Abstract
Purpose:
Microaggregates have often been observed during hemodialysis and are clearly associated with complications of hemodialysis therapy. In this study, we aimed to clarify the effects of two polysulfone membranes, with different abilities to activate blood cells, on the formation of these microaggregates; we also investigated their molecular mechanisms.
Methods:
Human whole blood was circulated through a mini-module dialyzer using the membranes in vitro; platelet–neutrophil complexes in blood were determined by flow cytometry. Isolated human neutrophils were incubated with the membranes in plasma, in the presence or absence of platelets, followed by flow cytometric analysis of intracellular reactive oxygen species and cell-surface activated CD11b on neutrophils.
Results:
CX-U, a conventional polysulfone membrane with remarkable cell activation, induced the formation of platelet–neutrophil complexes; however, NV-U, a new hydrophilic polysulfone membrane with slight or no cell activation, did not cause complex formation. Moreover, CX-U-induced reactive oxygen species production and the increase in activated CD11b expression on neutrophils were enhanced by platelets. On the other hand, NV-U hardly affected neutrophil activation, regardless of whether platelets were present or not. The enhancement of CX-U-induced neutrophil activations by platelets was greatly inhibited by anti-CD62P antibody.
Conclusion:
The ability of polysulfone membranes to activate blood cells is closely related to platelet–neutrophil interaction. Therefore, a biocompatible membrane, like NV-U, can be expected to prevent microaggregate formation during hemodialysis and avoid subsequent cell activation.
Keywords
Introduction
The formation of microaggregates has often been observed in blood sample of hemodialysis (HD) patients. Various studies have shown that the process involves interaction between blood cells and HD membranes, particularly HD membrane-induced platelet activation,1–4 and the platelet–neutrophil complex is primarily mediated by the interaction between CD62P on activated platelets and its ligand, P-selectin glycoprotein ligand-1 (PSGL-1), on neutrophils.5,6 In addition, several studies have suggested that direct physical interaction with platelets primes neutrophils to enhance neutrophil functions, such as production of reactive oxygen species (ROS), leading to oxidative stress-related pathological conditions. 4
A heightened incidence of microaggregates also exists in a variety of inflammatory, auto-immune and infectious diseases.7–9 Moreover, heightened occurrence of microaggregates has been encountered in cardiovascular disease, including atherosclerosis.10,11 This information suggested that microaggregates may contribute to the inflammatory and thrombotic processes in HD patients. In particular, atherosclerosis-related cardiovascular disease is a major cause of morbidity and mortality in HD patients, 12 and we believe that it is important to reduce such risks by preventing microaggregate-associated inflammation and thrombus formation.
Microaggregate formation during HD was reported to depend on the membrane materials. Regenerated cellulose (RC), and modified cellulosic membranes and polysulfone (PSf) membranes, were somewhat more active than ethylene vinyl alcohol and polyacrylonitrile.1–3 Among these materials, PSf is most commonly used to constitute HD membranes because of its high performance. Several different brands of PSf dialyzer are currently available commercially, and their membranes cannot be considered to be equivalent because we previously demonstrated their different activities against cells. 13 However, it is not clear whether such variability causes parallel differences in the formation of microaggregates. In the present study, we examined the effects of two PSf membranes, which have different abilities to activate cells, on the formation of platelet–neutrophil complexes and subsequent neutrophil activation.
Materials and methods
HD membranes
The HD membranes were obtained from commercial dialyzers: TORAYLIGHT® CX-U and TORAYLIGHT NV-U (Toray). The same PSf-based membrane (inner diameter: 200 μm, wall thickness: 40 μm, sterilization: Gamma-ray irradiation) is used in both CX-U and NV-U, but the surface of NV-U is improved through the use of a new hydrophilic polymer. 14
Reagents
Anti-CD62P antibody (clone: AK4), fluorescein isothiocyanate (FITC)-conjugated anti-CD33 antibody (clone: HIM3-4), allophycocyanin (APC)-conjugated anti-CD41 antibody (clone: HIP-8), and phycoerythrin (PE)-conjugated anti-activated CD11b antibody (clone: CBRM1/5) were purchased from BioLegend. Isotype control antibodies were purchased from Becton Dickinson and Company or BioLegend. We purchased 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) from Sigma-Aldrich.
