Abstract
Background:
Food insecurity (FI) is common globally and can have lifelong consequences. However, few studies have longitudinally examined how FI varies across gestation and the postpartum period (“the first 1000 days”); none have explored this in sub-Saharan Africa or in the context of HIV.
Objective:
To assess the prevalence and covariates of FI in the first 1000 days among Kenyan women.
Methods:
All pregnant women attending 7 clinics in western Kenya (n = 1247) were screened for HIV and FI (Individual Food Insecurity Access Scale) between September 2014 and June 2015. A subset of women (n = 371) was recruited into an observational cohort study and surveyed 11 times through 2 years postpartum (NCT02974972, NCT02979418). Data on FI, sociodemographics, and health were repeatedly collected. Severe FI was modeled using multilevel, mixed-effects logistic regressions (n = 346).
Results:
Of the 1247 pregnant women screened, 76.5% were severely food insecure in the prior month. Further, the prevalence of severe FI was higher among women living with HIV than those without (82.6% vs 74.6%, P < .05). In the cohort, the odds of being severely food insecure decreased monotonically after delivery. Each point higher on the Center for Epidemiologic Studies-Depression scale was associated with 1.08 times greater odds of being severely food insecure (95% CI: 1.05-1.10); each point higher on the Duke/UNC Functional Social Support Scale was associated with 0.97 lower odds of severe FI (95% CI: 0.94-0.99).
Conclusions:
Severe FI is prevalent during the first 1000 days in western Kenya. Services to mitigate the far-reaching consequences of this modifiable risk should be considered.
Introduction
Food insecurity (FI), or inadequate access to food in sufficient quantities and quality, is a growing global health crisis. 1 After decades of improvement, the estimated worldwide prevalence of severe FI has increased and remains stagnant at around 9%. 2 The burden of FI, however, is not equally distributed spatiotemporally. For instance, 24.7% of households in Eastern Africa experienced severe FI in 2019. 2 Intra-annual variations in rainfall and harvest also lead to seasonal fluctuations in FI, especially in regions where households primarily rely upon subsistence farming. 3 -5 Further, within communities and households, the negative impacts of FI on health and well-being are disproportionately experienced by marginalized groups and individuals. 2
Food insecurity may be exacerbated among women during the first 1000 days (ie, the year prior to delivery and 2 years postpartum) given increased physiological demands to support the developing fetus as well as physical and social factors that can limit an individual’s ability to produce, purchase, or prepare preferred foods. 6 -9 Women’s nutritional requirements increase throughout pregnancy and lactation, both in terms of macro- and micronutrients. 6,10 Further, expectant mothers can face diverse barriers that limit their ability to reliably access and prepare preferred foods, including loss of employment, pregnancy discrimination, illness, and increased caretaking responsibilities. 11 It is plausible that maternal FI peaks during late gestation, when a mother’s nutrient demand is at its apex, and declines postpartum. 12
Few studies, however, have assessed the prevalence of individual-level FI among mothers, 13 -15 which is important given that such information is critical for designing tailored interventions that address this potentially widespread and harmful experience. Indeed, the consequences of FI may be particularly deleterious in the critical period of the first 1000 days. 9 For example, women who experience FI during pregnancy have been found to be at greater risk of stress and disordered eating. 6,16,17 Infants in food-insecure houses are also more likely to have physical (eg, low birthweight) and chronic health issues, as well as cognitive problems like delayed learning that can persist into adulthood. 18 -21
The detrimental effects of FI during pregnancy and postpartum may be further compounded by HIV. 6,22 Food insecurity and HIV are co-occurring epidemics that have a bidirectional causal relationship. 23 Food insecurity increases the risk of HIV acquisition and progression through physiological, psychosocial, and behavioral pathways. Further, individuals living with HIV are more likely to experience FI due to the increased nutritional demands associated with the disease and (the side effects of) its treatment. 23,24 Previous research has found that pregnant women who experience HIV and FI have lower odds of sustained viral suppression, 25 experience adverse changes in body composition, 26 and are at greater risk of micro- and macronutrient deficiencies, which in turn are associated with low birthweight and vertical HIV transmission. 23,27
Despite the high prevalence of both HIV and severe FI in sub-Saharan Africa, as well as high fertility rates, the intersection of FI, HIV, and the first 1000 days has received minimal attention. Most research to date has only explored the relationship between pregnancy and FI using cross-sectional data from US mothers. 11,28 -30 Of the studies that have reported FI by gestational period, 16,31 -33 only two have been conducted among mothers outside the United States, 34,35 and none have considered differences by HIV status. Moreover, all previous longitudinal studies have only assessed FI at the household level, which masks critical intrahousehold variations in experiences of FI. 13,36,37 As such, there is a dearth of longitudinal data on FI in the first 1000 days, especially in contexts where the prevalence of HIV is high. Such data are essential for identifying modifiable risks that can be targeted by policies and programs to improve maternal and child health globally.
We therefore sought to fill this gap by examining trends in FI from pregnancy through 2 years postpartum among a cohort of Kenyan women with mixed HIV status. Our two objectives were to assess the prevalence of FI and HIV infection among pregnant women in this region and to identify covariates of FI during pregnancy and the postpartum period. In relation to the second objective, we hypothesized that (1) women living with HIV would experience greater FI during pregnancy relative to the postpartum period and (2) the odds of being severely food insecure would decrease after delivery.
