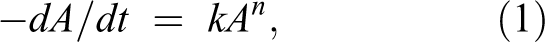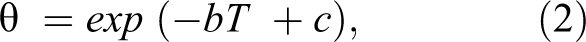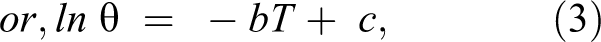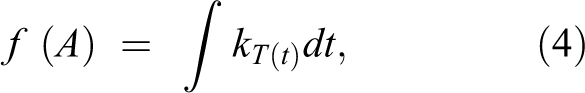Abstract
Background:
Providing nutritionally adequate complementary foods is essential in preventing malnutrition in young infants. Koko made from fermented corn dough constitutes the most common complementary food in Ghana. However, these do not meet the nutrient requirements.
Objective:
To develop and test the acceptability and shelf-life of an energy-dense complementary food supplement for improving the nutritional profile of traditional complementary foods.
Methods:
Surveys of families in urban and rural communities and stakeholder engagements with health professionals were done to identify the most predominant traditional complementary foods and young infants’ feeding practices. A food supplement, KOKO Plus, that would improve the nutritional profile of koko and other complementary foods needed to be developed. Linear programming was used in ingredient formulations for the food supplement based on locally available foods. The acceptability of the food supplement, KOKO Plus, in koko and soup was assessed using 14 trained assessors in a 2 × 2 crossover design. Shelf-life of the product was estimated using the difference in control sensory test method at different storage temperatures (24°C, 34°C, and 44°C).
Results:
Koko was the most predominant complementary food in many homes. The KOKO Plus food supplement improved the nutritional profile of koko to satisfy the nutrient intake requirements of young infants as recommended by World Health Organization. KOKO Plus was microbiologically safe, with estimated shelf-life of more than 12 months.
Conclusion:
The development of KOKO Plus as a food supplement provides a practical solution to improve the nutritional status of weaning children in Ghana.
Introduction
A preliminary survey on complementary feeding practices in Ghana revealed that koko (a thin porridge made from fermented corn dough) constitutes the most common complementary food in Ghana. 1 The overdependency on koko as the major complementary food raises issues of protein energy malnutrition and micronutrient deficiencies. To improve its nutritional quality, mothers, from the benefits of nutritional education, mix koko with legumes such as peanut, cowpea, or bambara groundnuts or add fish flour. These blended foods are usually not very suitable owing to the high levels of antinutritive factors, low-quality proteins, poor micronutrient profile, hygienic and safety issues, high bulk density or viscosity, and some organoleptic properties, particularly regarding the cereal–legume blends. 2 The convenience of processing such foods at the household level may also be a challenge. There are also economic constraints to some mothers. Stakeholder engagements across Ghana (with health-care professionals, nutrition officers, and mothers of child-bearing age) on strategies for improving complementary foods eventually led to the development of the complementary food supplement, KOKO Plus. 1,2 It is a food supplement designed to improve the nutrient profile of common traditional complementary foods for infants aged 6 to 24 months during the complementary feeding period. 1
Suri et al 1 described the use of linear programming methodology in the formulation of KOKO Plus to obtain a product that will improve the micronutrient contents and protein quality of traditional complementary foods. The original formulation was targeted at improving the nutrient content of koko, which is maize based. The concept was to develop a product by considering the nutritional constraints of koko, that is, to add a supplement made from locally available ingredients that will improve macronutrients, micronutrients, and energy content with high sensory acceptability. In the formulation of KOKO Plus, the use of legumes, including soybean, peanuts, and cowpea, in combination with the amino acid lysine, fat, sugar, and a micronutrients mix was predicted using linear programming to achieve the desired formulation based on the nutrient profiles of these ingredients. The final composition of the supplement consisted of roasted soybean flour, lysine, sugar, micronutrients mix, and vegetable oil. The product was packaged in 15-g, single-use sachets that drastically reduced the bulk and facilitated easy distribution to consumers and also increased its affordability. 2 The nutritional profile of KOKO Plus was designed to improve indigenous complementary foods to adequately complement breast milk in meeting the requirements of the target age-group as recommended by the World Health Organization (WHO). 3,4 KOKO Plus was to help meet at least 50% of the recommended daily allowance of micronutrient needs of the child. While it was originally developed based on the inadequate nutritional profiles of cereal-based porridges, it was also found to be applicable for most other indigenous complementary foods, including, soups, sauces, and many family foods introduced in the period of complementary feeding. Ghosh et al 2 discussed the conceptual bases of KOKO Plus as an innovation in infant and young child feeding involving a public–private partnership to develop and test the efficacy and effectiveness of the delivery of a low-cost complementary food supplement in Ghana.
The development of KOKO Plus followed typical product development strategies, from ideation, through to prototype development, testing, and then pilot scale production. 5 Factors such as nutritional needs of the target group, sensory properties, physiochemical properties, and safety were major considerations. Furthermore, the acceptability of the end product by mothers and caregivers as well as the infants was a very important consideration. Beyond these factors, another major and practical consideration was the shelf-life of the product. According to the Institute of Food Science and Technology Guidelines, 6 shelf-life is defined as the time during which the food product will (1) remain safe; (2) retain desired sensory, chemical, physical, and microbiological characteristics; and (3) comply with any label declaration of nutritional data when stored under specified conditions. Physical, chemical, and biological changes may occur during handling and storage which may lead to product deterioration in terms of nutritional, microbiological, or sensory quality. While it may seem necessary to determine the chemical or physical changes in the food for a full understanding of the deterioration process, they have a direct effect on sensory quality. Therefore, for any product, once the microbiological and nutritional hurdles have been ascertained, the remaining barrier to success depends very much on the sensory properties of the product. 7 Consumers are sensitive to even slight changes in sensory properties and will very easily reject products when such changes occur. Furthermore, some product properties are just too complicated to measure objectively, and physical or chemical measurement alone will not indicate consumer acceptability or rejection. Consequently, sensory shelf-life tests are popularly used in product development (or product rework). Sensory shelf-life tests are done using different approaches after varying product storage conditions and study duration. Common approaches include descriptive tests to measure quality changes 8 and hedonic testing to evaluate the impact of quality changes in consumers’ acceptability or preference. 9 Acceptance test or preference tests have also been used 10 to estimate shelf-life. Various applications 11,12 and analyses 10,13 of sensory shelf-life tests have been published.
One of the most popular approaches to sensory shelf-life estimation is the method of difference from control test, 14 which measures the degree of difference between stored samples and a control by a trained sensory panel using discriminative tests or intensity scales. 15 The magnitude of difference between stored samples and the control is modeled as a function of storage conditions (of time, temperature, relative humidity, and so on), and the shelf-life is estimated as the time at which the product reaches a predetermined difference from the control product.
The KOKO Plus food supplement is a low-moisture product that is packaged in laminated aluminum foil, and it is generally microbiologically stable. However, its shelf-life may be influenced by chemical changes (since it has significant amounts of oil) or by changes in the sensory characteristics of the product. While the chemical changes in the product might not be easily discernable, slight changes in sensory properties will be detected by consumers of the product. This work describes the development process and the sensory shelf-life studies of the KOKO Plus complementary food supplement.
Materials and Methods
Development of KOKO Plus
Product ideation
Two preliminary and related pieces of work that informed the product concept were carried out as follows: (1) a survey on complementary feeding practices in Ghana, which identified koko as the most common complementary food and (2) stakeholder engagements on complementary foods that brought out the need for the development of a complementary food supplement. The survey was conducted in 3 regions (Greater Accra, Upper West, and Central region) with the following objectives: (1) to understand the major issues associated with complementary feeding, health, and food security in the 3 regions; (2) to determine the different types of complementary foods used by mothers as well as those recommended by health workers in the 3 regions; (3) to examine the differences in the preparation of complementary foods and the use of different staples for preparing complementary foods in the Upper West, Greater Accra, and Central regions; and (4) to examine the potential acceptability for a ready-to-use complementary food product. Fifty-six respondents were interviewed in rural, peri-urban, and urban communities of the 3 regions. A total of 9 communities were engaged. Key informant interviews and focus group discussions were held with mothers, community elders, community health workers, regional directors, and district health service directors. Data were collected on infant feeding practices, food security, health and sanitation, infant and young child nutrition issues, complementary feeding practices, and use of specific complementary foods.
In the stakeholder engagement, several places, constituting a cross-section of the country, were visited by the research team. Communities visited are shown in Figure 1, and they were as follows: in upper west region (Wa,), northern region (Tamale, Langbesi, and Narilegu), central region (Cape Coast, Saltpond, and Elmina), ashanti region (Kumasi, Manso-Nkwanta, and Jachi-Pramso), eastern region (Koforidua, Asesewa, and Nnoyem), and Greater Accra region (Accra). In each region, the team met and interacted with the responsible officers for health and nutrition at the regional and district levels. The team also visited the basic nutrition and health centers, including the Community-Based Health Planning and Services (CHPS) Centers, to gather information about the norms and challenges of infant-feeding practices by mothers. This took the form of formal and informal meetings with parents of child-bearing ages at the CHPS centers to discuss the issues of malnutrition and complementary feeding practices and to elicit suggestions for improving the nutritional quality of infant foods, including the development of complementary foods and supplements. Prior to these interactions, approval from the Director General of the Ghana Health Services was sought and obtained.
