Abstract
Background:
Undernutrition is a leading risk factor for tuberculosis and is associated with adverse treatment outcomes. Energy-dense nutritional supplement (EDNS) may be helpful in managing undernutrition in tuberculosis patients.
Methods:
A longitudinal study was conducted among 102 newly diagnosed pulmonary tuberculosis patients in the Ballabgarh tuberculosis unit in Haryana, India, between September 2018 and February 2019. Participants were provided daily supplementation with 2 sachets of peanut-based EDNS providing 1000 kcal along with the tuberculosis chemotherapy for 2 months. Acceptability was assessed after one month of supplementation. Reported side effects were assessed every 15 days for 2 months and adherence, in terms of proportion of supplement consumed, was assessed on weekly basis for 2 months. Weight was measured at baseline and after 1 month and 2 months.
Results:
Mean age of participants was 33 years (standard deviation: 13.8) and 75.5% of the participants were male. Almost 100% of the participants found the taste, smell, consistency, and color of the EDNS acceptable. Three participants rejected it after one month of consuming EDNS and another 7 rejected after 2 months of consuming EDNS. After consuming EDNS for 2 months, 13.9% of the participants complained of nausea, 11.9% complained of vomiting, 10.9% complained of diarrhea, and 9.9% complained of pain in the abdomen and abdominal fullness or bloating anytime during the period of supplementation. The adherence to EDNS among the participants was reported as 92.6%.
Conclusion:
A peanut-based EDNS was highly acceptable and safe, and the adherence to it was high among patients with pulmonary tuberculosis.
Introduction
High prevalence of undernutrition has been reported among tuberculosis patients in India. A study reported that 55% of the tuberculosis burden can be attributed to undernutrition. 1 Undernutrition is associated with adverse outcomes among tuberculosis patients. 2 Nutrition supplementation can be an important adjunct to antituberculosis treatment to achieve better treatment outcomes and a supplementation of 800 to 1000 kcal/d has been recommended. 3 Nutritional supplementation has also been shown to improve the treatment adherence among tuberculosis patients. 4 Recognizing the role of nutritional therapy in management of tuberculosis, the Government of India has instituted a direct cash transfer of INR 500 (approximately US$7) every month to patients with tuberculosis for nutritional support. However, in an overall environment of food insecurity and poverty in the household, cash provided may not be sufficient to support the nutritional requirements in addition to the implementation challenges. 5 Energy-dense nutritional supplement (EDNS), in the form of a precooked, packaged, peanut-based paste made of sugar, vegetable oil, and skimmed milk powder enriched with vitamins and minerals, given along with the antituberculosis therapy may be one of the more efficient ways for nutritional supplementation among the tuberculosis patients. It is currently being used to treat malnutrition in severe acute malnourished children and is also widely being used in management of wasting among HIV-infected adults across the world.
A systematic review on the effect of nutritional supplementation on outcomes in patients with tuberculosis has reported lack of conclusive evidence in the form of randomized trials and recommended generation of more evidence. 6 Effectiveness of any intervention depends upon its acceptability and participant adherence to the intervention. Evidence on acceptability and adherence to EDNS in India among tuberculosis patients is limited 7 ; however, evidence in other population groups is equivocal. A study in India found the acceptability of EDNS in malnourished children to be 58%. 8 On the contrary, a study among children in Ethiopia, Ghana, Pakistan, and India, peanut-based EDNS was found highly acceptable. 9 However, acceptability among adults is likely to be different from children. Studies among adult patients in Senegal and Vietnam found variable acceptability but high adherence of peanut-based EDNS. 4,10 As a precursor to the trial on effectiveness of EDNS in improving treatment outcomes, we conducted a study to assess the acceptability and adherence to EDNS providing about 1000 kcal along with necessary micro and macronutrients, among adult malnourished pulmonary tuberculosis patients in Ballabgarh block of district Faridabad, Haryana, India.
Methods
This was a longitudinal study conducted among adults of age 18 years or more with pulmonary tuberculosis under Revised National Tuberculosis Control Programme in the Ballabgarh tuberculosis unit (TU) in district Faridabad, Haryana, India. Faridabad district has a population of around 1.8 million which are served by 9 TUs. Only those TB patients who were mild to moderately undernourished (body mass index [BMI]: 14-18.4 kg/m2) and permanent resident of Ballabgarh block were eligible for inclusion in the study. However, those with known allergy to any constituent of the nutritional supplement such as peanut, known diabetes, clinically icteric, pregnant or lactating, HIV infected, those with poor general condition, or inability to give valid consent were excluded. Eligibility was assessed through interview of the participants, clinical assessment, biochemical assessment, and measurement of their height and weight. A participant was excluded if on treatment for diabetes mellitus and/or if fasting blood sugar was more than 125 mg/dL. HIV testing is done routinely for all tuberculous patients; however, we did not specifically test for HIV. Urine pregnancy test was done for all female patients in the reproductive age group. The sample size for the study was 102 assuming an acceptability of 58% 8 and absolute precision of 10, confidence limit of 95% and nonresponse rate of 5%. All consecutive newly diagnosed pulmonary tuberculosis patients with at least 2 months of the antituberculosis treatment remaining on the tentative date of commencement of nutritional supplementation, in the first week of December 2018, were approached and were assessed for eligibility till the sample size was achieved.
