Abstract
Introduction:
Patients’ session-to-session variation has been shown to influence outcomes, making critical the monitoring of dialysis dose in each session. The aim of this study was to detect the intra-patient variability of blood single pool Kt/V as measured from pre-post dialysis blood urea and from the online tool Adimea®, which measures the ultraviolet absorbance of spent dialyzate.
Methods:
This open, one-armed, prospective non-interventional study, evaluates patients on bicarbonate hemodialysis or/and on hemodiafiltration. Dialysis was performed with B. Braun Dialog+ machines equipped with Adimea®. In the course of the prospective observation, online monitoring with Adimea® in each session was established without the target warning function being activated. A sample size of 97 patients was estimated.
Results:
A total of 120 patients were enrolled in six centers in China (mean age 51.5 ± 12.2 years, 86.7% males, 24.2% diabetics). All had an AV-fistula. The proportion of patients with blood Kt/V < 1.20 at baseline was 48.3%. During follow-up with Adimea®, the subgroup with Kt/V > 1.20 at baseline remains at the same adequacy level for more than 90% of the patients. Those with a Kt/V < 1.20 at baseline, showed a significant increase of Kt/V to 60% of the patients reaching the adequacy level >1.20. The coefficient of variation for spKt/V as evaluated by Adimea® was 9.6 ± 3.4%, not significantly different from the 9.6 ± 8.6% as blood Kt/V taken at the same time.
Conclusion:
Online monitoring of dialysis dose by Adimea® improves and maintains dialysis adequacy. Implementing online monitoring by Adimea into daily practice moves the quality of dialysis patient care a significant step forward.
Keywords
Introduction
The assessment of dialysis dose is important since extensive evidence has been accumulated on the dependency of survival on the adequacy of treatment. 1 In individual patients, patients’ session-to-session variation 2 has been shown to influence quality of treatment, so making the monitoring of dialysis dose in each session critical. Hemodialysis patient treatment quality is often sub-optimal, 3 with 15% to 20% of all patients receiving an inadequate dialysis dose.4,5 There are many reasons why the planned dialysis dose is not delivered: the prescribed treatment time is not achieved due to cardiovascular and hemodynamic instability or patient refusal. Inadequate blood pump speed is delivered in combination with sub-optimal needle placement, 6 as well as progressive access malfunction. 7 Regarding the dialysis time achieved per session, Martín Rodriguez et al. 8 analyzed all sessions during 2015 in 85 patients. They found that the median deviation between prescribed time with actual delivered time was of 9.01 min (1.1–28 min/session) and 15.5% of the patients showed a reduction higher than 10 min in at least 20% of the sessions during the year.
Most frequently, the lack of delivery of the prescribed treatment time is associated with central venous catheters as vascular access, older age and a higher Charlson comorbidity index. 8 In addition, patient factors such as frailty, non-adherence as well as organizational factors such as inflexibility of the care process may act as barriers. Online monitoring of each session highlights early adjustments needed for the quality of the delivered care. 9 Real-time integration of information technology systems with hemodialysis machines support optimizing the delivered dialysis dose, thereby in all probability improving patient morbidity and mortality. 8
The aim of this study was to detect the intra-patient variability of dialysis dose as estimated from pre-post dialysis blood urea derived single pool Kt/V 10 (blood Kt/V) compared to ultraviolet absorbance as a continuous assessment of spent dialyzate (Adimea® Kt/V). 11 The secondary aim was to examine the feasibility of using Adimea® Kt/V to improve dialysis adequacy as stated by the routine blood Kt/V,12,13 evaluating whether the proportion of patients reaching the treatment goal increased during the course of the study. Many years after the first publication of clinical practice guidelines, 14 the lack of prescribed versus delivered dialysis dose is still not a marginal problem. Online monitoring tools are available but not fully introduced in dialysis practice in all centers to support clinical staff in establishing reliable processes.
As a result, lower levels of dialysis adequacy in terms of weekly treatment time 15 and of dialysis dose15–18 are reported. The present study was conducted in the People’s Republic of China facilities not having previously used online monitoring tools, to raise attention in the routine daily practice for dialysis adequacy.
Patients and methods
This is an open, one-armed, prospective non-interventional study, evaluating patients on bicarbonate hemodialysis or on hemodiafiltration.
Patients had to meet the following inclusion criteria for enrollment: (1) treated on chronic hemodialysis for at least 6 months on 3 sessions/week schedule, (2) arterio-venous fistula as vascular access, (3) documented spKt/V calculated from pre-post-dialysis urea 10 from 1.0 to 1.4 over the last 3 months or (4) average of spKt/V < 1.35 out of three consecutive blood measurements, (5) age ⩾ 18 years, and (6) voluntary participation and written informed consent. Patients with the following conditions were excluded: (1) presence of severe hematologic disorders (e.g. multiple myeloma), (2) life expectancy less than 6 months, (3) treatment with single-needle dialysis, and (4) ongoing Kt/V monitoring by Adimea® (B. Braun Avitum, Melsungen, Germany). In the course of the prospective observation, online monitoring with Adimea® in each session was established without the target warning function of Adimea® being activated. Therefore, in case the operative conditions were not compatible with the adequacy target, the machine displayed the discrepancy, but did not generate an alarm. After informed consent, baseline and retrospective data collection, patients were observed for a period of 6 months prospectively; data from conventional hemodialysis or hemodiafiltration were collected (Figure 1). Adimea® Kt/V, blood Kt/V, blood values, prescription data, and treatment adjustments made during the therapy as well as any adverse events were recorded in an electronic clinical report form. Patients were treated according to prescription by their physician and examined according to their individual status.