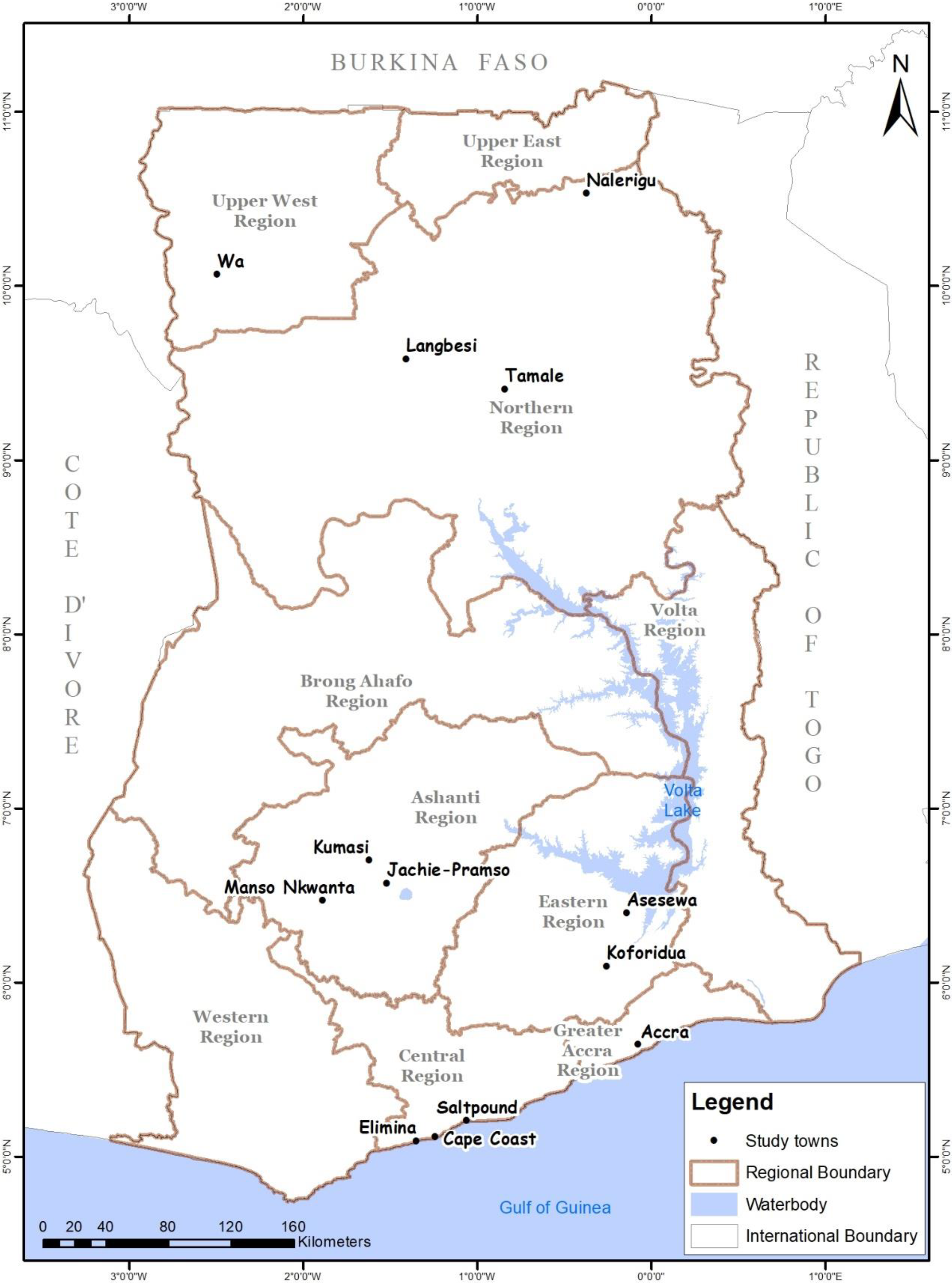
Study locations visited in Ghana during stakeholder engagement.
Product development process
The method used was as described by Suri et al 1 in which a ready-to-use food supplement using locally available raw materials to improve the traditional complementary food was developed for children aged 6 to 24 months. Briefly, 9 cereal–legume combinations (maize, sorghum, or millet combined with cowpea, peanut, or soybean) and koko (a traditional Ghanaian maize-based complementary food) were evaluated based on the macronutrient targets for a daily ration of complementary food for the age-group 12 to 24 months. Protein quality was assessed by the Protein Digestibility Corrected Amino Acid Score. Linear programming was then used to determine the amounts of additional oil, sugar, and lysine needed to meet macronutrient requirements. After optimization of the model, soybean–maize was the only combination to meet the constraints with the addition of oil, sugar, and lysine. However, since koko is usually made using fermented maize dough as the basic raw material, the cereal component in the formulation generated using linear programming was removed, and it was left with the soybean/amino acid/micronutrients/sugar and oil components. 2 Consequently, KOKO Plus was obtained as a complementary food supplement consisting of an optimized combination of vitamin–mineral mix, lysine, oil, and sugar with soybean as protein source. 2 It was packaged in laminated aluminum foil in 15-g sachets and used for sensory and shelf-life studies.
Sensory Studies
Selecting the appropriate complementary food for use with KOKO Plus food supplement
Two commonly used complementary foods were identified for use with the KOKO Plus supplement: maize-koko and light soup (a typical Ghanaian soup with tomato, onion, pepper, salt, and water as the basic ingredients). These 2 foods were selected for their universal usage as complementary foods in Ghana. Koko made using maize is widely used in many Ghanaian homes. It may be sweetened with an appropriate sweetener (usually sugar or honey) prior to feeding to the infant. Mothers and caregivers may also have milk, groundnut paste, and other supplements added to koko to improve its sensory characteristics and to some extent the nutrient profile. Light soup, which has the basic ingredients of onions, tomatoes, pepper, and salt, forms the basis for almost all savory dishes cooked in Ghana. Although a family food, it is frequently fed to infants during the complementary feeding period.
Sample preparation
Both matrices (ie, maize koko and light soup) were prepared by a professional caterer at the University of Ghana. Samples were prepared fresh each day about 1 hour prior to the start of the sensory tests. Samples were stored in a food flask to keep them warm throughout the sensory tests.
Maize koko
One portion of unsweetened maize koko had KOKO Plus added to it in the proportion of 15 g of KOKO Plus (ie, 1 sachet) to 200 mL of freshly prepared koko. Another portion of maize koko was sweetened with sugar, to the same level in grams as that found in KOKO Plus (ie, 6 g of sugar was added to 200 mL of freshly prepared koko).
Soup
The soup sample was also divided into 2 portions. To one portion, no sugar was added since soups are not normally sweetened in Ghana. However, KOKO Plus (15 g) was added to the second portion (200 mL) of the soup.
Recruitment of assessors
The target consumer group for KOKO Plus food supplement is infants between the ages of 6 and 24 months. However, for this test, adults, that is, mothers, and caregivers who were or had been in contact with infants within this age-group, were recruited to participate in the sensory test. Caregivers included grandparents, god parents, siblings, community care nurses and midwifes, students, and nursery/creche assistants. The rationale for using adult assessors was that in practice mothers and caregivers decide what to feed their infants and may select foods based on their own preferences. Assessors were recruited locally from within and around the University of Ghana community through electronic media, posters, fliers, and word of mouth.
Sample presentation to assessors
Samples presentation followed a 2 × 2 crossover design testing the acceptability of koko or soup with or without KOKO Plus supplement involving 14 trained assessors. In 1 arm of the study, the first randomly selected 7 assessors were served porridge in the first session followed by soup in the second session. In the second arm, the next 7 assessors started with soup in the first session followed by porridge in the second session. Within each session, samples with and without KOKO Plus were alternated. Each assessor was served about 20 mL of each sample. Samples were presented simultaneously. There was a 10-minute break between the first session and the second session, and assessors completed a demographic questionnaire during this period.
Test protocol
All tests were carried out in the sensory laboratory at the Department of Nutrition and Food Science at University of Ghana. All assessors completed an informed consent form prior to tasting. Assessors were oriented in the test protocol. Assessors first completed a paired preference test by circling their most preferred of the 2 samples presented to them. Assessors were conditioned to consider the preferences of their infants in selecting their preference. This was done to ensure a reflection of infant preferences was being obtained from the adults. Assessors were then asked to complete a 9-point hedonic scale indicating their degree of liking for the most preferred and the least preferred samples.