Nutritional supplement was provided in 92 g aluminium sachet providing 500 kcal, and participants were instructed to consume 2 sachets every day. Composition of supplement is presented in Table 1 and it adhered to all safety norms prescribed by Food Safety and Standards Authority of India. Energy-dense nutritional supplement sachets were produced commercially and adhered to the World Health Organization standards, except iron, for ready-to-use supplementary food for community management of acute malnutrition. 11 Supplementation for one week was provided by the medical social workers at the Directly Observed Treatment, Short Course (DOTS) center once a week for 2 months as per the fixed schedule. First sachet of the week was consumed in front of the study personnel and the remaining supply, that is, 13 more sachets for a week were given to the participant for consumption at home. Study personnel instructed participants on how to store and consume the supplement at home. Phone calls were made daily by the study personnel to remind the participants about consuming EDNS. Participants were instructed to return the empty sachet in their next visit for refilling the supply for the week.
Composition of the Energy-Dense Nutritional Supplement (EDNS).
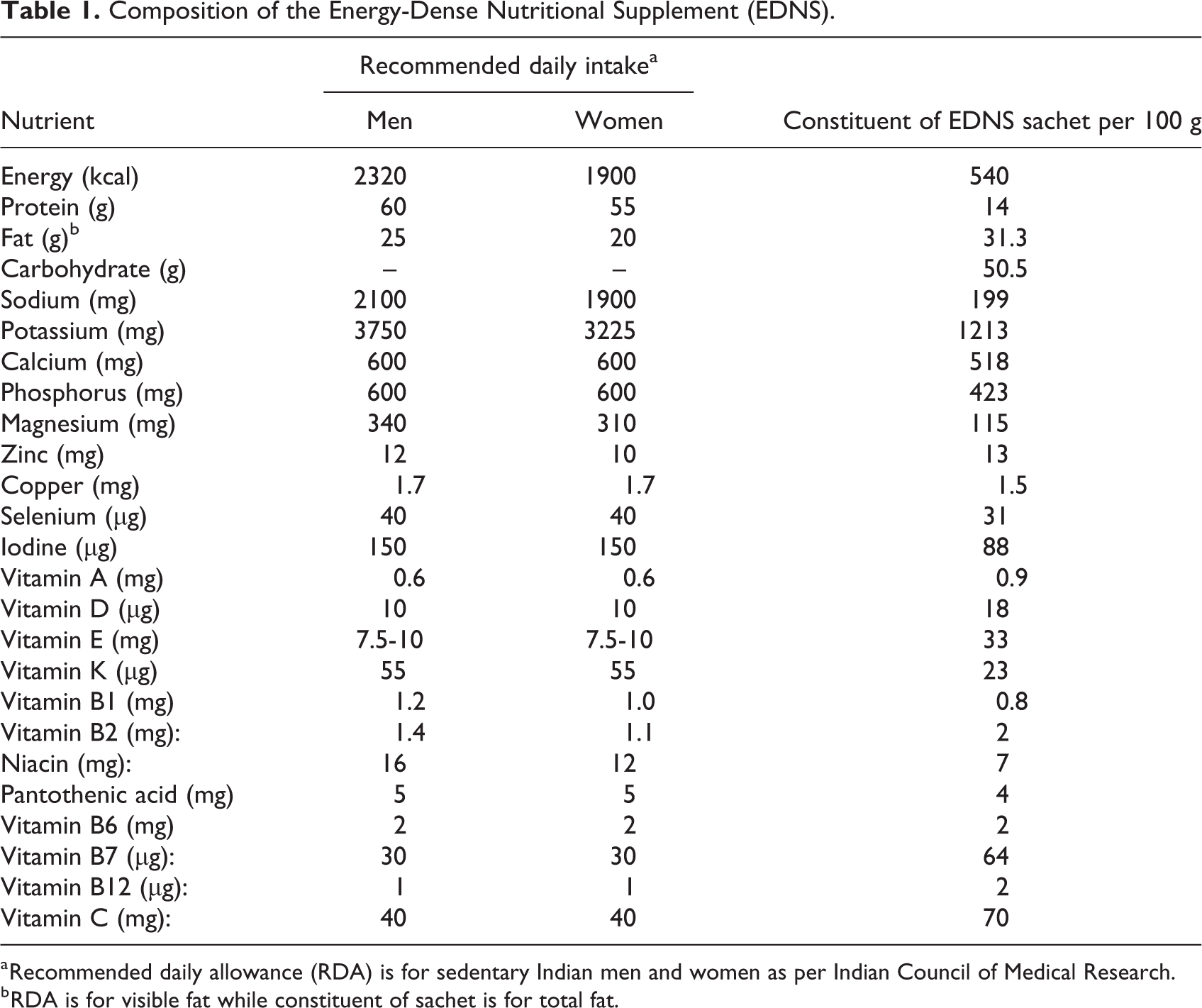
a Recommended daily allowance (RDA) is for sedentary Indian men and women as per Indian Council of Medical Research.