Methods
Study Setting and Population
All pregnant women attending 7 urban, periurban, and rural antenatal clinics (Kisumu, Macalder, Migori, Nyahera, Nyamaraga, Ongo, Rongo) engaged with Family AIDS Care and Educational Services (FACES) in the former Nyanza region, Kenya, between September 2014 and June 2015 were screened (n = 1247). These data can be considered representative of pregnant women in western Kenya because more than 96% of women in this region seek antenatal care (ANC). 38
A subset of women was then recruited into Pith Moromo (“enough feeding” in Luo), a cohort study examining the impacts of HIV and FI during the first 1000 days (NCT02974972). Women were eligible to participate if they were between 14 and 30 weeks of pregnancy, knew their HIV status, and intended to live in the catchment area until their infant(s) reached at least 9 months of age. Quota sampling was used to achieve equal numbers of women by HIV status (negative, known positive [ie, positive HIV test prior to current pregnancy], and newly diagnosed [ie, first positive HIV test during current pregnancy]) and FI tertile (measured using the 9-item Individual Food Insecurity Access Scale [IFIAS] 14 ; low [score: 0-9], moderate [10-18], and high [19-27]). Participants still in the study at 9 months postpartum were consented and enrolled into the Pii En Ngima (“water is life” in Luo) study and surveyed through 21 months postpartum (NCT02979418).
The former Nyanza region of Kenya was an appropriate study setting to test our hypotheses because of the high prevalence of FI and HIV. In 2014, more than 40% of households in the region lacked food or money to purchase food. 38 The estimated regional prevalence of HIV at this time was also the highest in the country at 15.1%. 39
Ethical Approval
Institutional Review Boards at Cornell University, Northwestern University, and the Kenya Medical Research Institute approved study procedures. All participants provided written informed consent at enrollment.
Data Collection
Survey data were collected by clinic-based study nurses using paper forms and tablet-based electronic surveys. All women attending clinics in the catchment area were administered a screening survey about their current pregnancy and self-reported HIV status. Those enrolled into the cohort study (n = 371) were interviewed during 9 in-person visits (2 during the index pregnancy and 7 postpartum) and 2 phone calls (at approximately 1 week and 6 months postpartum; Figure 1). 16,17 Given that psychosocial well-being is an important confounder between our exposures of interest and FI, 17,41,42 we restricted our analyses to data from time points at which both FI and mental health information were collected (baseline, 6 weeks, 9, 12, 15, and 21 months postpartum).
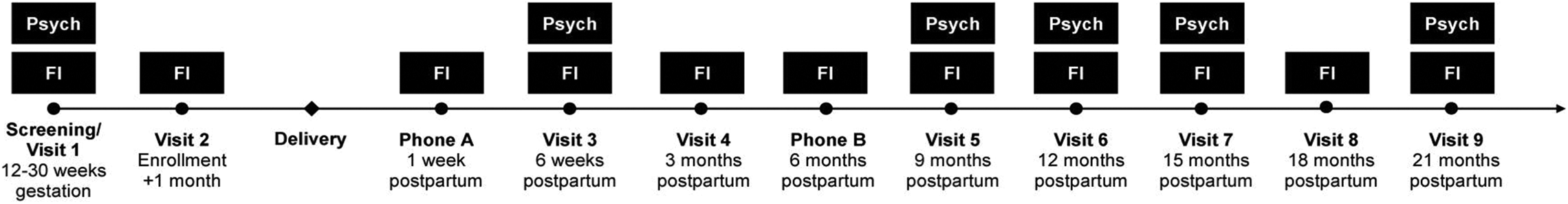
Time points at which food insecurity and psychosocial measures were collected among Kenyan women enrolled in the Pith Moromo/Pii En Ngima study. Adapted from the study by Tuthill et al. 40 FI indicates food insecurity; Psych, psychosocial measures (Center for Epidemiologic Studies-Depression scale and the Duke/UNC Functional Social Support Scale).
Dependent variable: FI
Food insecurity in the prior month was measured using the 9-item IFIAS. 14 The IFIAS was used at screening and all subsequent study visits (Figure 1). It is an adaptation of the Household Food Insecurity Access Scale (HFIAS) 43 ; participants are queried about their personal experiences with access to and use of food rather than those of the household. Each item has 4 response options (never = 0, rarely = 1, sometimes = 2, often = 3) that are summed together (range: 0-27). The IFIAS has been validated for use among pregnant women elsewhere in East Africa. 14 The internal consistency for the IFIAS among all women at screening was high, with a Cronbach α of 0.95.
Individuals were categorized as food secure, mildly food insecure, moderately food insecure, or severely food insecure following the HFIAS guidelines. 43 Given that the majority of respondents were severely food insecure, we created a binary variable for FI that classified individuals as severely food insecure or not at each visit.
Primary independent variables: pregnancy and HIV status
Gestational age was estimated based on reported last menstrual period at screening. For those enrolled into the cohort, pregnancy was reassessed at every visit after the 3-month postpartum survey using human chorionic gonadotropin urine tests. Months postpartum was calculated by subtracting the interview date from the reported delivery date. We then used these data to categorize individuals into critical developmental periods: second trimester (14 to 26 weeks gestation), third trimester (27 or more weeks gestation), 0 to 5 months postpartum, 6 to 11 months postpartum, 12 to 17 months postpartum, and 18 or more months postpartum.
HIV status was self-reported at screening and confirmed using antenatal clinic booklets. HIV status was then assessed with colloidal gold rapid tests for all women eligible for enrollment and reassessed at every visit for mothers in the study who previously tested negative.