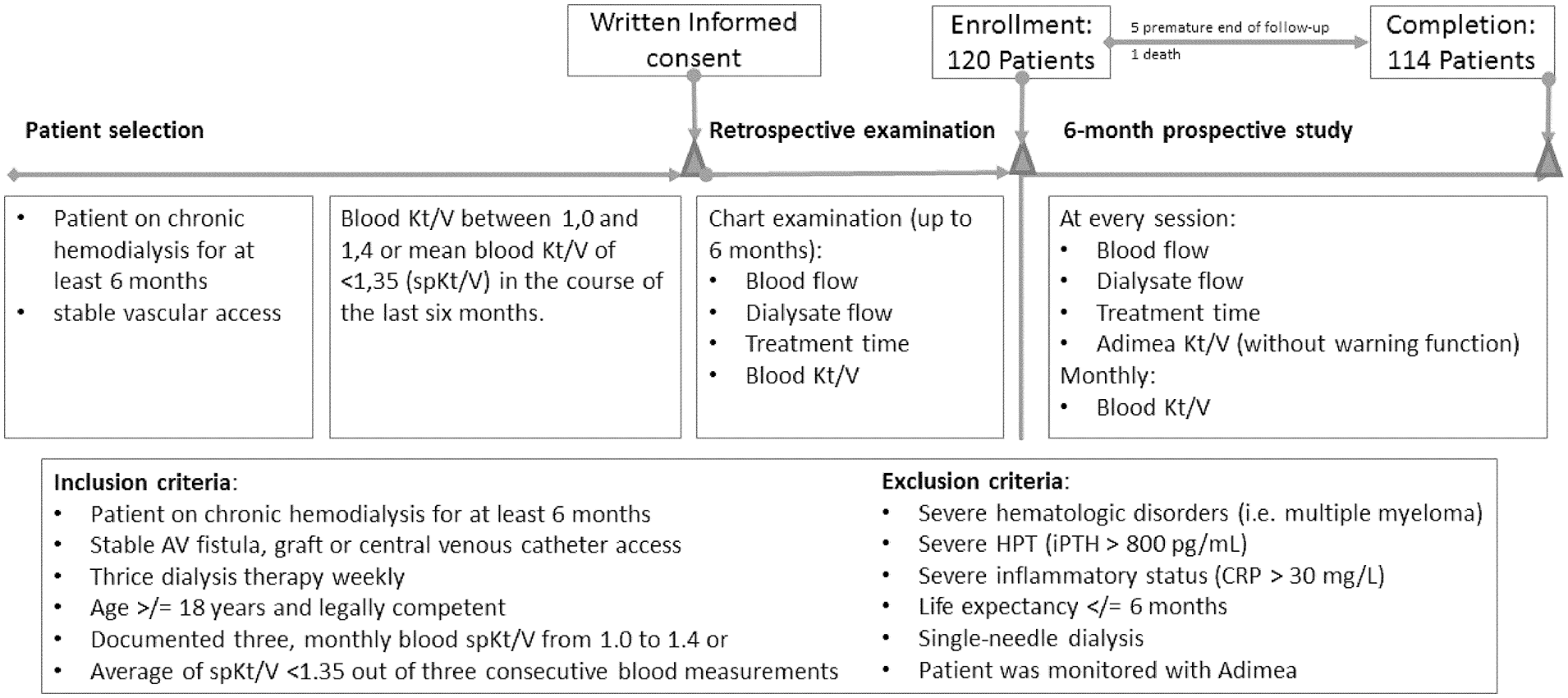
Study flow-chart including selection criteria and study population.
All data captured during the study were obtained from routine clinical care assessments. Dialysis was performed with B. Braun Dialog+ machines equipped with Adimea®. The data collection was strictly anonymous.
Adimea® Kt/V
Adimea® system utilizes the principles of spectroscopy. A light source transmits ultraviolet (UV) light through the dialyzate. Some molecules contained in the dialyzate, which are removed from the plasma during dialysis, absorb the light. This absorption is measured by a sensor. It has been proven, 19 that the UV absorption measurements can be used to determine the dialysis dose as there is a very close linear correlation between the measured UV absorption signal and the urea in the dialyzate. Adimea® has been validated for hemodialysis, hemodiafiltration, and single needle dialysis. 11
Study approval and patient consent
Trial registration was not compulsory because of the observational nature of the study. The protocol was submitted and approved by the Ethical Committee of each participating hospital from April 2, 2013 to March 14, 2014.
All participating patients signed a written informed consent.
Sample size
A sample size of N = 97 patients had 90% power to detect a difference in within-patient standard deviation of 0.01 (assuming a within-patient standard deviation of 0.07 for Adimea® Kt/V measurement and of 0.08 for blood Kt/V) and taking a common standard deviation of the differences of 0.03, using a paired t-test with a 0.05 two-sided significance level. To account for early discontinued patients with less than three monthly blood and Adimea® Kt/V measurements, a total of N = 120 enrolled patients were planned.