b RDA is for visible fat while constituent of sachet is for total fat.
Acceptability was assessed after one month of supplementation through participant interview using semistructured questionnaire which included perceptions of taste, smell, consistency, color, side effects, packaging, consumption (accepted readily, forced to ingest it, or rejected completely), time taken to consume nutritional supplement, and general appreciation of nutritional supplement. The participants responded to questions about acceptability parameters (taste, smell, consistency, color, packaging) as Yes or No, and if their response was No, they were further asked to explain the reason for the same. Similar approach has also been used in the past. 12 The nutritional supplement was considered acceptable if the patient did not perceive problems of undesirable taste, smell, color, consistency, did not feel that they were being forced to take supplement, or did not completely reject the supplement after one month of intake. Information on side effects including symptoms of food allergy was collected every fortnight through participant interview, and adherence was assessed through weekly participant interview about numbers of sachets consumed and supplement count during the period of supplementation of 2 months. Information on sociodemographic and clinical characteristics was collected at the baseline. All interviews were conducted by pretested semistructured questionnaires. In addition to the measurement of weight at baseline to assess eligibility, the weight was measured twice at monthly interval. Weight was measured to the nearest 100 g using calibrated electronic scales. The study was conducted between September 2018 and February 2019.
Statistical Analysis
Data were entered in Microsoft Excel and analyzed using Stata version 12 (StataCorp). Descriptive analysis was done for acceptability, frequency, and proportion of various acceptability parameters are presented. Side effects of supplement were presented as frequency and proportion of participants experiencing side effects any time during the period of follow-up. Adherence to the supplementation was reported as mean of percentage adherence with total possible number of sachets which could be consumed in 2 months of supplementation, that is, 120 as denominator and actual number of sachet consumed as numerator. Adherence for those who discontinued supplementation was counted as zero from the point of discontinuation. All means were reported with standard deviation. Independent t test and analysis of variance were used to assess the significance of difference in mean percentage adherence between various subgroups of participants at 5% significance level. We also assessed the correlation between adherence and weight gain.
Ethical Considerations
Ethical approval for the study was obtained from the Institute Ethics Committee of the All India Institute of Medical Sciences, New Delhi. Written informed consent was obtained from all participants.
Results
A total of 179 patients with pulmonary tuberculosis were approached to assess eligibility for inclusion in the study, of which 102 were found eligible. Mean age of study participants was 33 years (standard deviation [SD]: 13.8) and 43% were in the age group 25 to 49 years. Most of the participants (75.5%) were male and currently married (49%). Mean BMI of male participants was 16.5 kg (SD: 1.3 kg, range: 14.0-18.4 kg) and the mean BMI of female participants was 17.1 kg (SD: 1.2 kg, range: 14.6-18.4 kg). Sociodemographic details of the participants are presented in Table 2. Of the 99 participants who were still consuming the supplement at the end of one month of supplementation, 99% reported that they were consuming it as per instructions and 96% reported that they were eager to consume it. Everyone found the taste, smell, consistency, and color to be acceptable while 99% perceived an improvement in health after consuming the nutritional supplement (Table 3). After consuming EDNS for 2 months, 13.9% of the participants reported nausea, 11.9% reported vomiting, 10.9% reported diarrhea, and 9.9% reported pain abdomen, and abdominal fullness or bloating anytime during the period of supplementation (Table 3). Other symptoms included breathing difficulty, skin allergy, and oral ulcers, each symptom was reported by 2 participants. After one month, 3 participants had discontinued consumption and another 7 discontinued during the second month of supplementation; reasons cited for discontinuation were side effects (4), migration out of study area (2), dislike of taste (1), lack of interest (2), and unrelated medical condition (1).