Other covariates
A number of sociodemographic variables previously found to be associated with FI and pregnancy or FI and HIV were assessed. 11,14,39 Maternal ethnic group (Luo or other ethnic group), age (in years), education (primary or less vs greater than primary), marital status (married or cohabiting vs single), and number of individuals living in the household were captured at enrollment. Household wealth was derived from a principal component analysis of self-reported household asset ownership at approximately 9 months postpartum using an adapted version of the Kenya Integrated Household Budget Survey Questionnaire 44 and made into quintiles. Month of interview was used as a proxy for seasonal factors that influence food availability, for example, rainfall and harvest schedule.
Psychosocial well-being was measured at 6 time points (Figure 1). Social support was measured using a 10-item scale adapted from the Duke/UNC Functional Social Support Scale and assessed as a continuous score (range: 0-30), where higher scores indicate greater social support. 45 Depression was assessed using the Center for Epidemiologic Studies-Depression scale (range: 0-60), 46 which has been shown to be suitable for use among pregnant women living with HIV in low-resource settings. 47 Higher scores indicate greater depressive symptomatology. Cronbach α for the social support and depression symptomatology scales at baseline (0.93 and 0.85, respectively) suggested high internal consistency.
Missing Data and Multiple Imputation
There were approximately 1% to 3% missing data across time-variant variables. Data were missing due to nonresponse and attrition. Complete case analysis (CCA) only includes women who have data on at least one variable at each time point, which reduces sample size and discards useful data points when cases are incomplete. 48 We therefore used multiple imputation to generate data points for these variables of interest and to address potential bias caused by missing data.
We imputed 10 data sets with multiple imputation using chained equations. Ordinal logistic regressions were used to impute wealth quintiles and responses to the IFIAS, and linear regressions were used to impute social support and depression symptomatology scores. We report results from multiple imputation; results from CCA are included in Supplementary Material S1. The two analyses gave results that are similar in direction of associations and significance at P < .05.
Data Analysis
Analyses were conducted using Stata 15 (StataCorp). For our first objective, we calculated the population prevalence of maternal FI by self-reported HIV status (n = 1247) and examined differences using Pearson chi-square test. We also assessed the relationship between severe FI and HIV status using a multilevel, mixed-effects logistic regression that accounted for the hierarchical data structure (ie, individuals nested within clinic sites) and controlled for month of interview.
For our second objective, we restricted our analysis to individuals who were enrolled into the cohort study. Given our interest in how FI varies across the gestational period, we excluded 22 individuals who were only interviewed at one time point (Figure 2). We also excluded 3 individuals who seroconverted during the study period to allow for subgroup analysis by HIV status, such that our final analytic sample size was 346 women. We first characterized the population using descriptive statistics (Wilcoxon rank-sum test, t test, Pearson chi-square test). We then assessed the bivariate relationships between severe FI and household, sociodemographic, and health characteristics using multilevel, mixed-effects logistic regressions. Variables that were significant at P < .2 were included as controls in the final adjusted model of severe FI. Given our purposive sampling strategy, we did not include HIV status as a control variable but instead explored HIV status as an effect measure modifier in subgroup analyses.
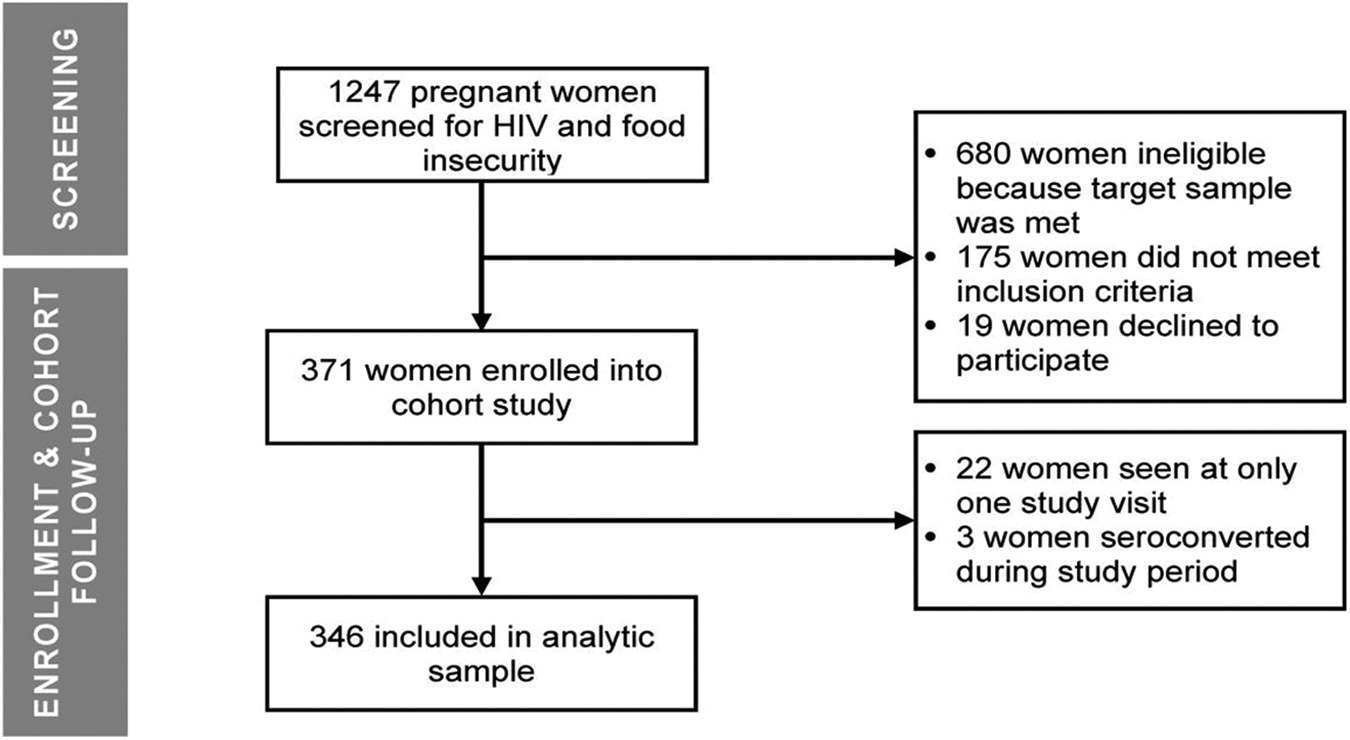
Study flow of participants through the Pith Moromo/Pii En Ngima study.