Shelf-Life Studies
Storage test
Storage test was conducted at 3 different temperatures of 24°C, 34°C, and 44°C at 78% relative humidity for a period of 12 months. The samples were packaged in the same manner as the final product “KOKO Plus” (ie, 15 g/sachet, in laminated aluminum foil) for the shelf-life tests. During the tests, sensory evaluation was carried out at time periods of 1, 2, 3, 4, 6, 8, 10, and 12 months for the samples stored at 24°C and 34°C. However, the sensory test for samples stored at 44°C was evaluated for only 6 months after starting.
Modeling sensory data to estimate shelf-life: Time–temperature tolerance
Theoretical background
Foods deteriorate during storage and distribution, and it has been established 16 that the change in any quality index A with time (dA/dt) is represented by:
where k is the rate constant and n is reaction order.
The equation is also written as: f(A) = kt, where f(A) is the quality function.
However, most data related to quality deterioration do not provide rate constants (k) but rather are in the form of overall shelf-life as a function of storage temperature. 14 Since the temperature range is usually quite narrow, the relationship between shelf-life and storage temperature is:
where θ is the shelf-life at temperature T and b is the slope of the semi-log plot of θ versus T and c is the intercept or reference temperature. 16
The value of the quality function f(A) at time t under changing environmental conditions can be estimated from
where T (t) is the temperature as a function of time.
To estimate the quality change (ie, shelf-life) under variable temperature conditions, the quality function f(A) is solved using time–temperature tolerance procedures. 16
Sensory shelf-life evaluation
A trained panel (n = 5) was used to assess the freshness of the samples during storage. Products stored at 5°C were used as control. The samples were prepared as 10% water dispersions of the stored product. Panelists scored the product on appearance, aroma, and taste on a 5-point scale, relative to the control sample. A score of 5 meant that the product was as good as the control, 3 referred to the product acceptability limit, and scores ranging from 0 to 2.9 referred to products that were not acceptable.
The data obtained from storage tests were modeled using the Time/Temperature/Tolerance method. 16 Under the experimental conditions of specified temperatures and time durations at 78% relative humidity, the sensory scores for taste and flavor reduced faster than the other sensory scores. Consequently, the scores for taste and flavor were used to estimate the shelf-life of the product. In modeling the sensory data for shelf-life estimation, a sensory score of 3.0 was considered as the limit of product acceptability.
Microbiological Analyses
Samples of KOKO Plus were produced and analyzed for microbial load, primarily to determine their safety and wholesomeness. Analysis included enumeration of aerobic mesophiles (total plate count), coliforms (total coliform counts), and yeast and molds (yeast and mold counts) as well as for the detection of Escherichia coli. Total plate count was by ISO 4833-2:2013 APC/30°C/72HR/PCA Cfu/g); total coliform count by ISO 4832:2008; yeast and mould count by ISO 7932, 25°C/7 days/PDA cfu/g); and detection of E coli by ISO 7251 2005, 44°C/48HR/l TB/ECB.
Ethical considerations during the study
Permission to conduct this study was obtained from the Ghana Health services. Ethical approval was obtained from the Noguchi Memorial Institute for Medical Research, University of Ghana (031/14-15) for sensory evaluation of the product, after the food was approved to be used for the study by the Ghana Food and Drug Authority. Participation in the study was voluntary, and written informed consent was sought from all the respondents engaged in the stakeholder interactions with the research team and in the sensory evaluation of the product. Respondents who were literate gave signed consent forms, while verbal consents were documented in the presence of a third-party witness for those who were illiterate after it was explained to them in the local language
Data Analyses
The data for sensory analyses were subjected to χ2 analyses (at P < 05) to determine whether the acceptability of soup and koko was significantly associated with the addition of KOKO Plus to the product. The data for sensory shelf-life testing was fitted into appropriate models (semi-log transformation of sensory data as a function of storage time and temperature) for shelf-life estimation of the product.
Results
Product Idea Generation and Preliminary Interactions With Stakeholders
Survey on koko consumption by children aged 6 to 24 months
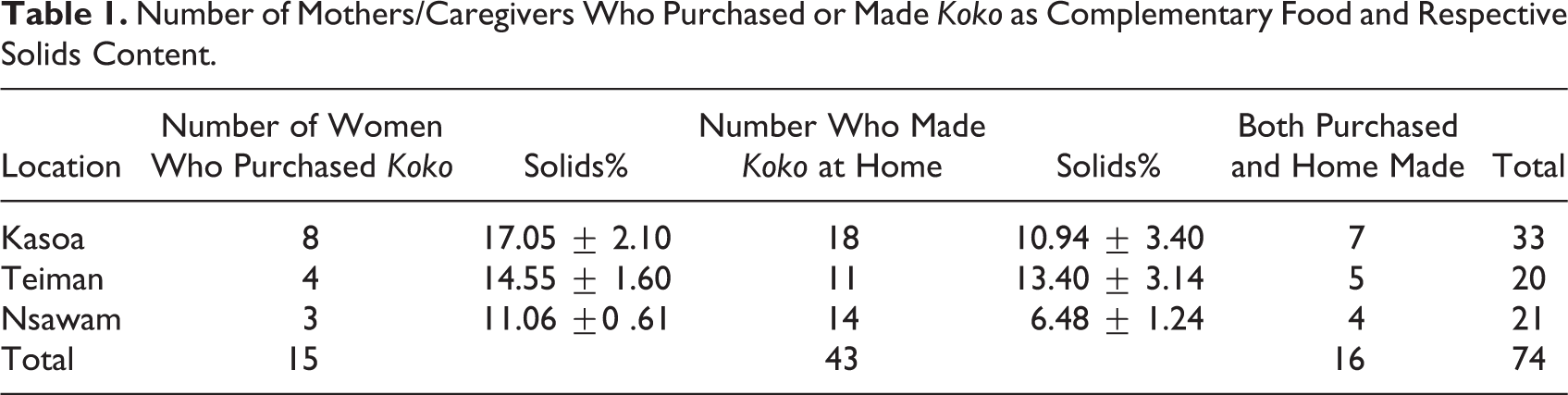
Mothers were randomly sampled from 3 locations (Kasoa, Teiman, and Nsawam) in Central, Greater Accra, and Eastern Region of Ghana. They consisted of a total of 74 mothers and caregivers with infants aged 6 to 24 months. All mothers and caregivers confirmed that they fed koko to their children as complementary food. While some of them purchased koko from local vendors, others made their own koko at home. Other parents also purchased or made their own at home (Table 1). The χ2 analyses showed that significantly higher numbers of mothers and caregivers prefer to feed their children with homemade koko than that purchased from local vendors. Koko samples (both homemade and from vendors) were obtained and the volumes and solids content determined. The results show that the consistency of koko obtained from the various sources varied significantly and ranged between 6.48% and 14.55% solids (Table 1). The homemade koko was generally of lower solids content than that purchased from the community vendor, suggesting that mothers and caregivers prefer lower consistency koko for their young infants. The community-vended koko had higher consistency because they are prepared for adult consumers.
Number of Mothers/Caregivers Who Purchased or Made Koko as Complementary Food and Respective Solids Content.
The volume of koko consumed by infants varied greatly with age (Figure 2.). Infants aged 6 to 9 months predominantly consumed less than 100 mL of koko during feeding. As they got older (14-17 months), their koko consumption volumes increased drastically, reaching as high as 200 to 400 mL per feeding time for 22- to 25-month-olds, probably due to the low solid content of koko. Generally, however, 200 mL of koko was sufficient across all age categories within 6 to 24 months.