Sociodemographic and Clinical Profile of the Study Participants With Pulmonary Tuberculosis in Ballabgarh Block, India.

Acceptability and Safety of Energy-Dense Nutritional Supplement (EDNS) Among Pulmonary Tuberculosis Patient in Ballabgarh, India.

a Attrition of 3 participants at the end of one month when acceptability was assessed.
b Attrition of one participant before side effect was assessed at the end of first fortnight.
As per participant interview, 11 340 sachets were consumed, and 11 274 sachets were returned out of a total possible supplementation of 12 240 sachets. Overall adherence after 2 months was 92.6% as per the participant interview and 92.1% as per supplement count. Mean number of sachets consumed was 111.2 (SD: 22.8). Adherence was higher among males, 18 to 24 years old, having a monthly family income of INR 5000 to 10000, and having BMI between 17 and 18.4. However, there was no statistically significant difference between various subgroups (Table 4). Mean weight gain was 4.2 kg (SD: 2.8) after one month of supplementation and 6.0 kg (SD: 3.3) after 2 months of supplementation, and there was a weak correlation between adherence and weight gain (correlation coefficient: 0.14).
Adherence to Energy-Dense Nutritional Supplement (EDNS) Among Pulmonary Tuberculosis Patient in Ballabgarh, India.
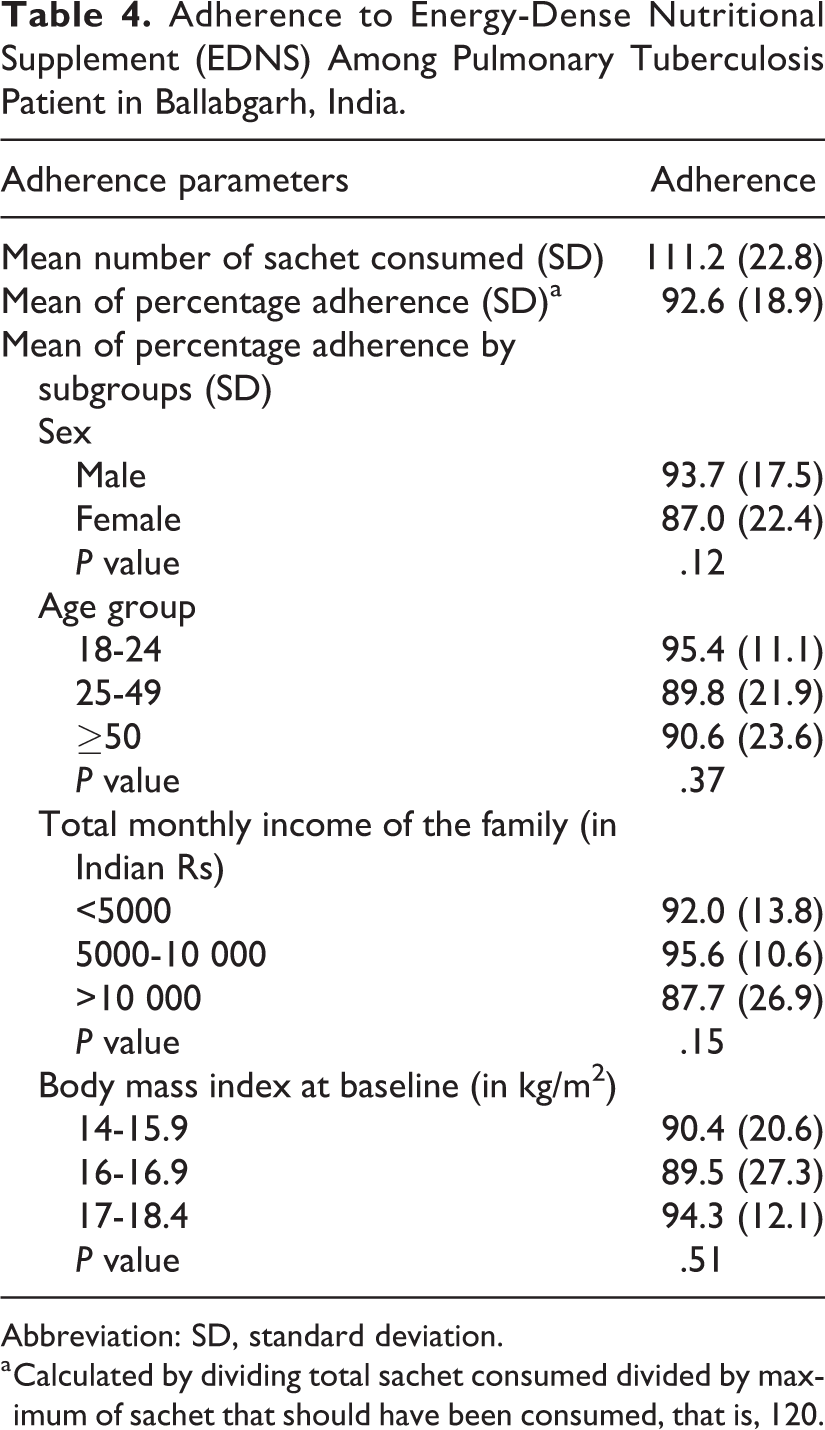
Abbreviation: SD, standard deviation.
