Abstract
Background:
The obesity epidemic among women in Africa is a health problem, and many studies attribute it to childbearing. However, most studies of postpartum weight retention (PPWR) occur in high-income countries.
Objective:
Therefore, this review sought to identify the potential factors affecting PPWR among African women.
Methods:
Four databases were searched from January 2000 to December 2020: Medline/PubMed, Google scholar, Ajol research, FreeFullPDF. The quality of included studies was assessed using the Newcastle Ottawa Scale.
Results:
Fifteen studies (5 from west, 4 from south, 3 from east, 2 from central, and 1 from north) were included: 8 cohort and 7 prospective cohort studies. Two studies examined the effect of obesity and weight gain during pregnancy on PPWR, 3 studies assessed the effect of childbirth, 4 examined the effect of breastfeeding, 4 assessed the impact of morbidities such as HIV, and 2 looked at food insecurity. Five studies demonstrated that postpartum weight is due to residual pregnancy weight gain and childbirth weight gain and is accentuated as parity increases (n = 2). Breastfeeding has a controversial effect, while morbidity (n = 4) and food insecurity (n = 4) contributed to weight loss. The variation in weight was also influenced by cultural practices (n = 1), prepregnancy weight (n = 1), and socioeconomic status (n = 1). On all domains, only 3 included studies were of good quality.
Conclusions:
Pregnancy weight gain, childbirth, breastfeeding, morbidity, and food insecurity were associated with PPWR. However, preexisting factors must be considered when developing PPWR modification strategies. In addition, due to the limited number of studies included, robust conclusions cannot be drawn.
Introduction
Obesity is a significant global health challenge. 1 In most low- and middle-income countries, over 50% of women enter pregnancy with a body mass index (BMI) above the recommended range. 2,3 Excessive gestational weight gain in pregnancy is associated with postpartum weight retention (PPWR), 4,5 contributing to long-term obesity risk for women, 6 particularly when they have a high prepregnancy BMI. 7,8 It also increases their risk of gestational diabetes, complications, and mortality in pregnancy 9 -12 and impacts the offspring’s future health and obesity risks. 13,14 On the other hand, PPWR averages 0.5 to 3 kg; however, this is highly variable, with up to 20% of women retaining > 4 kg at 1 year postpartum. 15 A PPWR may occur up to 15 years after the first postpartum year, 16 increasing the risk of preeclampsia, gestational diabetes mellitus, stillbirth, congenital anomalies, and low birth weight during subsequent pregnancies. 17,18 Therefore, PPWR represents a significant economic burden regarding increased health care costs, reduced productivity, and disability rates over time. 19,20
A variety of factors contribute to PPWR, including lifestyle behaviors, psychological, and sociodemographic factors. Beside excessive gestational weight gain as the primary risk factor, 21,22 other factors include high prepregnancy BMI, primiparity, short duration of breastfeeding, high energy intake during and after pregnancy, and low physical activity after pregnancy. 23,24 In addition, Knudsen et al found that dietary glycemic index and glycemic load play a role for excessive gestational weight gain and PPWR, which may be more pronounced among women affected by overweight and obesity. 25,26 Psychological factors such as depression and sleep disorders have also been linked to PPWR, due to hormonal changes, social adjustment, and changes in family structure. 27 -29 Furthermore, researchers have found that dissatisfaction with body image early in the postpartum period (at 3 and 6 months) is related to postpartum weight gain. 30 Finally, there is some evidence that weight retention may be associated with age, 24 marital status, and race. 31
Particularly, Africa has always been portrayed as the continent with the most undernutrition. However, nowadays, sub-Saharan Africa (SSA) face the challenge of an increasing trend of obesity. Indeed, SSA is recognized as having a “double burden” of malnutrition, that is, the coexistence of undernutrition and overnutrition. 32 Eight of the top 20 countries ranked for adult obesity are found on the continent. 33 More than 10% of adult women in 18 African countries are affected by obesity. In West Africa, from 2000 to 2004, 60% of urban women were affected by overweight or obesity. 34 Therefore, African women are more likely to enter pregnancy overweight or obese. Furthermore, they are more likely to remain overweight following pregnancy, with excessive gestational weight gain and PPWR being key risk factors as the society focuses more on child than on the mother. Snacking increased, mothers have less time for sport, poorer body image due to pregnancy, and depression in some women, 35,36 resulting in increased PPWR, a source of metabolic pathologies over time.
To our knowledge, there have not been any systematic review to summarize factors that may contribute to PPWR in Africa, where there is strong evidence of a disproportionate burden of obesity and weight-related complications associated with pregnancy. Considering the prevalence of infectious and noninfectious diseases in poor environments characterized by ill-health systems, a clearer understanding of factors associated with PPWR in African women is very necessary. Therefore, the present review aims to synthetize the evidence concerning postpartum weight change factors among women in Africa over the past 20 years. This review is anticipated to identify determinants that must be considered in managing postpartum weight, ensuring that public health problems associated with overweight are reduced. The evidence summary will be useful for researchers, health care practitioners as well as intervention and health promotion designers.
Methods
The review followed standard systematic review methods and the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA). 37 This study did not require approval from the Internal Review Board because it used data from published studies.
Search Strategy
We searched the following databases from January 2000 through December 2020: MEDLINE, PUBMED, Google scholar, Ajol research, and FreeFullPDF. The searches were conducted in February 2021 on carefully selected keywords and terms. After repeated attempts and adjustments, a final search strategy was built, as shown in Figure 1. In addition, separate searches were conducted for specific African regions: West Africa, East Africa, Central Africa, Southern Africa, or Maghreb.

Search Strategy for identification of studies.
Inclusion and Exclusion Criteria
For inclusion, studies needed to (i) report postpartum weight in women in Africa; (ii) be published in English or French; (iii) be quasi-experimental, randomized controlled trials, or cohort studies; and (iv) present original research findings. However, studies focused only on infant postpartum weight or placental retention were excluded.
Study Selection
A study eligibility form was used to screen studies for inclusion. First, 2 reviewers (JK and JL) independently assessed each citation’s title and abstract for eligibility. After that, the full texts of eligible studies were independently reviewed to determine each study’s eligibility. Where there were uncertainties regarding eligibility, all reviewers participated in the decision.