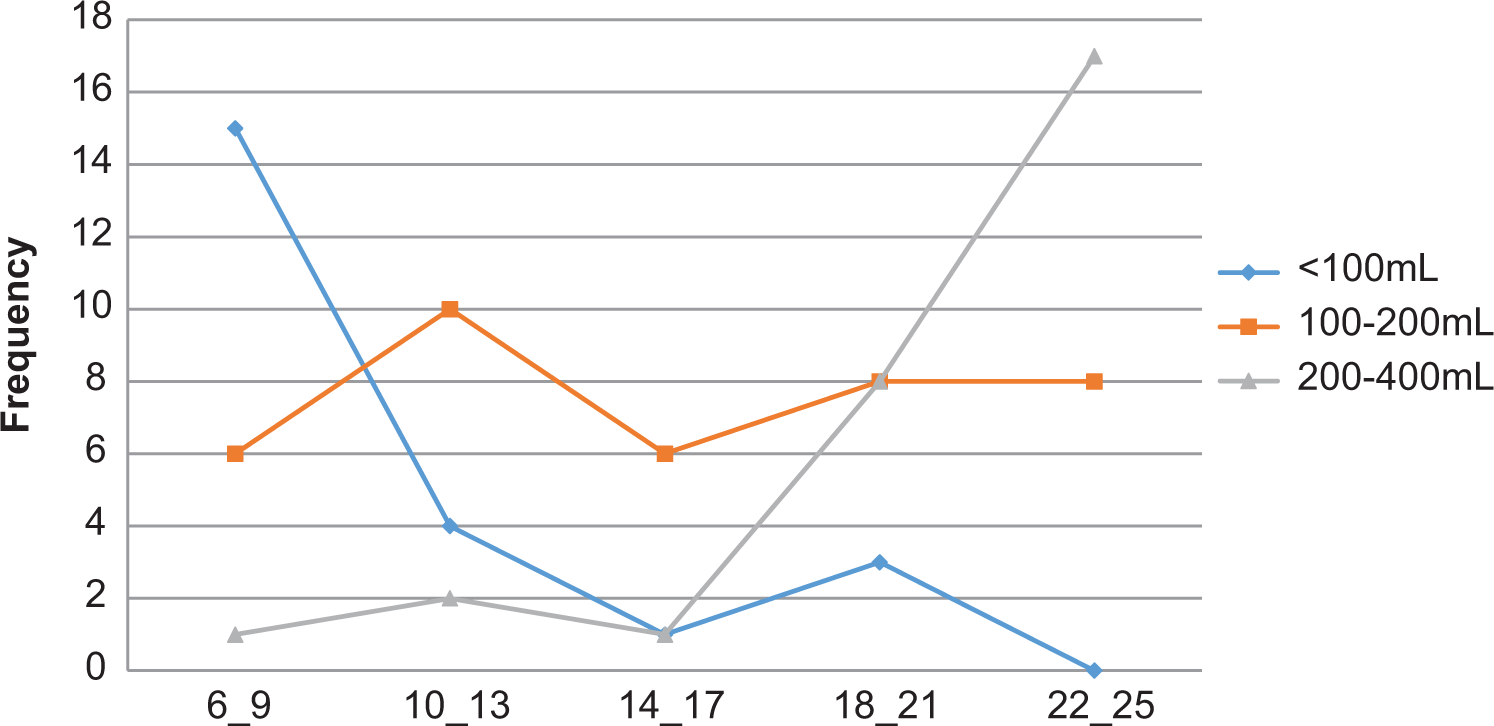
Volume of koko consumed as a function of child’s age in months.
A follow-up study to confirm mothers’ and caregivers’ preference for koko consistency was done using 15 randomly selected mothers and caregivers. They were recruited from among workers of the University of Ghana. Mothers and caregivers were presented with 4 coded samples and instructions explained to them. They were asked to score the products on a 7-point hedonic scale. The results (Table 2a) show that koko made to 10% solids was far more preferred by mothers and caregivers over the other consistencies. Similarly, most mothers and caregivers preferred koko that was only slightly sweetened (Table 2b)
Mothers’ Preferred Koko Consistency and Sweetness for the Child.a
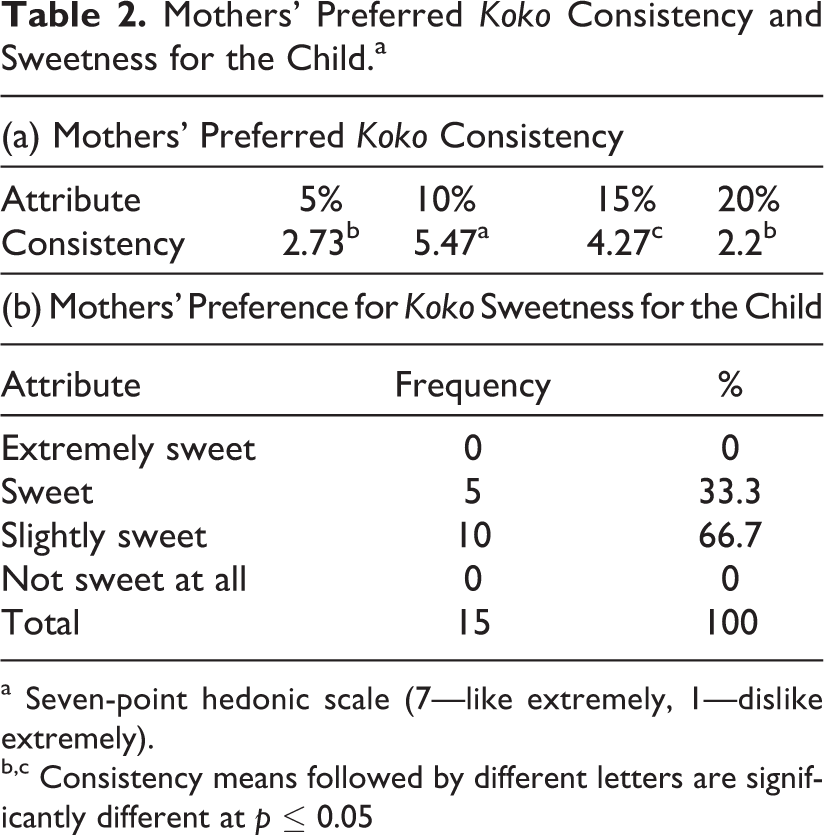
a Seven-point hedonic scale (7—like extremely, 1—dislike extremely).
b,c Consistency means followed by different letters are significantly different at p ≤ 0.05
Mothers and caregivers efforts to improve the nutritional quality of koko as complementary food
During the stakeholder interactions, it was clear that mothers and caregivers are aware that koko alone as a complementary food may be inadequate in meeting the nutritional needs of the infants. Mothers were said to be educated on how to improve the nutritional quality of koko by the district and community nutrition officers by adding milk, eggs, and legumes. Figure 3 shows that at the 3 locations that were sampled, mothers and caregivers admitted to adding milk or koose (deep fat fried cowpea paste) to improve the protein and calorie content.
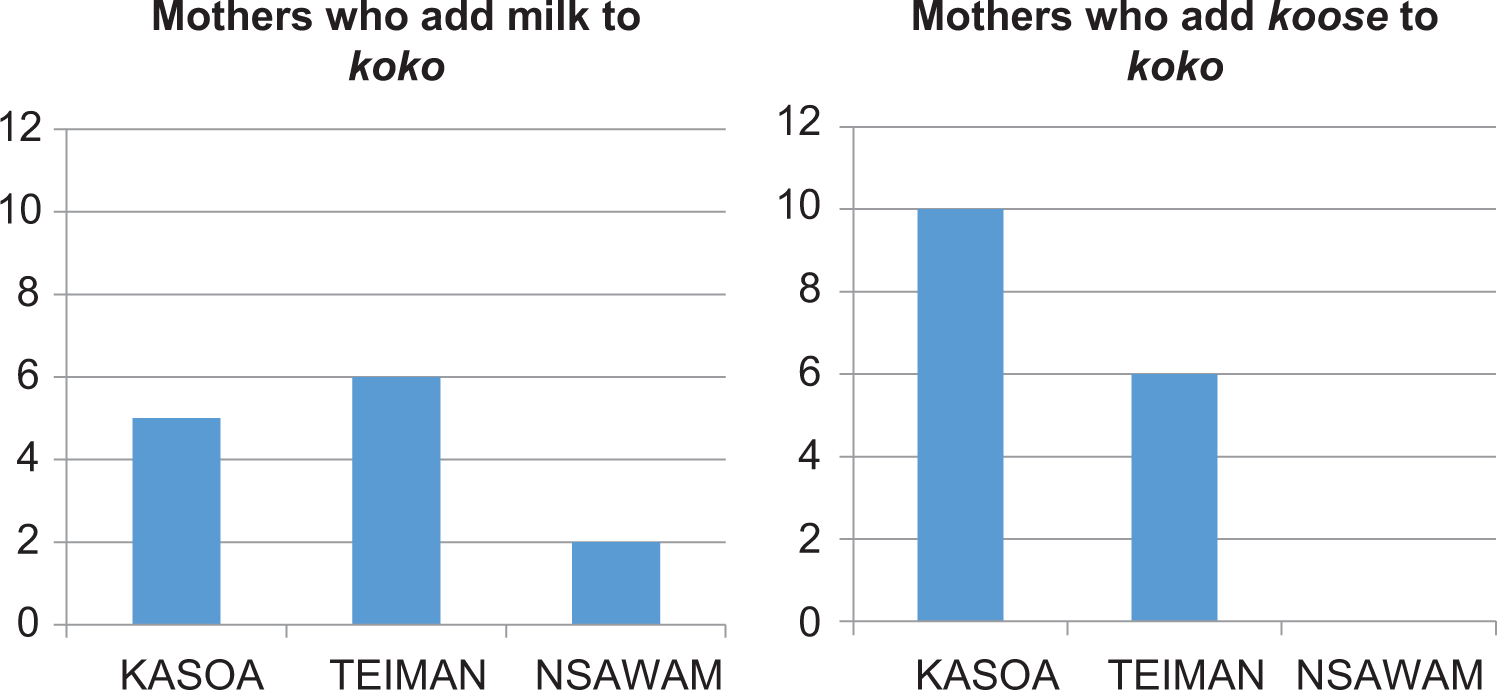
Efforts at improving the nutritional quality of koko by mothers and caregivers.