This sample size was also considered sufficient to detect a difference of 0.037 between the within-patient mean values of the two procedures, using a paired t-test and assuming an overall standard deviation of 0.13, a power of 80%.
Statistical analysis
Descriptive parameters are presented according to the type of variable evaluated (mean, standard deviation, median, upper and lower quartile for continuous data; counts, absolute and relative frequencies for categorical and ordinal data).
Primary endpoint
The primary endpoint was the session to session variability of Adimea® Kt/V and by blood Kt/V, assessed by the within-patient standard deviation (SD) and the derived coefficient of variability (SD/mean × 100) of the values using all available time points.
The mean of the SDs were calculated and statistically compared using a paired t-test.
In addition to the paired t-test, also 95% confidence intervals (CI) for the mean SD Adimea® and blood Kt/V were calculated.
The level of statistical significance is assumed to be α = 0.05. Statistical analyses were conducted using IBM® SPSS® statistics version 24.
Results
Table 1 reports the characteristics of the 120 patients enrolled in six centers in China. The mean age was 51.5 ± 12.2 years and 86.7% were males. The proportion of diabetics was 24.2% and all of them were with an AV-fistula for vascular access. Stratifying the patients by their monthly baseline blood Kt/V lower or greater/equal than 1.20, the proportion of patients with inadequate dialysis dose <1.20 Kt/V was 48.3%. This group of patients included a significant greater proportion of males (95% vs 79%), patients significantly taller (174 vs 169 cm), and with greater dry body weight (73 vs 62 kg).
