Abstract
Background:
Cambodia has a high prevalence of moderate acute malnutrition and severe acute malnutrition (SAM). The SAM treatment requires ready-to-use therapeutic foods (RUTFs), whereas ready-to-use supplementary foods (RUSFs) are used for prevention of acute malnutrition. Three locally produced fish-based products were developed: an RUTF paste (NumTrey-Paste) for treatment and 2 wafer versions, one for prevention (NumTrey-RUSF) and one for treatment (NumTrey-RUTF).
Objective:
To assess the acceptability of NumTrey-Paste and NumTrey-RUSF in comparison to a standard biscuit product (BP-100) used for the treatment of SAM.
Methods:
Acceptability of NumTrey-RUSF and NumTrey-Paste was tested in a nonblinded crossover taste trial among children (n = 52), aged ≥ 6 months to 18 years, and their caregivers. Eight organoleptic qualities were assessed on a 5-point hedonic scale, as well as a ranking test. A score of 1 to 3 was categorized as acceptable. The acceptability of NumTrey-RUTF was assessed using the caregivers’ perception during an SAM treatment intervention.
Results:
Taste trial: The proportion of children categorizing products as overall acceptable was lowest for NumTrey-Paste compared to for BP-100 and NumTrey-RUSF (21% vs 43% [BP-100] and 36% [NumTrey-RUSF]). No difference was found in the proportion of children who ranked BP-100 or NumTrey-RUSF as “liked most” (P > .05). Acceptability of NumTrey-RUSF ranked highest in appearance and taste (caregiver), whereas acceptability of NumTrey-Paste was ranked lowest in appearance and smell among the products. Intervention trial: The acceptability of NumTrey-RUTF increased from 72% to 86%.
Conclusions:
The overall acceptability was ranked lowest for a pure paste product. However, filling the paste into a wafer made the product more acceptable.
Introduction
Acute malnutrition is still a major public health problem throughout the developing world, including Southeast Asia. Like many developing countries, Cambodia has seen improvements in poverty rates and food security over the recent decades, but many children continue to suffer from acute malnutrition. 1 The latest national survey highlighted the need for urgent action, with 12% of children < 5 years being acutely malnourished (weight-for-height [WHZ] < −2). 2
Acute malnutrition can be classified as moderate or severe depending on anthropometric and clinical status. Moderate acute malnutrition (MAM) is defined as a WHZ between −2 and −3 standard deviation (SD) and/or a mid- upper arm circumference (MUAC) between 115 and 125 mm. Severe acute malnutrition (SAM) is defined as WHZ < −3 SD and/or an MUAC < 115 mm and/or presence of nutritional edema. 3
The severe acute malnutrition (SAM) is a critical condition for children and requires immediate treatment with specialized therapeutic foods. According to the SAM treatment protocol in Cambodia, children with SAM and with complications needs to be treated in a hospital-based setting. In hospitalized children, SAM treatment starts with fortified milk products, where the caregiver is required to stay with the child at the hospital. 4,5,6 For uncomplicated SAM cases, home-based treatment can be as successful as hospital-based treatment, which eliminates problems arising from hospitalization, such as interruption of the caregiver’s work and care of other children and hospital-acquired infections. 7
Successful home-based treatment relies on availability of ready-to-use therapeutic foods (RUTFs). The RUTFs are nutrient- and energy-dense foods that provide all the energy and nutrients required to promote growth. 8 They do not require preparation prior to consumption, making them practical in settings where facilities are limited and hygiene is an issue. 7
Ready-to-use supplementary foods (RUSFs) are similar to RUTFs but have a slightly lower energy density. The RUSFs have been tested for their effectiveness in prevention of acute malnutrition. 9,10 Locally produced versions of RUTFs and RUSFs have been developed and tested for acceptability in various settings worldwide. 11 -17 The RUTFs are widely used in the treatment of SAM worldwide, while programs for preventive use of RUSF products have been difficult to establish. There is currently no national program for prevention of acute malnutrition in Cambodia, 8 for example, by supporting children with MAM with RUSFs. Such products are also not available for purchase by the children’s caregivers. Therefore, the development of a locally produced RUSF which either can be incorporated into a national program for prevention of acute malnutrition or be available for purchase by caregivers could contribute to preventing acute malnutrition in Cambodia.
