Abstract
Background:
Adult vitamin B12 (B12) deficiency may present itself with nonspecific mainly neurological symptoms, and thus plasma biomarkers are often judged to be of major importance in the further diagnostic process. Four biomarkers are of special relevance: total B12, holotranscobalamin (the part of B12 bound to the active transport protein, transcobalamin, also named holoTC or active B12) and the 2 so-called metabolic markers that accumulate if B12 is lacking, methylmalonic acid (MMA) and homocysteine.
Objective:
This article briefly reviews the inherent limitation of biomarkers, discusses its use in establishing the diagnosis and cause of B12 deficiency, and when following or discontinuing treatment with B12.
Methods:
The review is based on published papers, but also on knowledge gained from working within the area.
Conclusion:
It is concluded that a combination of a B12 and a metabolic marker, for example, total B12 and MMA, may prove most useful in daily practice. An unexpectedly high level of total B12 is most often of no clinical importance, though sometimes related to the presence of underlying cancer. Measurement of total B12 is of limited value in patients on treatment with pharmacological doses of B12 but may be helpful if B12 treatment is discontinued.
Plain language title
Vitamin B12-Related Blood Tests
Plain language summary
Blood-testing is considered an important part of the diagnostic procedure in patients suspected to suffer from B12 deficiency. A deficiency is supported by a low level of plasma B12, and confirmed by a high level of methylmalonic acid, judged according to age and kidney function. Alternatively, a high level of homocysteine may support the diagnosis. Treatment of B12 deficiency is mainly guided by improvement of symptoms, with a very limited need for further blood testing. If B12-treatment is discontinued, B12 status should be judged every 6 months for approximately 2 years to detect a possible reoccurrence of a deficient state. An unexpected high level of plasma B12 is most often of no clinical implication.
Keywords
Introduction
Biomarkers play an essential role when identifying patients with an impaired vitamin B12 (B12) status. If the patient is anemic, a full hematologic examination should be performed. A macrocytic anemia in a patient not suffering from folate deficiency strongly supports the diagnosis (review). 1
However, B12 deficiency has many faces, and the diagnosis is challenging in non-anemic patients, disclosing a plethora of primarily neurological and/or neuropsychological-related symptoms (review). 1,2 Over time 4 plasma biomarkers, total B12, holotranscobalamin (holoTC, active B12), methylmalonic acid (MMA), and homocysteine (Hcy) have come into play to explore a relationship between the symptoms of the patient and a possible B12 deficiency. 1
This review presents a general introduction to the use of biomarkers followed by a summary concerning each of the 4 B12-related biomarkers. This, as an introduction for discussing the use of B12-related biomarkers to diagnose and follow patients with B12 deficiency. The review is based on published papers and a long-lasting experience within the field.
Biomarkers in Clinical Medicine
A method suitable for measurement of the biomarker in question must be developed and validated. Next, a reference interval must be established including confounding factors such as age, ethnicity, sex, and kidney function. The reference interval traditionally covers 95% of results obtained in a nondiseased population; thus 2.5% of such individuals will have levels below the reference interval. In other words, the diagnostic specificity is 97.5% if the lower limit of the reference interval is employed as a decision limit, and lower if a higher decision limit is chosen (review). 3
The ideal biomarker will have a diagnostic sensitivity of 100%, meaning for example that all suffering from B12 deficiency would have a biomarker value below the lower limit of the reference range or the chosen decision limit. However, biomarkers show overlapping values for individuals with or without the disease in question, and thus choosing a decisions limit with a high diagnostic sensitivity will imply a low diagnostic specificity and vice versa.
Most often the decision limit chosen for total B12 will favor a high diagnostic sensitivity—though still below 100%. This implies that even though a very low level for total B12 may strongly support B12 deficiency, a value close to the decision limit may be of limited value. Likewise, a value above the lower limit of the decision limit may not rule out a deficiency.
The diagnostic stringency may be improved by using several biomarkers, but in relation to B12 deficiency—like in perhaps all other conditions—the diagnosis cannot be based on biomarker values alone. The diagnosis should always be based on an interplay between symptoms, clinical observations, and biomarker values.