Demographics, comorbidities and main baseline lab results of enrolled patients by baseline Kt/V.
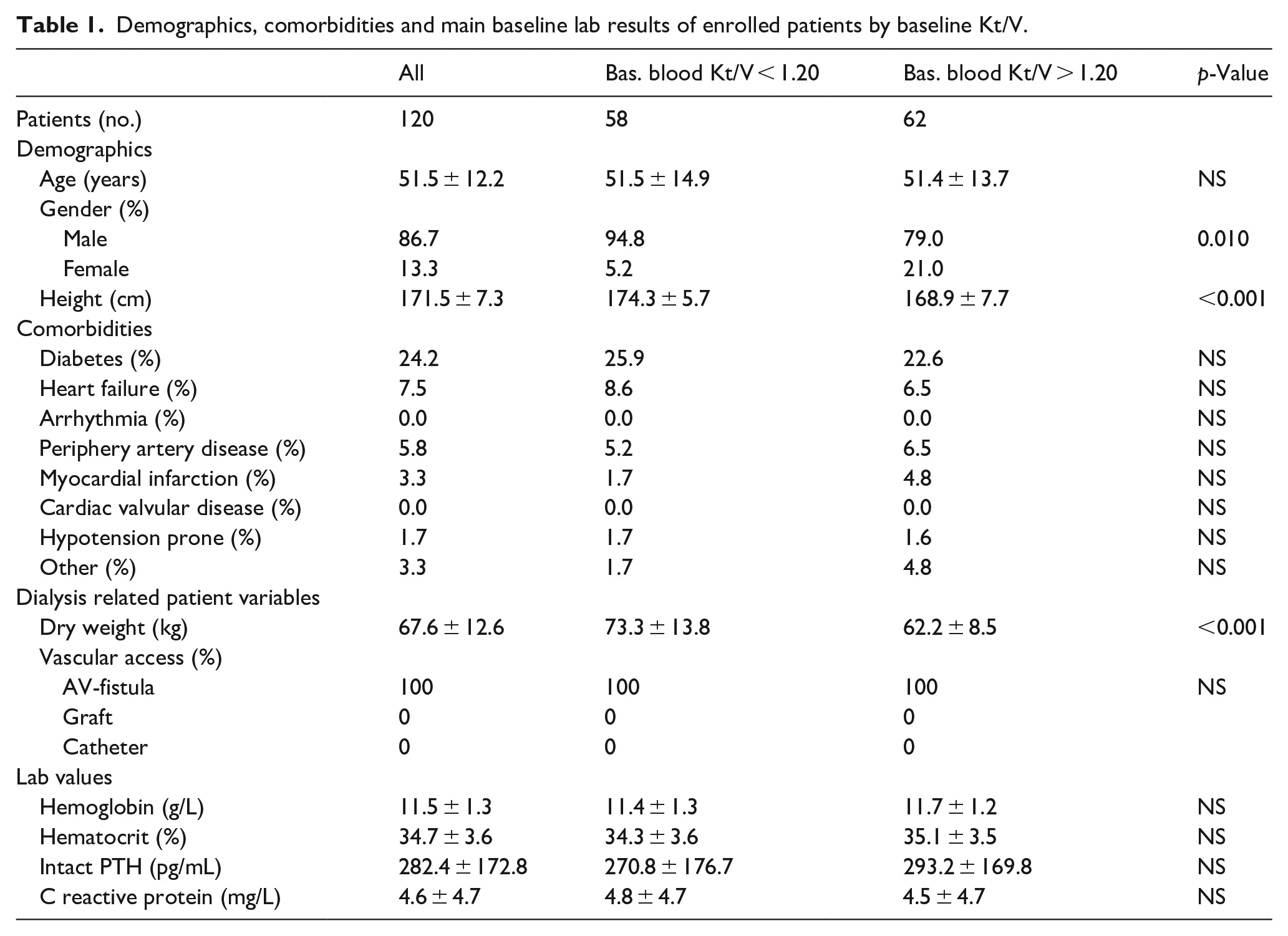
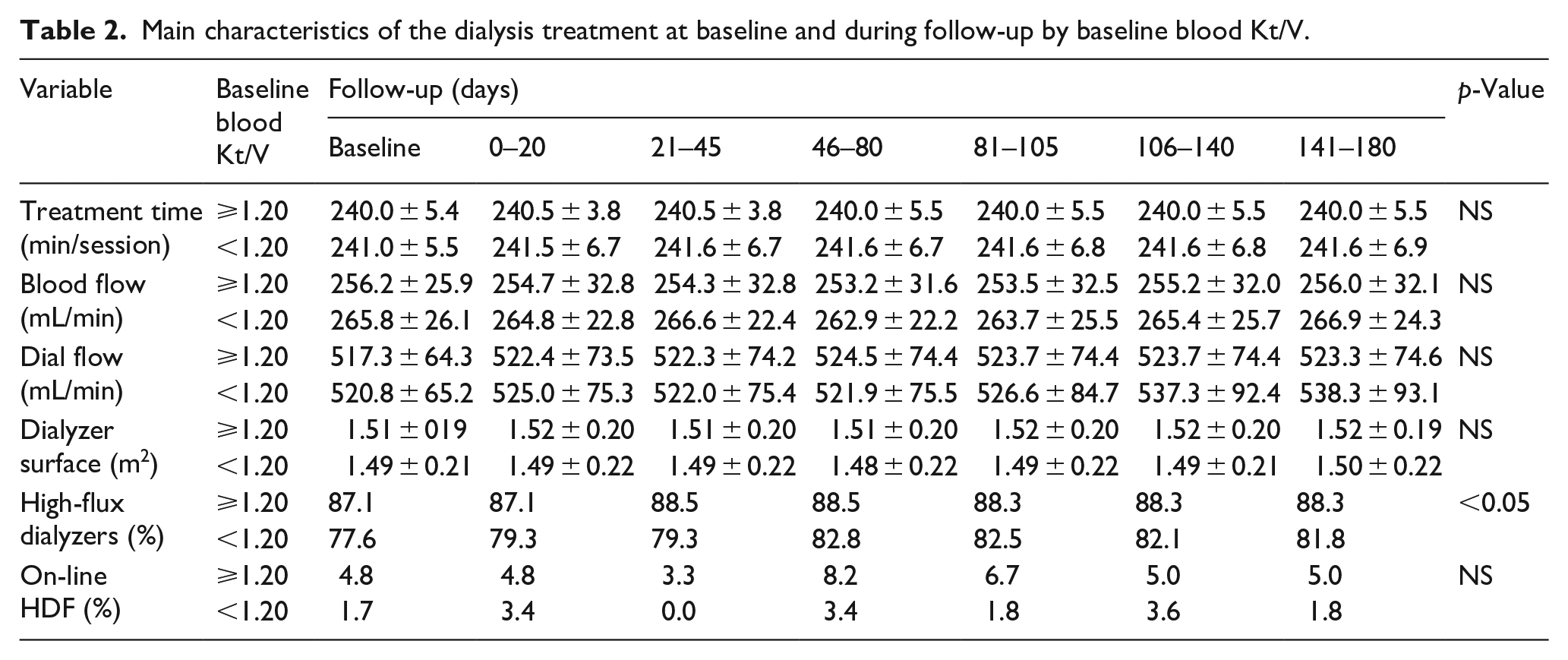
Table 2 reports the main features of the extracorporeal dialysis treatment at baseline and during the 6-month follow-up stratified by baseline blood Kt/V. At baseline, 95.8% of the patients were on 4-h treatment/session and 3.3% were on 4.5 h treatment/session. The mean blood flow was 261 ± 28 mL/min. Dialyzer surface area ranged between 1.20 (17.5%) and 1.80 m 2 (24.2%). About 80.0% of the patients were on high-flux dialysis, 4.2% on on-line hemodiafiltration, with only marginal differences between the two groups stratified by level of dialysis dose (Table 2). Blood Kt/V was 1.21 ± 0.14, with 62/120 having a blood Kt/V ⩾ 1.20 and 58/120 below the target. Figure 2 shows the development of blood Kt/V during follow-up. The group ⩾1.20 at baseline increased on average and remained ⩾1.20 adequacy level for more than 90% of the patients. The other group, with all patients with a blood Kt/V < 1.20 at baseline, showed a significant increase of blood Kt/V in the first 20 days of follow-up with dialysis sessions monitored by Adimea®. As a result, the proportion of patients in the adequacy level ⩾1.20 increased from 0% to about 60% and was maintained for the full follow-up. Means and standard deviations of Adimea® Kt/V during follow-up in patients with baseline blood Kt/V ⩾ 1.20 and those with blood Kt/V < 1.20 are shown in Figure 3.
Main characteristics of the dialysis treatment at baseline and during follow-up by baseline blood Kt/V.
