Abstract
Background:
Dietary vitamin B12 (B12) deficiency is common in Indians. Long-term compliance to tablet supplementation is poor in asymptomatic individuals.
Objective:
To study efficacy of B12 fortified nutrient bar and yogurt in improving plasma B12 concentrations in children and adults.
Methods:
Two double-blind, placebo-controlled directly observed therapy randomized controlled trials were conducted for 120 days: (1) Healthy children (10-13 years) were fed nutrient bar fortified with B12 (2 μg), multiple micronutrients B12 (1.8 μg) or placebo. (2) Healthy adults (18-50 years) were fed yogurt fortified with B12 (2 μg) or Propionibacterium (1 × 108 cfu/g) or placebo. B12, folate, homocysteine, and hemoglobin concentrations were measured before and post intervention.
Results:
We randomized 164 children and 118 adults; adherence was 96% and 82%, respectively. In children, B12 fortified bars increased B12 concentrations significantly above baseline (B12 alone +91 pmol/L, B12+ multiple micronutrients +82 pmol/L) compared to placebo. In adults, B12 fortified yogurt increased B12 significantly (+38 pmol/L) but Propionibacterium and placebo did not. In both trials, homocysteine fell significantly with B12 supplementation. Rise of B12 and fall of homocysteine were influenced by dose of B12 and folic acid. There was no significant difference in change of anthropometry and hemoglobin between groups.
Conclusions:
B12 fortified foods are effective in improving B12 status in Indian children and adults. They could be used to improve B12 status in the national programs for children, adolescents, and women of reproductive age. They could also be used as over-the-counter products.
Introduction
Low vitamin B12 (B12) status is widely prevalent in vegetarian populations. 1 Although asymptomatic in many individuals, deficiency may be associated with hematologic, neurocognitive, and cardiovascular manifestations in some. 2 Low maternal B12 status during pregnancy is linked to increased risk of neural tube defects, preeclampsia, gestational diabetes, preterm delivery, fetal growth restriction, as well as to increased risk of future adiposity and insulin insensitivity in the child. 3,4
Only bacteria synthesize B12 naturally, which is incorporated by animals into their tissues. 5 Humans obtain B12 from consumption of such animal derived foods (meat, liver, fish, eggs, and milk). Plant foods do not contain B12. Low B12 status in developing countries is largely attributed to low intake of animal-origin foods, either because of cultural and religious practices (Hindu, Jain, and Buddhist) or due to poverty, which precludes expensive nonvegetarian foods from diet. 6 Low B12 status is common in Indians, both in those living in India as well as those migrated abroad. 7 This is largely attributable to vegetarianism but not to pernicious anemia (malabsorption due to lack of intrinsic factor). Our research showed that B12 deficiency is common in and around Pune, India, 7 despite normal B12 absorption in > 85% of the population. 8 Increasing intake of animal origin foods to improve vitamin B12 status has obvious practical limitations in Indians. In our community-based trials, B12 status improved by supplementation with capsules containing 2 µg B12 per day 9 or drinking 400 mL of milk daily. 10
Although low-dose supplements are successful in improving B12 status in a trial setting, they are unlikely to have a big impact in public health owing to poor long-term adherence in asymptomatic individuals. Food fortification may be an effective way to improve B12 status in Indians and other vegetarian populations. Fortification of breakfast cereals and milk with B12 is common in many countries but not in India. In recent years, probiotics including Propionibacterium have been claimed to improve B12 status. 11 This could also be of help in Indians and warrants further research.
As part of our research to improve B12 status of the population, we tested 2 B12 fortified food items: (1) A nutrient bar in school children, and (2) Yogurt in adults, for their efficacy to improve B12 status. We also tested a Propionibacterium species probiotic, claimed to produce substantial quantities of B12, as a separate fortificant for yogurt.
Methods
Study Design and Intervention
We conducted 2 double-blind, placebo-controlled, randomized, directly observed therapy (DOT) trials. We excluded those with chronic medical illness and those with very low B12 concentration (< 100 pmol/L) or anemia (hemoglobin < 10 g/dL) for ethical reasons and advised them appropriate treatment. We also excluded those on regular vitamin supplementation (> 10 days). Randomization was computer generated, and stratification was based on baseline B12 and hemoglobin concentrations (below and above median) to ensure comparable distribution. All participants were instructed not to take any vitamin supplements during the trial. Intervention products were consumed under observation (DOT). Adherence was calculated for each participant by calculating the number of days of attendance during the trial. Adverse events were recorded.