Other foods used for complementary feeding
Although koko was the predominant food fed to young infants, other foods were also used. Table 3 shows that in rural, peri-urban, and urban communities in the Greater Accra, central, and upper west regions, family foods such as tuo zaafi (TZ), banku, Tom Brown, and light soup were fed to children. Other complementary foods including legume-cereal blends (weanimix) were also used as complementary food. In focus group discussions, most mothers would patronize ready-to-use supplementary foods if available. Similarly, the health professionals would also support the use of ready-to-use complementary foods, but it should be inexpensive for mothers to afford it.
Complementary Foods Commonly Used in Study Communities.
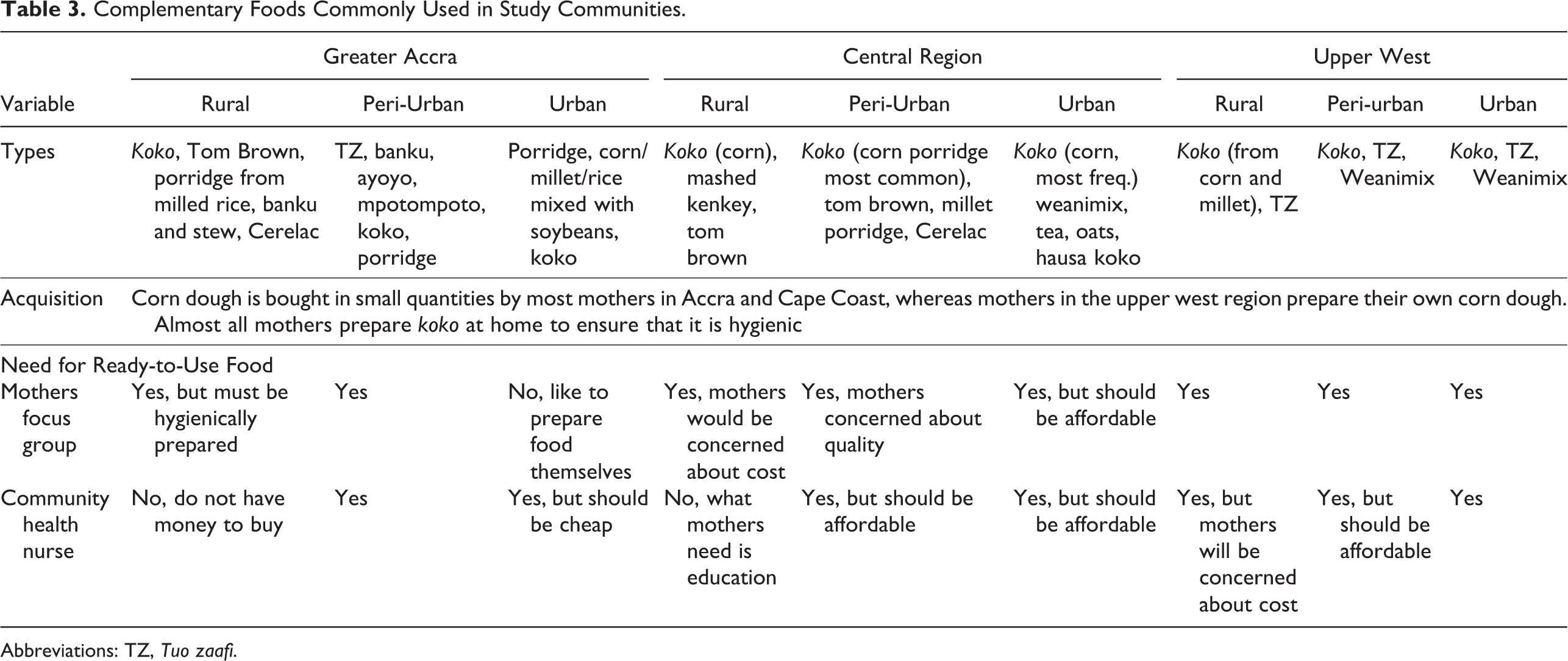
Abbreviations: TZ, Tuo zaafi.
Preliminary formulations of KOKO Plus and acceptability studies
From the foregoing, it was clear that koko is a major complementary food in most of the communities across Ghana. Mothers and caregivers were clear about the characteristics of koko they require for infant feeding. They require it at a specific consistency (at about 10% solids), and it should not be very sweet. Koko however is nutritionally inadequate and needs to be improved to WHO standards as a complementary food. Consequently, preliminary formulations of KOKO Plus was made (1) and used for acceptability tests among a randomly selected group of mothers and caregivers.
Three koko samples were prepared as follows: sample A (400 mL) was prepared at 15% solids using dehydrated corn dough. To this sample, 0.5 g of millet malt was added at about 60°C and stood for 30 minutes to obtain a thinner consistency before mixing with 1 sachet (15 g) of KOKO Plus. Another batch of koko was prepared to a volume of 800 mL using dehydrated corn dough at 10% solids. It was divided into 2 portions of 400 mL each. To the first koko portion of 400 mL, nothing else was added and was labeled as koko sample B. It served as the control sample. A sachet of KOKO Plus (15 g) was mixed with the second portion of 400 mL of koko and labeled as sample C (Table 4). Fourteen mothers and caregivers were recruited from among workers of the University of Ghana. They were presented with the 3 coded samples and instructions explained to them. They were asked to score the products on a 7-point hedonic scale. The results (Table 4) show that products B and C which had similar consistencies (10% solids) without and with experimental KOKO Plus as supplement were equally acceptable to mothers and caregivers.
Mean Hedonic Scores for Koko With and Without Experimental KOKO Plus Supplement.ab
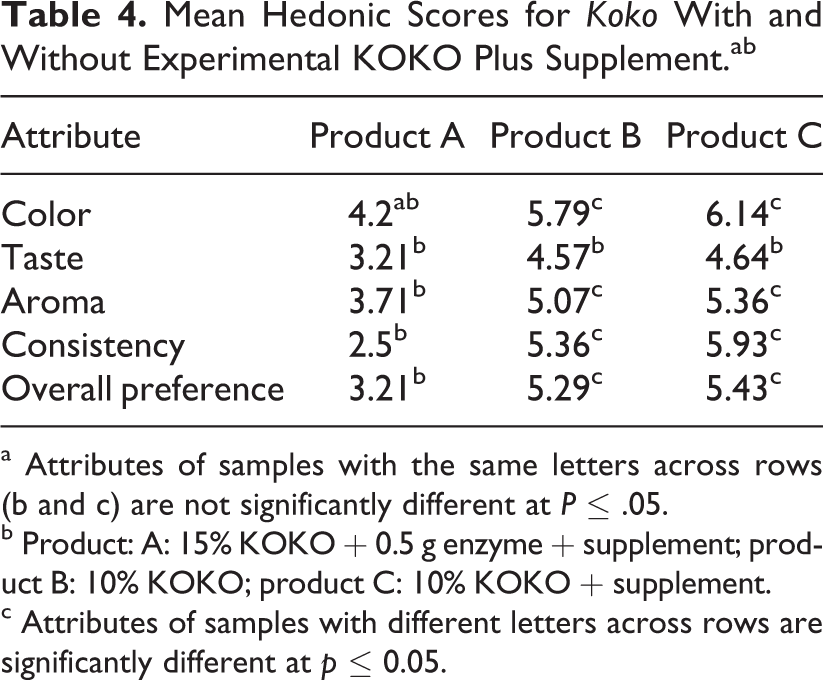
a Attributes of samples with the same letters across rows (b and c) are not significantly different at P ≤ .05.
b Product: A: 15% KOKO + 0.5 g enzyme + supplement; product B: 10% KOKO; product C: 10% KOKO + supplement.
c Attributes of samples with different letters across rows are significantly different at p ≤ 0.05.
Formulation and nutritional profile of KOKO Plus prototype
As stated by Ghosh et al, 2 the supplement formulation was based on the assumptions that the target child (1) is between 6 and 24 months of age, (2) consumes an average amount of breast milk for his or her age-group, and (3) consumes an average amount of complementary food for his or her age-group. Furthermore, the final formulation of KOKO Plus was designed to meet the requirements as outlined by Lutter and Dewey 3 and in consonance with the revised amino acid requirements published by the WHO. 17
Based on the positive outcome of the sensory acceptability tests, the formulation for the KOKO Plus supplement was considered adequate and acceptable to consumers, and it was validated using different sets of mothers and caregivers. The final product of 15 g/sachet of KOKO Plus contained 7.3 g soybean (48.8%), 0.98 g palm oil (6.5%), 5.6 g sugar (37.3%), 0.112 g lysine (0.75%), and 1 g micronutrients mix (6.7%). The formulation of the supplement is presented elsewhere, 2 and its nutritional composition was evaluated 1,2 and presented in Table 5. It shows the nutritional profile as well as the proportion of the requirements for the infants’ nutritional needs to be met.