Commonly used RUTFs are Plumpy’Nut, produced by Nutriset (Malanunay, France), and BP-100, produced by Compact (Søftland, Norway). The peanut-based Plumpy’Nut was introduced in Cambodia in 2008, but the introduction was unsuccessful, partly due to an insufficient implementation strategy. 18 BP-100, a compressed bar, based on cereal, milk powder, vegetable oil, and added vitamins and minerals, 19,20 is currently the standard product used in the treatment of SAM in Cambodia. 21 However, in an unpublished acceptability trial comparing BP-100 with locally produced RUTFs from Vietnam, BP-100 was shown to have lower scores on organoleptic qualities than 2 locally produced Vietnamese products, indicating that BP-100 might also not be well accepted in Cambodia. 22 To address this gap in availability of ready-to-use products meeting the Cambodian preferences, we aimed to develop 2 locally produced ready-to-use foods (both an RUTF and an RUSF) from local ingredients.
Most critical was the identification of a suitable protein source. The majority of commercial produced ready-to-use products contain dried skimmed milk (DSM) as a protein source, but costs of DSM are high and rely on import. The current price for DSM is ∼US$7/kg, whereas fish in Cambodia can be procured for as little as US$1.5/kg in the high season. The costs of imported RUTF also include shipment, insurance, and duty and taxes. Reducing the current costs for the procurement of an imported RUTF (∼US$6.4/kg) with the local production of an RUTF, using locally available fish as a protein source, would enable the Cambodian government to treat more children.
Therefore, to reduce the cost of the products and to adapt the taste to local preferences, milk powder was replaced by dried powdered small fish from the Mekong River. Small-sized fish have high nutritious value, specifically calcium from the fish bones, 23 and fish is both a suitable and sustainable replacement of milk powder in Cambodia. We had previously successfully incorporated dried powdered fish in instant porridge products for children in Cambodia. 24
In 2014, a pretrial was conducted on the acceptability of an initial version of fish-based paste in comparison with BP-100 in children attending a preschool day care center in Phnom Penh. The pretrial showed poor acceptability of both products. 25 Based on this trial, the fish-based product was further developed for improved acceptability.
Here, we report the further development of the locally produced, fish-based RUTF and RUSF products in Cambodia and the acceptability of the products among Cambodian children and their caregivers.
Materials and Methods
Development of NumTrey-RUSF and NumTrey-RUTF
The fish-based RUSF and RUTF products were developed in a stepwise process, based on the experiences from the development of a local RUTF in Vietnam. 12 First, the initial development of this locally produced fish-based product was a paste tested among preschool-aged children in Phnom Penh (2014). 25 Hereafter, pilot products were produced. Among the initial pilot products was a solid “cube” which resembled the successful “mooncake” from Vietnam. 12 The cube faced challenges, however, in both the texture (too soft) and taste. Therefore, the fish-based paste product was redeveloped as an RUTF (NumTrey-Paste). At the same time, an RUSF paste was developed, which became inspired by a common wafer snack in Cambodia. Thus, the RUSF paste was filled into a crispy elongated hollow wafer made from rice flour, eggs, water, sugar, salt, and coconut meat (NumTrey-RUSF).