Four B12-related Biomarkers
Total B12
Measures of total B12 by microbiological assays were introduced in the mid-1950s 4 and at the time presented a milestone in the diagnosis of B12 deficiency. Later, competitive protein binding assays were introduced, and since the mid-1990s, such assays have been available on most automatic laboratory platforms. 5
Total B12 represents the sum of several molecular forms of B12 bound to haptocorrin (∼3/4 of circulating B12), a protein of unknown function, and transcobalamin (∼1/4 of circulating B12), the protein that ensures cellular internalization of B12. 6
Prior to analysis, the serum/plasma sample is exposed to an extreme pH and to cyanide ions to liberate B12 from its binding proteins and to convert the various forms of the vitamin to cyano-B12. Most current assays can be classified as protein competition assays where plasma B12 and an added B12 derivative compete for binding to the B12 binding protein intrinsic factor. Despite assay similarities, reference intervals differ slightly among the methods. Because of that, a B12 measure should always be judged in relation to the method specific local reference interval or decision limit. 5,7
Initially, the level of circulating total B12 was considered to mirror the body-content of B12. 8 Still, later studies have revealed that results for total B12 measures far from always mirror B12 status, 9,10 and this in turn led to the development of additional biomarkers.
Methylmalonic Acid (MMA) and Homocysteine (Hcy)
MMA and Hcy are often referred to as the metabolic markers of B12 status. 11 B12 is a coenzyme for the conversion of homocysteine to methionine and the metabolism of methyl malonyl-CoA. If the vitamin is lacking, one would therefore expect MMA and Hcy to accumulate in the bloodstream. 1 Methods suitable for routine use were developed during the 1980s employing HPLC/GC/MS methodologies. Such methods are still widely used for the measurement of MMA, 12,13 while assays adaptable for automated laboratory platforms have been developed for Hcy (review). 14
An increased level of MMA proved to be superior to a low total B12 for supporting a diagnosis of B12 deficiency. 11 MMA and Hcy is still considered to be superior to the other 3 biomarkers, provided age and renal function is taken into consideration when judging the result. 15 Significant drawbacks are analytical cost and availability. Analysis of MMA is far more expensive than analysis of total B12, and while total B12 is widely available, analysis of MMA is performed only in a limited number of laboratories.
Hcy is increased not only in B12 deficiency but also in individuals lacking folate or vitamin B6. In addition, it is influenced by a common genetic polymorphism in the MTHFR gene. 14 Despite these limitations, Hcy is still considered a potentially helpful biomarker of B12 status, especially in settings where MMA is unavailable and where the population is folate replete.
Holotranscobalamin (holoTC, active B12)
HoloTC (Holotranscobalamin) represents plasma B12 available for the cells of the body, and because of that, it was speculated to be superior to total B12. This could be tested once reliable methods had been launched early in this century. A methodological advance is that holoTC can be measured without the harsh pretreatment of the samples requested to measure total B12 (review). 16 Today, direct sandwich immunoassays for holoTC are available on automated laboratory platforms, but so far, the assay has yet to outplay the measurement of total B12, except for a few situations. While total B12 declines during pregnancy due to a decline in haptocorrin, holoTC remains stable. 17 Thus, holoTC or MMA rather than total B12 should be used to judge B12 status in pregnant women. In addition, holoTC may be used to judge a patient’s ability to absorb vitamin B12, as demonstrated by employing the CobaSorb test. The test assesses the absorption of B12 by judging the increase in holoTC following a dosing regimen of 9 µg cyano-B12 thrice daily for 2 days as compared to the baseline level. 18
The Combined Indicator of Vitamin B12 Status (cB12)
Within the last decades an algorithm that uses 2 or more of the 4 biomarkers has been proposed under the name of xcB12, where x denotes the number of biomarkers included. 19 4cB12 includes results for all 4 biomarkers (total B12, holoTC, MMA, and Hcy). It is currently considered the best biomarker stratification for B12 status and has proven very useful in the research setting, 20 but its robustness and cost-effectiveness in the clinical setting is yet to be explored.