Results
Population Prevalence of FI
Among all women attending 7 ANC clinics in western Kenya between September 2014 and June 2015 (n = 1247), only 5.4% were classified as being food secure in the prior month (Figure 3). More than three-quarters (76.5%) of women were severely food insecure. The prevalence of severe FI was highest at the Nyamaraga clinic (88.0%) and lowest at the Kisumu clinic (64.7%). The prevalence of HIV among pregnant women across all sites was 23.9%.
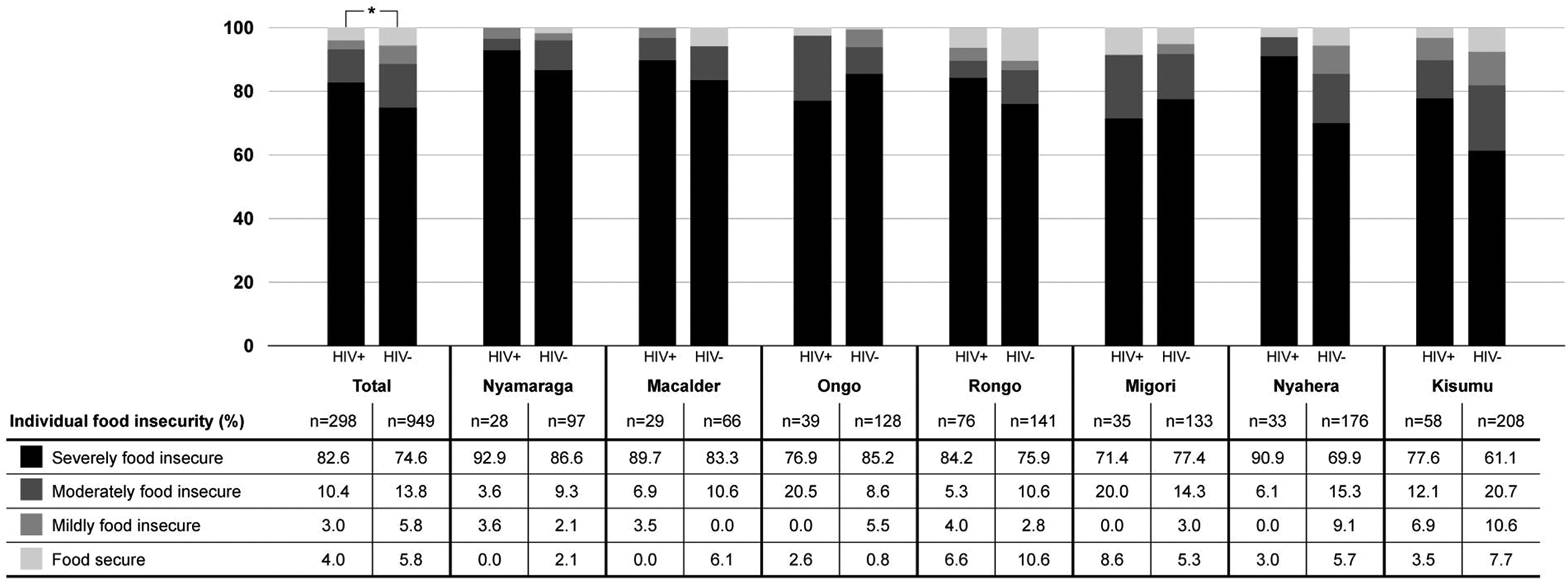
Population prevalence of food insecurity among pregnant women attending 7 antenatal clinics in western Kenya, by HIV status, between September 2014 and June 2015, measured using the Individual Food Insecurity Access Scale (IFIAS; n = 1247). 14 * P < .05.
Maternal FI differed by HIV status (Figure 3). Comparing across all sites, 82.6% of women living with HIV were severely food insecure compared to 74.6% of mothers without HIV. In a multilevel, mixed-effects logistic regression of severe FI controlling for month of interview, pregnant women living with HIV had 1.63 (95% CI: 1.15-2.31) times greater odds of being severely food insecure compared to women without HIV (P = .006).
Cohort Characteristics
The Pith Moromo cohort had a higher prevalence of severe FI (84.4%, n = 346) at baseline than the entire population screened (76.5%, n = 1247) due to our quota sampling strategy. At baseline, women with and without severe FI differed across most characteristics (Table 1). For instance, a greater proportion of women with severe FI were in the lowest wealth quintile (23.1% vs 5.4%) and had greater depressive symptomatology (mean [standard deviation]: 19.2 [8.4] vs 15.3 [7.5]) than women without severe FI.