The NumTrey-RUSF version was developed with the potential for being used in the prevention of acute malnutrition. The NumTrey-Paste was developed with a nutrition composition aimed to match the requirements for RUTF products, with the exception that milk powder was replaced by fish powder. 4
Both the NumTrey-Paste and NumTrey-RUSF were included in a taste and acceptability trial, where they were compared with BP-100 and each other. A second generation of the wafer (NumTrey-RUTF) was developed based on the taste trial. The ingredients of the tested products are shown in Table 1. The indigenous small, freshwater fish species Cyprinidae, for example (scientific name/local name/common name), Henicorhynchus siamensis/Trey Riel/siamese mud carp, Paralaubuca carroini/Trey Slak Russey, Rasbora myersi/Trey Changva muol/Myer’s silver rasbora, and Puntioplites proctozystron/Trey Chrakeng/Snail eating barb was supplied by local fish sellers from Phnom Penh, Cambodia. The fresh fish was gutted and delivered on ice to the Royal University of Agriculture (RUA), Phnom Penh. At RUA, the fish was washed in 3 different steps: first with clean water, secondly chlorinated water (200 ppm), and last water with added turmeric powder (15-20 g or 3-4 teaspoons mixed with 4 L of clean water). The cleaned fish was then placed into a greenhouse-type solar dryer for small-scale dried food industries built by Bayer (Phnom Penh, Cambodia) and Silpakorn University (Bangkok, Thailand) within the SHARE project (Sustainable, Hygienic And Resource Efficient Solar Dryers for Cambodians) at RUA. 26 The fish was solar dried for approximately 1 to 2 days until the fish was dry. At a local food processing factory, Vissot, the dried fish was baked and ground. The fish was baked in 2 rounds. First, the fish was baked at 60°C to 70°C for 15 minutes. For the second round, the fish were turned and baked at 60°C to 70°C for another 10 minutes. The fish was baked in single-door, 2-tray oven (model RL 2; CG Machines, China). After being baked, the fish was ground into small particles using a Quanda grinder (model QD; Shanghai Quanda Food Machinery Co, Ltd, China), grinding 2 times for 3 to 4 minutes on high speed (2000 rpm).
Ingredients and Nutrition Composition of the Fish-Based NumTrey Products and BP-100 Per 100 g.
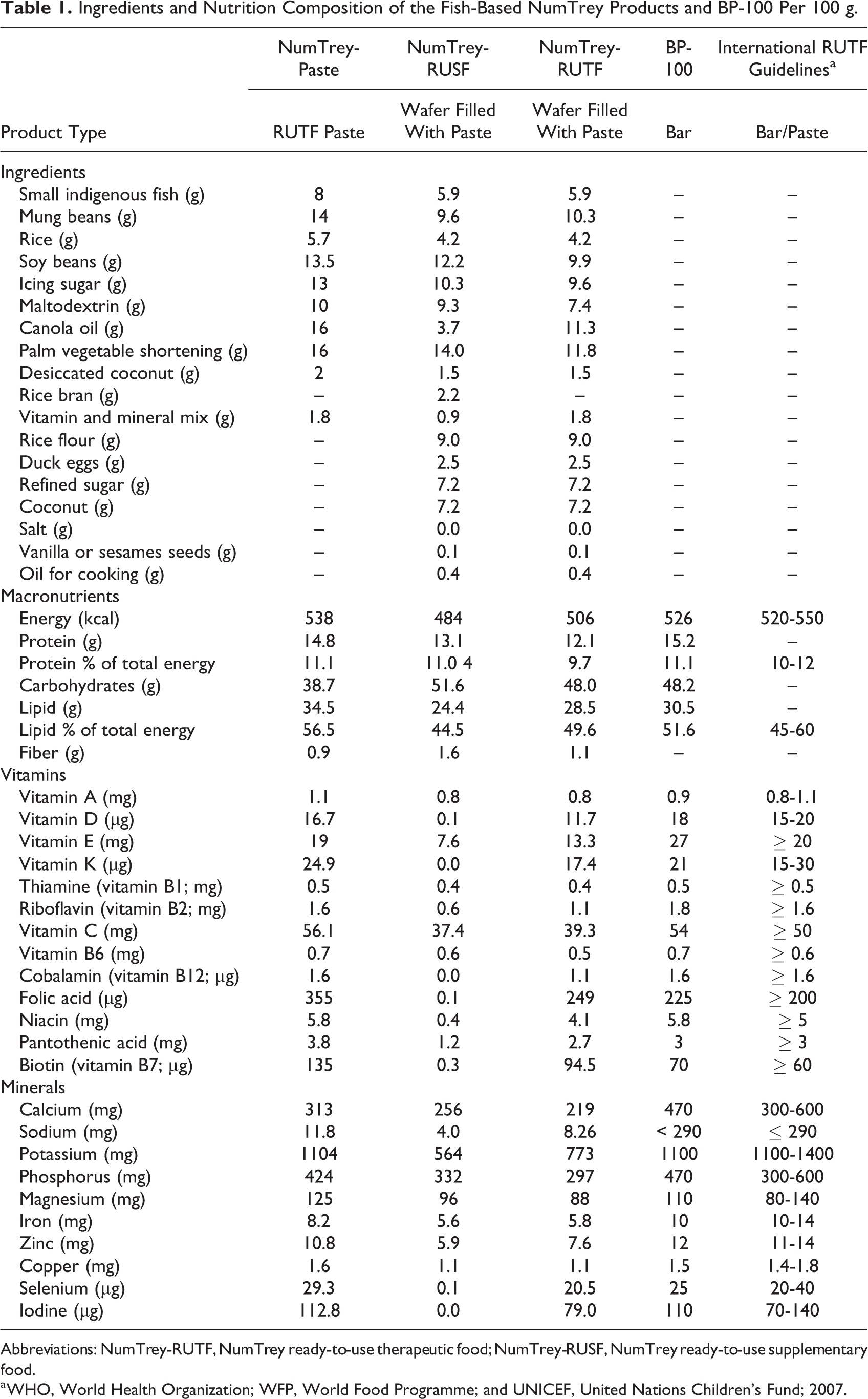
Abbreviations: NumTrey-RUTF, NumTrey ready-to-use therapeutic food; NumTrey-RUSF, NumTrey ready-to-use supplementary food.