Nutrient Bar Trial
This was conducted in a village school (Pabal, Pune District, ∼70 km from Pune) in children aged 10 to 13 years. School authorities approved participation in the trial. We arranged a meeting with the school children and their parents to explain the trial and invited them to sign an assent and consent. Included children were randomized to 3 groups to receive one bar per day, fortified with (a) 2 µg B12 or (b) multiple micronutrients (MMN), including 1.82 µg B12 (United Nations Children’s Emergency Fund guidelines) 12 (Supplementary Table 1), or (c) no added micronutrients (placebo). All bars were similar in appearance and taste. The children ate the bars in the school in the morning recess, directly observed by research staff. The duration of supplementation was 120 consecutive school days, from September 2011 to March 2012.
Yogurt Trial
The yogurt study was conducted in volunteers aged 18 to 50 years at the KEM Hospital, Pune. Hospital staff members and their friends were invited to enroll through notice board advertisement. Eligible participants were randomized into 3 groups to receive 100 gm yogurt fortified with (a) 2 µg B12, (b) 1 × 108 cfu/g Propionibacterium, and (c) without any additions (placebo). The strain used for fortification (Propionibacterium freudenreichi subsp. freudenchii ATCC 6207, GRAS certified) was provided by the National Dairy Research Institute and was recommended because it was known to produce B12 in vitro. 13 The 3 yogurt preparations were similar in appearance, taste, flavor, and smell. The yogurt was eaten under supervision for 120 consecutive working days between December 2013 and May 2014.
Ethics
In nutrient bar trial, informed written assent was obtained from the children, and consent was signed by the parents. In yogurt trial, participants provided written consent. Both trials were approved by KEM Hospital Research Centre Ethics Committee. For the nutrient bar trial, permissions were obtained from the school authorities and the District Health Officer. The trials were registered with the Clinical Trials Registry of India (CTRI/2012/07/002799 -nutrient bar, CTRI/2015/04/005703 -yogurt).
Measurements
Height was measured to the nearest 0.1 cm using a stadiometer (CMS Instruments) and body weight to the nearest 0.01 kg using an electronic weighing scale (Model no. HD-358, Tanita Corporation). At baseline, nonfasting venous blood sample was collected in ethylenediaminetetraacetic acid vacutainers for measurements of hemogram, B12, folate, and homocysteine. Hemogram was measured on a Beckman Coulter Analyzer (AC.T diff) on the same day. Plasma aliquots were stored (−70 °C) until further analysis. B12 was measured by a microbiological assay using a colistin sulfate-resistant strain of Lactobacillus leichmannii (Coefficient of variation (CV) < 8%). 14,15 Plasma total homocysteine (homocysteine) was measured by fluorescence derivative of monobromobimane using HPLC (inter and intra batch CV < 4%). Plasma folate was measured by a microbiological assay using a chloramphenicol-resistant strain of Lactobacillus Casei (inter and intra CV < 8%). 16,17 The blood and clinical measurements were repeated at the end of the trial.
Statistical Analysis
For children, we calculated sample size required in each group to demonstrate a 50% rise in plasma B12 concentrations at 5% significance level and found that 55 individuals allowing a dropout rate of 10% will provide a power of more than 90%. For adults, we calculated sample size required in each group to demonstrate a 30% rise in plasma B12 concentrations at 5% significance level and found that 45 individuals allowing a dropout rate of 10% will provide a power of more than 80%.
Data are presented as median (25th-75th percentile) for continuous variables and as percentages for categorical variables. Skewed variables were log normalized before analysis. Significance of the differences between baseline and end of trial levels of clinical and biochemical measurements were tested by paired t test for continuous variables and by χ2 test for categorical variables. Significance of difference from placebo group was tested by t test. Analysis was by intention-to-treat method. The participants who were lost to follow-up were analyzed by “last observation carried forward” method.
We used multivariate regression analysis to calculate predictors of change in B12 and homocysteine concentrations after intervention. We normalized the variables by converting them to standard deviation scores. We used age, sex, BMI, baseline concentrations of B12 or homocysteine, and dose of B12 and folic acid in intervention group as determinants of change. In the yogurt trial, there were no folic acid supplements; therefore, we used change in folate concentrations.
Results
Nutrient Bar Trial
We approached 180 school children and their parents to participate in the trial, of whom 178 (99%) agreed. Fourteen children were excluded: 13 with B12 <100 pmol/L and 1 with anemia; they were prescribed appropriate treatment (Figure 1A). The 164 randomized children (57% girls) were 11.3 (10.9-12.0) years old, 139.4 (134.5-144.5) cm tall and with a weight of 28.8 (25.2-33.4) kg; there was no significant difference between groups. Plasma B12, folate, and homocysteine concentrations were comparable (Table 1).