Baseline Characteristics of Women Enrolled in Pith Moromo Who Did Not Seroconvert During The Study Period and Were Interviewed at More Than One Time Point, by Individual Food Insecurity Status.a
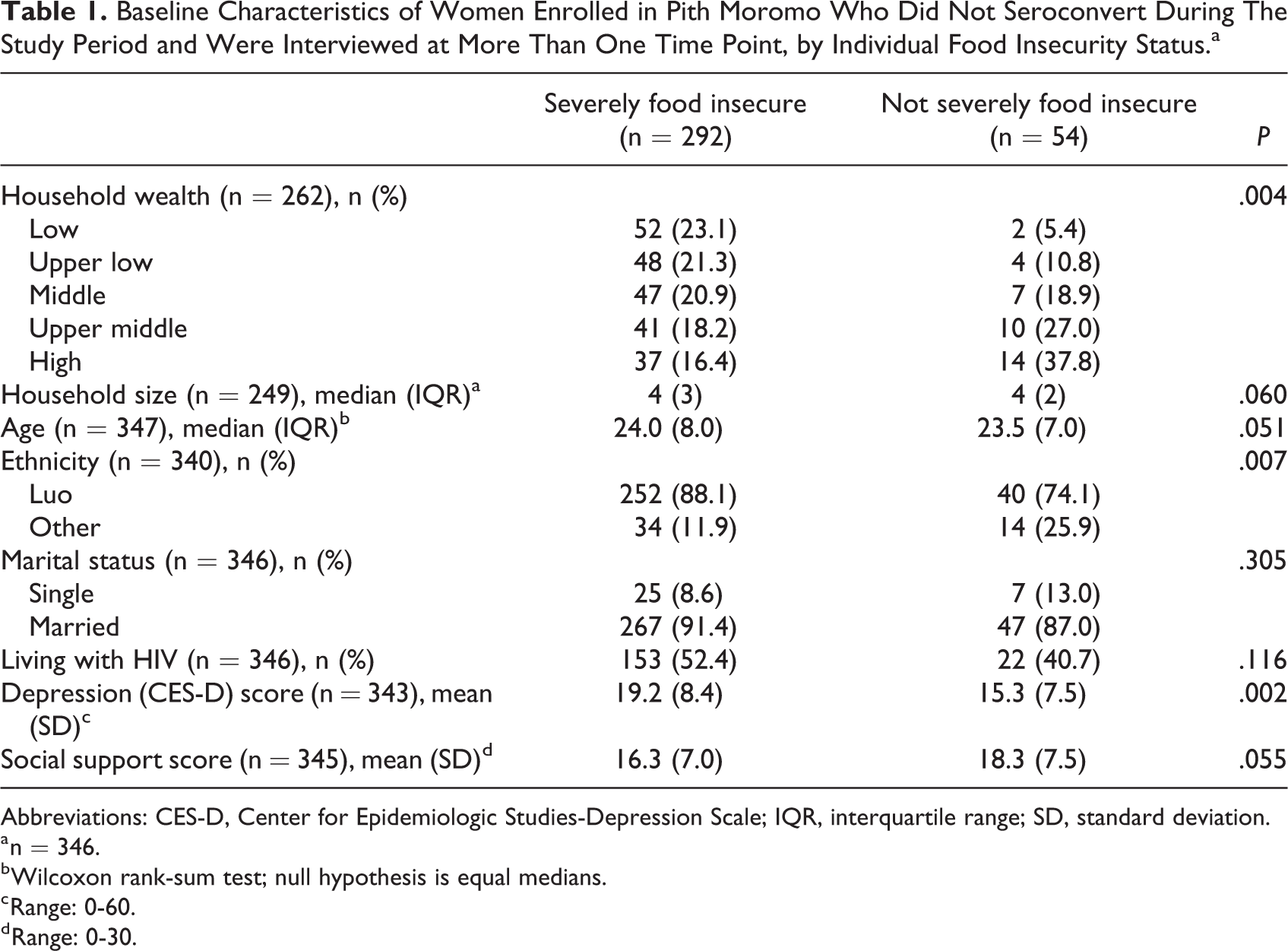
Abbreviations: CES-D, Center for Epidemiologic Studies-Depression Scale; IQR, interquartile range; SD, standard deviation.
a n = 346.
b Wilcoxon rank-sum test; null hypothesis is equal medians.
c Range: 0-60.
d Range: 0-30.
Covariates of FI Across the Perinatal Period
In multilevel, mixed-effects logistic regressions, factors associated with severe maternal FI were similar for women living with and without HIV (Table 2). Among all women, the odds of being severely food insecure monotonically decreased as women progressed through the postpartum period (Figure 4 and Table 2). For instance, women who were 6 to 11 months postpartum had 79% lower odds of being severely food insecure compared to women in the second trimester of pregnancy (95% CI: 0.12-0.37). The observed effect was stronger among women without HIV.
Multilevel (Individuals Nested Within Clinic Sites), Mixed-Effects Logistic Regressions of Severe Food Insecurity Across Pregnancy Through 2 Years Postpartum Among Kenyan Women in Pith Moromo/Pii En Ngima, by HIV Status.