a WHO, World Health Organization; WFP, World Food Programme; and UNICEF, United Nations Children's Fund; 2007.
Soybean and mung bean were purchased preroasted and were grounded at Vissot. Rice, the fish powder, and coconut were extruded before being mixed with the other ingredients.
The ingredients were extruded using a snack food extruder machine (model SLG65; Jinan Datong Machinery Co Ltd, China) at a local factory (Lyly Foods, Phnom Penh, Cambodia) premises separately. The temperature for the extrusion ranked from 150°C to 170°C for 4 to 8 seconds. The extruded products were packed and transported back to Vissot the same day. When arrived back at Vissot, the extruded products were grinded further 2 times 4 to 5 minutes until a fine mass was obtained. The rest of the ingredients were hereafter added and mixed well using a 25-kg Sinmag mixer (model SM, Taichung, Taiwan). Vitamin and mineral mix (DSM) was added to the NumTrey-Paste and the NumTrey-RUSF to increase the micronutrient content and to follow United Nations Children’s Fund (UNICEF) and World Health Organization specifications for RUTF products. 4
The production process and staff were trained and employed good hygienic practices and manufacturing practices. The NumTrey-Paste was packed in a portion-sized sachet with 92 g of paste in each sachet to be squeezed out for consumption without prior preparation required.
Nutrition Composition of the Products
The nutrition composition of the NumTrey products and BP-100 is shown in Table 1. The nutrition composition of the NumTrey-Paste for the purpose of being an RUTF met the international guidelines for most minerals and vitamins, except the paste contained 8% small whole fish powder instead of milk powder. BP-100 is produced by Compact and was imported from Norway. Information on BP-100’s nutritional composition was obtained from the label on the packaging.
The addition of the wafer slightly changes the nutrition composition of the final product. The NumTrey-RUTF differs from the NumTrey-RUSF by having higher energy, higher lipid, and higher micronutrient density to fulfill the requirements for RUTF products.
Microbiological safety was analyzed at the Pasteur Institute in Cambodia for every fifth batch of final product produced to ensure they complied with WHO/UNICEF requirements for the microbiological safety of RUTF products. All 19 tests met WHO/UNICEF requirements for ready-to-use products and were bacteriologically satisfactorily.