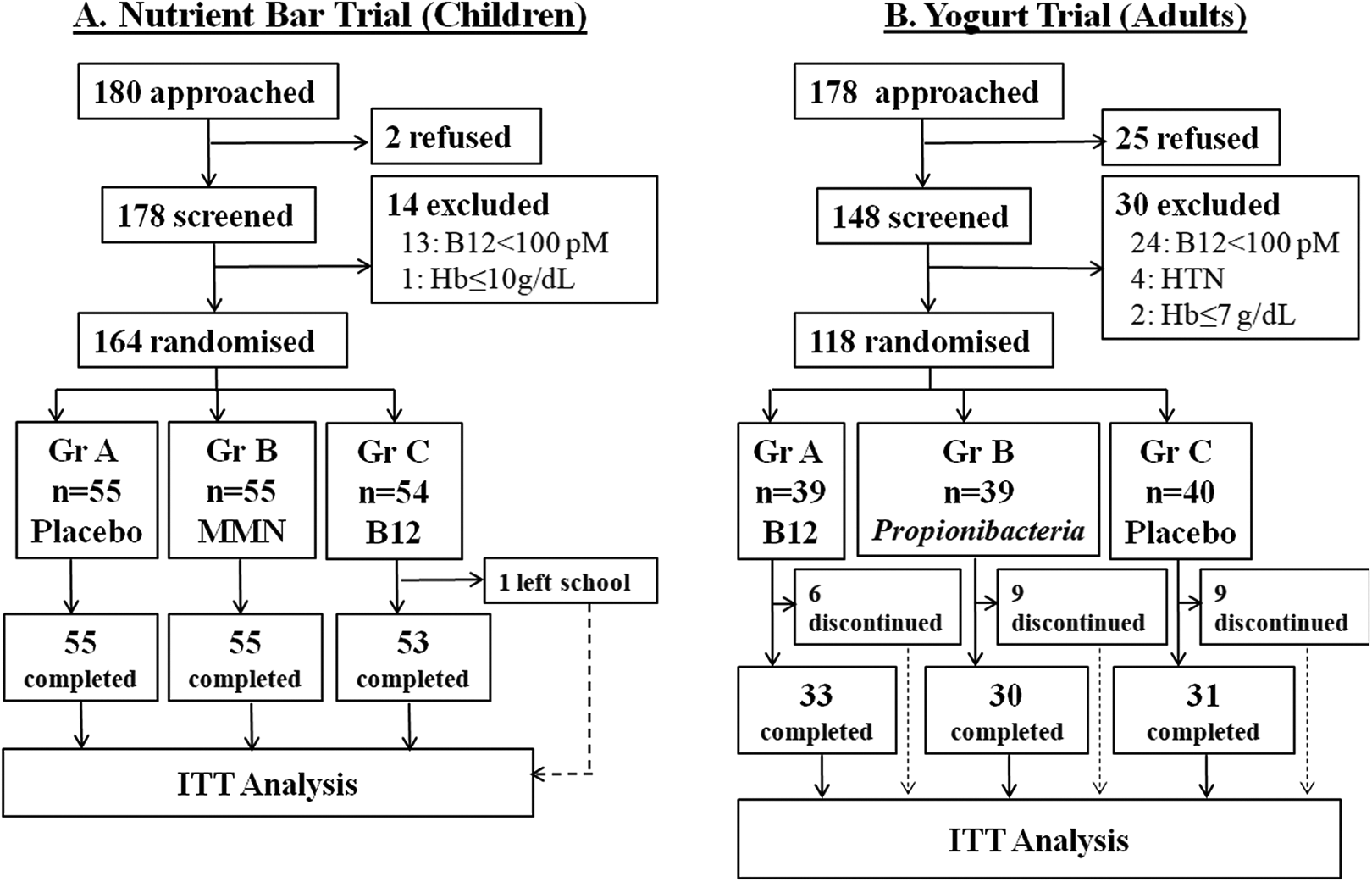
Flow of participants in 2 trials. (A) Nutrient bar trial was conducted in village school children. (B) Yogurt trial was conducted in healthy adult volunteers in an urban setup.