Data Extraction
Using a modified Cochrane Collaboration’s data extraction form, 38 2 reviewers independently extracted data from eligible studies. Data were extracted on study setting, design, participants’ characteristics, interventions, controls, and duration of follow-up. Study sample size, age, and data collection methods were also recorded (Tables 1 –5).
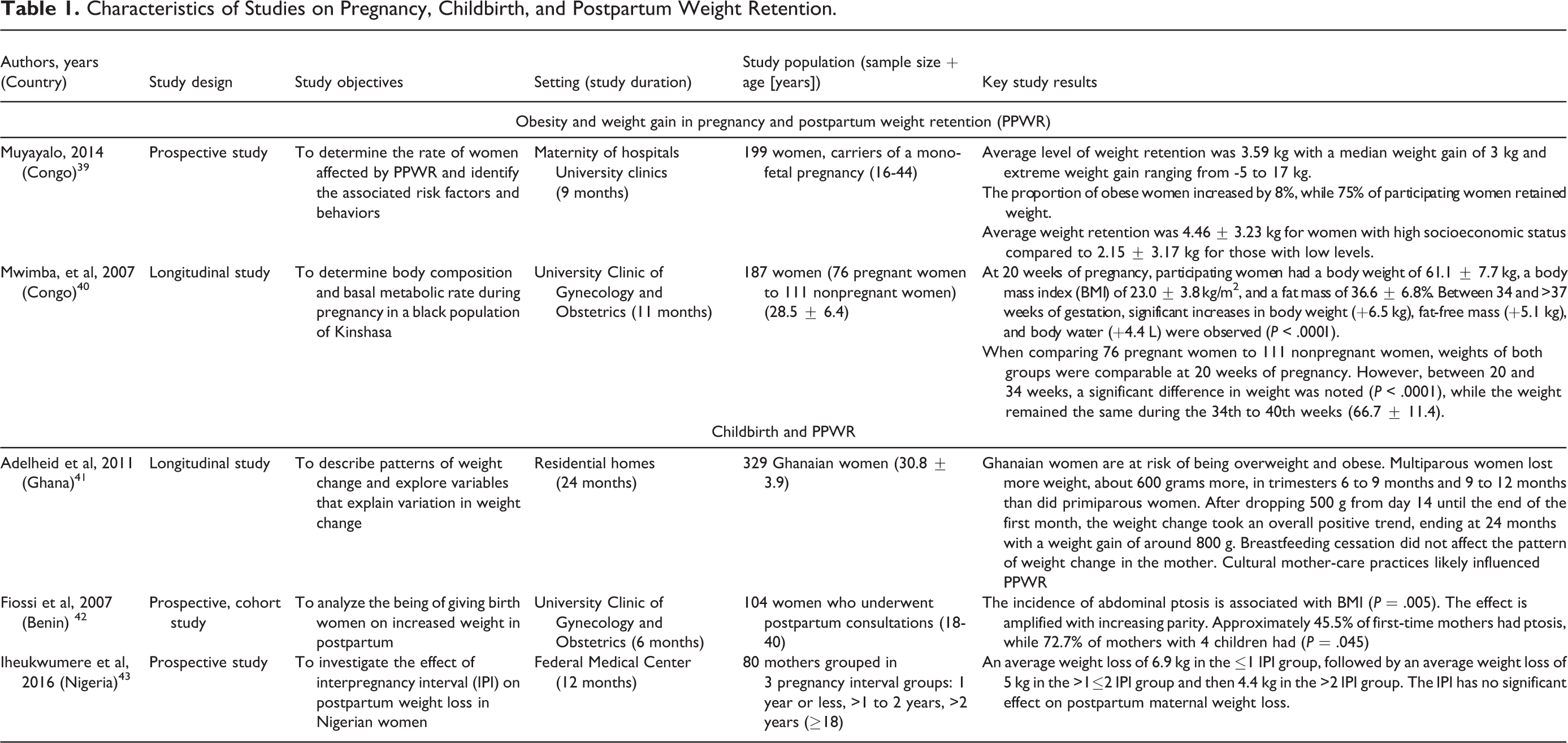
Characteristics of Studies on Pregnancy, Childbirth, and Postpartum Weight Retention.
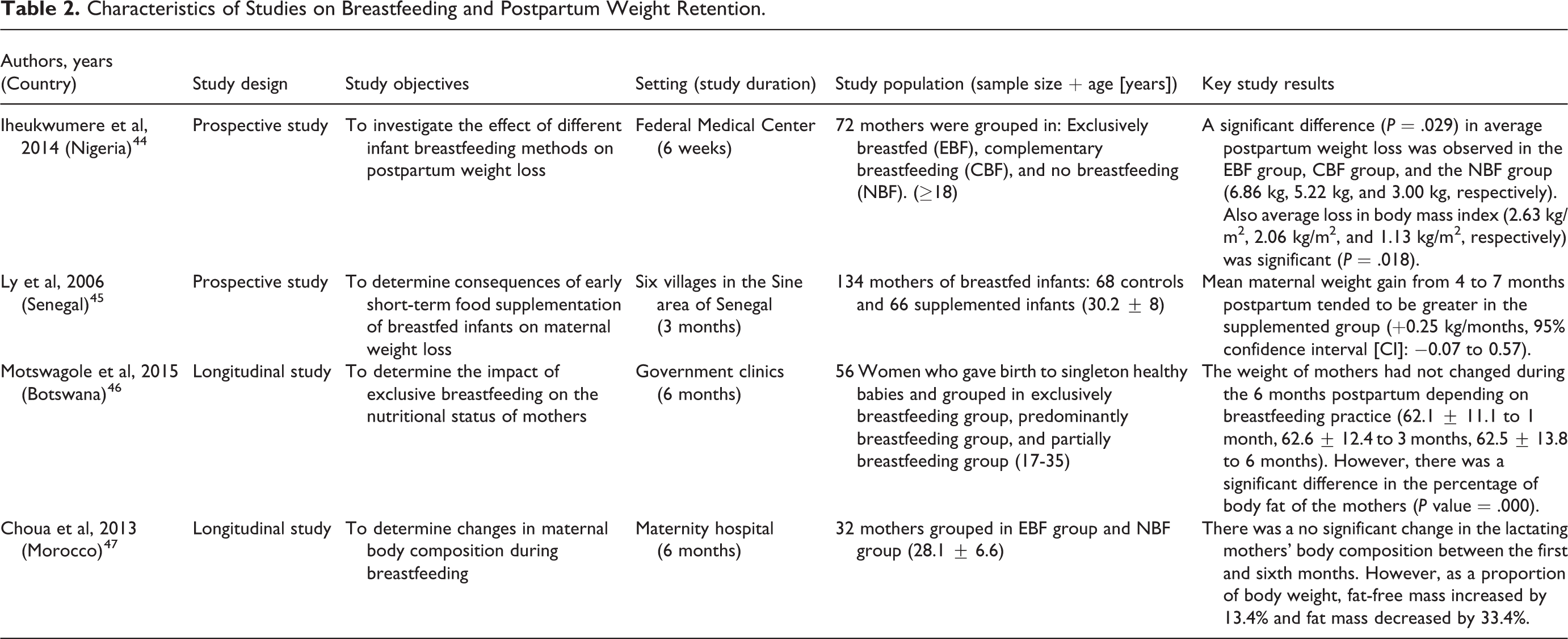
Characteristics of Studies on Breastfeeding and Postpartum Weight Retention.