Preparation of whole blood, platelets, and neutrophils
Venous whole blood anticoagulated with heparin (50 U/mL) was obtained from healthy blood donors and used within 30 min of blood collection. Data obtained with whole blood/cells from one blood donor were considered as one experiment. Platelet-rich plasma (PRP) was prepared by centrifuging heparinized blood at 150 g for 10 min. To ensure autologous, cell-free plasma collection, heparinized blood was centrifuged at 1400 g for 10 min, followed by filtration with a 0.2 µm filter. Heparinized human blood used for the preparation of neutrophils was mixed with an equal volume of 2% dextran in saline, and most of the erythrocytes were allowed to sediment out for 40 min. The leukocyte-rich supernatant was then subjected to Ficoll-Paque PLUS (GE Healthcare UK Ltd.) density gradient centrifugation at 1300g for 20 min. Residual erythrocytes were removed by hypotonic lysis (BD Biosciences) with a resulting purity of >95% neutrophils as assessed by flow cytometric analysis of the expression of CD33 and CD11b.
This study was reviewed by the Human Tissue Samples Ethics Committee for R&D Toray Industries, Inc. and approved by the chairperson of the committee in compliance with the Human Tissue Samples Ethics Rules for R&D Toray Industries, Inc. All blood donors were informed about the study procedure and provided informed consent.
Hemoperfusion of mini-module dialyzer in vitro
We prepared a mini-module dialyzer (miniMD) containing 50 hollow fiber membranes. 13 Distilled water was circulated through the miniMD at 1 mL/min for more than 5 min, followed by the circulation of phosphate buffered saline (PBS) for 30 min to clean it. Subsequently, whole blood (5 mL) was circulated at a flow rate of 1 mL/min through the circuit at 37°C for 10 min, and blood samples were collected for analysis. A similar silicon-tube circuit without the miniMD was used for sham perfusion.
Treatment of platelets and neutrophils with small pieces of HD membrane
The HD membranes were cut into small pieces 1–2 mm in length, rinsed with sterile saline, and stored in sterile saline until use. The small pieces of membrane (derived from 100 cm lengths of hollow fibers) were added to 2 × 106 neutrophils suspended in 200 μL of autologous plasma or PRP and incubated at 37°C for 15 min, followed by flow cytometric analysis of intracellular production of reactive oxygen species (ROS) and cell-surface activated CD11b, as described later. In the study of molecular mechanisms, neutrophils were pretreated with 20 μg/mL anti-CD62P antibody for 10 min at 37°C before adding small pieces of membrane.
Flow cytometric analysis of platelet–neutrophil complexes, intracellular production of ROS, and cell-surface activated CD11b
The blood sample collected after hemoperfusion in vitro was stained with FITC-conjugated anti-CD33 antibody and APC-conjugated anti-CD41 antibody. The blood was then fixed to prevent spontaneous activation of platelets and lysed by addition of FACS lysing solution (BD Biosciences); the cells were subjected to flow cytometry analysis within 1 h following sample preparation.
Isolated neutrophils treated with small pieces of HD membrane in plasma were stained with PE-conjugated anti-activated CD11b antibody and DCFH-DA (0.1 mM) at 37°C for 15 min. After diffusion into the cell, DCFH-DA is deacetylated by esterases to the non-fluorescent compound DCFH, which is later oxidized by ROS into a fluorescent compound. The neutrophils were fixed with 1% paraformaldehyde in PBS at room temperature for 15 min and subjected to flow cytometry analysis.
The fluorescence intensity of the cells was analyzed using a Gallios flow cytometer (Beckman Coulter). CD41 positive and CD33 low-positive cells (CD41+CD33lo) were identified as the platelet–neutrophil complexes (Figure 1(a) and (b)). Results of formation of platelet–neutrophil complexes were expressed as the percentage of CD41-positive cells (%) among CD33 low-positive cells. The isolated neutrophils were gated according to their size and granularity on a forward-scatter versus side-scatter dot plot. ROS production was detected by measuring the intracellular oxidized form of DCFH on flow cytometric analysis.