Nutrient Bar Trial—Comparison Within and Between Groups.a
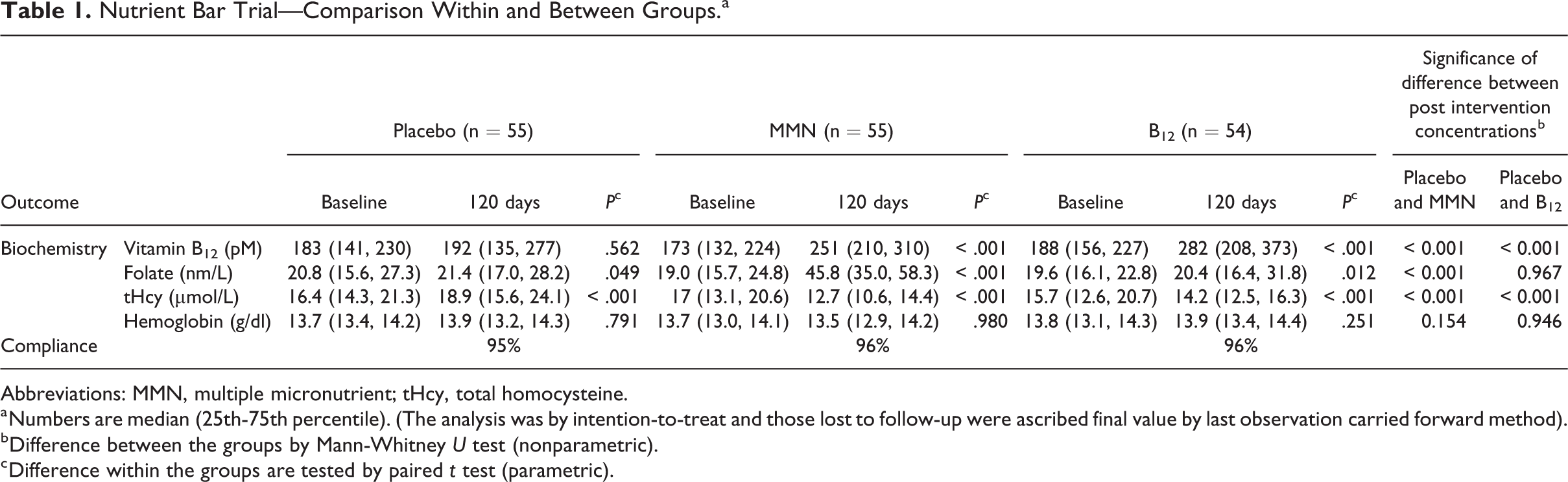
Abbreviations: MMN, multiple micronutrient; tHcy, total homocysteine.
a Numbers are median (25th-75th percentile). (The analysis was by intention-to-treat and those lost to follow-up were ascribed final value by last observation carried forward method).
b Difference between the groups by Mann-Whitney U test (nonparametric).
c Difference within the groups are tested by paired t test (parametric).
After intervention, B12 rose significantly by median 91 pmol/L in B12 alone group and by 82 pmol/L above the baseline in B12 + MMN group (P < .001, both), while there was no change in the placebo group (Figure 2). Folate concentrations rose significantly in the MMN group. Homocysteine reduced by median 1.4 and 3.8 μmol/L in the B12 alone and B12+ MMN groups, respectively (P < .001), and increased by median 1.9 μmol/L (P < .001) in the placebo group (Figure 2). Hemoglobin concentrations did not change with the intervention. There was an average gain of 4.3 (3.6-5.2) cm in height and 2.9 (2.0-4.4) kg in weight post intervention but no significant difference between the groups.
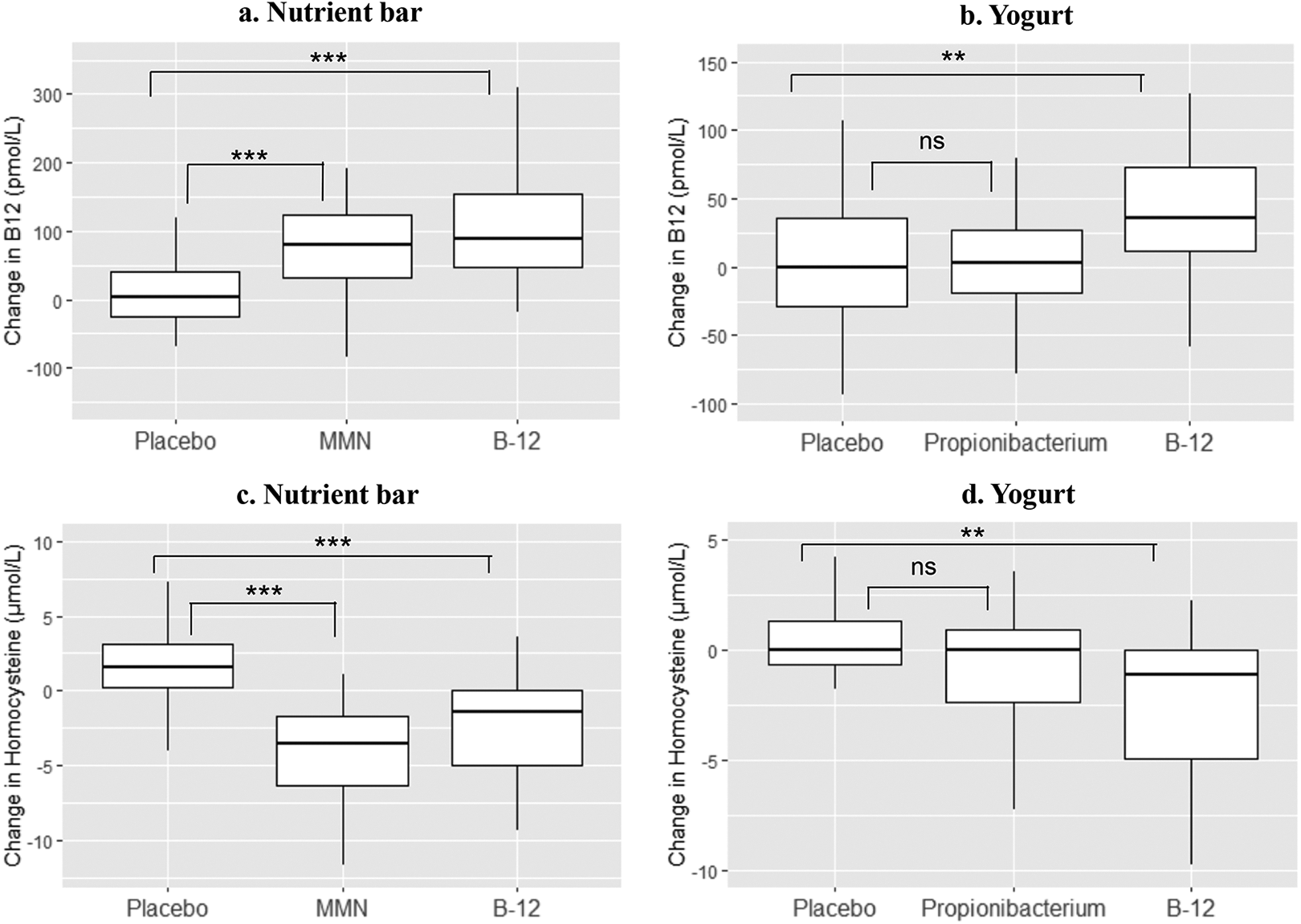
(Box plot) Change in plasma vitamin B12 and homocysteine concentrations after supplementation with fortified food products for 120 days. Differences between the groups are tested by Mann-Whitney U test. P values are given as *< .05, **< .01, ***< .001. B-12, vitamin B12 (2.0 μg); MMN, multi micronutrient (1.8 µg vitamin B12); NS, nonsignificant; Propionibacterium: Propionibacterium freudenreichi (1 × 108 cfu/g).
Comparison with Placebo
The post intervention concentrations of B12 and rise above baseline were significantly higher in both B12 supplementation groups compared to those in the placebo group. Rise and post intervention concentrations of folate were higher in the MMN group compared to the placebo and B12 alone group. Fall in homocysteine concentrations was significantly greater in both the B12 and MMN groups compared to the placebo group (Figure 2), the fall was greater in the MMN group compared to the B12 alone group (−3.8 vs −1.4 μmol/L, P = .001).
Mean adherence to daily nutrient bar consumption was high and similar in 3 groups (≥ 95%). One child dropped out during the trial because of change of school (Figure 1A). There were a total of 4 serious adverse events (hospital admissions for incidental illnesses), none related to the intervention product.
Predictors of B12 and Homocysteine Change
Apart from dose of B12, higher baseline concentrations of B12 predicted larger rise in B12 concentrations. Similarly, fall in homocysteine concentration was predicted by a higher B12 and folic acid dose, and higher baseline homocysteine concentration, especially in females (Table 2).
Multivariate Regression for Change in B12 and Homocysteine.