Use of the four B12-related Biomarkers in the Clinical Setting
Is the Patient B12 Deficient?
The clinical use and cutoff limits of the abovementioned biomarkers differ among countries and laboratories caused by test methodologies, availability, and tradition. Various studies and recommendations have suggested using more than one biomarker, 19 -21 such as total B12 or holoTC combined with MMA or Hcy. Most often, a value for total B12 well below the local reference interval is judged to indicate B12 deficiency. However, in our experience, this should nevertheless be supported by the measurement of MMA before interpretation (see below). A result for total B12 around the lower limit of the reference interval is of limited value, and it is recommended to supplement such a result with a measurement of MMA. A high level of an age-adjusted MMA, considering renal function, further supports B12 deficiency, while a low level makes it less likely. A high level of B12 makes B12 deficiency less likely, but on the bottom line, clinical symptoms should always overrule biochemistry. HoloTC may be used as first-line testing, and in folate-replete populations, Hcy may be used instead of MMA.
The algorithm employed at Aarhus University Hospital, Denmark, for evaluation of adult patients with suspected B12 deficiency is shown in Figure 1. The figure is based on total B12 measured employing Atellica (Siemens), with a reference interval of 200 to 600 pmol/L. 22 The decision limit chosen to indicate the need for further analysis is 250 pmol/L. 23 MMA is measured employing an in-house liquid chromatography–tandem mass spectrometry method, essentially as previously described. 12 The upper limit for the reference interval for adults aged 18 to 65 years is 0.40 µmol/L and 0.46 for adults aged >65 years, 22 and these are also the values used as cutoff points (Figure 1). Notably, the choice is to measure MMA in all patients with total B12 levels below the decision limit of 250 pmol/L. The choice has been made to increase diagnostic accuracy and thereby minimize the risk of unjustified lifelong overtreatment with pharmacological doses of B12.
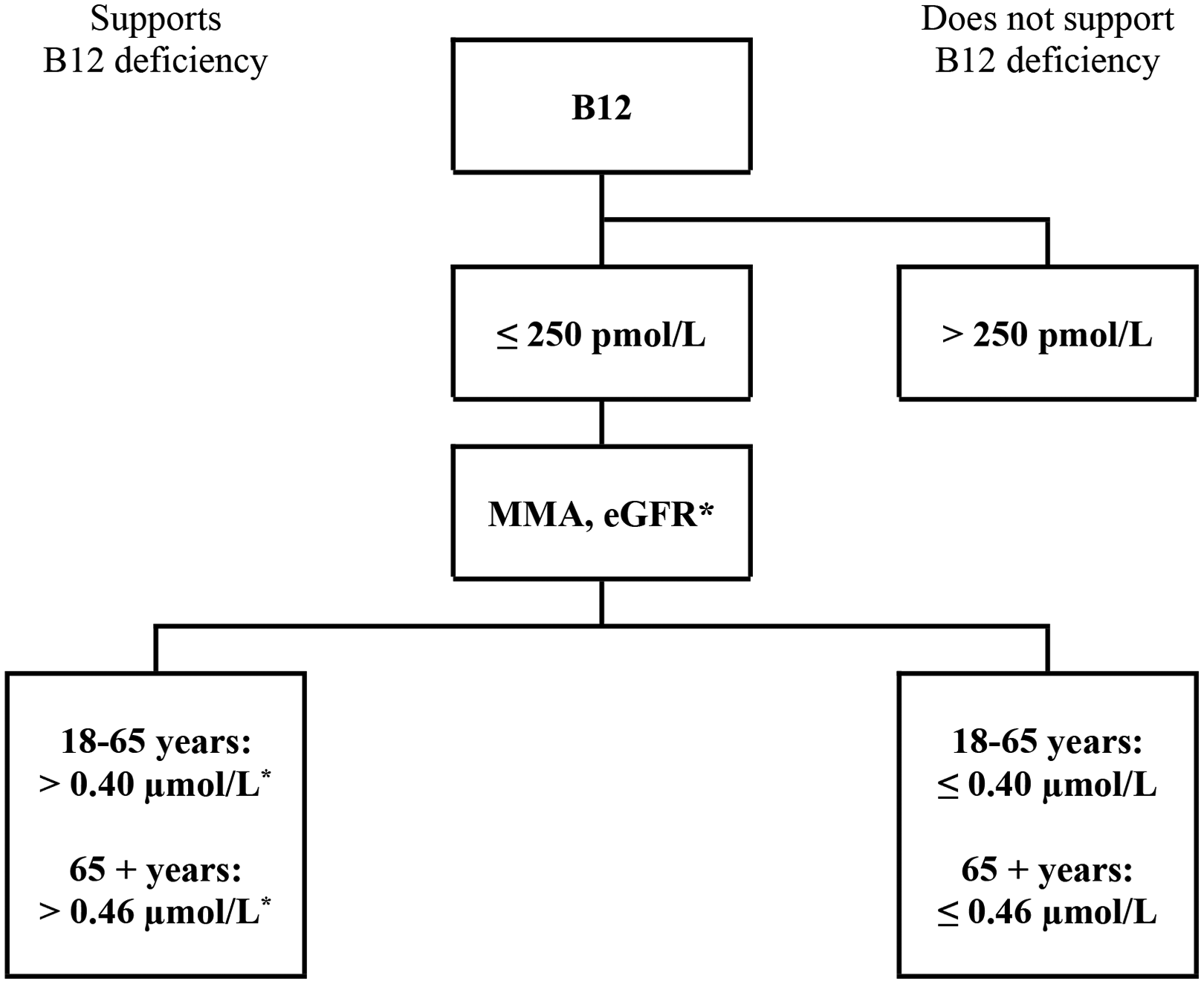
Biomarkers to support B12 deficiency in adult patients. Total B12 and MMA is included in the figure. As noted in the text, holoTC may replace total B12 as a first line test, and homocysteine may be used instead of MMA, notably in folate replete populations. Cutoff limits should be decided based on local reference intervals. The figure is based on current practice at Aarhus University Hospital, Denmark. *For patients with an eGFR below the reference interval (≤60 