Characteristics of Studies on HIV and Postpartum Weight Retention.

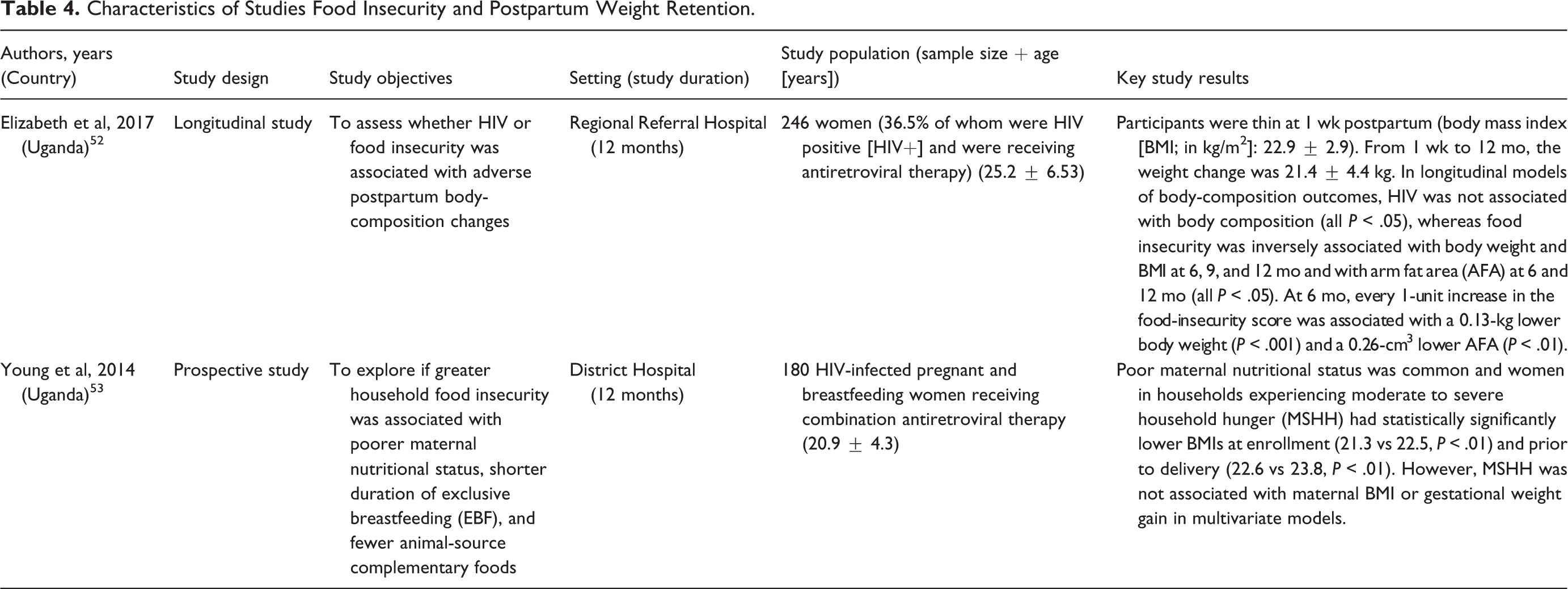
Characteristics of Studies Food Insecurity and Postpartum Weight Retention.