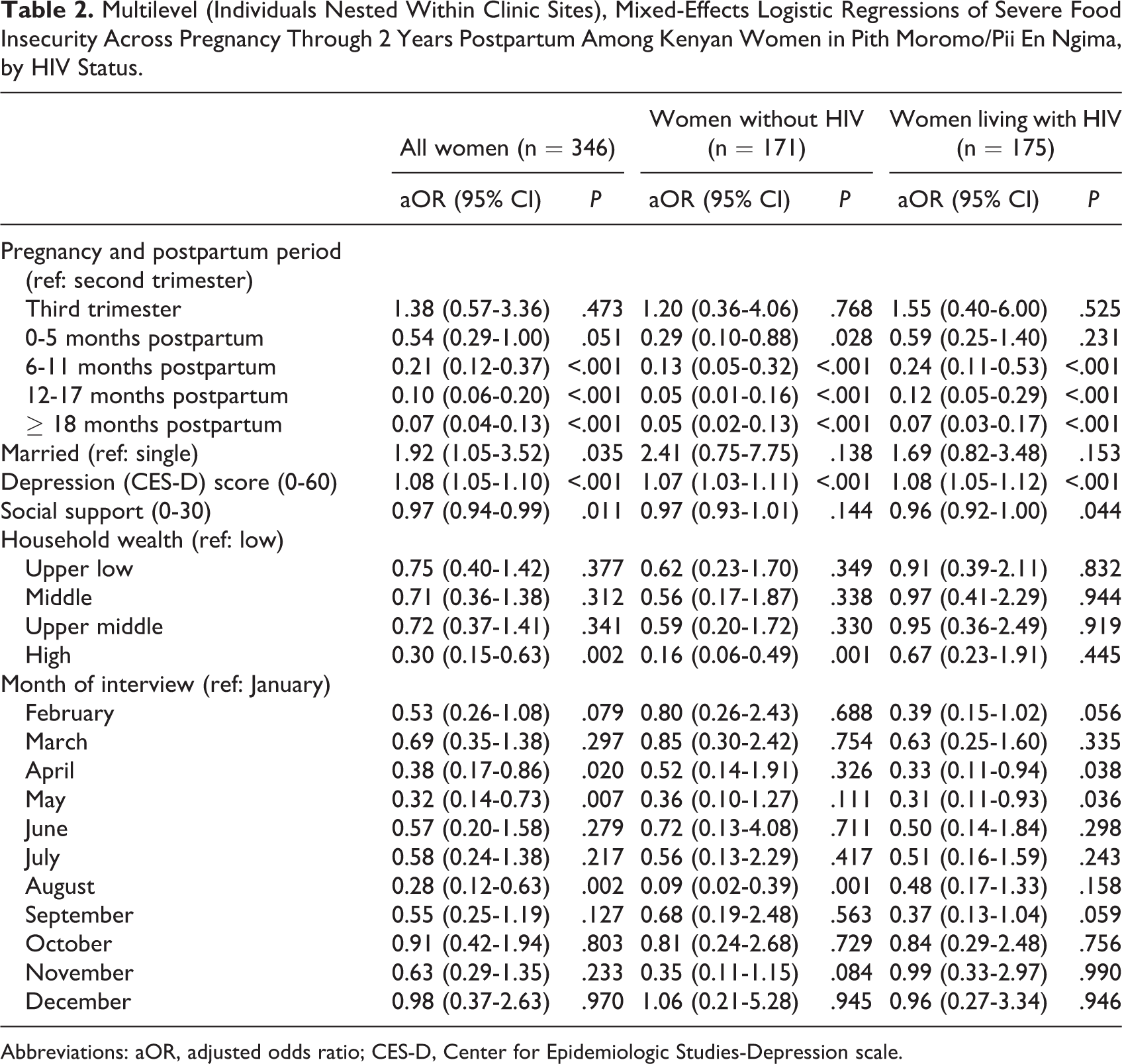
Abbreviations: aOR, adjusted odds ratio; CES-D, Center for Epidemiologic Studies-Depression scale.
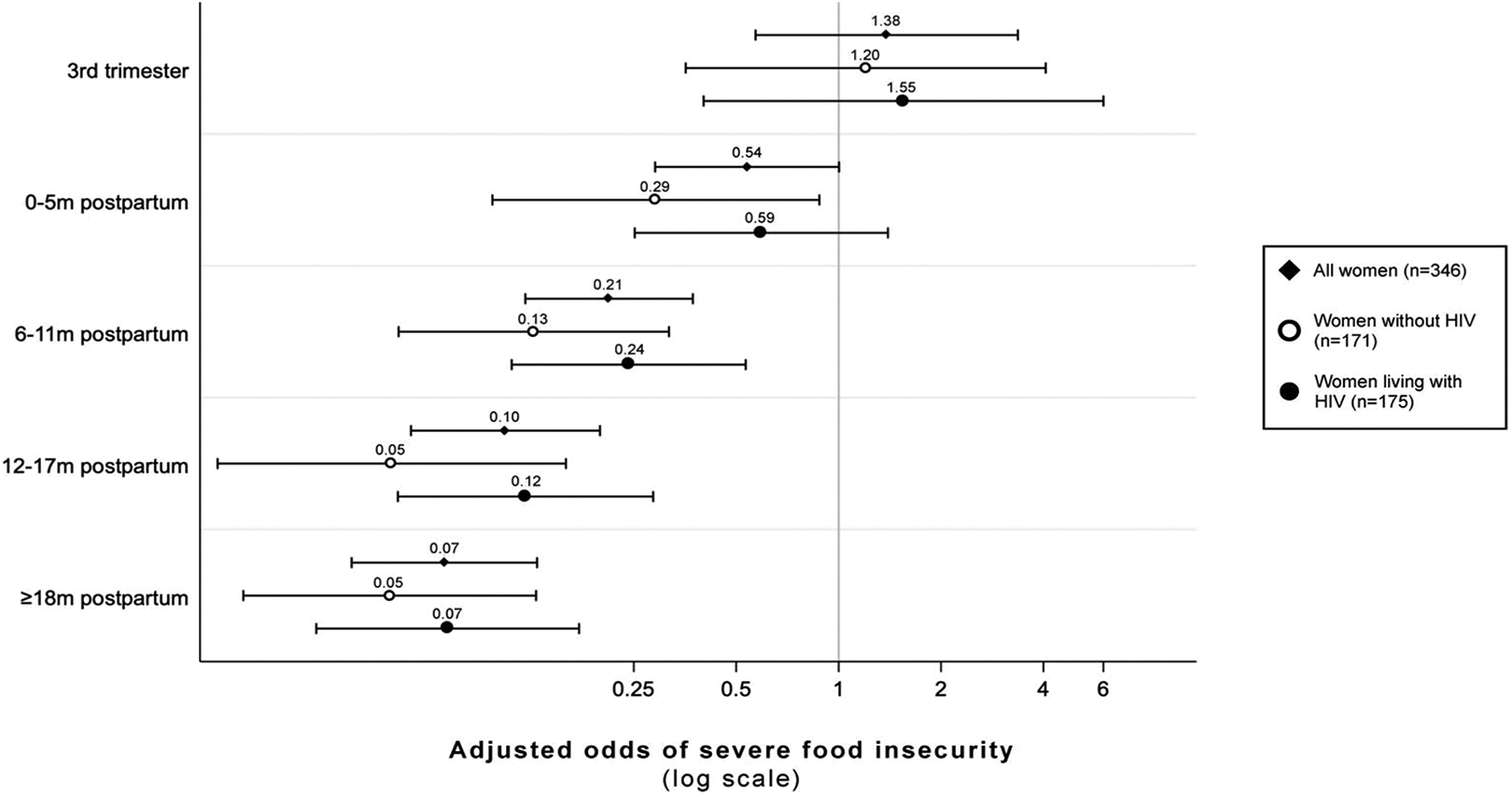
Adjusted odds and 95% CIs of severe food insecurity across pregnancy and postpartum among women in Pith Moromo/Pii En Ngima, relative to the second trimester of pregnancy. Adjusted for marital status, depression (CES-D) score, social support score, wealth, and month of interview. CES-D indicates Center for Epidemiologic Studies-Depression scale.
Psychosocial well-being was negatively associated with severe FI (Table 2). Specifically, among all participants, each point higher on the CES-D Scale was associated with 1.08 times greater odds of being severely food insecure (95% CI: 1.05-1.10). This held in subgroup analyses among women living with HIV and those without. Each point higher on the social support scale was associated a 3% decrease in the odds of being severely food insecure among all women (95% CI: 0.94-0.99; Table 2). In subgroup analyses, social support was inversely associated with odds of being severely food insecure among women living with HIV only.
Discussion
In this longitudinal study of predictors of maternal FI across the first 1000 days, we found that the prevalence of severe FI among pregnant women in western Kenya (76.8%) was higher than the national prevalence in the general population (19.1% for 2014-2016). 2 The prevalence of HIV among screened pregnant women was 23.9%, which was higher than the regional prevalence (15.1% for 2012-2013). 39 Using a multilevel, mixed-effects logistic regression, we found that living with HIV was associated with greater odds of being severely food insecure. We also found that the odds of severe maternal FI decreased throughout the postpartum period, and that greater depressive symptomatology and lower social support were associated with greater odds of being severely FI.