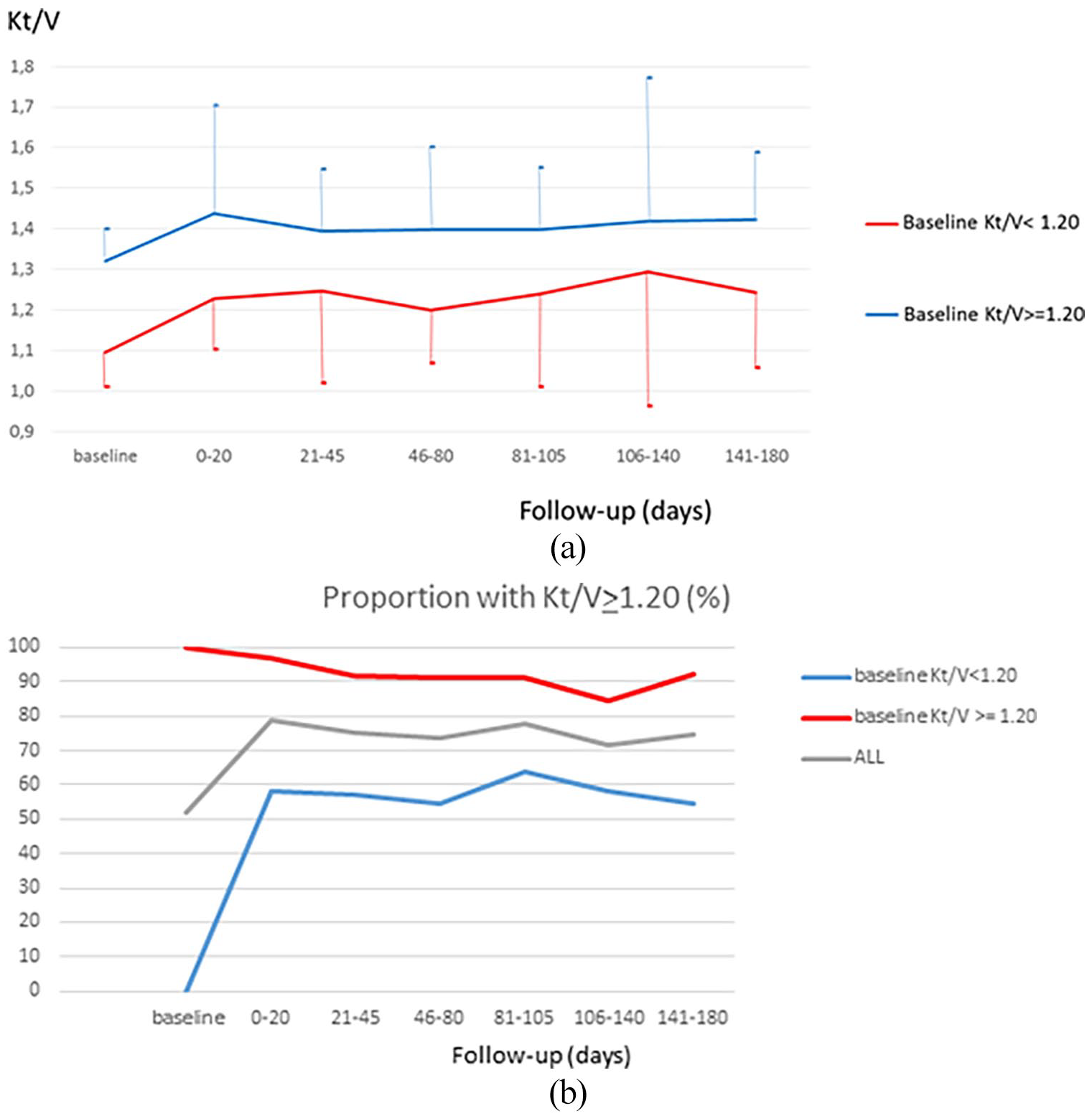
Development of blood Kt/V from baseline (without Adimea® monitoring) and during Adimea® monitoring follow-up by baseline blood Kt/V: (a) mean and standard deviation of blood Kt/V and (b) proportion of patients reaching the blood Kt/V target of 1.20.