Nutritional Profile of Koko With and Without KOKO Plus.
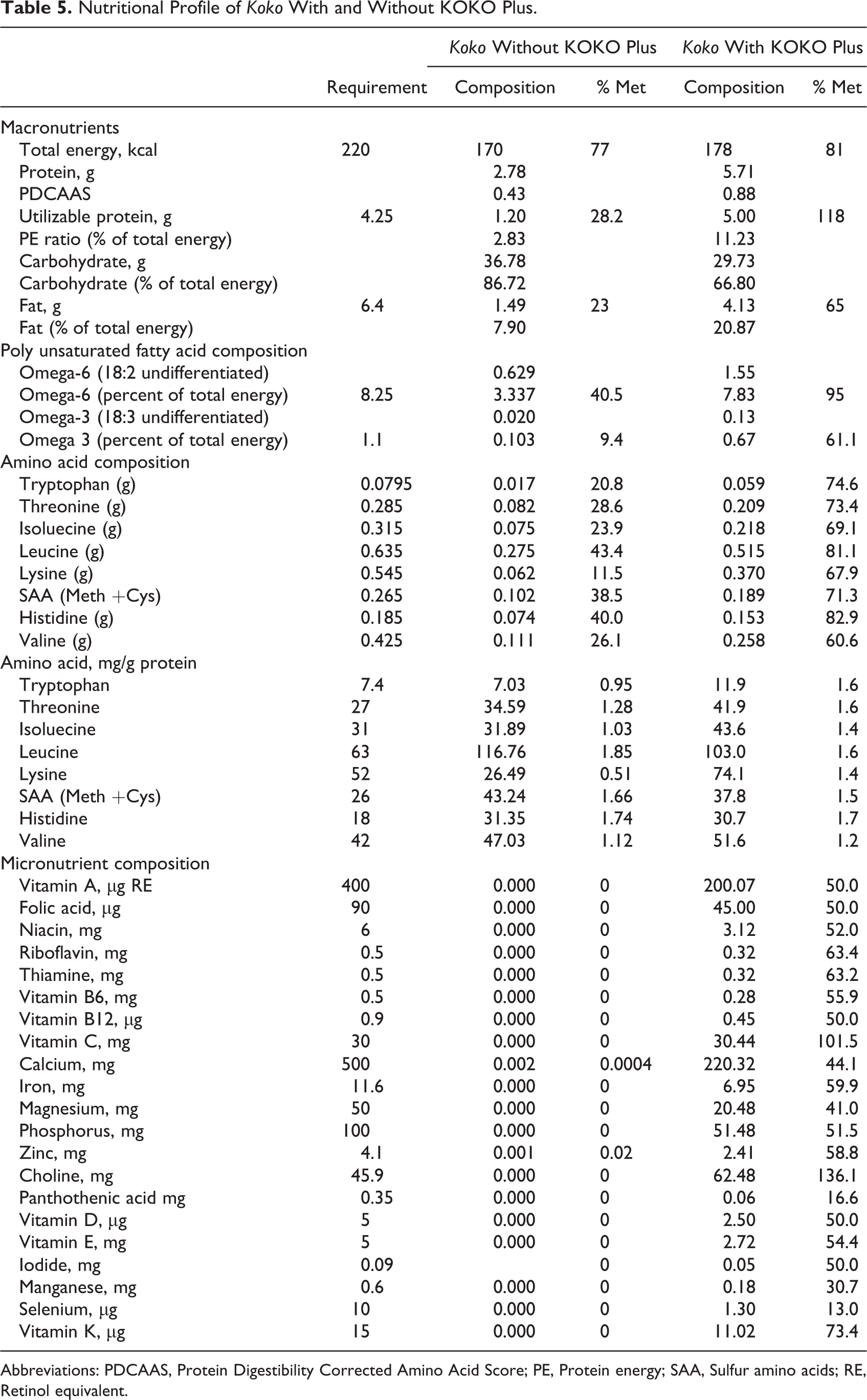
Abbreviations: PDCAAS, Protein Digestibility Corrected Amino Acid Score; PE, Protein energy; SAA, Sulfur amino acids; RE, Retinol equivalent.
Sensory Evaluation for KOKO Plus in Different Complementary Food Matrices
The sensory acceptability test for KOKO Plus in soup and in koko (data not shown) showed that there were no significant differences in preference for either porridge or soup with or without KOKO Plus. This is consistent with the earlier findings during the developmental stage of the product. This suggests that adding KOKO Plus to the complementary food will not detract mothers and caregivers from feeding their children with them based on sensory acceptability.
Microbiology of KOKO Plus
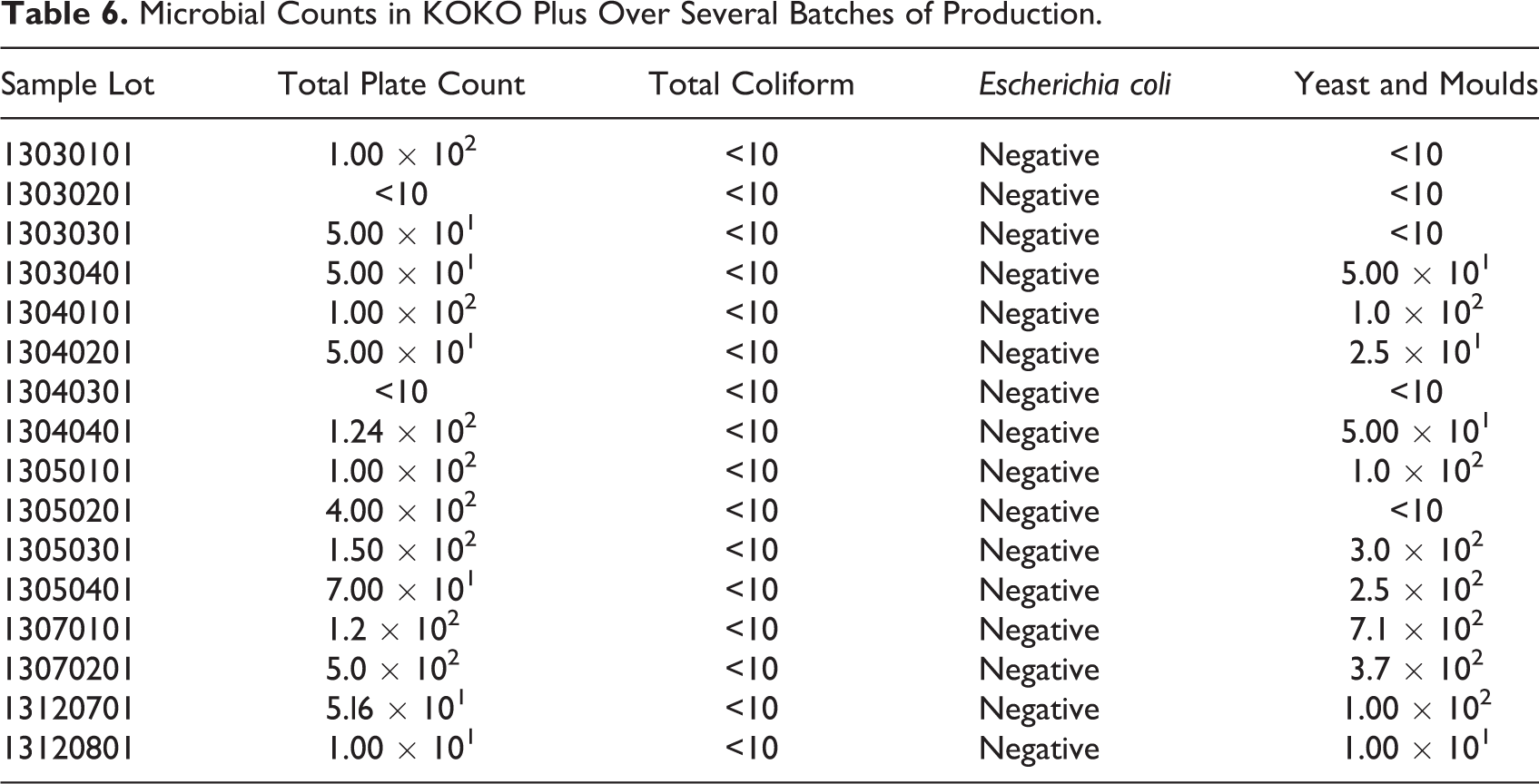
Several batches of the KOKO Plus food supplement was produced and tested in the field for its acceptance by mothers and caregivers. Batches of production were randomly sampled from the field trials for microbiological stability and safety tests. The results (Table 6) showed low counts of yeasts and molds (<2 × 102) as well as low total plate counts (<1.5 × 102), indicating that the product will be microbiologically shelf stable. Furthermore, low total coliform counts (<10) and no detectable E Coli suggest that the product was produced under good hygienic conditions, and it is microbiologically safe for consumption, especially by the target group.