In support of our first hypothesis, we found that maternal FI was higher among women living with HIV (82.6% vs 74.6%, P < .05, Figure 3). This aligns with previous qualitative and quantitative work that has found a bidirectional relationship between HIV and FI. 23,49,50 Importantly, FI is often described as a barrier to antiretroviral therapy (ART) adherence. 24 Given that ART during pregnancy is critical for suppressing viral loads and reducing the risk of vertical HIV transmission, 51 FI should be screened for during ANC visits. Programs that help food-insecure mothers access diverse foods in sufficient quantities, especially in lean seasons, may also improve ART adherence.
In support of our second hypothesis, we found that the odds of being severely food insecure did not vary during pregnancy but did decrease after delivery, even when controlling for other critical determinants of FI such as seasonality (Figure 4 and Table 2). This aligns with previous cross-sectional work in Kenya that found that FI status did not vary across gestation. 35 Interestingly, the odds of severe FI during 0 to 5 months postpartum relative to the second trimester of pregnancy were not different for women with HIV but decreased for women without HIV (Table 2). This differential effect may due to the fact that women living with HIV in this region often breastfeed for longer periods than their maternal counterparts without HIV and thus have higher nutritional demands 52 ; further research is needed to understand these pathways. Regardless of HIV status, the odds of severe FI decreased at all time points after 6 months postpartum. At 6 to 11 months postpartum, mothers have typically recovered from delivery and are encouraged to transition from exclusive breastfeeding to mixed feeding that incorporates complementary foods. 53 As breastfeeding frequency and duration decrease postpartum, maternal micro- and macronutrient requirements return to prepregnancy levels. 54 As such, the physical and nutritional demands of pregnancy and lactation may, in part, explain these observed trends, although there are undoubtedly social dynamics at play as well.