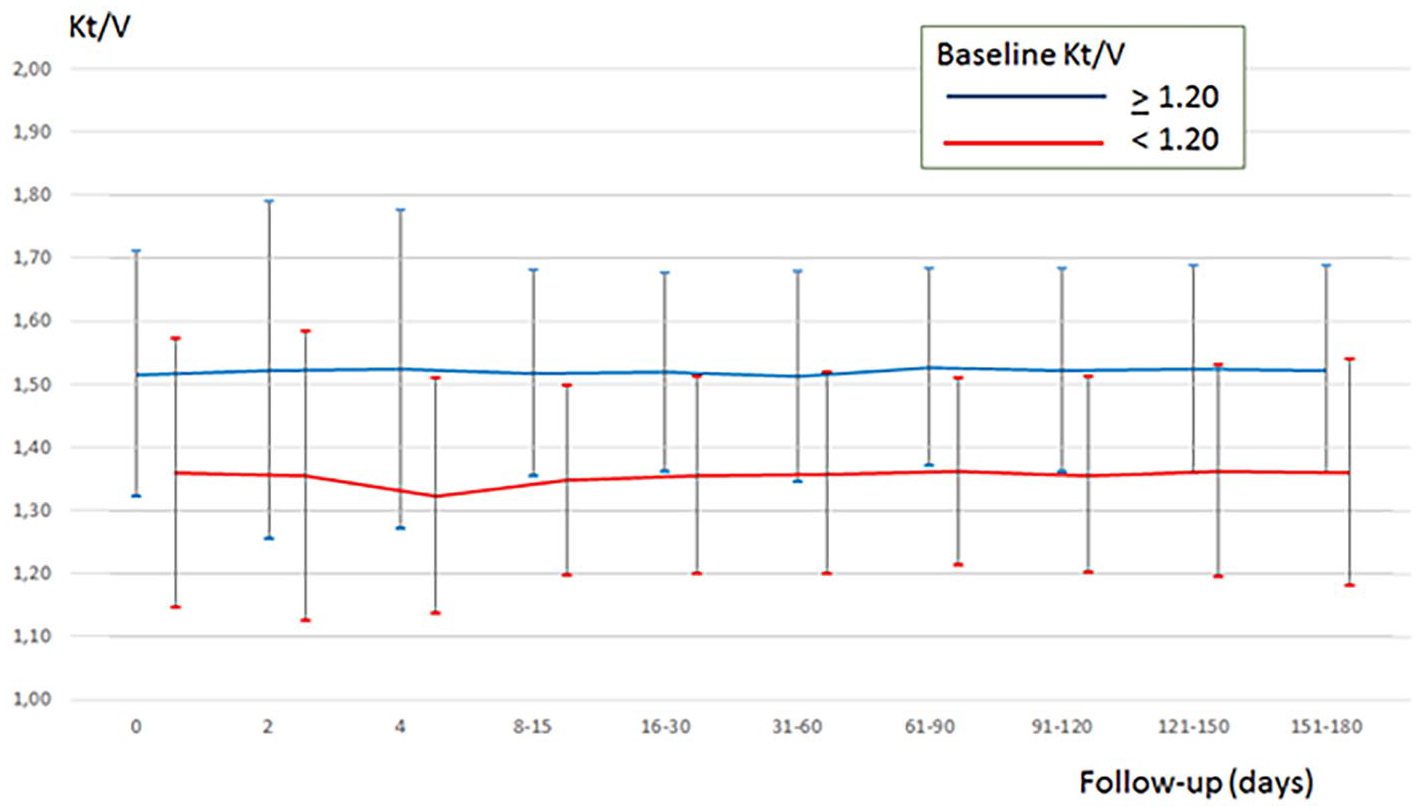
Development of Adimea® Kt/V during Adimea® monitoring follow-up by baseline blood Kt/V.
Figure 4 shows the Bland and Altman graph of those sessions evaluated by Adimea® Kt/V as well as blood Kt/V (see also supplemental material). According to the primary aim of the study, the coefficient of variation (CV) for Adimea® Kt/V was measured and compared to the blood Kt/V. The results of Adimea Kt/V were 9.6 ± 3.4%, not significantly different from the 9.6 ± 8.6% of blood Kt/V. The distribution of the CV of Adimea® Kt/V was very similar to that of blood Kt/V, with most of the patients below 10% of CV (data not shown).
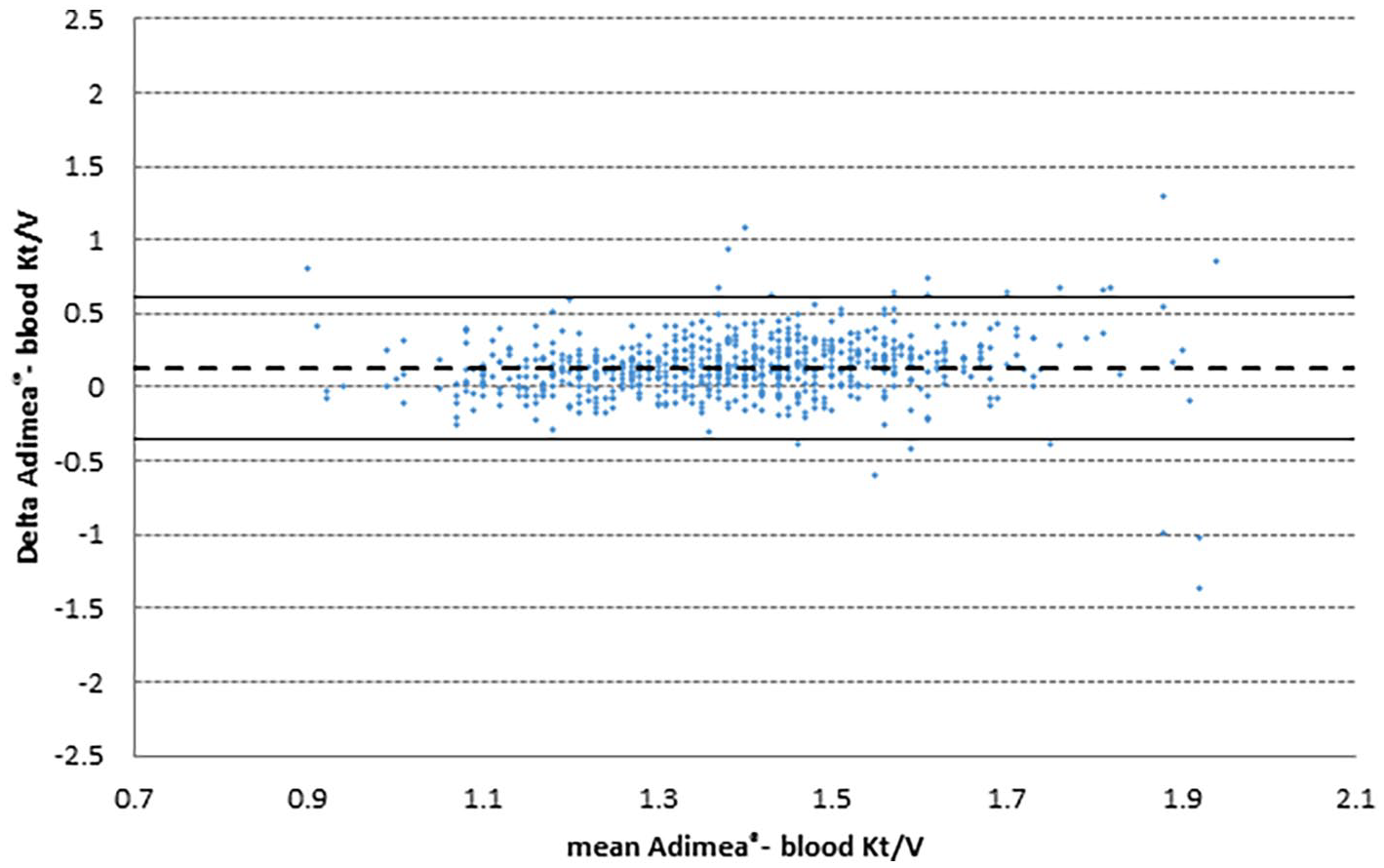
Bland and Altman graphs showing distribution of difference between Adimea® and blood Kt/V in respect of their mean. Lines represent mean (- - -) and 95% confidence interval (-----).
Discussion
This study evaluated the reliability of monitoring the dialysis dose by Adimea® Kt/V in each session and the intra-patient variability of dialysis dose in comparison to the reference method of blood Kt/V, confirming that online monitoring by Adimea® according to Bland and Altman graph (Figure 4) is a reliable method in monitoring dialysis dose and essentially contributes to improvement and maintaining of dialysis adequacy (Figure 2). 20
The main aim of this study was to compare the variability of blood KT/V and online Kt/V dialysis dose by Adimea®. The result is a non-significant difference between the standard deviations of Adimea® and blood Kt/V. In addition, the coefficients of variation were of 9.6 ± 3.4% and 