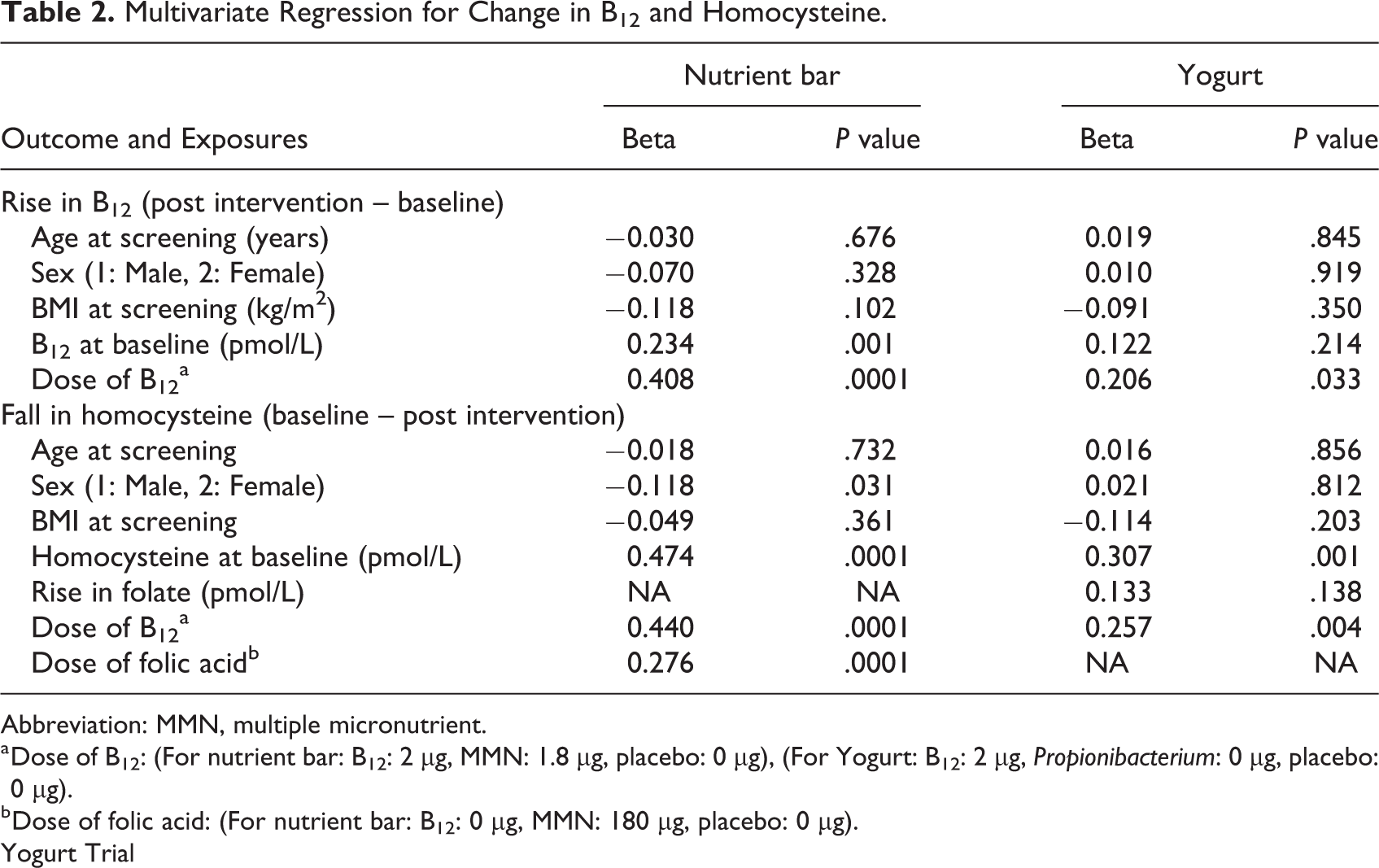
Abbreviation: MMN, multiple micronutrient.
a Dose of B12: (For nutrient bar: B12: 2 μg, MMN: 1.8 μg, placebo: 0 μg), (For Yogurt: B12: 2 μg, Propionibacterium: 0 μg, placebo: 0 μg).
b Dose of folic acid: (For nutrient bar: B12: 0 μg, MMN: 180 μg, placebo: 0 μg).
Yogurt Trial
Of 178 adult volunteers, 148 consented to participate. Thirty were excluded: 24 with B12 < 100 pmol/L, 2 with anemia and 4 due to medical disorders (Figure 1B). The 118 randomized adults (81% women) were 27.0 (27.8-35.0) years old, 157.5 (152.8-162.5) cm tall, with a weight of 56.5 (49.4-64.6) kg and BMI of 22.9 (19.7-26.0) kg/m2, there was no significant difference between groups. Plasma vitamin B12, folate, and homocysteine concentrations were comparable (Table 3).