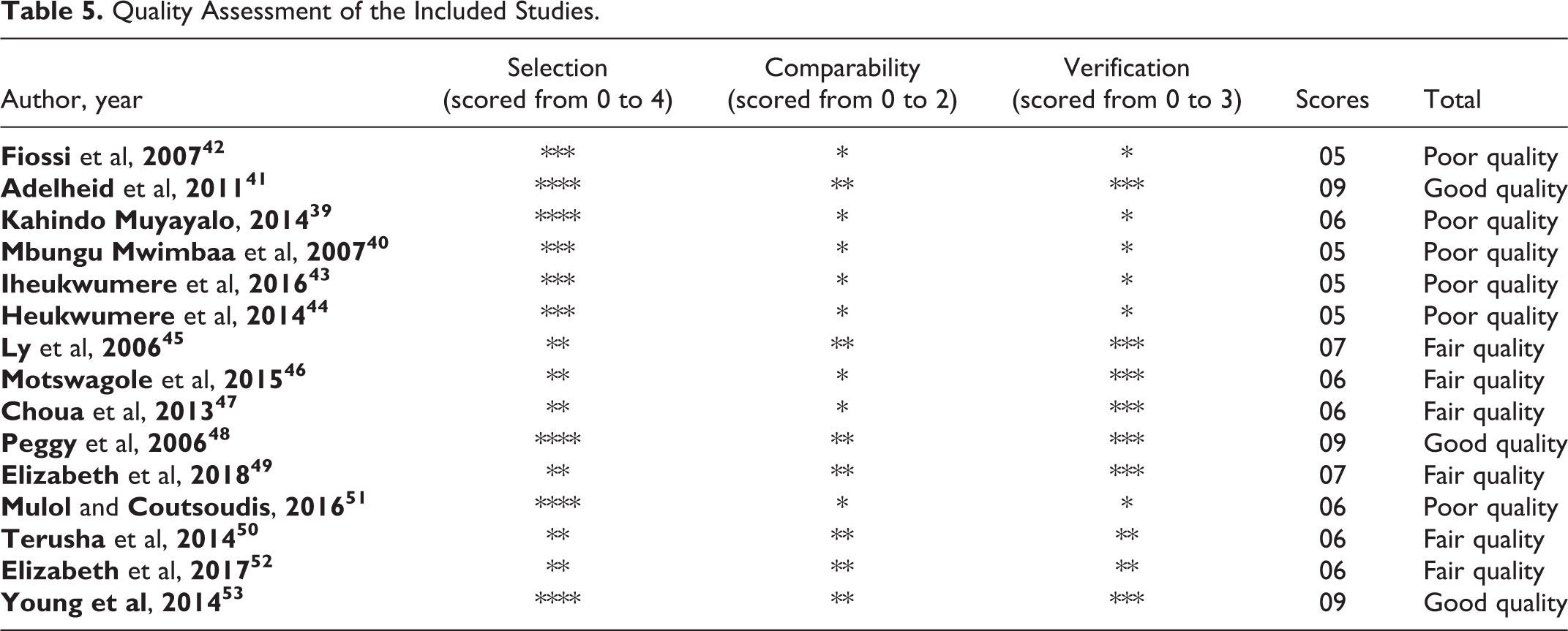
Quality Assessment of the Included Studies.
Quality Assessment
Two reviewers assessed the quality of included studies using the procedures described by Li et al. 54 Specifically, cohort studies were assessed using the Newcastle Ottawa Scale. We evaluated the representativeness of the exposed cohort in the study setting, the selection of the nonexposed cohort, the determination of exposure, demonstrating the absence of the outcome of interest at the start of the study, the comparability of cohorts based on design and analysis, and the adequacy of outcome assessment and follow-up. The thresholds for converting Newcastle-Ottawa scales to Agency for Healthcare Research and Quality standards were good, fair, and poor. 55 To be considered good quality, the study should have 3 or 4 stars for the selected domain, 1 or 2 stars for the comparability domain, and 2 or 3 stars for the outcome/exposure domain. Studies of fair quality scored 2 stars in the selection domain AND 1 or 2 stars in the comparability domain AND 2 or 3 stars in the outcome/exposure domain. The study was rated poor quality with 0 or 1 star in the selection domain OR 0 stars in the comparability domain OR 0 or 1 stars in the outcome/exposure domain. When necessary, disagreements between reviewers were resolved by discussion and consensus, with arbitration by a third reviewer.
Data Analysis
We did not perform statistical meta-analyses because very few studies qualified for inclusion. In addition, the included studies had marked heterogeneity in their design and methodology, and most did not provide appropriate statistical data for meta-analysis. Thus, we summarized, compared, and contrasted the extracted data from 15 eligible studies.
Results
The search identified 143 titles, whose screening and abstracts led to 29 potentially eligible studies. After a full-text review, 14 articles were excluded for not meeting the inclusion criteria, leaving 15 for this review (Figure 2).
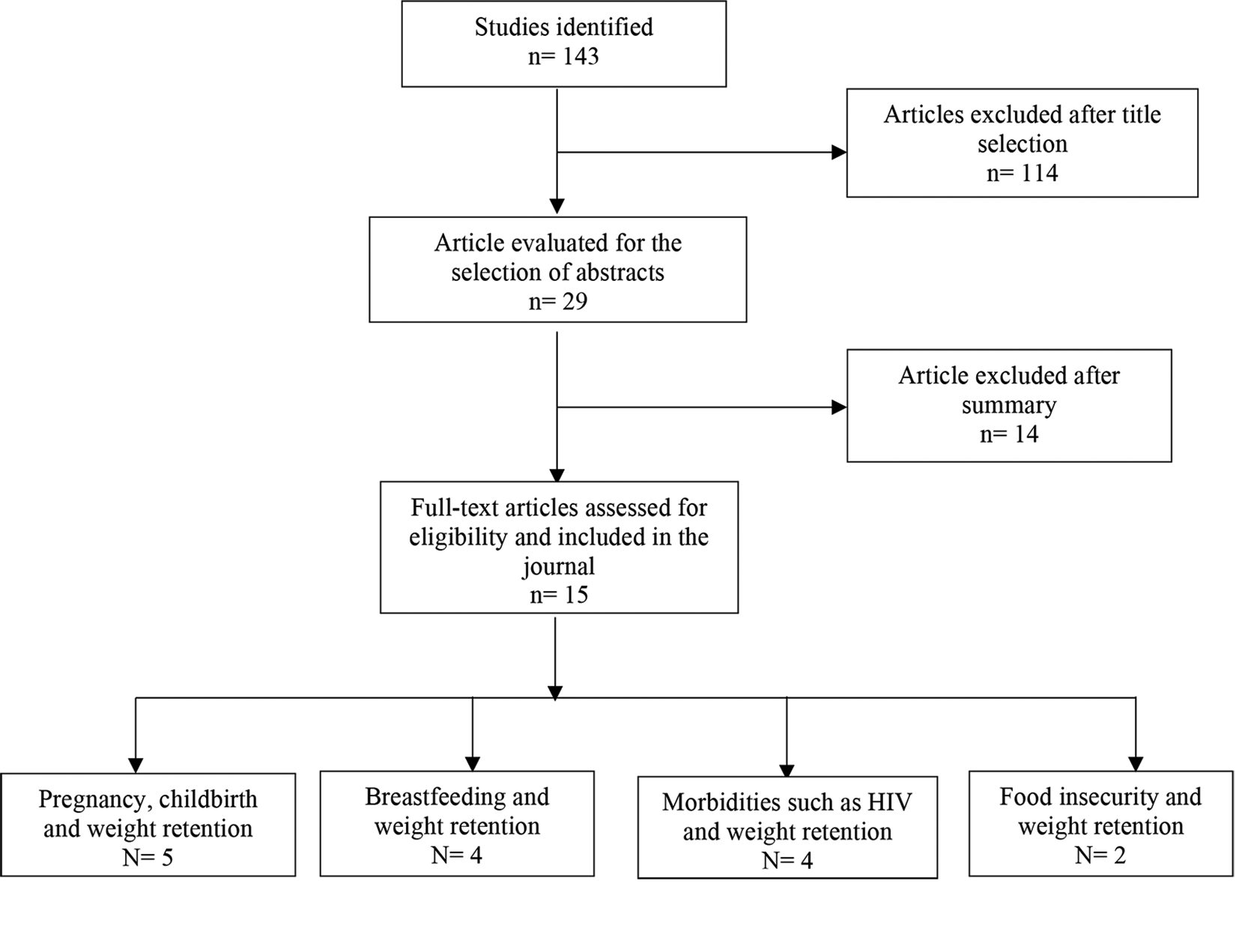
PRISMA flow chart of search strategy and selection process.