9.6 ± 8.6% by the online and the blood based method, respectively (p = NS). The range is comparable to evaluation by McIntyre et al. in 26 patients with ionic dialysance (13%) and pre-post dialysis urea blood Kt/V (11%) with no statistical difference.
What are the components of intra-patient variability in Kt/V? Dealing with blood Kt/V, the accuracy of the measurement of dialysis dose depends on the accuracy and timing of drawing the sample and laboratory errors. 21 By using Adimea® throughout the full course of the dialysis session it was expected to decrease intra-patient variability, but the results of this study did not confirm the hypothesis. A partial answer to this apparent contradiction is probably the frequency of sampling. The intra-patient variability when dialysis dose is monitored at every session can be higher mainly because of the variability in delivered treatment time and blood flow, as pointed out by Lambie et al. 22 Other factors subject to practice variability (i.e. needle insertion 6 ), needle size), patient related factors (i.e. hemodynamic condition, blood flow rate). Organizational factors in dialysis units (e.g. shift planning 23 ) may affect delivered dialysis dose by indirectly affecting delivered treatment time. Another study, 24 following-up dialysis adequacy of 11 patients on hemodiafiltration with online urea monitoring by an experimental (but reliable) system based on a urease cartridge, 25 found high intra-patient variability, and reported in six stable patients a coefficient of variability of 9.5% and 5.2% for urea generation rate and Kt/V, respectively.