Yogurt Trial: Comparison Within and Between Groups.a
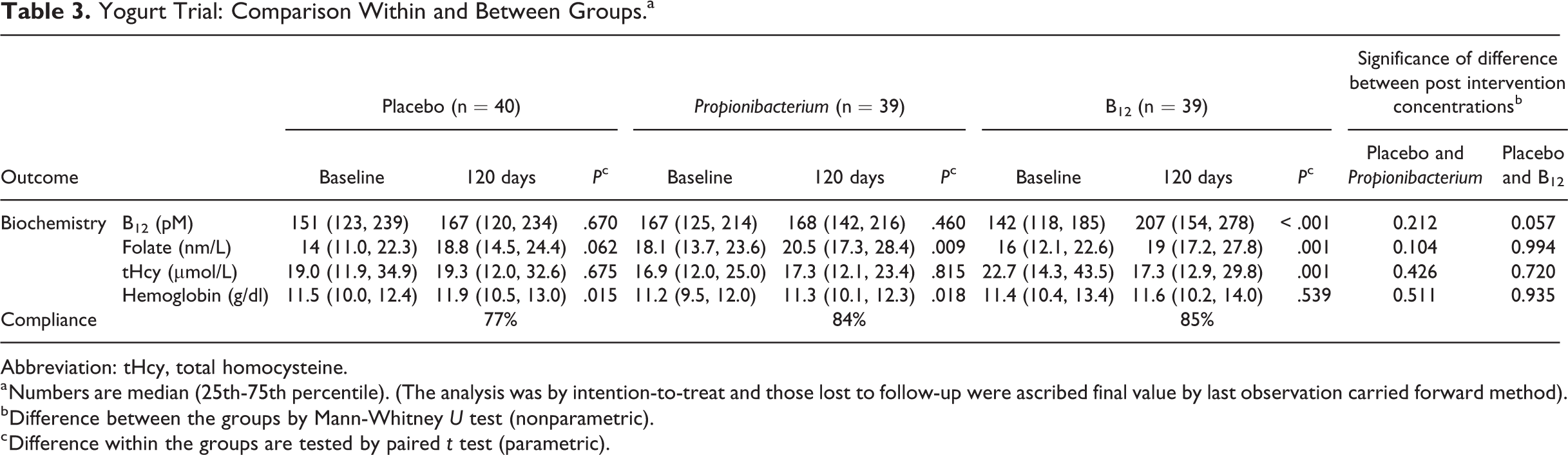
Abbreviation: tHcy, total homocysteine.
a Numbers are median (25th-75th percentile). (The analysis was by intention-to-treat and those lost to follow-up were ascribed final value by last observation carried forward method).
b Difference between the groups by Mann-Whitney U test (nonparametric).
c Difference within the groups are tested by paired t test (parametric).
After intervention, B12 rose significantly by median 38 pmol/L above the baseline in the B12 group (P < .001) and homocysteine decreased by median 2.7 µmol/L (P < .001; Figure 2). There were no significant changes in B12 or homocysteine in the Propionibacterium and the placebo groups. There was a small rise in folate concentrations in all groups, but no significant changes in hemoglobin concentrations and weight in any group.
Comparison with placebo
The rise in B12 concentration and fall in homocysteine concentration from baseline was higher in B12 fortified yogurt group compared to the placebo and Propionibacterium fortified groups (Figure 2).
During the intervention 6 participants discontinued from the B12, 9 from Propionibacterium and 9 from plain yogurt group; all for reasons not related to intervention. Ninety-four (82%) participants completed the trial (Figure 1B). Mean adherence for daily yogurt consumption was similar in 3 groups (≥ 82%). There was one serious adverse event (hospital admission for incidental illness) not related to the intervention product.
Predictors of B12 and homocysteine change
Dose of B12 in the intervention groups predicted larger rise in B12 concentrations. Similarly, fall in homocysteine concentration was predicted by the B12 dose, and higher baseline homocysteine concentration, but not by change in folate levels (Table 2).
Discussion
Our results show that regular consumption of B12 fortified nutrient bar or yogurt for 4 months significantly improved B12 status. Our population has high prevalence of B12 deficiency, and we used near Recommended Dietary Allowance doses in our trials to make it public health relevant. Directly observed therapy design ensured high adherence and allowed us to explore the full potential of fortification with these doses. Placebo comparison helped investigate the specific effects of vitamin B12 interventions. Nutrient bar and yogurt are 2 commonly eaten food items in India and were well accepted by the participants. Our results make it an attractive public health proposition. Rather disappointingly, the Propionibacterium probiotic at the recommended dose did not influence B12 status.