We found relationships between psychosocial well-being and FI. Our finding that greater depressive symptomatology was associated with higher odds of severe FI aligns with previous work among pregnant and postpartum populations in low- and middle-income settings. 17,40 -42 It is unclear, however, whether depressive symptomatology precedes or is a product of FI. Previous work in the United States supports a potential bidirectional relationship: The stress of FI increases the risk of depression, and depression may lead to FI by reducing an individual’s social and economic capital. 55,56 Given the plausible causal relationships, future studies should more rigorously explore the relationship between FI and depression in low- and middle-income countries using longitudinal data that incorporates biomarkers of stress.
Our finding that greater social support is associated with lower odds of severe FI is consistent with previous studies that have found that social support moderates the relationship between FI and depression (ie, greater social support mitigates the negative impacts of FI on mental well-being). 57,58 Social support may reduce FI through emotional (eg, reducing feelings of uncertainty and worry), instrumental (eg, gifts of food, agricultural inputs), appraisal (eg, empowering individuals to identify new solutions), and informational (eg, advice on how to make food last, information about how to get food aid) mechanisms. 59 These were not assessed in this study, but knowing how social support may shape FI could be valuable for designing cost-effective interventions.
Subgroup analysis suggests that women living with HIV may be more likely to benefit from increased social support (Table 2). This is perhaps due to the additional burdens HIV infection presents when caring for a newborn, 60 but this too requires further exploration. Future work should investigate which types of social support are most effective at reducing FI, and whether the types of support offered to women differ by maternal HIV status or change throughout the perinatal and postpartum periods.
Taken together, our findings suggest that FI during the perinatal period in western Kenya is a prevalent public health issue. Given the diverse and sometimes lifelong health consequences associated with FI during the first 1000 days for both women and their infants, 6,16 -21 FI should be screened for at the first ANC visit, and interventions should prioritize mitigating FI among this vulnerable group. Further, policies and programs should account for potentially co-occurring mental health conditions that may exacerbate FI and its negative sequelae.
Strengths and Weakness
This study is novel for its concurrent investigation of HIV and maternal FI across gestation in a population outside the United States. There are also several weaknesses. As with most panel studies, there was some systematic missingness in our data. Observations across visits decreased across time, yet the percentage of missing data was under 10% for the primary outcome and most covariates. Further, we used multiple imputation to account for missing data and found similar results for models using imputed and complete-case data. Additionally, a greater proportion of women enrolled in the cohort were severely food insecure compared to the general pregnant population as a result of the quota sampling strategy, meaning that our findings may not generalize to other populations.
Given that FI is dependent upon local food availability, 61 we controlled for month of interview in our models to account for seasonal fluctuations in food production and harvest. Month of interview, however, may not have adequately captured other key community and environmental determinants of FI. We therefore recommend that when possible, future studies examining FI in populations dependent on subsistence farming collect information on monthly rainfall, crop production (including amount grown and stored), market conditions, and water insecurity, which often co-occurs with and can interact multiplicatively with FI to undermine maternal well-being. 62 -64
Conclusion
This study provides an important contribution to the literature by documenting the prevalence and patterns of maternal FI among women with and without HIV during the first 1000 days. We found that FI decreases after delivery and is positively associated with HIV infection, greater depressive symptomatology, and lower social support among women in western Kenya. As the first 1000 days become formalized into development and policy agendas, it is imperative to both recognize the prevalence and far-reaching consequences of FI in this period, as well as to provide care and services to mitigate it.
Supplemental Material
Supplemental Material, sj-docx-1-fnb-10.1177_0379572121999024 - Prevalence and Covariates of Food Insecurity Across the First 1000 Days Among Women of Mixed HIV Status in Western Kenya: A Longitudinal Perspective
Supplemental Material, sj-docx-1-fnb-10.1177_0379572121999024 for Prevalence and Covariates of Food Insecurity Across the First 1000 Days Among Women of Mixed HIV Status in Western Kenya: A Longitudinal Perspective by Gloria G. Alvarez, Joshua D. Miller, Marianne V. Santoso, Pauline Wekesa, Patrick Mbullo Owuor, Maricianah Onono and Sera L. Young in Food and Nutrition Bulletin
Footnotes
Acknowledgments
We thank the Kenya Medical Research Institute for providing space within the FACES clinics for the work to be conducted, ethical and logistic support to conduct this research, and oversight in Kenya. We warmly thank study nurses and study trackers, as well as the mothers and their infants who participated in this study. We thank Shalean Collins for assisting with data collection and management, as well as Irene Tsai for her preliminary analyses that helped inform this work. Finally, we are grateful to Emily Tuthill, Shalean Collins, Maggie Butler, and the entire Young Research Group for providing feedback on earlier versions.
Authors’ Note
GGA and JDM are the co-first authors and contributed equally to the article. GGA and JDM led data analysis, created figures and tables, wrote the original draft, and critically revised the subsequent versions. JDM was responsible for data cleaning and management. MVS assisted with data analysis and interpretation, as well as manuscript revision. PW, PMO, and MO provided insights into data interpretation and critically revised subsequent versions. MO assisted with design and supervisions of data collection. SLY conceived the study, acquired funding, designed data collection, helped supervise data collection, assisted with the writing of the original draft, and critically revised subsequent versions. The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsements, either expressed or implied, of the US Government or the National Institutes of Health.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institutes of Health K01 MH098902 and R21MH108444. SLY and MVS were supported by the generous support of the American people provided to the Feed the Future Sustainable Intensification Innovation Lab (SIIL) through the US Agency for International Development Cooperative Agreement AID-OAA-L-14-00006.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