a Calculated by dividing total sachet consumed divided by maximum of sachet that should have been consumed, that is, 120.
Discussion
This study among malnourished tuberculosis patients reported a very high level of acceptance and adherence of EDNS, few side effects and substantial weight gain. There are very few studies which have assessed the acceptability of peanut-based EDNS among adult tuberculosis patients. A study among HIV-TB coinfected patients in Senegal reported that 41.6% of the participants felt that the taste could be improved; 16.6% felt it was too salty, 16.6% felt it was both too salty and too sweet, while 8.3% felt it was too sweet. 4 The adherence to peanut-based EDNS in this study was 100%. However, the study was constrained by very low sample size. Another study in Vietnam among HIV patients reported an acceptability of peanut-based EDNS similar to locally produced EDNS bar and had an adherence of 81%. 10 Another study among community health workers in Bangladesh also found high level of dissatisfaction with taste, consistency, and smell of peanut-based EDNS. 12 In contrast, our study reported almost a universal acceptance of EDNS. This high acceptability and adherence could have been influenced by the perceived therapeutic benefit of EDNS as well as the persistent follow-up by the study personnel. We did not find any significant difference between various subgroups in adherence to the supplementation, probably due to inadequate power. An earlier study in India reported locally cooked EDNS as a low cost and feasible option for nutritional supplementation with a high level of compliance. 7
There was a weight gain of 6.0 kg after 2 months of supplementation which is more than what was reported in earlier trials from India and abroad. 7,13,14 However, there was weak correlation between adherence and weight gain and it is difficult to attribute the weight gain to supplementation without a comparator group as some weight gain is expected due to the antitubercular treatment. Supplementation in our study was delivered through DOTS centers, wherein supplements were provided for one week along with the antituberculous chemotherapy. As it comes prepacked and has long shelf life at room temperature, we did not face any operational challenge in its distribution through DOTS channel. The total cost of supplement for each patient per month was INR 1200 ($17).
There are certain limitations in the study in light of which the study findings should be interpreted. Adherence to the supplement was assessed through interview and sachet count. Although the first sachet was consumed in front of the investigator, there is a possibility that the sachets given for consumption at home could have been consumed by other family members. We also didn’t collect information on quantity of EDNS from the sachet consumed at home and it was assumed that the full quantity was consumed by the participant. The study was not powered to detect the difference in adherence between subgroups of the participants. As there was no control group, it is not possible to attribute the weight gain or reported side effects entirely to the supplement and as the period of follow-up was for only 2 months, it was not possible to assess whether the weight gain is sustainable. However, high level of acceptability and adherence to a peanut-based precooked prepackaged EDNS was demonstrated in our study. The effectiveness of EDNS in improving treatment outcomes and sustainable weight gain should be assessed through a randomized controlled trial. If proved effective, nutritional supplementation through DOTS centers could be a low cost and feasible option. The cost of supplement is also expected to decrease significantly if used on a large scale.
Studies have shown that incentives such as nutritional supplementation increases adherence to the antituberculosis treatment, 4,15 and this strategy can be used to decrease default rates and improve adherence in our settings. India has set a target to eliminate tuberculosis by the year 2025. 16 High levels of moderate to severe undernutrition have been reported in TB patients, both men and women, in India. From rural areas, the poor and marginalized sections, lower weights have been reported in TB patients. 3 This intervention, if found effective may contribute toward elimination of tuberculosis by improving treatment outcomes among large number of malnourished patients.
Conclusion
A peanut-based EDNS was highly acceptable and safe, and the adherence to it was high among patients with pulmonary tuberculosis.
Footnotes
Acknowledgments
The authors acknowledge the support of all the investigators and coinvestigators, our research staff, study participants, and other members of the community who helped us in conducting this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is funded by a grant from Indian Council of Medical Research.