At baseline, in our study a high proportion of patients (48.3%) did not reach the minimum level of dialysis adequacy (according to blood Kt/V ⩾ 1.20). The high proportion of patients with blood Kt/V < 1.20 is likely due to study selection criteria (spKt/V in the range 1.00–1.40 or an average <1.35). Patients not reaching the level of adequacy were more likely to be males, were significantly taller and with greater body weight. However, despite their higher therapy needs in term of treatment time, blood flow and/or dialyzer surface, all dialysis parameters were quite similar to those of the other group adequately treated since baseline. During follow-up 60% of the patients originally with a blood Kt/V < 1.20 (58 out of 120) reached the target level of adequacy. Table 2 shows that the prescription of treatment time, blood flow etc. remained stable with minimal modifications and therefore, other causes might have increased delivered dialysis dose. A variable level of compliance to the prescription of the physician due to several reasons.26–28 Each additional minute of effective treatment time is associated with a significant 3.6% higher probability to achieve the level of dialysis adequacy. 29 Rocco and Burkart 30 described that a loss of 30 min of treatment time translates into an effective delivered Kt/V of 0.88 instead of the originally planned 1.05 according to former guidelines. A monitoring system such as Adimea® is of practical benefit because it allows to uncover those sessions not going to reach the Kt/V target value whilst the session is still running. In fact, the prediction of delivering an inadequate dialysis dose during dialysis enables a modification of the critical treatment parameters, including prolonging the session those few minutes required to fulfill the quality requirements. By showing the prediction graph on the machine screen to the patient, forecasted dialysis dose and implications on the actual operative conditions can be explained to the patient. Accounting for changes in treatment parameters, for example after increasing blood flow, in a few minutes the new target route will be promptly displayed. 31 Therefore, at the beginning of the session, if the initial lower blood flow has not been increased, the system will alarm showing the negative forecast of the final Kt/V. The same situation occurs if the blood flow is decreased during the treatment and not upregulated again, or in case of disturbances due to needle placement or arm position, or the dialyzer is losing performance because of fibers clotting. Even if in this study the alarm option of Adimea® was not activated, the efficacy of the system can be demonstrated by the sustained achievement of dialysis dose during the follow-up. It is interesting to note that Adimea® Kt/V of the sessions selected for blood Kt/V evaluation were not statistically different in respect of those other sessions in the same weekdays (data not shown). Doubts on the representativeness of once a month Kt/V have been raised by several publications.7,32,33 Without on-line monitoring, the testing day is the session nurses and patients know that they are under scrutiny and not surprisingly, this session is very likely to have a high focus. Two main options are commercially available to estimate online dialysis dose. 34 On the base of the equivalence of urea clearance to ionic dialysance, Kt/V has been calculated using the mean dialysance 35 intermittently estimated every 45 min, that is 6 times over 4-h treatment. 36 The ionic dialysance approach relies on the assumption that plasma conductivity remains stable during the measurement period, and actual alterations in plasma conductivity could have a significant impact on the accuracy of the measurement. 36 However, this approach may cause the risk of salt loading due to the spiking of dialyzate sodium up to 155 mEq/L as required for these measurements. 37 Even if on average previous studies did not find any evidence of clinically relevant salt loading during ionic dialysance measurements,38,39 patients with lower predialysis sodium concentration had most net gain during the sessions. 39 The intermittent approach of this method could miss problems occurring between the dialysance tests, such as alarms causing blood pump stop and/or dialysis fluid bypass. With a continuous method as the UV absorbance spent dialyzate monitoring, the immediate recognition of low clearances can trigger prompt interventions correcting or mitigating possible issues arising during treatment. 40 It can be applied not only in double but also in single needle hemodialysis as opposed to the ionic dialysance method. 41
This study has strengths and limitations. The main limitation as a consequence of its observational nature is that the sequence of sessions monitored or not by Adimea® dialysis dose were not randomized. Thus, the benefit is judged on the base of the improvement from baseline. Because of the inclusion criteria of the last three spKt/V evaluations in the range 1.00–1.40 and exclusion of patients with catheters, the study likely selected patients with lower baseline Kt/V variability, thereby decreasing the probability to detect differences in variability between blood Kt/V and Adimea® Kt/V. The enrolled patients, being relatively young and mainly males with AV-Fistula for vascular access, might not fully represent the current population on dialysis in other countries. Considering improved quality of treatment by monitoring dialysis dose in this young population, in elderly frail patients even higher benefit is expected. 42 Older patients are at higher risk for receiving lower dialysis dose, 43 but when adequately dialyzed they have a good quality of life. 44 Given that their variability of dialysis dose is expected to be greater, the real-time transparency on delivered dialysis dose should support nurses to govern those modifiable factors such as treatment time or blood flow during the therapy.
In summary, this study confirms that simple to use integrated clinical tools, 45 that are able to monitor continuously delivered dose of dialysis have an absolute clinical utility, even in a young population with AV fistula. The 60% of this population not reaching Kt/V target at baseline were able to consistently achieve Kt/V adequacy during the study in monitoring naïve centers. Thus, a major component of the success is an improved understanding of the staff and the patients, empowering both groups for correct shared decision making. In frail patients, with vascular instability and frequent episodes of intradialytic hypotension as well as a high proportion of catheters used for vascular access, monitoring is expected to produce even higher benefit.42,46 The dialysis machine using the Adimea tool alarms for potential missing dose targets and follows changes with new target prognosis. An automatic control continuously adjusting factors as blood flow, dialyzate flow and treatment time (within single patient prescribed limits) to ensure the achievement of target dose might be integrated. 47
Supplemental Material
sj-pdf-1-jao-10.1177_03913988211059841 – Supplemental material for Ensuring hemodialysis adequacy by dialysis dose monitoring with UV spectroscopy analysis of spent dialyzate
Supplemental material, sj-pdf-1-jao-10.1177_03913988211059841 for Ensuring hemodialysis adequacy by dialysis dose monitoring with UV spectroscopy analysis of spent dialyzate by Li Zhang, Wenhu Liu, Chuanming Hao, Yani He, Ye Tao, Shiren Sun, Marten Jakob, Daniele Marcelli, Claudia Barth and Xiangmei Chen in The International Journal of Artificial Organs
Footnotes
Acknowledgements
The authors wish to thank Mr. Phillip Orlik for editorial assistance
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CB, DM, and MJ are full time employee of B. Braun Avitum.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