Assessment of Acceptability
The locally produced products were assessed for acceptability in 2 separate trials: a taste trial and during an SAM intervention trial among children receiving treatment for SAM.
Taste Trial
The trial was a crossover study using a sensory scoring test, followed by a ranking of the products to compare 2 fish-based products NumTrey-Paste and NumTrey-RUSF with BP-100. The results of the taste trial were used for subsequent development of the NumTrey-RUTF wafer, and it was therefore not included in the taste trial assessment. The composition of the 2 fish-based products is described previously.
Participants and taste trial
All children aged 6 months to 18 years and their caregivers visiting the National Pediatric Hospital in Phnom Penh, Cambodia were invited to participate in the trial in the period of October 26-30, 2015. The rationale behind this age range is that all children, that is, everyone below 18 years of age, who came to the National Pediatric Hospital were eligible. In Cambodia, acute malnutrition in older children is quite common, due to underlying infections, for example, HIV and tuberculosis. Therefore, the hospital was also interested to know whether the product could be used for older children with acute malnutrition. Children and caregivers with a fish or soybean allergy and children who were too ill to taste the products (eg, children with high fever or nausea) were excluded.
First, the child undertook the sensory test and product ranking, then the caregiver undertook the same sensory test and ranking. In cases where children were below 3 years of age, the caregiver evaluated and ranked the products based on the child’s nonverbal communication. The approach of using nonverbal communication as an indication of infants and young children’s acceptance of food products is a commonly used method in the baby food industry. 27 The method has previously been used in acceptability trials of food products for young children but is less reliable than results obtained through verbal communication with older children. 28,29 This evaluation occurred before the caregiver had tasted the products. For ranking in these cases, the caregiver was asked to explain which reaction the child had made in order for them to rank the product as they did.
Sensory testing and ranking
The child and subsequently the caregiver were presented with one product at a time. The products were presented in a clear box with a white spoon, in order to avoid any potential influence the colors of the materials may have on the perception of the products. 30
The child and caregiver were asked to taste each product a minimum of 2 times before evaluating a set of organoleptic qualities: appearance, color, smell, looks appetizing, taste, texture, swallowing, and overall which was the overall impression of the product, given on a scale of 5. This was to justify continuing the intervention trial, though it does not indicate acceptability over a longer period of time, with a high daily intake, as required in the treatment of SAM.
A 5-point hedonic scale, starting from 1 = very good, 2 = good, 3 = neutral, 4 = bad, to 5 = very bad was used. The scale was presented as “smiley” faces. The 5-point hedonic scale has been used as a standard tool in previous studies on food acceptability and it has been found to be appropriate for children between 4 and 10 years and free from cultural biases. 31 A score of 1 to 3 (very good to neutral) was assumed to be the minimum level of acceptance of the organoleptic qualities.
The products were presented in random order in 6 different combinations, to prevent the order of presentation influencing the child’s or caregiver’s evaluations. After each evaluation of a product, the child and caregiver were asked to rinse their mouth with water.
The ranking was conducted after the sensory test. The participants were presented with all 3 products at the same time. They were asked to rank the products on a 3-point scale using smiley faces from 1 = liked best, 2 = liked second best, and 3 = liked least.
Acceptability During SAM Intervention Trial
Participants and intervention trial
The results from the taste trial showed a lower acceptability of the NumTrey-Paste compared with wafer: NumTrey-RUSF. These results were used to develop the wafer NumTrey-RUTF filled with RUTF paste. After the development of the NumTrey-RUTF, this was used in an intervention trial. The interventional trial tested the effectiveness of the NumTrey-RUTF in comparison with the currently used RUTF BP-100 in the treatment of SAM among Cambodian children aged 6 and 59 months. The results from the effectiveness trial will be reported separately.
The trial was conducted at the National Pediatric Hospital from September 2015 to January 2017 among children visiting the outpatient treatment for SAM. At the first phase of the trial, the NumTrey-Paste was piloted among children aged from 6 months to 18 years before the taste trial was conducted. Children with complications were admitted to the hospital for SAM treatment. They were initially stabilized at the hospital following standard protocols. