Abstract
Background:
Evidence suggests lack of understanding of the association of specific nutrients with different time points of linear growth trajectory.
Objective:
We investigated the role of dietary macro- and micronutrients on length-for-age z (LAZ) score trajectory of children across first 24 months of their life.
Methods:
The MAL-ED Bangladesh birth cohort study recruited 265 healthy newborn children after birth. The linear growth trajectory of those children was modeled using latent growth curve modeling (LGCM) technique.
Results:
Dietary magnesium intake at 9 to 11 months was positively associated (coefficient β = 0.006, P < .02) with LAZ at 12 months. Animal protein intake at 15 to 17 months, in turn, was positively associated (β = 0.03, P < .03) with LAZ at 18 months. However, vitamin D intake at 15 to 17 months was negatively associated (β = −0.06, P < .02) with LAZ at 18 months. Other micro- and macronutrients did not show any statistically significant association with the linear growth trajectory. We also found that birth weight (β = 0.91, P < .01), treating water (β = 0.35, P < 0.00), and maternal height (β = 3.4, P < .00) were positively associated with intercept. Gender had a significant negative association with the intercept, but a positive association with the slope (β = −0.39, P < .01; β = 0.08, P < .04), respectively. Conversely, birth weight had negative association with the slope (β = −0.12, P < .01).
Conclusions:
Dietary magnesium and animal protein were positively and vitamin D was negatively associated with the linear growth trajectory. Maternal height, birth weight, gender, and treatment of drinking water also played significant roles in directing the trajectory.
Keywords
Background
Physical growth is the most reliable global indicator of well-being and development in children. 1 The World Health Assembly 2025 global targets include multiple growth-related targets for stunting, wasting, and overweight during early and late infancy. 2 Stunting or linear growth faltering not only results in mortality, morbidity, and impaired cognitive development, but also results in poor school attainment and chronic diseases in later life. 1 Globally, a total of 150.8 million children are stunted. 3 Although the rate of stunting is gradually declining, at current pace it is anticipated we would be able to alleviate only 44% of the burden by 2030. 3
Stunting or linear growth faltering starts during the intrauterine period and can continue until 24 months of age and beyond. 4 Maternal malnutrition, frequent infections, and inadequate dietary intakes are known to play crucial roles in determining the trajectory of linear growth of a child. 5
Multiple studies have reported the role of different macro- and micronutrients on linear growth trajectory. Inadequate complementary feeding is an important contributor to poor growth among 6 to 23-month-old children living in developing countries. 6 An analysis of data from 2 large cohort studies has reported the importance of dietary protein for ensuring adequate growth of children. 7 The MAL-ED study found that lower percentage of energy derived from dietary protein intake had strong association with lower length-for-age. 5 However, in addition to poor energy intake, diets that are not adequately diversified and poor in bioavailability of micronutrients can equally hinder length gain. 8 Zinc, an essential nutrient, is a component of many enzymes that regulates cell growth and so is important for the metabolism of growth. 9 Zinc, iron, and magnesium deficiencies contribute to anorexia and therefore are important risk factors for growth retardation as a result of reduced intake of protein and energy. 10
Adequate intake of dairy products can influence calcium balance and bone mineralization and thus ensures proper bone growth, consequently linear growth. 11 Different population-based intervention studies have also identified strong associations between dietary vitamin A, vitamin D intake, and growth. 10,12 Zinc, iron, and vitamin A deficiencies are also associated with reduced immune function and increased risk of morbidity which can also contribute to growth impairment. 13 Accordingly, micronutrient deficiencies have both direct and indirect effects on growth by increasing both the prevalence and severity of disease and debility.
However, very few studies have measured the association of dietary nutrients with different time points of linear growth trajectory. To address this gap we aimed to investigate the role of macro- and micronutrients on linear growth trajectory of children across the first 24 months of age by applying latent growth curve models to the MAL-ED birth cohort data.
We wanted to address how the intake of specific dietary macronutrients and micronutrients at different time points in this interval may affect growth and so provide insight into the optimal timing and dosage of supplementation to improve growth. This analysis was partly based on the underlying effect these macro- and micronutrients may have on the biology of bone growth and related processes. Proteins and micronutrients such as zinc, potassium, magnesium, and vitamin D have been reported to have an effect on growth hormone and insulin-like growth factor 1 that are key hormones involved in bone development and growth at critical points in this period.
Methods
Setting
The MAL-ED (Etiology, Risk Factors, and Interactions of Enteric Infections and Malnutrition and the Consequences for Child Health) study is a longitudinal birth cohort study which was conducted in Bauniabadh slum of Mirpur, Dhaka, Bangladesh. 14 Following a well-defined protocol, field workers visited households between February 2010 and February 2012 and enrolled 265 healthy newborn children within the first week of birth. Field workers were able to follow 211 children for 24 months which was 80% of total recruitment. The study was approved by the research and ethics review boards of icddr, b, and informed written consents were collected from the mothers/caregivers of the children.
Data Collection
Monthly anthropometric measurements were conducted according to the standard anthropometric methodology. 15 Children were weighed with very minimum clothing using a Seca digital scale (model no. 345, Hamburg, Germany, 10 g precision), and a Seca infantometer (model no. 417, Hamburg, Germany) was used to measure the recumbent length to the nearest 1 mm. The length-for-age z (LAZ) score for each child was determined using the World Health Organization (WHO) 2006 child growth standards. 16
From study participants, 24-hour food intake data were collected monthly beginning at 9 months of age to assess nutrient intake of the children using a 24-hour multiple-pass dietary recall approach. 17 Trained research staff used visual aids (pictures of portion sizes and standardized measuring utensils) and interviewed the mothers to quantify the amount of different foods their children consumed during the previous 24 hours. A second dietary recall interview was conducted on nonconsecutive days with 10 to 20 randomly selected participants. The mothers were not notified in advance in order to remove potential bias and to ensure maximum quality of the collected data. A locally adapted food composition table was used to convert the dietary intake data to nutrients. 18
Information on water, sanitation and hygiene (WASH), and socioeconomic status (SES) was collected using pretested questionnaires. Demographic and SES questionnaires were adopted from the Demographic and Health Survey (DHS) questionnaires, and water and sanitation sources were defined as improved (or not) based on the WHO criteria. 19
Data Management
The precise estimate of nutrient intake depends on the availability of a food, source of a nutrient, and other population specific factors. Accuracy of such estimate is inversely related to the amount of within-subject variation of nutrient intake. Because of this variation, a single 24-hour recall could provide imprecise estimates of usual intake. 20 It is assumed that, in presence of such a greater variability, the data collection process should be replicated regularly to achieve a higher level of precision. And, it is also assumed that the precision of an individual’s energy intake averaged from multiple dietary recalls is quite high. 20 The MAL-ED study collected one 24-hour dietary recall per child each month and 15 such measures were recorded from each child from 9 to 23 months. We took this opportunity and averaged the data on dietary macro- and micronutrient intake for the 3-month periods of 9 to 11, 12to 14 and 15 to 17, 18 to 20, and 21 to 23 to capture more precise information of dietary intake over the entire time points. Associations of the 3-month averages of macronutrient and micronutrient intake with LAZ score (outcome variable) of the subsequent month were then determined. The averaged dietary intake factors were assumed to vary between individuals with time. All the other covariates were kept as time invariants.
Statistical Analysis
We reported the maternal-, child-, and household-level characteristics of the study participants using means and standard deviation (SD) for normally distributed continuous variables, and frequency and percentage for categorical variables. A conceptual framework was developed to describe the role of dietary and other factors on LAZ score trajectory. Bivariate linear regression was used to explore the associations of each of the dietary macro- and micronutrients, household-level factors, and LAZ. Animal protein, fat, carbohydrate, iron, zinc, calcium, magnesium, phosphorus, vitamin A, and vitamin D—a total of 10 nutrients were found to have statistically significant (P value < .2) association with LAZ score. The growth trajectory model was then estimated under the statistical framework of LGCM. 21 Latent growth curve modeling is a form of longitudinal analysis within the framework of structural equation modeling that estimates growth trajectories of repeated measures of dependent variables as a function of time and other measures. 22 Latent growth curve modeling reports the intercept factor of a model at the level of the outcome variable at which the time variable is zero. The slope factor, on the other hand, presents the linear change of outcome variable with time. Initially an unconditional model (model 1; Supplementary Figure 1) was developed which included only the intercept and slope as latent variables for the trajectory of the outcome variable LAZ score without adjusting for any covariates. A quadratic term was then added to determine whether this trajectory could be modeled as an upturn or downturn over time beyond a linear effect and so provide a better model fit (model 2; Supplementary Figure 2). Model selection was based on Akaike information criterion (AIC), Comparative fit index, root mean square error of approximation, and standardized root mean square residual. According to the goodness-of-fit statistics, the model with quadratic term showed a better fit than the initial model (Supplementary Table 1). The unconditional model with quadratic term was used for building the subsequent latent growth curve models.
The child-, maternal-, and household characteristics that showed significant relationship with LAZ in the bivariate analysis was added to this unconditional model as time-invariant covariates. We included the household characteristics collected at baseline as we found that data were essentially unchanged between the 2 time points that they were collected. Individual 3 month-lagged macro- and micronutrient intakes were then added incrementally as time-varying covariate keeping the time-invariant covariates fixed. To control for WASH factors in child growth, WASH household variables were included as part of the household characteristics. Model selection at this step was based on AIC and model showing lowest AIC value was selected for subsequent analysis (model 3, Supplementary Table 1). We also tried to measure the association of the time-invariant covariates gender and maternal height with the macro- and micronutrient covariates (model 4, Supplementary Table 1). But the model did not prove to be a better fit than the model (model 3) reported at this manuscript. The LGCM analyses were conducted using MPlusv8. 23
Results
Sociodemographic Characteristics
Table 1 describes the sociodemographic characteristics of the participants. Mean (SD) of birth weight and birth length of the cohort was 2.82 (0.42) kg and 48.30 (2.02) cm, respectively. Mothers had a mean (SD) height of 149.03 (5.05) cm. At 24 months, the overall male-to-female ratio was 1.04. Only 10.9% of children were exclusively breastfed (received only breast milk for the entire first 6 months). Majority of study households were headed by a male family member. A total of 84.4% mothers were between 18 and 30 years old, 39.8% mothers were younger than 18 years during their first pregnancy, and only 19.4% mothers were illiterate. Approximately, 44.5% households had a monthly income less than 100 USD and 64% of the households treated the drinking water to make it safe.
Sociodemographic Characteristics of the Participants.
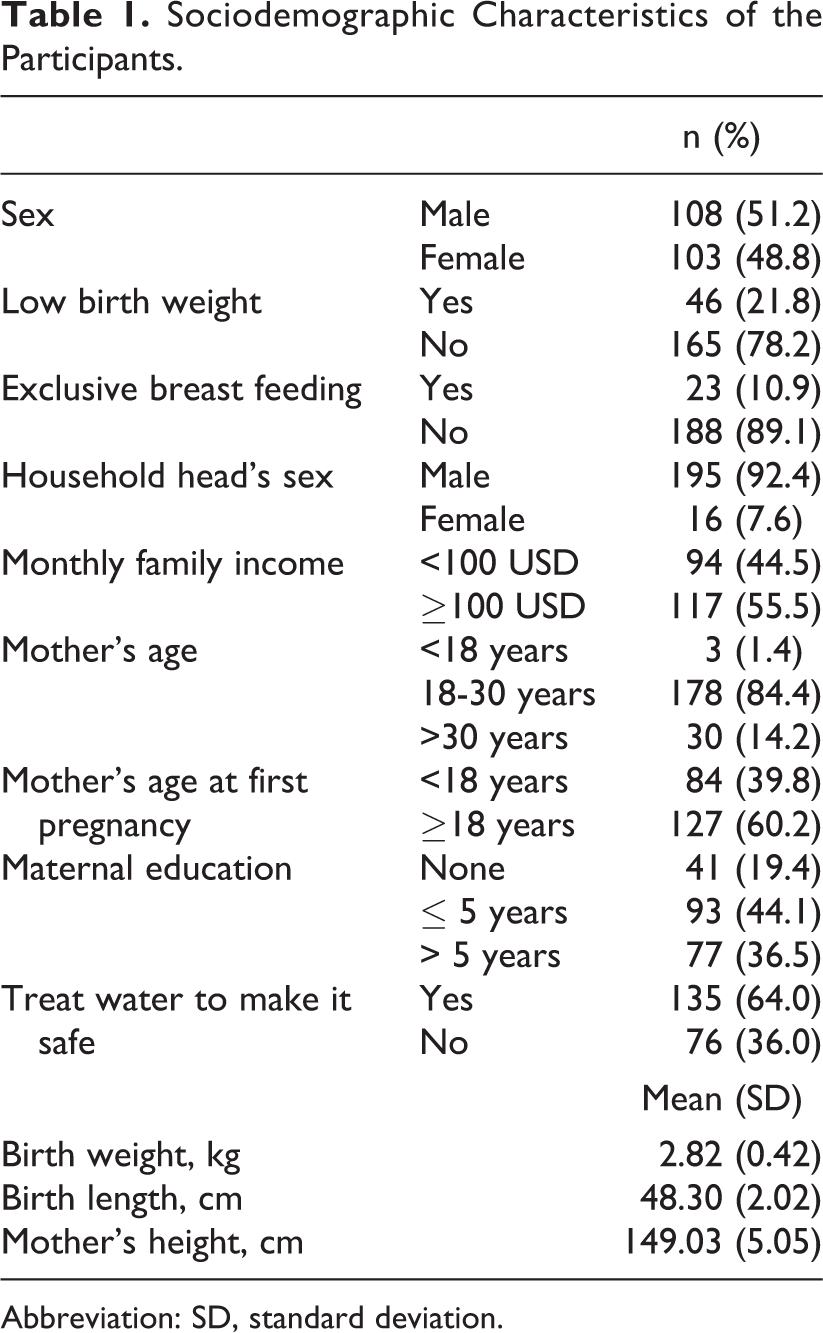
Abbreviation: SD, standard deviation.
Centile Curves of LAZ Score
Figures 1 and 2 present the centile curves (0.5th-99.5th percentiles) of LAZ trajectory of male and female children, respectively, from birth to 24 months. The curves show that female cohort started with an LAZ score (median LAZ −0.97) very close to their male counterparts (median LAZ −1.03). They also experienced more changes in their slopes than the male children. The male–female gap widened with time and at 24 months of their age female cohort’s median LAZ score was −1.91 and male’s median LAZ was −2.08.
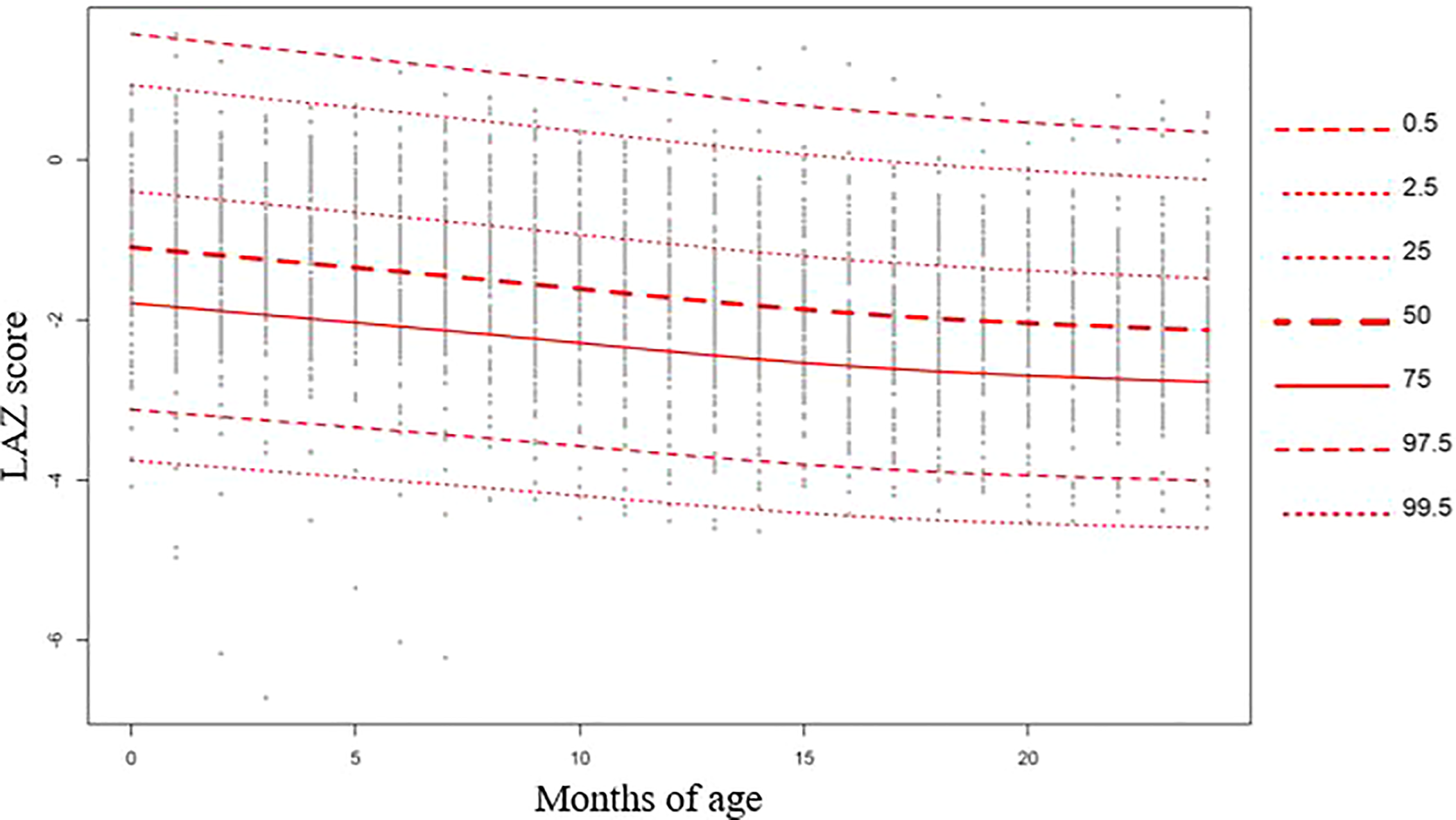
Centile curves (0.5th-99.5th percentiles) of LAZ trajectory of male children from birth to 24 months.
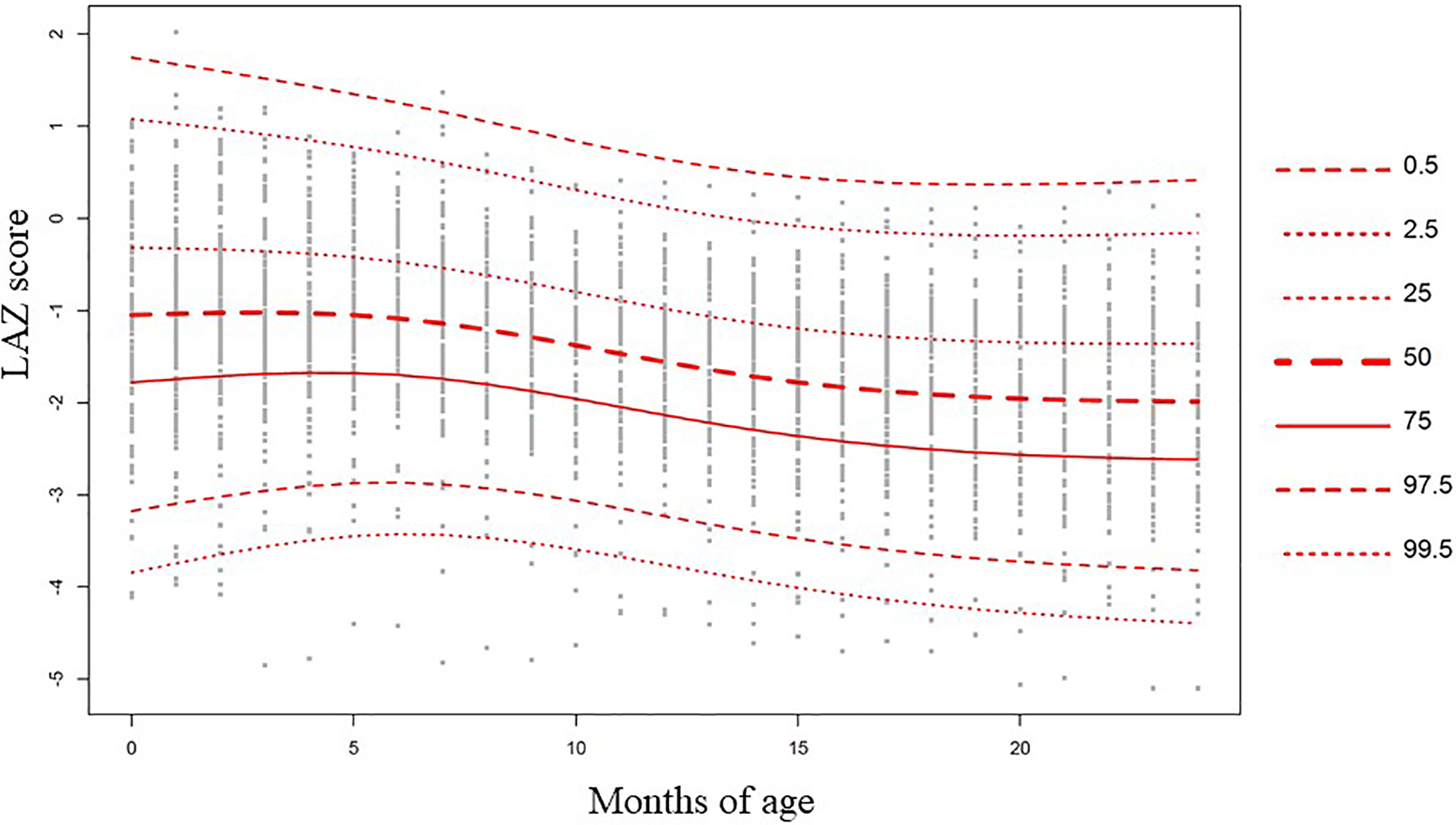
Centile curves (0.5th-99.5th percentiles) of LAZ trajectory of female children from birth to 24 months.
Latent Growth Curve Model
Table 2 reports the parameters of association of LAZ score trajectory with dietary macro- and micronutrients and household level factors and Figure 3 presents the explanatory variables those had statistically significant (P < .05) association with the outcome variable. In the LGCM analysis, male gender had a significant negative association with the intercept but a positive association with the slope factor (coefficient b = −0.39, P < .01; b = 0.08, P < .04, respectively). In contrast, birth weight had a positive association with the intercept but negative associations with the slope (b = 0.91, P < .01; b = −0.12, P < .01). Water treatment (b = 0.35, P < .00) and maternal height (b = 3.4, P < .00) were positively associated with intercept. The testing of relationship between individual 3 month-lagged macro- and micronutrient intakes with LAZ found that dietary magnesium intake at 9 to 11 months was positively associated (b = 0.006, P < .02) with LAZ at 12 months. Animal protein intake at 15 to 17 months, in turn, was positively associated (b = 0.03, P < .03) with LAZ at 18 months. However, vitamin D intake at 15 to 17 months was negatively associated (b = −0.06, P < .02) with LAZ at 18 months. The LAZ score was not associated with intake of any macro- or micronutrients at the later phases of infancy (21-24 months).
Estimated Parameters of Association of LAZ Score Trajectory With Dietary Macro- and Micronutrients and Household Level Factors.
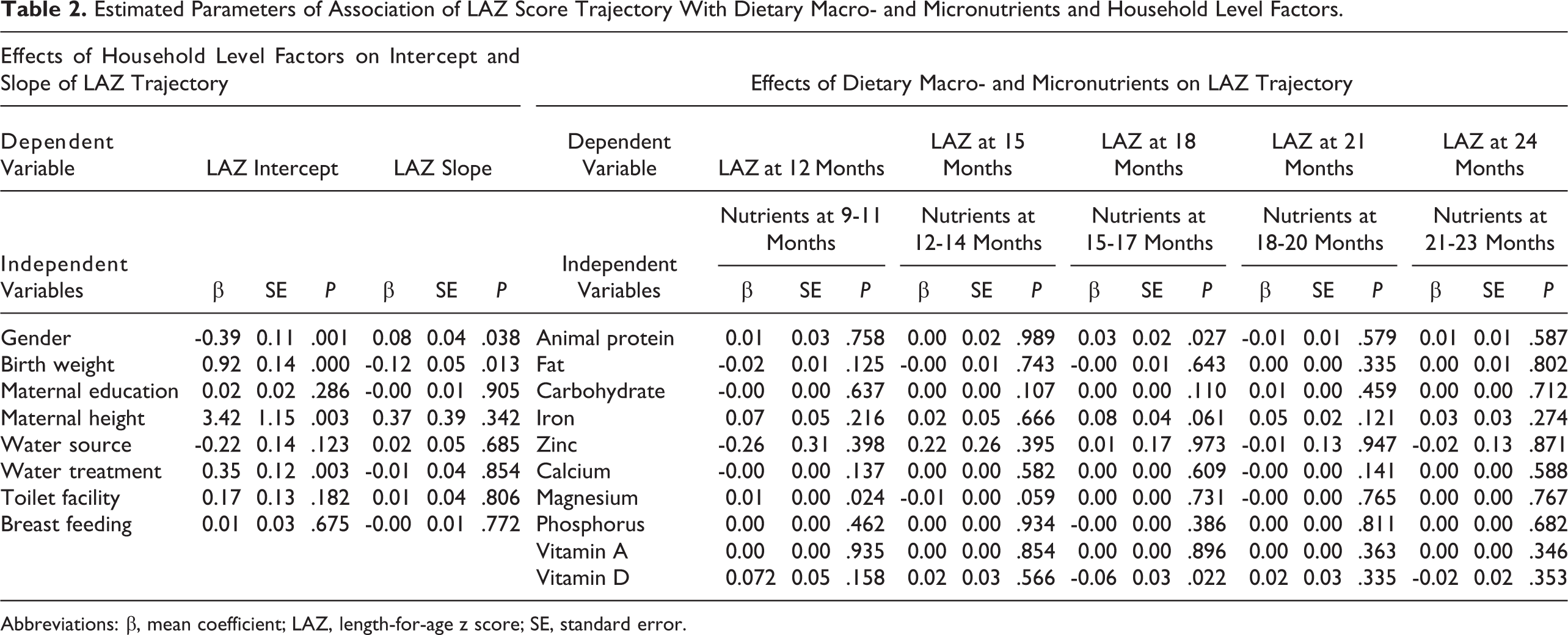
Abbreviations: β, mean coefficient; LAZ, length-for-age z score; SE, standard error.
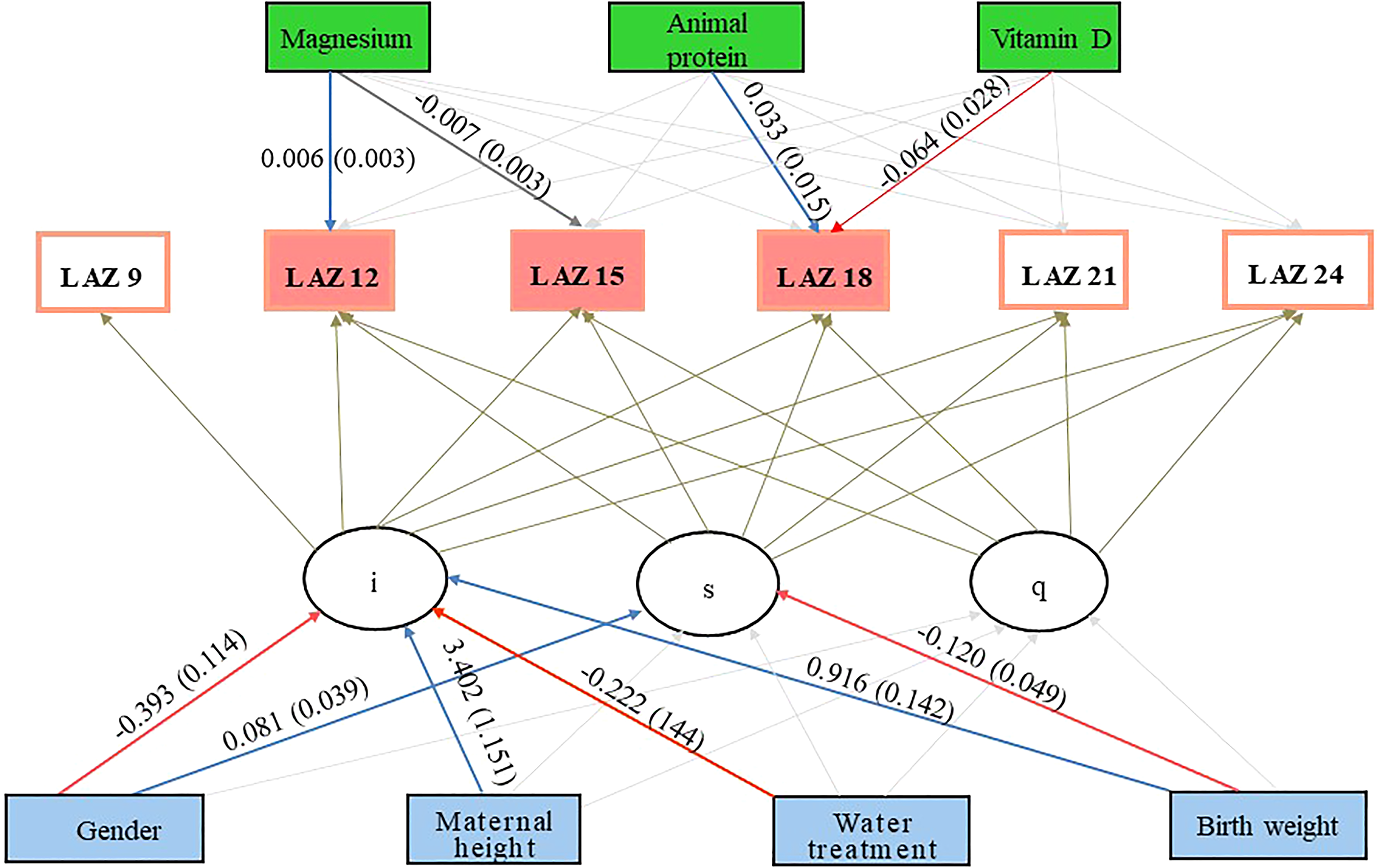
Conditional latent growth curve model showing the association of dietary macro- and micronutrients and sociodemographic factors on linear growth trajectory of 9 to 24 months old Bangladeshi children; i = intercept; s = slope; q = quadratic term. Positive associations are annotated by “blue” arrows and negative associations are annotated by “red” arrows.
Discussion
We have applied an LGCM analytical approach to investigate the role of different macro- and micronutrients on linear growth trajectory of children across the first 24 months of their life. Previous analyses of the MAL-ED Bangladesh cohort data using population specific “generalized estimating equation” models did not find associations of any of the macro- or micronutrients with growth. 18,24 But, in addition to modeling the repeated measures of a variable, LGCM also took into account the 2 other aspects of change—intercept and linear slope factors. With the aid of the LGCM, we have found that dietary magnesium and animal protein were positively associated with LAZ scores, while vitamin D intake was negatively associated to it. Other micro- and macronutrients did not show any statistically significant association with the linear growth trajectory. We also found that birth weight, treatment of drinking water, and maternal height were positively associated with intercept or the model factor at which the time variable is zero. Gender had a significant negative association with the intercept, but a positive association with the slope or the linear change of outcome variable with time. Conversely, birth weight had negative association with the slope.
Dietary magnesium was found to have a significant association with LAZ score among children at 12 months of age. There are few studies reporting on the relationship between magnesium intake and child growth that would support our findings. However, Guatemalan children who were having edematous protein caloric malnutrition had an increased rate of recovery following magnesium supplementation. 25 Another study carried out by Singla et al reported that malnourished children had low magnesium levels in relation to their nonmalnourished counterparts. 26
Magnesium is known to be the second most important intracellular cation of the human body. 26 It has critical roles in the glycolytic pathway, cyclic adenosine monophosphate formation, and fatty acid and protein synthesis. 27 Deficiency of magnesium is known to result in a compromised primary- and secondary immune response. 28 In animal models, magnesium deficiency has been found to be associated with low plasma Insulin-like growth factor 1, which reverts to normal levels once the deficiency has been corrected. 29 Severe magnesium deficiency can modify the secretion and/or skeletal responsiveness to vitamin D metabolites. 30 Moreover, reduction of dietary magnesium was reported to be related to altered formation or action of 1,25(OH)(2)-vitamin D in rats. 31
Vitamin D plays crucial roles in protecting human body from infections and inflammatory diseases. 32 It is known to regulate the metabolites of bone mineral metabolism and hence skeletal development. 33 Results published from the MAL-ED Bangladesh project have reported that the dietary intake of micronutrients in this cohort was inadequate. 24 Data from the same setting also revealed that older aged underweight and normal-weight children had higher odds of vitamin D deficiency than their younger counterparts. 32 Moreover, low calcium and high phytate content of Bangladeshi diet may accelerate vitamin D utilization—the outcome of which is a relative increase in vitamin D demands. 34 These results may partially explain the findings of our analysis that vitamin D intake at 15 to 17 months was negatively associated with LAZ score at 18 months. However, as the role of vitamin D in linear growth is intertwined with that of dietary calcium and parathyroid hormone, in the absence of information on the latter variable, it is difficult to explore the apparent paradoxical relationship. Community-based descriptive studies are needed to provide further insight into these relationships. Such studies should also explore the role of genetic polymorphisms of vitamin D receptor protein for predicting bone mineral density as Yoshizawa et al reported that mice lacking the vitamin D receptor failed to thrive after weaning. 35
We found that dietary intake of animal protein during 15 to 17 months was positively associated with LAZ score at 18 months of age of the children. This finding is supported by the multicountry analysis of MAL-ED birth cohort data, which reported that lower percentage of energy from dietary protein was strongly associated with a decline in length-for-age. 5 Moradi et al also reported that access to animal protein better predicts height than other energy sources. 36 Esteban et al conducted a study to compare the relative importance of proteins in children’s diets and found that interventions concentrating on protein intake might have the greatest beneficial effect on linear growth of children. 7 This result is supported by an additional study that found that early introduction of eggs, which is a good source of animal protein, can significantly improve growth in young children. 37
The relationship between maternal height, birth weight, and linear growth during early childhood is well established. 5,18 Our study has provided additional unique findings that show that birth weight and maternal height had significant positive associations with the intercept term while birth weight was negatively associated with the rate of LAZ score change. These results can be interpreted to mean that the children with greater birth weight and whose mothers were of greater height had better LAZ score at baseline. This finding also suggests that, though such children had more chances to attain a better length-for-age, such odds could be altered by the presence of other factors such as morbidity, infection, and dietary inadequacy. 1,5 Given this, we hypothesize that the growth-promoting role of the inborn factors could be modified or distorted by the presence of growth-limiting factors and those factors could act more proximally to growth trajectory than the birth characteristics.
We also found that treatment of drinking water was positively associated with intercept of the linear growth trajectory. Unsafe water acts as a major barrier to the safe preparation of complementary foods, and hence can inversely modify the potential required for linear growth accretion. 38,39
The strength of this analysis is efficient application of LGCM to explore the role of different micro- and macronutrients on intercept and slope factors of linear growth trajectory over time. But, we do have limitations to report. Our analysis revealed that male gender—the reference category of gender for this analysis—was negatively associated with the intercept but positively with the slope or rate of change of linear growth trajectory. Centile curves for LAZ score shows that female children had a better LAZ score at birth but experienced more changes in the trajectory. We could not explain this relation. Moreover, except dietary magnesium, animal protein, and vitamin D, other micro- and macronutrients did not show any statistically significant association with the linear growth trajectory. As statistical associations do not implicate causality, the absence of such statistically significant associations might be due to the absence of related interactions among the nutrients. Hence, making concrete conclusions regarding those relations might not be possible.
Conclusion
In summary, this study found that dietary magnesium and animal protein were positively associated with LAZ score trajectory, while vitamin D intake was negatively associated to it. We also found that maternal height, birth weight, gender, and treatment of drinking water can play deterministic roles in directing the linear growth trajectory. However, the association is not implicating causality and intervention studies are required to determine that.
Supplemental Material
Supplementary_figure_1 - Dietary Magnesium, Vitamin D, and Animal Protein Intake and Their Association to the Linear Growth Trajectory of Children from Birth to 24 Months of Age: Results From MAL-ED Birth Cohort Study Conducted in Dhaka, Bangladesh
Supplementary_figure_1 for Dietary Magnesium, Vitamin D, and Animal Protein Intake and Their Association to the Linear Growth Trajectory of Children from Birth to 24 Months of Age: Results From MAL-ED Birth Cohort Study Conducted in Dhaka, Bangladesh by Subhasish Das, J. Johanna Sanchez, Ashraful Alam, Ahshanul Haque, Mustafa Mahfuz, Tahmeed Ahmed and Kurt Z. Long in Food and Nutrition Bulletin
Supplemental Material
Supplementary_figure_2 - Dietary Magnesium, Vitamin D, and Animal Protein Intake and Their Association to the Linear Growth Trajectory of Children from Birth to 24 Months of Age: Results From MAL-ED Birth Cohort Study Conducted in Dhaka, Bangladesh
Supplementary_figure_2 for Dietary Magnesium, Vitamin D, and Animal Protein Intake and Their Association to the Linear Growth Trajectory of Children from Birth to 24 Months of Age: Results From MAL-ED Birth Cohort Study Conducted in Dhaka, Bangladesh by Subhasish Das, J. Johanna Sanchez, Ashraful Alam, Ahshanul Haque, Mustafa Mahfuz, Tahmeed Ahmed and Kurt Z. Long in Food and Nutrition Bulletin
Supplemental Material
Supplementary_table_1 - Dietary Magnesium, Vitamin D, and Animal Protein Intake and Their Association to the Linear Growth Trajectory of Children from Birth to 24 Months of Age: Results From MAL-ED Birth Cohort Study Conducted in Dhaka, Bangladesh
Supplementary_table_1 for Dietary Magnesium, Vitamin D, and Animal Protein Intake and Their Association to the Linear Growth Trajectory of Children from Birth to 24 Months of Age: Results From MAL-ED Birth Cohort Study Conducted in Dhaka, Bangladesh by Subhasish Das, J. Johanna Sanchez, Ashraful Alam, Ahshanul Haque, Mustafa Mahfuz, Tahmeed Ahmed and Kurt Z. Long in Food and Nutrition Bulletin
Footnotes
Authors’ Note
S.D. and J.J.S. are the co-first authors. S.D., J.J.S., T.A., and K.Z.L. conceived the study. S.D., J.J.S., M.A.A., M.A.H., M.M., T.A., and K.Z.L. developed the overall analysis plan. M.A.A. managed the data set and provided technical support. M.A.A. and S.D. analyzed the data. S.D. and K.Z.L. developed the tables/graphs and wrote the initial draft of the manuscript. All authors critically reviewed the manuscript and gave intellectual inputs.
Acknowledgments
The authors would like to thank all the participants and their parents for sharing their time and providing consent and information necessary for the successful completion of the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This research protocol (MAL-ED birth cohort study) was funded by University of Virginia (UVA) with support from MAL-ED Network Investigators in the Foundation of National Institute of Health (FNIH), Fogarty International Centre (FIC) with overall support from the Bill & Melinda Gates Foundation (BMGF), grant number GR-681; icddr, b acknowledges with gratitude the commitment of BMGF to its research efforts. icddr, b is also grateful to the Governments of Bangladesh, Canada, Sweden, and the UK for providing core/unrestricted support.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