mL/min), it is noted that an increased level of MMA may be caused also by the impaired kidney function. B12 indicates total plasma B12; MMA, methylmalonic acid; eGFR, estimated glomerular filtration rate. The decision limits are based on studies by Abildgaard et al, 22 Hvas et al, 23 and Nielsen et al. 24 For further details see: https://www.analysefortegnelsen.dk/AnalyselisteZoom.asp?Lok=AUH&Id=AAA00281 (In Danish).
Finally, it must be emphasized that biomarker values are difficult to interpret in patients who have been self-medicating with high-dose B12 before testing, and enquiry concerning this should always be done.
An Unexpectedly High Level of Total B12
Interpreting an unexpectedly high level of total B12 is a challenge (review). 25 Autoantibodies against intrinsic factor have been claimed to cause a spurious high total B12 level due to analytical interference. However, a carefully performed study documents it to be of no importance for routine measurement of total B12, 26 possibly because the autoantibodies are destroyed during the harsh pretreatment of the samples.
Another issue is a suggested presence of an ill-defined high molecular form of B12 (macro-B12). It has been reported that almost 50% of samples with a high level of total B12 contain macro-B12, and that this in turn leads to an incorrect high level for total B12. 27 However, currently, it is unresolved whether this is correct-or caused by nonspecific methodologies used for detection of the so-called macro-B12.
Based on methods that specifically quantify the B12 binding proteins, it can be concluded that an increased level of total B12-not explained by treatment with the vitamin-is caused by an increase in haptocorrin, but sometimes also in transcobalamin (review). 28
An unexpected high level of total B12 is often of no known clinical importance. 28 However, an increased level of total B12 caused by increased levels of haptocorrin or transcobalamin has been related to various types of cancer, notably hematological in origin, but also including liver, lung, and breast tumors. This is mirrored in the risk of a cancer diagnosis within 1 year of finding an increased measurement of total B12. The risk is 11% in patients with a high total B12 (>800 pmol/L), but notably, it is also relatively high, 6.7%, in patients with values covered by the reference interval for total B12 (200-600 pmol/L). 29 It should be stressed that it is the cancer that causes the high level of haptocorrin/transcobalamin and thereby of total B12, and not the opposite. In accord with this, a high intake of B12 is not associated with cancer. 30
Why Is the Patient B12 Deficient?
