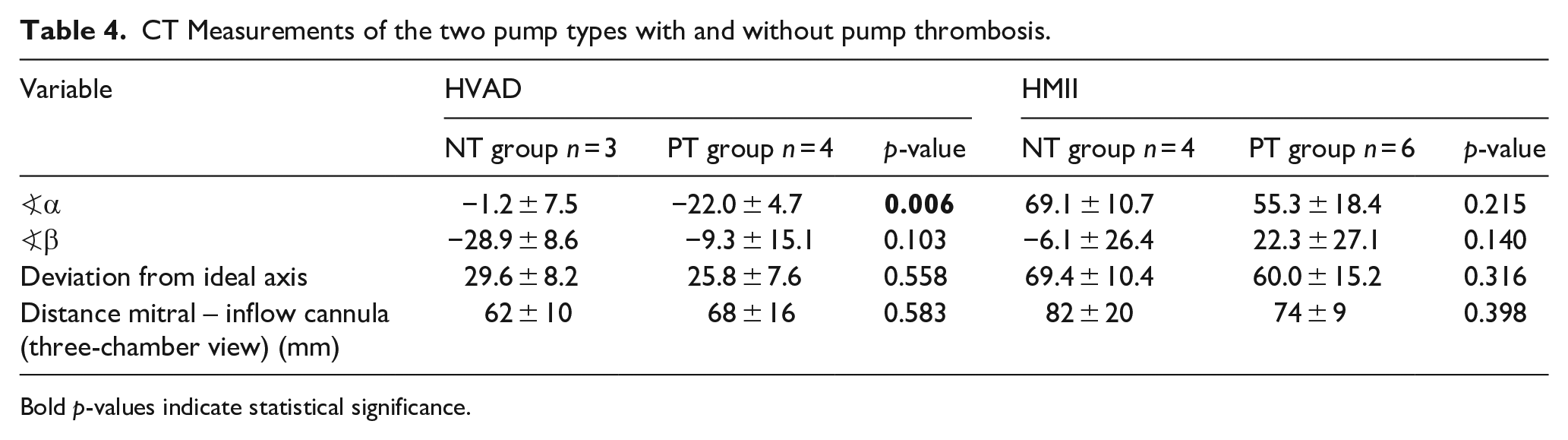
Bold p-values indicate statistical significance.
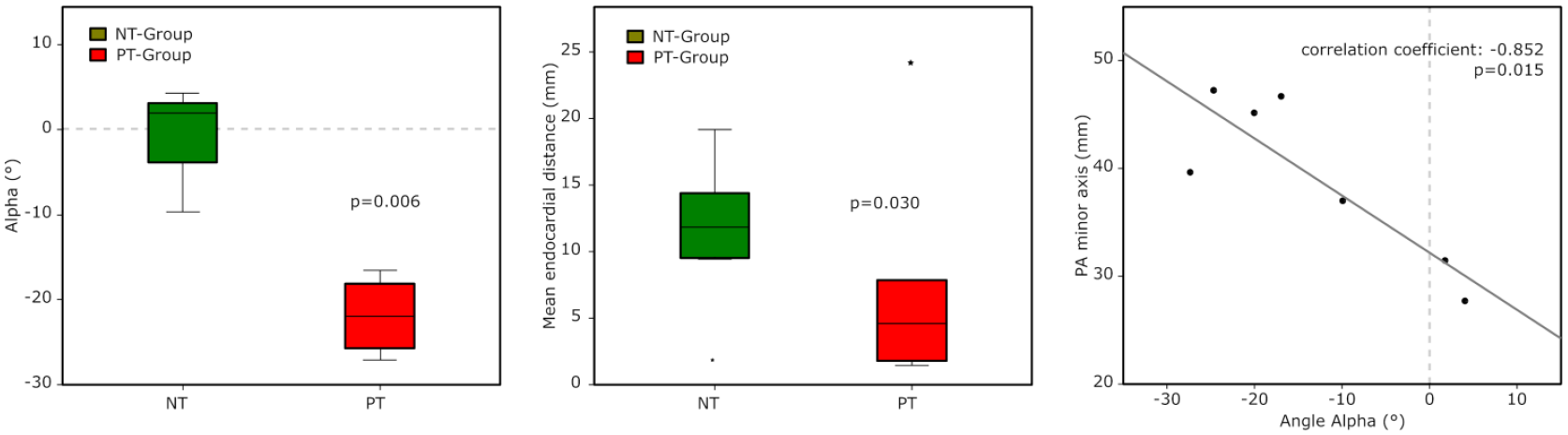
Angle α measured in the multiplanar reconstruction (HVAD patients), distance of the inflow cannula to the endocardial wall (all patients) and correlation of the PA minor axis with angle α.
Correlation of radiographs and CT data
For the HVAD, in the frontal radiographs the minor axis of the pump ellipse (see Figure 3), the projected area and in the lateral radiographs the cannula visibility correlated well with the CT angle α. For the angle β further correlations were found for the PA and sagittal IC-horizontal angles (see Table 5).
Correlation table of selected X-ray parameters to values from CT-scans.
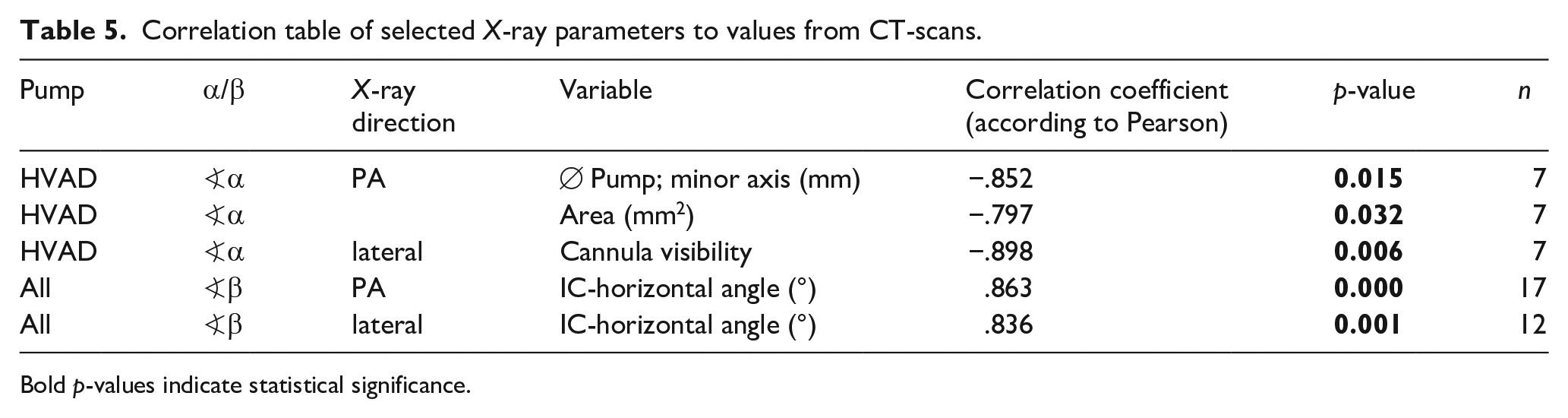
Bold p-values indicate statistical significance.
Discussion
Malpositioning of VADs and inflow cannulas potentially leads to a vicious circle of flow disturbance, suction (followed by low flow or tissue irritation), all of which may ultimately contribute to the adverse events profile of these devices. 22 However, by surgical techniques and careful implantation this risk could be reduced and influenced.
In this study, routinely obtained imaging data were used to identify malpositions from routinely obtained radiographs and CT scans and to correlate them with pump-thrombosis.
HMII positioning
Several geometric measurements (e.g. angle of inflow relative to the horizontal or to the pump, pump depth, angle between the pump, and the outflow graft) have been suggested which are closely related to a higher risk of thrombosis, lower readmission-free survival and poor unloading.7,8,10,13,29 Differences reported for HMII7,8 between PT and NT patients were not found in this study, probably because in both groups, the angle of the IC to the pump body was above the proposed risk angle 8 of >55° (NT 57.0 ± 14.7 vs PT 64.1 ± 21.3, p = 0.534), and further no differences were found in pump depth either (NT 90.2 ± 29.0 vs PT 103.1 ± 22.8 (p = 0.406), as also found in Han et al. 29 Overall, the analyzed data showed that the HMII pumps and ICs in our cohort were well positioned outside the proposed risks measures. 8
HVAD positioning
Positioning aspects of HVAD are less well understood but adequate HVAD pump positioning seems to be easier compared to HMII because no pump pocket needs to be created and the pump can be placed intrapericardially. 22
Large IC-horizontal angles of the HVAD inflow cannula (>65°) have been associated with reduced right ventricular function, reduced LV discharge and LV flow, more heart failure readmissions and increased risk of gastrointestinal bleeding (GIB).11,12,36 However, in our study, the mean IC-horizontal angles were generally much smaller (PT 6.1 ± 15.3° vs NT 5.0 ± 6.1°, p = 0.890) compared to literature data (49.3 ± 32.2°, range 4°–104°) 11 and no patients were found who were above the mentioned value. Low IC-horizontal angles seem to be favorable as they point more in the direction of the mitral valve. 11
Anterior or posterior rotation of the HVAD is seen as a large projected pump body area on the PA radiographs. PT patients tend to have larger areas (PT 1711 ± 214 mm² vs NT 1490 ± 261 mm², p = 0.072) and simultaneously the small minor axis of the projected ellipse was significantly larger (PT 41.6 ± 4.8 mm vs NT 34.9 ± 6.0 mm, p = 0.026). In the lateral radiographs, IC visibility was used to identify anterior or posterior rotation, and this can be used as a simple parameter to predict pump thrombosis with reasonable sensitivity (80%) and acceptable specificity (75%). A posterior deviation from the coronal plane has been identified as a risk factor for pump thrombosis and can be easily identified on PA (large projected area) and lateral (cannula visibility) radiographs.
CT scan parameters and correlation with radiographs
Chest or cardiac CTs are usually not performed routinely but represent a useful diagnostic tools for LVAD patients.20,23 and CTs has been used to identify pump malposition in up to 53% of patients in Sorensen et al. 31 Clinical radiographs on the other hand are standard for outpatient visits and therefore, we have tried to use these data complementary and not separately.
In the multiplanar reconstructions the angle α, the deviation of the IC to the mitral valve in the three-chamber view differed between PT and NT patients in HVAD patients. Several X-ray parameters correlated well with the angle α (PA radiographs: area, minor elliptical axis; lateral radiographs: visibility of the cannula). This is an important clinical implication as it makes it easy to identify a PT risk on cheap X-rays, of which a much larger number are routinely acquired with a low radiation dose and no contrast agent exposure compared to CT scans. Future research should focus on whether cut-off values should be used to identify at-risk individuals in order to optimize clinical outcomes with intensified follow-up and risk-adapted modifications (e.g. increase INR target range).
Overall, the HVAD appears to be better aligned with the mitral-tip axis compared to the HMII (27.4 ± 7.5° vs 59.7 ± 13.8°), which was similarly found in another study 22 and might be explained by the different pump designs.
The distance of the IC to the endocardial wall was smaller in PT patients, possibly indicating more wall contact and/or suction events, 37 which might lead to flow reduction and disturbances. 11 In numerical models, this mechanism could be explained by reduced blood washout and higher stagnation at low wall distance, 14 but further clinical data on suction and position are needed.
Contribution of other risk factors
In a subanalysis of the same cohort of patients, pump thrombosis was associated with a lower percentage of INR time in the therapeutic range of 2.0 to 2.5 within 60 days before pump thrombosis (36 ± 26% vs 65 ± 28%). 34 Pump misplacements appear to be more of a long-term risk factor, whereas INR abnormalities appear to be more of a short-term risk factor but can add up. Besides other known patient and management factors (infections, compliance to medication)6,8–11,13,15,17 the presented data clearly shows that the pump position is another aspect that, in combination with poor anticoagulation therapy, contributes to the development of pump thrombosis.
Limitations
Due to the restrospective single-center character of this study, the number of PT patients was limited and not all patients routinely underwent postoperative CT imaging. This hindered the quantification of pump position from CT scans for all patients. This limitation could be eliminated in a larger prospective multi-center study. Remodeling of the left ventricle over time and cardiac contraction—although hugely reduced in this cohort—are possible additional factors that are likely to have an impact on the pump position measurements and are certainly potential subjects for further studies.
Conclusion
The importance of an appropriate LVAD pump position was illustrated in this study. On routinely performed radiographs and CT scans, position parameters were associated with pump thrombosis for HVAD patients. Deviations of the inflow cannula from the mitral-apical axis favor pump thrombosis. This was not only detected in CT scans, but the sagittal rotation of the HVAD, which was identified on lateral radiographs as visibility of the HVAD inflow cannula, was a predictive parameter for pump thrombosis.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TS, DZ are consultants for Medtronic and Abbott. TS, DZ, HS, and FM received research grants from Medtronic and/or Abbott not related to this work. All other authors have no financial relationship related to this article to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the Austrian National Bank (Jubiläumsfonds #17314) and the Austrian Research Promotion Agency FFG (Project M3dRES Number 858060).