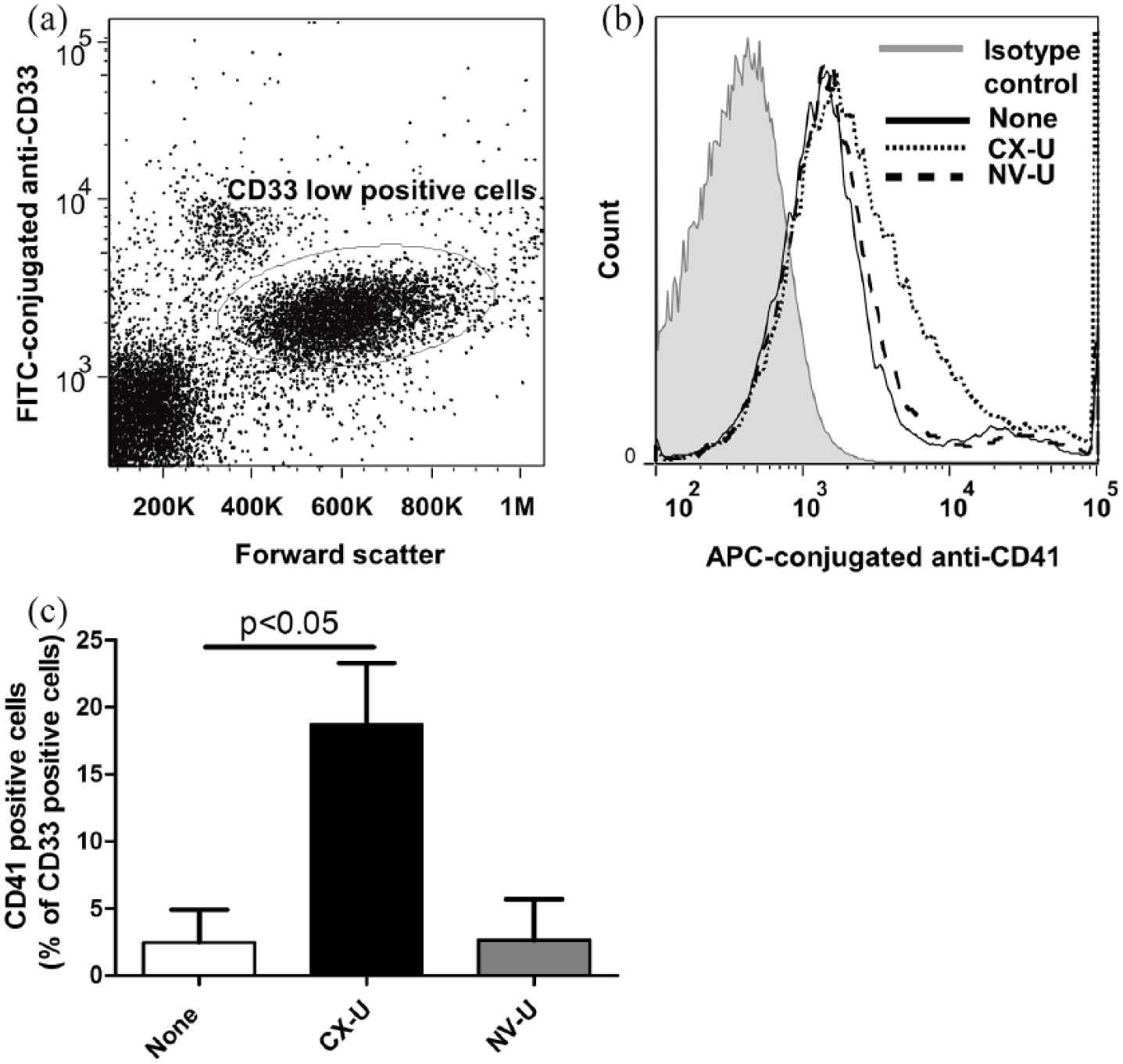
Formation of platelet–neutrophil complexes induced by CX-U and NV-U in whole blood. (a) A dot plot of forward scatter versus log fluorescence of the FITC-conjugated anti-CD33. The CD33 low-positive cells were identified as neutrophils. (b) A representative flow cytometry histogram plot showing expression of CD41 on CD33 low-positive cells. (c) The percentage of CD41-positive cells among CD33 low-positive cells (platelet–neutrophil complexes) was determined. Data presented as mean ± SEM of three independent experiments are shown. The p value shown was obtained by Dunnett’s multiple comparison.