Clarifying the cause of B12 deficiency is mandatory for choosing the optimal treatment. Patients unable to actively absorb B12 will need a lifelong treatment with pharmacological doses of the vitamin, as is the case in patients with autoimmune gastric atrophy. Such patients lack intrinsic factor, a protein produced in the stomach’s parietal cells, and mandatory for the active uptake of B12. 1 If an autoimmune gastritis is suspected, measuring intrinsic factor and parietal cell autoantibodies may be helpful. 31
Back in time, the Schillings test was widely used to judge the patient’s ability to absorb B12. Schillings test 1 tested the capacity for an active uptake of B12, and Schillings test 2, explored whether an impaired uptake could be improved by the coadministration of intrinsic factor. 32 These tests are no longer available, but the CobaSorb test (explained above) has been suggested to replace the Schillings test 1. 18 A significant limitation for this test is that it needs to be performed before treating the patient with B12.
Biomarkers for Following Treatment Response
Once treatment is initiated, the question is how best to ensure its effectiveness. In the anemic B12 deficient patient, hematological parameters including iron and folate should be tested weekly during the first weeks of treatment. Obviously, a possible iron and/or folate deficiency developing because of the increased blood formation should be treated. 1
Measurement of total B12 is often used to follow patients treated with B12. However, this is more confusing than helpful and cannot be recommended. Patients on treatment with pharmacological doses of B12 are likely to have a high level of total B12, also at the time of the next injection. 18 Despite this, many patients request additional injections to remain symptom-free. 33 These 2 observations have been interpreted to indicate that the measurement of total B12 may be misleading when judging the effect of treatment with B12. We currently lack studies that relate posttreatment biomarker values to clinical response in patients treated with pharmacological doses of B12. Because of that, it is advisable to adjust treatment according to symptoms rather than to treat “biochemistry.” 2
Discontinuation of B12 Treatment
Today, many patients receive B12 injection treatment on a questionable basis. 18 Most likely, the same accounts for other types of high-doses B12 treatments, like oral administration. To diminish unneeded treatment, a discontinuation trial may be warranted in patients with an unclear primary diagnosis and in patients where impaired absorption of B12 is no longer an issue.
Discontinuation of treatment should be done in a manner that ensures early detection of a possible reoccurrence of a B12 deficient status. Obviously, the patient should be asked to report any reoccurring symptoms, and in addition it may be reasonable to request the biomarkers employed to detect B12 deficiency approximately every 6 months for at least 2 years. B12 is lost slowly from the body, 34 and thus it will take time before a possible deficient state will give significant symptoms and/or marked alterations in biomarker values. If no sign of B12 deficiency has emerged after around 2 years, the patient is unlikely to need continuous treatment with B12. 18
Conclusion
Today, biomarkers related to B12 such as total B12, holoTC, MMA, and Hcy represent essential tools, mainly in relation to diagnosing B12 deficiency and less so in relation to treatment control. The combination of the 4 into the so called 4cB12 is currently the best we have regarding a biochemical definition of B12 deficiency, but the combined use of total B12 (alternatively holoTC) and MMA (alternatively Hcy) is considered sufficient in daily practice.
Biomarkers cannot be trusted to give the complete picture of the patient’s condition. They should always be used in combination with the symptoms of the patient and the observations made by the attending physician. On the bottom line, a B12 deficient patient is a patient who improves upon treatment with B12.
Supplemental Material
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721241227114 - Vitamin B12-Related Biomarkers
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721241227114 for Vitamin B12-Related Biomarkers by Ebba Nexo and Tina Parkner in Food and Nutrition Bulletin
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