Microbial Counts in KOKO Plus Over Several Batches of Production.
Shelf-Life Estimation
The data for sensory evaluation for shelf-life estimation of KOKO Plus is shown in Table 7a. The score of taste and flavor reduced and approached the critical value of 3 faster than other sensory attributes, and therefore, the data for taste and flavor were used in the shelf-life estimation for KOKO Plus (Figure 4 and Table 7b).
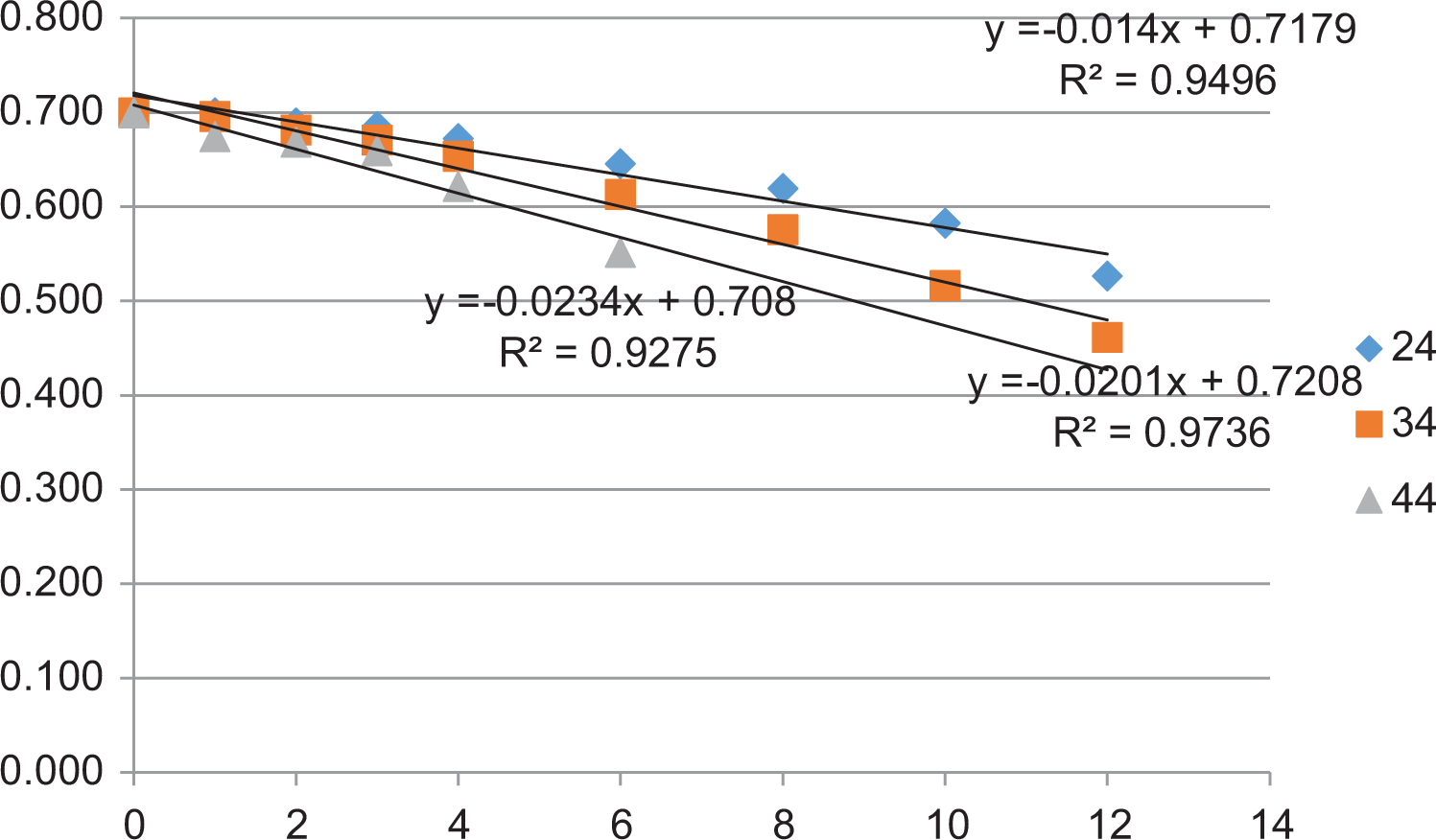
Log sensory scores as a function of storage time in months.
Sensory Scores for Taste and Flavor and Estimated Shelf-Life of KOKO Plus as a Function of Temperature of Storage.
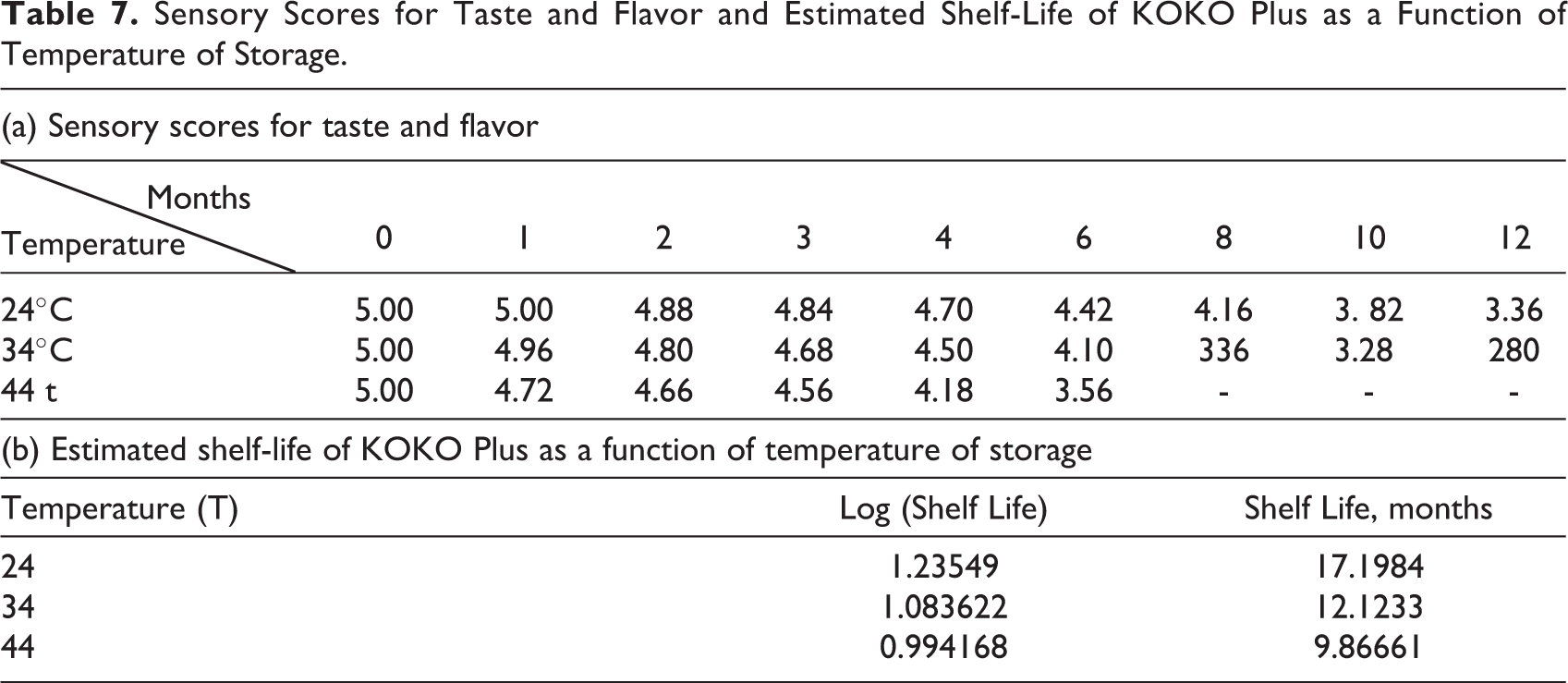
The shelf-life of KOKO Plus was estimated to be about 15 months at 28°C by regressing shelf-life on temperature. However, it was indicated as 12 months for safety reasons, since assumptions were made for narrow fluctuations in temperature during storage. The adequacy of shelf-life of 12 months of KOKO Plus in the distribution channels in Ghana was validated through further experimental studies.