Sample Characteristics
As shown in Tables 1 –5, the included studies were published in French and English between 2006 and 2018. One (1) study was conducted in Benin, 2 in the Congo, 2 in Nigeria, 1 in Ghana, 1 in Senegal, 1 in Kenya, 1 in Botswana, 1 in Morocco, 2 in Uganda, and 3 in South Africa. A total of 8 cohort, and 7 prospective cohort studies were included. Nine of the 15 studies were conducted in medical centers, 3 at the University clinic, 2 in rural settings, and 1 in a residential home. Sample characteristics showed significant heterogeneity: size (32-2340 women), age (15-45 years), and follow-up period (6-28 months). Finally, 2 studies examined the relationship between PPWR, obesity, and weight gain in pregnancy, 3 examined childbirth, 4 looked at breastfeeding, 4 focused on morbidities, namely HIV, and 2 examined food insecurity.
Obesity and Weight Gain in Pregnancy and PPWR
Two studies examined obesity and weight gain in pregnancy and postpartum (Table 1). In the study of Muyayalo, 199 women carrying mono-fetal pregnancies were recruited during antenatal care (starting no later than 20 weeks after amenorrhea) and followed until 6 weeks after delivery. 39 Pregnancy weight gain (P < .001) and socioeconomic level (P < .05) were the main factors associated with weight retention 6 weeks postpartum. An average PPWR of 3.59 kg was reported, a median weight gain of 3 kg, and extreme weight gains ranged from 5 to 17 kg. The proportion of women affected by obesity increased by 8%, while 75% of participating women retained weight. Finally, the average weight retention was 4.46 ± 3.23 kg for women with high socioeconomic status compared to 2.15 ± 3.17 kg for those with low levels.
Similar results were found in a longitudinal study by Mwimba et al in Congo involving 76 black women in Kinshasa. 40 At 20 weeks of pregnancy, the participating women had a body weight of 61.1 ± 7.7 kg, a body mass index of 23.0 ± 3.8 kg/m2, and a fat mass of 36.6% ± 6.8%. Between 34 and > 37 weeks of gestation, significant increases in body weight (+ 6.5 kg), fat-free mass (FFM) (+5.1 kg), and body water (+ 4.4 L) were observed (P < .0001). When comparing 76 pregnant women to 111 nonpregnant women, the authors found that the weights of these 2 groups were comparable at 20 weeks of pregnancy. However, between 20 and 34 weeks, a significant difference in weight was noted (P < .0001), while the weight remained the same during the 34th to 40th weeks (66.7 ± 11.4).
Childbirth and Postpartum Weight Retention
Three studies examined the relationship between childbirth and PPWR. Onyango et al described weight change patterns in the 6 sites of the World Health Organization Multicentre Growth Reference Study and explored variables that explain variation in weight change within and between sites. 41 The authors found that, on average, all groups of mothers gained weight in Ghana. They had the highest median BMI of over 27 kg/m2 in the postpartum period compared to Brazilian, Indian, Norwegian, and United States mothers. However, after losing 0.5 kg from day 14 to the end of the first month, the weight change took on an overall positive trend ending at 24 months with a net gain of 0.8 kg. The overall tendency was for the normal BMI group to gain double or triple the weight gained by the other groups. The authors suggested that cultural practices related to maternal care affect PPWR. Looking at the influence of parity on weight loss, in the first half of infancy, primiparas had higher weight losses than multiparas; however, the opposite applied in 6 to 9 months and 9 to 12 months, where Ghanaian multiparas lost about 0.6 kg more than their primiparous counterparts.
Similarly, Fiossi-Kpadonou et al study found that parity was associated with higher PPWR. 42 Of 104 women who underwent postpartum consultations, the abdominal ptosis rate was 45.5% among first-time mothers with a BMI between 20 and 29 kg/m2 and 72.7% among multiparous mothers whose BMI was between 20 and 40 kg/m2 (P = .024). The authors also explained early PPWR (after delivery) as residual pregnancy weight gain. However, it is unclear whether the interpregnancy interval (IPI) length affects PPWR. A prospective study of Nigerian women found an average weight loss of 6.9 kg in the ≤ 1 IPI group, followed by an average weight loss of 5 kg in the > 1≤ 2 IPI group and then 4.4 kg in the > 2 IPI group. However, there was no significant difference in weight loss among the 3 groups. 43
Breastfeeding and Postpartum Weight Retention
Four studies investigated the relationship between breastfeeding and PPWR (Table 2). In the study of Iheukwumere et al in Nigeria, 72 pregnant women in labor were recruited, and anthropometric measurements were taken 24 hours after delivery and then repeated 6 weeks after. 44 Mothers were grouped under the following: exclusively breastfed group (EBF), complementary breastfeeding (CBF), and no breastfeeding (NBF) group. A significant difference (P = .029) in average postpartum weight loss was observed in the EBF group, CBF group, and the NBF group (6.86 kg, 5.22 kg, and 3.00 kg, respectively). This study indicates that reduction in maternal postpartum weight was associated with breastfeeding even on a short-term basis, especially if practiced exclusively.