The response of circulating B12 concentrations was proportional to the dose of B12, and in turn reflected in reduction of circulating homocysteine concentrations. This indicates an improved methylation status of the body in both the children and the adults. Addition of other micronutrients (folic acid, B6, and B2) to B12 had an additional effect on lowering of homocysteine in children. In addition to the expected effect of the dose of B12, higher baseline concentration of B12 predicted a higher rise in B12 concentration. The latter finding was also seen in our previous study which showed that higher baseline B12 concentration predicted higher absorption of the supplemented vitamin. 8 Higher fall in homocysteine concentration was predicted by its higher baseline concentration and by dose of B12 and folic acid. In our trials, B12 supplementation had no effect on hemoglobin concentration which might be because of its satisfactory levels at baseline or a requirement for relatively higher doses of B12 for hematological effects. In another study of more severely B12 deficient adolescent girls, we were able to show improvement in hemoglobin concentration (and peripheral nerve function) with similar daily dose of vitamin B12 but continued over a longer period of 11 months. 18 Our results support fortification of commonly eaten food items as vehicles for improving B12 status in populations with low B12 status who do not have the problem of defective absorption.
There are only a few studies of B12 fortification to improve the vitamin status. They have been done in preschool children (MMN) 19,20 and in elderly population (B12), 21 the 2 groups commonly thought to be at risk of low B12 status. The vehicles for B12 were rice, milk, wheat flour (consumed as bread), common salt, and breakfast cereals. 19,20,22 -24 In a population with high burden of low B12 status, a multipronged food fortification program may be more successful than isolated food product fortification to improve the B12 status. In our own and others’ experience, supplementation with tablets in asymptomatic individuals suffers rapid reduction in adherence. Such supplementation also has complex logistic requirements in field operations in large populations.
We conducted our trials in a population with high prevalence of low B12 status, making it relevant to many such populations in India and other developing countries. 25 Participation rates were high. The placebo-controlled, randomized design ensured an unequivocal result and high-quality evidence for both trials. The use of DOT approach ensured near complete compliance (≥ 95%) in children, thus allowing full potential of the intervention to be exploited. Our findings will be directly applicable to vulnerable populations (children, adolescents, young adults, and pregnant women), where low B12 status may have major implications for growth, development, and reproductive health. Possible weaknesses include small sample size and a relatively short period of intervention. Relatively lower adherence in a DOT trial in the adults (average 82%) is a reflection of multiple demands on time in working middle class and not a reflection of the investigational product. Even at this attendance, there was a rewarding improvement in B12 status.
In summary, we used fortified versions of 2 commonly consumed food items to improve B12 status in deficient populations. This approach appears superior to use of vitamin tablets, which has proved difficult to sustain over longer periods of time in asymptomatic population. Both food items additionally provide calories, proteins, and other nutrients, and therefore could be easily adopted in national programs of feeding malnourished children and adults, as well as in the Mid-day Meal programs in schools. Both food items could be made at low cost by small-scale home industry promoting local economy. These facts will help the policy-makers to make appropriate decisions in deserving populations. Further research should investigate benefits of B12 supplementation to physical, cognitive, and reproductive outcomes in deficient populations.
Supplemental Material
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721211025448 - Efficacy of B12 Fortified Nutrient Bar and Yogurt in Improving Plasma B12 Concentrations—Results From 2 Double-Blind Randomized Placebo Controlled Trials
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721211025448 for Efficacy of B12 Fortified Nutrient Bar and Yogurt in Improving Plasma B12 Concentrations—Results From 2 Double-Blind Randomized Placebo Controlled Trials by Chittaranjan Yajnik, Sonal Kasture, Vaishali Kantikar, Himangi Lubree, Dattatray Bhat, Deepa Raut, Nilam Memane, Aboli Bhalerao, Rasika Ladkat, Pallavi Yajnik, Sudhir Tomar, Tejas Limaye and Sanat Phatak in Food and Nutrition Bulletin
Footnotes
Authors’ Note
C.Y., S.T., and P.Y. conceptualized and planned the study. S.K., V.K., H.L., R.L., P.Y., and C.Y. were involved in the trial conduct. D.B., D.R., and N.M. performed all the laboratory measurements. Statistical analysis was done by T.L. and A.B. C.Y., T.L., S.P., and D.B. wrote the manuscript.
Acknowledgments
The authors thank the team at Diabetes Unit, KEM Hospital Research Centre, Pune, for practical assistance during the trial (Charudatta Joglekar, Sonali Wagle, Vidya Mudliar, Komal Adawani, Rucha Wagh). The authors thank Dr Arun Nanivadekar and Dr Mohan Gupte for their guidance. The authors are grateful to the participants of both the trials.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Department of Bioechnology (DBT), New Delhi, India. DSM India kindly provided nutrient bars for the study. Hi Tech Biosciences India Limited, Pune, produced yogurt for the trial.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