Discussion
Product Ideation and Concept Development
The survey of communities and engagement with stakeholders including nutrition and health professionals as well as mothers and caregivers of infants was quite revealing. Complementary feeding of infant children in all communities is predominantly done using koko. There was a general awareness among mothers and caregivers that the nutritional quality of koko was inadequate for the developmental needs of the infants. Furthermore, the (10% solids) consistency of koko that was generally preferred by mothers provided inadequate calories. Consequently, mothers added products such as milk and koose to improve the nutritional quality of koko. It was also observed that as the child grew older, the volume of koko required per feeding increased tremendously to support the energy needs. However, the average consumption volume of the infants across the age-group of 6 to 24 months was 200 mL. Indeed these observations support the assertion that koko did not meet the recommended complementary food nutrient density nor the Food and Agriculture Organization of the World Health Organization (WHO/FAO) protein and amino acid requirements for infants aged 6 to 24 months. 1
While koko was the predominant complementary food in most communities, mothers and caregivers also introduced (soft) family foods such as soups, TZ, and banku. This is consistent with the observation that most infants are able to consume home-available family foods during the second year of life, probably by about 12 months of age, and that special foods with liquid or semisolid consistency may be required only during the period from 6 to 11 months. 18 Weanimix, which is a cereal-legume blend, was another popular food that was introduced to the young infants. It may seem to be a far better complementary food than koko. Ghosh et al 2 however reported that analysis of commonly used cereal–legume complementary food blends showed that many did not meet either the protein-quality needs or the fat and essential fatty acid needs of young infants. The authors further noted that most fortified blended foods (eg, corn–soy blend) are inadequate for feeding young and malnourished children because of high levels of antinutrients, lack of high-quality protein sources (eg, milk), suboptimal micronutrient content, and many other factors. These observations led to the concept of development of the food supplement, and mothers and caregivers’ input were strongly sought in the prototype development and testing. Sensory testing of the prototype supplement indicated that there was no significant difference in the acceptability of koko or soup with or without the addition of the food supplement. Consequently, the drivers for adding it to infant food may not just be sensory acceptability alone. If mothers are convinced that adding the food supplement to koko or soup would provide a nutritionally superior complementary food, its chances of acceptability would be greatly enhanced. Indeed, mothers and caregivers as well as the health professionals confirmed that they will be willing to use a ready-to-use food supplement to improve the quality of koko if it is available and the cost is affordable. These revelations were considered to be quite insightful, especially when considering accessibility and affordability by underprivileged, very low-income mothers and caregivers.
Nutritional Profile of KOKO Plus Supplement
It has been established that adequate nutrition during the first 2 years of life is critical to ensure optimal physical and mental development of infants and young children. 3 The observation that koko is the predominant complementary food for most mothers in Ghana confirms the nutritional risks in depending solely on it because koko is poor in energy density and protein quality. Furthermore, it is also devoid of micronutrient and has been implicated in protein energy malnutrition. 2 Table 5 clearly shows that koko alone does not meet the nutritional requirements of the infants and young children (6-24 months). Consequently, mothers through the benefits of nutritional education sometimes blend koko with legumes or use available cereal legume blends as complementary foods (Table 3). However, Suri et al 1 reported that blends containing maize, millet, or sorghum as cereals and cowpea, peanut, or soybean as legumes had improved protein quality and energy but still do not meet all micronutrient recommendations for complementary foods. Dewey and Brown 18 identified “problem nutrients” as those for which there is the greatest discrepancy between their content in complementary foods and the estimated amount required by the child. The authors contend that multiple micronutrients are likely to be limiting in the diets of children aged between 6 and 24 months in developing countries. They suggested that with linear programming, it was possible to achieve optimal complementary food blends that met energy and macronutrient requirements by the addition of other ingredients, including oil, sugar, and lysine, but micronutrients for the most part remained deficient and would need to be supplemented. The development of KOKO Plus was predicated upon the fact that nutrient-dense complementary foods with supplemented micronutrients and amino acids can improve the profile of these problem nutrients and promote long-term benefits to the young infants. 2
The final product of 15 g/sachet of KOKO Plus contained 7.3 g soybean (48.8%), 0.98 g palm oil (6.5%), 5.6 g sugar (37.3%), 0.112 g lysine (0.75%), and 1 g micronutrients mix (6.7%). Table 5 also shows that when a sachet containing 15 g of KOKO Plus is mixed with 200 mL of koko at 10% solids (dry matter basis), the total energy content changed from 170 to 178 kcal. This is an improvement in meeting the daily calorie requirements of the infant from 77% to 81%. Similarly, there was an improvement in meeting the requirements for protein. The utilizable protein requirements for infants is 4.25, but koko made using fermented corn dough provided only 1.20 (ie, only 28.2% of the requirement). When a sachet of KOKO Plus (15 g) was mixed with 200 mL of koko, the utilizable protein content increased to 5.00, raising the utilizable protein to 118%. Similarly, the fat content improved from 1.49 for koko alone to 4.13 when KOKO Plus was added. With the requirement of fat being 6.4 g, this was an increase in total requirements met from 23% to 65%.
The fatty acid as well as amino acid profile also showed that mixing KOKO Plus in koko improved the polyunsaturated fatty acid profile as well as essential amino acids profile and composition. There was a general improvement in the profile of omega 3 and omega 6 fatty acids as they met 61% and 95% of the requirements, respectively. Similarly, there was a general improvement in the amino acids when KOKO Plus was added to koko, as it met 60% or above for the requirements of essential amino acids (Table 5). Table 5 also shows that KOKO Plus improved the micronutrients profile (vitamins and minerals) in koko by 40% to 60%. These observations are clear demonstrations that when a sachet of (15 g) KOKO Plus was added to 200 mL of koko (10% solids), the nutritional profile improved to fully or partially meet the daily requirements for many of the “problem (micro) nutrients.” 18
Microbiology of KOKO Plus
Microbial growth and activity are some of the major factors affecting food deterioration during storage. These depend upon the physiochemical characteristics of the food product, moisture content, pH, temperature of storage, relative humidity, and water activity. The microbial counts (Table 6) were very low, and fecal coliforms (represented by E coli) were not detected. These indicate that proper hygienic precautions had been taken during production and packaging of the KOKO Plus complementary food supplement. It is also evident from the low microbiological counts (Table 6) that the supplement being a dried product did not support microbiological growth and multiplication. These observations therefore signify the wholesomeness and safety of the KOKO Plus complementary food supplement and the integrity of the packaging during storage. 19,20 This is important for the food to be presented in a safe manner to the infants and young children, as the risk of contamination with pathogens was clearly minimal. The microbiological analysis of the product was also important for the reason that it was usually done before the shelf-life determination by sensory analyses to ensure that the product was safe for consumption.
Shelf-Life Estimation
Shelf-life is determined based on product physical, chemical, microbiological, or sensory characteristics. The factor that mostly affects these characteristics is environmental temperature. In this work, the temperature range of 24°C to 44°C is the yearly temperature range in Ghana that the product would be exposed to during distribution and storage. For dry products such as KOKO Plus, the shelf-life could be defined by either chemical changes (because it has significant amounts of fat) or by the sensory characteristics of the product with varying environmental temperature conditions. It has been suggested 21 that consumer study is the most suitable means of determining the shelf-life of baby foods. The sensory score for taste and flavor (Table 7a) reduced and approached the critical value of 3 faster than other sensory attributes. Therefore, the data for taste and flavor were used in the shelf-life estimation for KOKO Plus.
The shelf-life of KOKO Plus was estimated from transforming the sensory scores data and using the regression of log shelf-life versus temperature (Table 7b) to obtain the following equation:
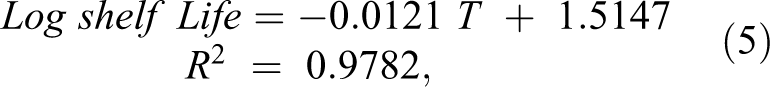
where, T is the storage temperature (in °C) of the KOKO Plus complementary food supplement. The equation obtained had a high regression coefficient (R2 = .9782) and can be used to estimate the shelf-life of the product at different storage conditions within the temperature ranges of 24°C to 44°C. The shelf-life of KOKO Plus was estimated to be 15 months at 28°C and at relative humidity of 78%. However, for uncertainty factors (since a number of assumptions were made in the sensory methodology), the shelf-life would be stated as 12 months. It is worth noting, however, that nutritional analyses were not done during the shelf-life study to ascertain the stability of some of the nutrients, and this could be considered as a limitation in the study.
Conclusion
The development of a nutrient-dense complementary food supplement (KOKO Plus) followed typical stages of new product development from product idea and concept development, prototype generation, and testing through to sensory and shelf-life estimation. The surveys and stakeholder engagement were useful in identifying koko and other soft products (such as soup) as complementary foods. While the addition of KOKO Plus complementary food supplement did not significantly influence mothers’ and caregivers’ acceptability of koko, it greatly improved its nutritional profile in both macro- and micronutrients. The product was microbiologically safe and had a shelf-life of at least 12 months.
Footnotes
Acknowledgments
The authors are grateful to Ajinomoto Co. Inc, Japan, for funding the research from which the study emanated, to Dr Yasuhiko Toride, Satoshi Kitamura, and all staff of Ajinomoto who supported the study. The authors are also grateful to the Nevin Schrimshaw International Nutrition Foundation and the University of Ghana, the collaborators of the study, for the opportunity for the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Ajinomoto Co. Inc, Japan.