Statistical analysis
Data were presented as the mean ± standard error of the mean (SEM). Statistical significance was determined using the SAS System (SAS Institute). Statistical analysis was performed using an F-test followed by a t-test for two groups or using Bartlett’s test followed by Dunnett’s comparison for three groups. Differences were considered significant when the p value was <0.05.
Results
We investigated the effects of two PSf HD membranes, CX-U and NV-U, on the formation of platelet–neutrophil complexes in whole blood. To evaluate the interaction among blood cells under flow conditions similar to those found in the clinical setting, we used an in vitro miniMD system that pumps whole blood through HD membranes. Circulation of whole blood through the miniMD with CX-U increased the formation of platelet–neutrophil complexes compared to sham circulation without a miniMD. On the other hand, NV-U had no effect on the formation of platelet–neutrophil complexes (Figure 1).
We then explored the influence of platelet–neutrophil complexes on cell activation and its mechanisms. In this experiment, we used purified neutrophils and PRP to conduct experiments under well-defined conditions suitable for assessing the interaction between platelets and neutrophils. When neutrophils were incubated with CX-U in the absence of platelets, CX-U induced ROS production of the neutrophils and increased activated CD11b expression on the neutrophils (Figures 2 and 3). In the presence of platelets, the CX-U-induced neutrophil ROS production and activated CD11b expression were further increased (Figures 2 and 3). On the other hand, NV-U did not affect neutrophil activation whether platelets were present or not (Figures 2 and 3).
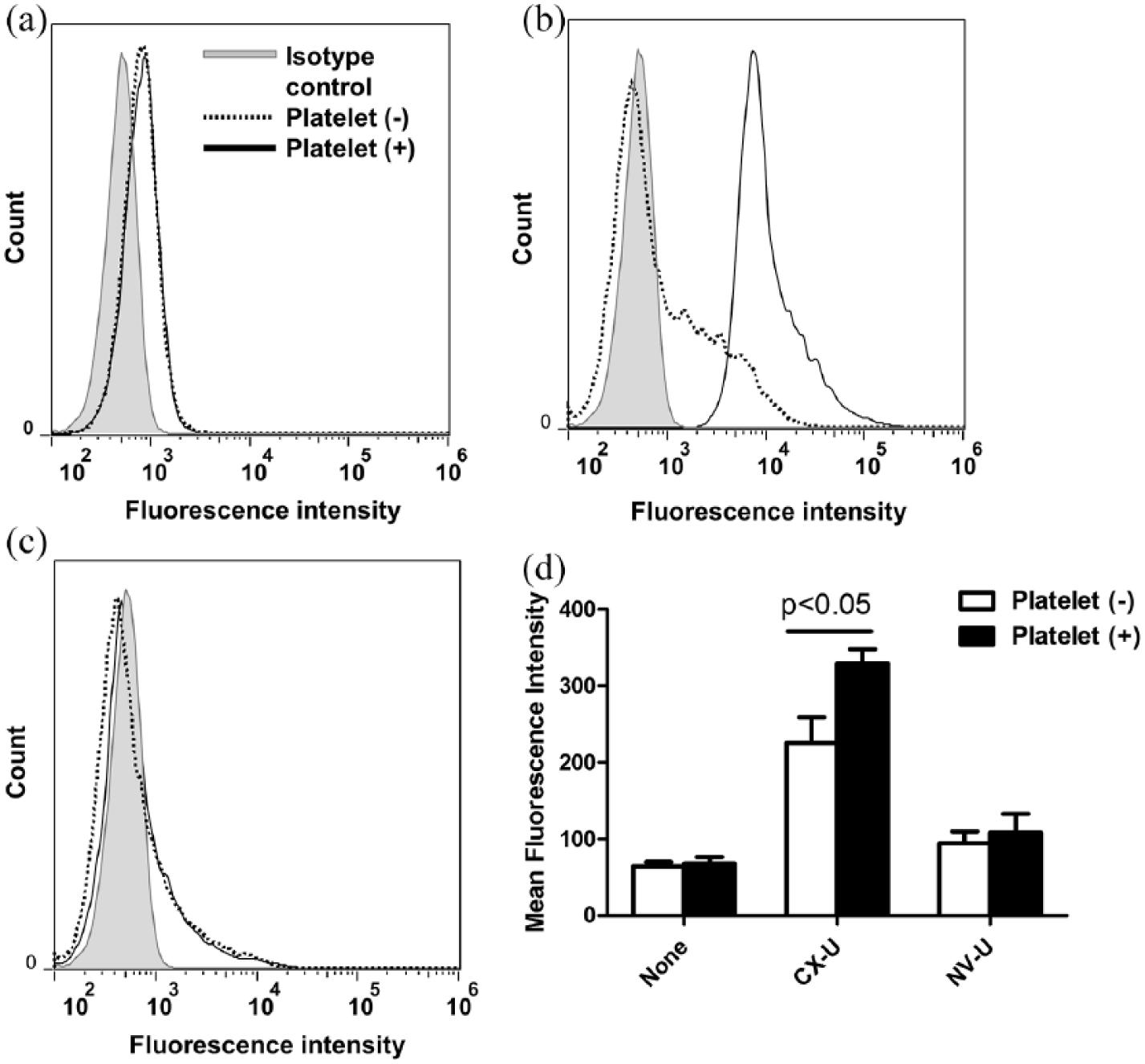
Enhancement of hemodialysis (HD) membrane-induced neutrophil ROS production by platelets. Isolated neutrophils suspended in plasma (platelet (–)) or platelet-rich plasma (platelet (+)) were treated without (none) or with an HD membrane, and then stained with DCFH-DA. Representative flow cytometry histogram plots: (a) none, (b) CX-U, and (c) NV-U. (d) Data presented as mean ± SEM of four independent experiments. The p value was obtained from a t-test.
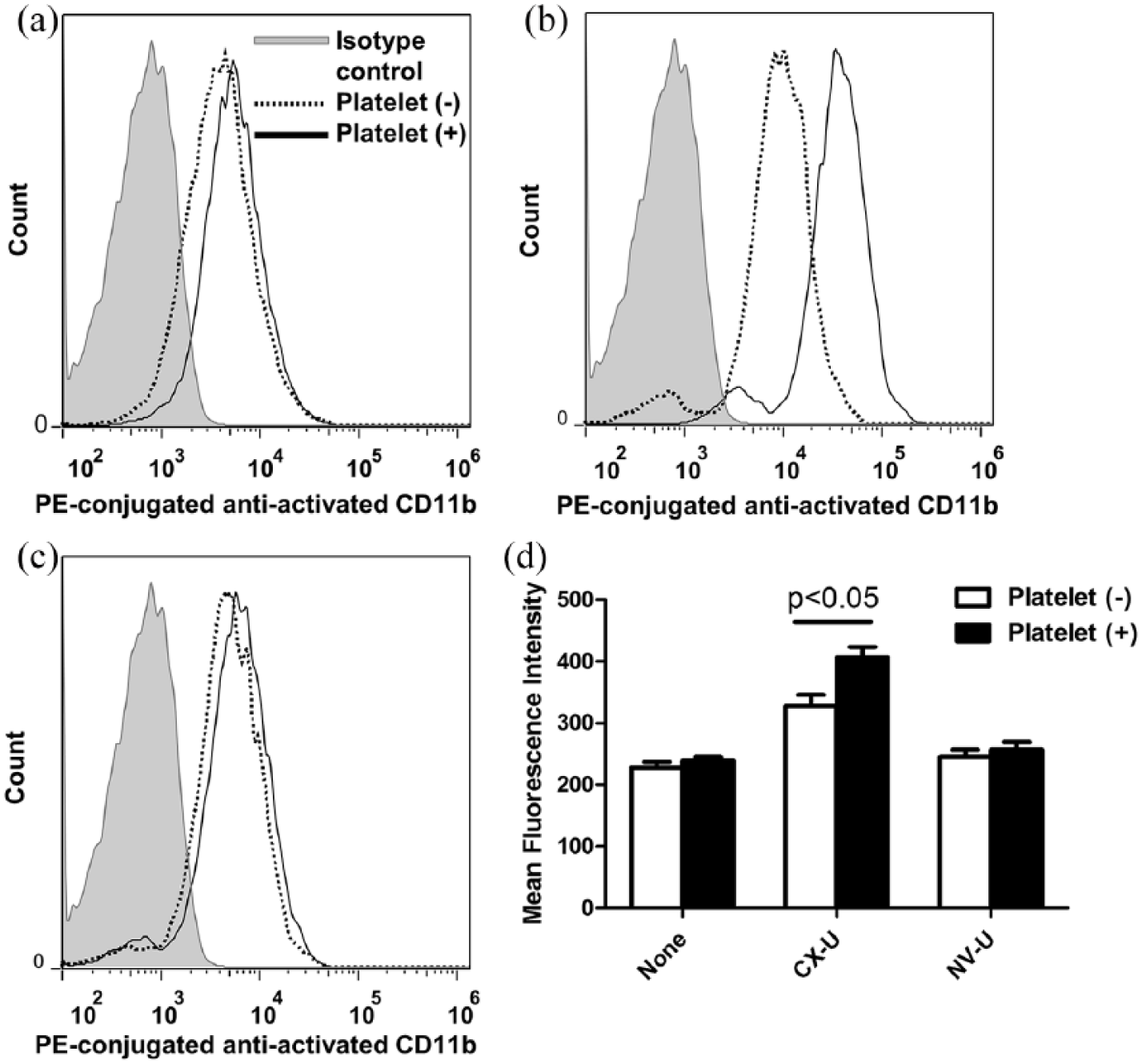
Enhancement of activated CD11b expression of neutrophils by platelets. Isolated neutrophils suspended in plasma (platelet (–)) or platelet-rich plasma (platelet (+)) were treated without (none) or with an HD membrane, and then stained with PE-anti-activated CD11b antibody. Representative flow cytometry histogram plots: (a) none, (b) CX-U, and (c) NV-U. (d) Data presented as mean ± SEM of three independent experiments. The p value was obtained from a t-test.
Because CD62P, also called P-selectin, is known to mediate platelet–neutrophil adhesion,5,6,15 we then examined the involvement of CD62P in the enhancement of neutrophil activation induced by platelets. Anti-CD62P antibody reduced CX-U-induced neutrophil ROS production in the presence of platelets to the same level of production seen in the absence of platelets (Figure 4(a)). In addition, anti-CD62P antibody reduced activated CD11b expression in the presence of platelets (Figure 4(b)). In the presence of platelets, there were no statistically significant differences between controls and anti-CD62P in terms of ROS production (p = 0.09) or activated CD11b expression (p = 0.10).
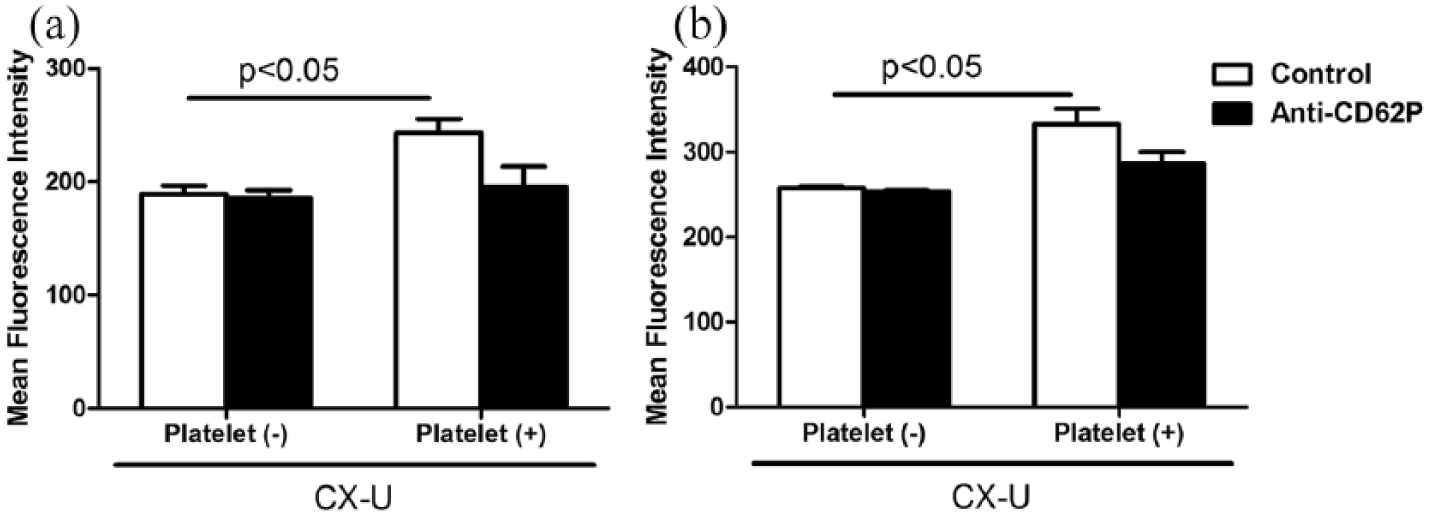
Effects of anti-CD62P antibody on hemodialysis (HD) membrane-induced neutrophil activation. Isolated neutrophils suspended in plasma (platelet (–)) or platelet-rich plasma (platelet (+)) were treated with or without small pieces of CX-U in the absence or presence of 20 μg/mL anti-CD62P antibody, and then stained with PE-conjugated anti-activated CD11b and DCFH-DA. Data are presented as mean ± SEM of triplicate measurements obtained from one representative experiment. The p values shown were obtained from t-tests. (a) ROS production and (b) activated CD11b expression.
Discussion
Clinical evaluation reports have shown that the number of platelets adhering to membrane surfaces was lower with the NV-U dialyzer than with CX-U. 16 Our in vitro studies also supported these clinical observations, as we were able to confirm that CX-U caused remarkable activation of platelets and NV-U did not. 13 CX-U and NV-U have very different abilities to activate blood cells; we thus further investigated their biocompatibility by assessing their effects on the formation of microaggregates and subsequent cell activation in vitro.
Our results can be summarized as follows: (1) CX-U increased the formation of platelet–neutrophil complexes, and platelets promoted CX-U-induced neutrophil activation; (2) the enhancement of CX-U-induced neutrophil activation by platelets was greatly suppressed by the blockade of CD62P; (3) NV-U did not provoke the formation of platelet–neutrophil complexes and neutrophil activation, even when co-incubated with platelets. On comparing CX-U and NV-U, the cell activation induced by the membranes seems to correlate with the formation of platelet–neutrophil complexes induced by the membranes because the formation of cell complexes was induced to a great degree by CX-U, with minimal changes induced by NV-U.
Regarding platelet activation, previous studies suggested that the formation of microaggregates induced by HD membranes depends on the ability of the HD membranes to activate platelets.2,5 This is because CD62P is usually stored in a membrane protein granule in platelets and rapidly expressed on the cell surface to establish cell adhesion, but only when platelets are activated. 15 In agreement with this information, it is thought that the different abilities of CX-U and NV-U to stimulate platelets led to different CD62P expression on the surfaces of platelets, followed by differential formation of platelet–neutrophil complexes.
Regarding neutrophil activation, the redistribution of a CD62P ligand, PSGL-1, on neutrophils is thought to contribute to the formation of platelet–neutrophil complexes. 6 In particular, with RC membranes, complement-induced redistribution of PSGL-1 on neutrophils is considered to be the main cause of microaggregate formation. This is because neutrophils constitutively express PSGL-1 and the RC membrane showed a slight tendency toward platelet activation. 5 PSf membranes activate complement poorly 17 and it is quite unlikely that complement-induced redistribution of PSGL-1 occurred. Our previous study showed that fibrinogen adsorbed on PSf membranes can activate neutrophils 13 ; therefore, CX-U-induced neutrophil activation via fibrinogen may cause redistribution of PSGL-1 and subsequent microaggregate formation.
Microaggregates of blood cells are often observed in a variety of pathological disorders including HD treatment, and inflammatory disease. The clinical outcomes associated with increased microaggregates have not been clearly elucidated; however, they play important roles in cell activation, ROS production, cell adhesion, and phagocytosis.4,18 In fact, we confirmed that interaction with platelets augmented CX-U-induced neutrophil ROS production. Thus, HD-membrane-induced microaggregates are most likely linked to HD-associated complications by inducing chronic inflammation and oxidative stress, and we believe that it is extremely important to reduce these risks.
In summary, we demonstrated that varying platelet and neutrophil activation among HD membranes causes parallel differences in the formation of microaggregates. It is thus of paramount importance to prevent cell activation if we are to prevent microaggregate formation. Consequently, the use of HD membranes with low inducibility for cell activation, like NV-U, is expected to decrease the formation of microaggregates in HD patients and has some benefits in HD treatment by reducing the risk of various HD-associated complications.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of Toray Industries, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