However, in Senegal, where Ly et al used a randomized intervention cohort study to determine the consequences of early short-term food supplementation of breastfed infants on maternal weight loss. 45 Supplementation with high-energy, nutrient-dense food from 4 to 7 months of age, twice daily under the supervision of field workers. Both controls and supplemented infants were free to eat other complementary foods. The authors found that the mean maternal weight gain from 4 to 7 months postpartum tended to be greater in the supplemented group (+ 0.25 kg/month, 95% confidence interval [CI]: 0.07-0.57). They concluded that early short-term infant supplementation tended to decrease maternal postpartum weight loss, but it increased, rather than shortened, the duration of breastfeeding.
Contrary to both studies, in 2015, Motswagole et al assessed the effects of exclusive breastfeeding on the nutritional status of mothers in Botswana. 46 Breastfeeding exclusivities were determined by the deuterium-oxide dose-to-mother technique, and nutritional status was assessed using anthropometric measures (height and weight) in 56 mother–infant pairs at 6 weeks and again at 3 and 6 months postpartum. The authors found that participating mothers’ mean BMI was within the healthy range of 23.9 kg/m2. However, there was a significant difference in the mothers’ body fat percentage during the 6-month follow-up (P value < .001). The mean FFM decreased from 45.5 kg at 6 weeks to 42.9 kg at 6 months, while the body fat percentage increased from 24.8% at 6 weeks to 28.3% at 6 months. In addition, they found that mothers overestimated exclusive breastfeeding rates at all of the time points. According to the deuterium-oxide dose-to-mother technique, at 3 months, 61% of the mothers were exclusively breastfeeding, 13% were predominantly breastfeeding, and 27% were partially breastfeeding. At 6 months, none of the mothers were exclusively breastfeeding.
Similarly, in 2013, a Moroccan longitudinal study involving 32 mother–baby pairs examined the changes in body composition after pregnancy. 47 The women were recruited by a pediatrician at a maternity hospital in Rabat, Morocco, before giving birth during the third prenatal visit (38th week of pregnancy). After delivery, Deuterium oxide doses were applied to mothers, and measurements regarding breast milk consumption and body composition measures were made at the first, third, and sixth months. Mothers’ mean BMI was 26.2 ± 4.5 kg/m2 at the first month and 25.1 ± 3.4 kg/m2 at 6 months. There was no significant change in BMI during the 6 months of the study. However, there was a significant change in body composition between the first and sixth months. There was 13.4% increase in FFM, (P = .003) and 33.4% decrease in fat mass (P = .0001), this decrease was significant (P = .04) from the third month postpartum. Results also showed that the exclusive breastfeeding rate was 33.3% at the first month, 26.7% at the third, and 12.5% at the sixth, and in exclusively breastfeeding mothers, fat mass decreased significantly (P = .04) from the third month.
HIV Status and Postpartum Weight Retention
A study published in the Journal of Acquired Immune Deficiency in 2006 42 measured the body composition of South African lactating women in relation to HIV status (Table 3). Fat-free mass and fat mass (FM) using bioimpedance spectrometry and anthropometric measurements were obtained at 8 and 24 weeks postpartum in 92 HIV-infected (HIVpos) and 50 HIV-uninfected (HIVneg) lactating mothers. At 8 weeks, HIVpos and HIVneg mothers were not significantly different in weight (62.7 vs 63.9 kg). However, between 8 and 24 weeks, HIVneg mothers had an average weight loss of 1.4 kg, unlike a weight gain of 0.4 kg in the HIVpos mothers (P < .01). There were no significant group differences with regard to change in FFM (0.3 vs 0.1 kg; P = .9) and fat mass (21.5 vs 20.3 kg; P = .2).
Elizabeth et al found similar results in Kenya, where they evaluated the role of HIV-infection on body composition during pregnancy and lactation. A cohort of pregnant women (n = 333; 50.5% HIV+, receiving antiretroviral therapy [ART]) were enrolled at 7 clinics in western Kenya. Two prenatal (mean ± standard deviation: 23.6 ± 4.4 and 33.4 ± 2.0 weeks gestation) and 3 postpartum (6, 14, and 36 weeks) measurements included height, weight, FM, and FFM by bioimpedance analysis. In longitudinal models, the authors found that HIV-infected women had lower weight (ß = −3.0 kg, P = .003), fat mass (ß = −1.5 kg, P = .02), FFM (ß = −1.5 kg, P = .01).
However, in another cohort study 38 conducted in South Africa where 2340 HIV-infected and uninfected women were followed from birth to 24 months of infant age to determine the effect of infant feeding practices on weight change postpartum. Women with HIV (−0.2 kg CI: −1.7 to 1.3 kg; P = .81) and uninfected women (−0.5 kg; 95% CI: −2.1 to 1.2 kg; P = .58) experienced a similar nonsignificant weight loss over the 24-month period. The authors concluded that weight change postpartum was not associated with 5-month breastfeeding modality among HIV-infected and -uninfected women. Similarly, in the study of Mulol et al, the breastmilk volumes were high in HIV-infected mothers: 831 ± 185 g/day at 6 weeks, 679 ± 281 g/day at 9 months, and 755 ± 287 g/day at 12 months. However, these high output volumes had no negative impact on the mother’s FFM. 44
Food Insecurity and Postpartum Weight Retention
Elizabeth et al assessed whether HIV or food insecurity was associated with adverse postpartum body-composition changes in Ugandan women. 52 A cohort of 246 women with 56.5% seropositive and receiving ART were followed up to 12 months after childbirth. HIV was not associated with body composition (all P > .05), while food insecurity was inversely associated with body weight and BMI at 6, 9, and 12 months and arm fat area at 6 and 12 months (P < .05). At 6 months, each unit increase in food insecurity score was associated with a lower body weight of 0.13 kg (P < .001). Body-composition changes are minimal during lactation. HIV is not associated with body composition; however, food insecurity is associated with changes in body composition during lactation (Table 4).
However, in Young et al study, 53 where the authors explored if greater household food insecurity was associated with poorer maternal nutritional status, shorter duration of EBF and fewer animal-source complementary foods. 53 Poor maternal nutritional status was common and women in households experiencing moderate to severe household hunger (MSHH) had statistically significantly lower BMIs at enrollment (21.3 vs 22.5, P < .01) and prior to delivery (22.6 vs 23.8, P < .01). However, MSHH was not associated with maternal BMI or gestational weight gain in multivariate models. MSHH was not associated with EBF at 6 months or breastfeeding at 12 months. However, among those women still EBF at 4 months (81.0% of population), those experiencing MSHH were significantly more likely to cease EBF between 4 and 6 months (adjusted Hazard Ratios: 2.52, 95% CI: 1.03-6.19).
Quality Assessment
Table 5 shows that the quality assessment scores of the reviewed studies. Three studies were of good quality, 6 of fair quality, and 6 of poor quality. Although the 3 domains were all scored, 9 of the 15 included studies had 3 to 4 stars for the “selection” domain. However, 8 studies had 1 star for the “comparability” domain as there was no control for confounding factors in the analyses. Similarly, only 7 studies had the highest “verification” score (3 stars) because, in the other studies (n = 8), the loss of follow-up or the follow-up time was not long enough.
Discussion
This review found limited (n = 5) but promising evidence that pregnancy weight gain and childbirth contribute to PPWR among African women. However, other factors such as breastfeeding (n = 4), morbidity (n = 4), and food security (n = 2) must be considered for PPWR in the region. The PPWR averages 0.5 to 3 kg. However, the influence of breastfeeding on PPWR was controversial. Among the 4 included studies, reduction in maternal postpartum weight was associated with breastfeeding even on a short-term basis, especially if breastfeeding was practiced exclusively. As for morbidity, the authors found that HIV status resulted in a weight loss of 1.4 kg after 8 weeks after delivery. Based on the 2 studies on food security, there was a significant correlation between food insecurity and less PPWR even when morbidity and/or breastfeeding were present. Additionally, we found some preexisting factors that affect PPWR, including parity (n = 2), cultural practices after delivery (n = 1), prepregnancy BMI (n = 1), and socioeconomic status (n = 1). However, the quality of included studies was mainly of fair or poor quality. At the same time, survey sites and sample characteristics (size, participants’ age, and length of follow-up) varied significantly.
According to the included studies, weight changes in pregnant women are significant between 4 and 8 months. Interestingly, even in Central Africa, women’s weight at 6 weeks after pregnancy predicted postpartum weight gain. 39 These findings indicated the need for culturally adapted physical activity and diet interventions to prevent gestational weight gain. A review of trials examining such strategies showed lower gestational weight gain in intervention groups than in control groups. 56 In addition, given that many women with a normal prepregnancy weight become increasingly overweight or obese by 1 year after delivery, interventions to prevent excessive pregnancy weight gain are likely to be more effective in normal-weight women than in women who are already overweight. Finally, although breastfeeding has mixed effects on PPWR, exclusive breastfeeding in infants tends to reduce maternal weight gain after childbirth, as observed by Baker et al, 57 suggesting that health care providers should communicate this substantial association during pregnancy and postpartum.
A variety of preexisting factors still understudied in Africa, such as parity, IPI, socioeconomic status, cultural practices after delivery, and food insecurity, were associated with PPWR. Pregnancy and its recurrence are significant risk factors, as most women affected by obesity (50%-80% of cases) attribute their obesity to pregnancy and its recurrence, with an average weight gain after 3 pregnancies of more than 10 kg. 58 However, the IPI did not affect postpartum weight and may not help reduce the weight gained during pregnancy. 59 The disparity in the return to prepregnancy weight by socioeconomic status is consistent with the actual situation in SSA, where poverty affects the onset of obesity. 60,61 During the last 2 decades, there has been a steady increase in urbanization rates, acculturation, and westernization through epidemiological, demographic, and nutritional trends. 62 -65 Furthermore, previous studies suggest that ethnicity and cultural practices may contribute to PPWR. 66 According to Parker and Abrams, 67 black mothers are more likely to retain or gain more weight in the postpartum period than white mothers. Finally, the severity of food insecurity influenced the nutritional status of pregnant and lactating women infected with HIV, suggesting further investigation of the causal relationship between these phenomena.
A Conceptual Framework for PPWR
In line with this review findings, our PPWR conceptual framework (Figure 3) summarizes factors that may contribute to PPWR in African women. This framework builds on previous research frameworks 68 by integrating social, environmental, behavioral, and biological factors contributing to PPWR. The principal theory underlying our framework is that PPWR is due in part to a combination of suboptimal living environments (e.g., social and physical), differential exposure (morbidity), and health risk behaviors.
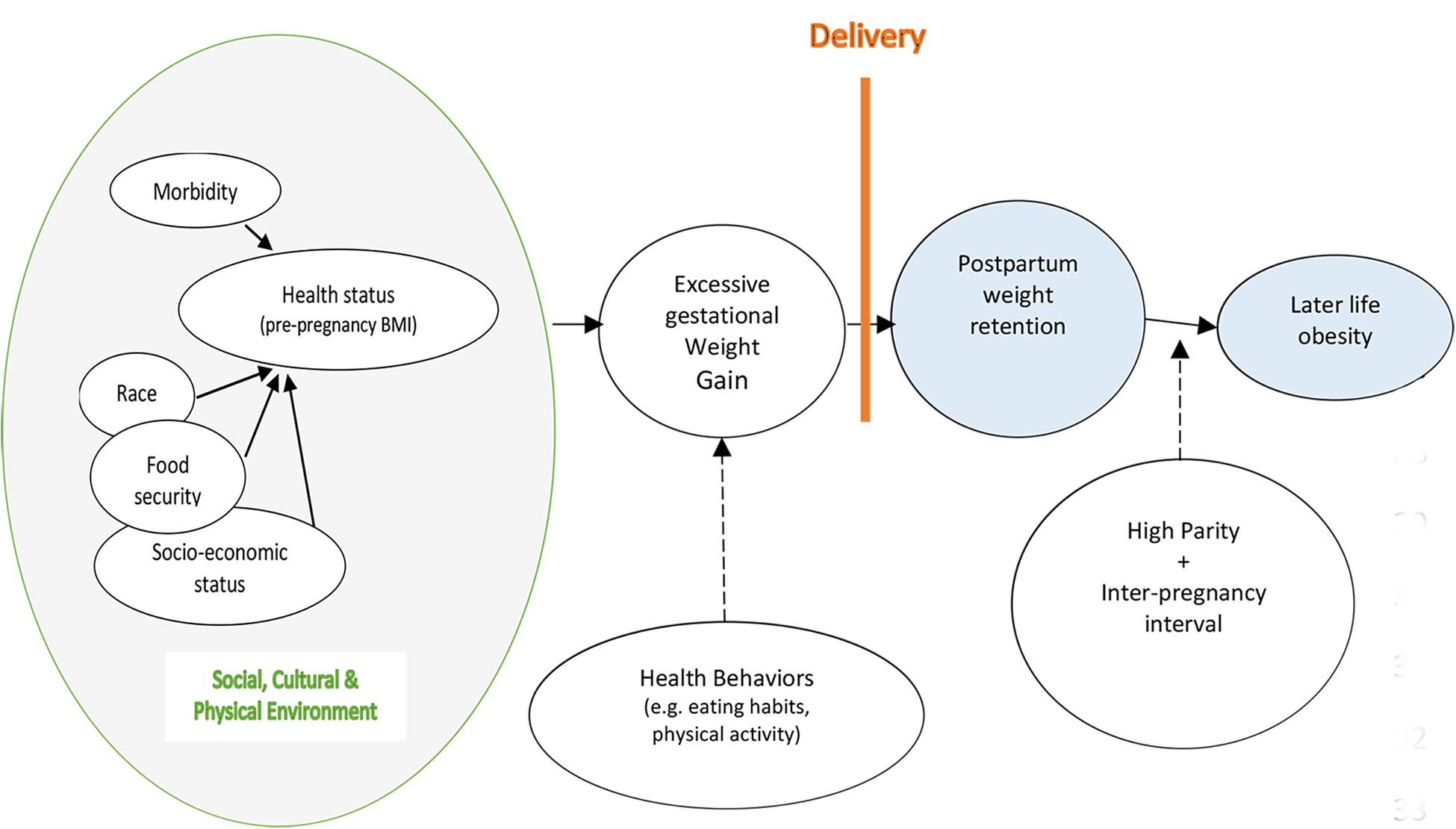
Conceptual model for postpartum weight retention. (Hypothesis represented by dotted line and Review evidence represented by solid line).
Limitations and Implications for Practice and Future Research
The paucity of high-quality eligible studies limits the strength of the evidence in this review. Only 3 good-quality cohort studies demonstrated that parity, morbidity (HIV status), and household food insecurity contribute to PPWR. Twelve of the 15 included studies were assessed as having poor or fair quality in all domains. In addition, we found only 3 studies that assessed the influence of low socioeconomic status, cultural practices, and the low interval between pregnancies despite being prevalent in Africa. These findings underscore the need to consider these preexisting factors when developing interventions to reduce maternal postpartum weight gain.
Low-calorie intake was identified as a factor predicting weight loss, 23,24 but none of the included studies examined participants’ diets. The failure to consider foods rich in nutrients and calories, such as starches, which African women primarily consume, as contributors to a healthy rise in milk consumption is a limitation. Additionally, maternity leave and mothering the new child can result in a sedentary lifestyle, and specific eating habits postpartum that may pose weight gain risk factors 69 have not been investigated. For example, in the study of Boardley et al, 60 higher consumption of total and fat calories was associated with weight retention among black mothers who exercised less than white mothers. Therefore, to fully understand the causes of postpartum weight gain, it is necessary to collect information regarding the mother’s food consumption and activity levels in the region.
Further, previous studies have found that psychological factors (such as depression or sleep disorders) and sociodemographic factors (such as age, marital status, and race) also play a role in PPWR. 27 -29 In SSA, the overall estimated level of postpartum depression was 18.6%. 70 Moreover, research has shown that early body image dissatisfaction after childbirth (at 3 and 6 months) is associated with weight retention 9 months after birth. 30 According to our review, however, these factors have not been studied in relation to weight retention in the region. Therefore, these factors should be investigated to understand their role in PPWR.
Even though EBF was associated with lower PPWR, stopping breastfeeding does not seem to influence this variation, while the ability of breastfeeding to reduce postpartum weight is mixed. The study methods showed that mothers’ evaluation of breastfeeding practices would have influenced maternal weight variation. Nigeria and Senegal used the 24-hour recall method, which may have biased the results. For example, a mother practicing mixed breastfeeding could be considered exclusive breastfeeding. To limit bias, it is recommended to utilize reference methods such as deuterium-dose-to-mother to assess the effectiveness of practice and its effect on maternal weight. 71 However, when these rigorous methods were used in Botswana, 46 breastfeeding did not change weight. The authors explained the result by the fact that most people have access to adequate resources such as water, sanitation, and health care in urban areas.
Conclusion
A lack of studies on factors associated with weight change in the postpartum period in Africa indicates that future studies must employ strong study designs to evaluate the impact of pregnancy and other preexisting factors on weight change. The impact of breastfeeding, socioeconomic status, cultural practices, and the interval between pregnancy needs further research before widespread adoption can be recommended. In addition, it will be essential to study the relationship between weight retention, diet, physical activity, and psychosocial and behavioral factors in future studies. Finally, there is a dearth of research on maternal weight gain and body composition postpartum in Africa, where obesity-related comorbidity rates are steadily increasing.
Footnotes
Authors’ Note
JK conducted the literature search and data interpretation for a PhD thesis at the University and prepared the draft manuscript. HA, WAH, and JL participated in the conceptualization and design of the study and reviewed and edited the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
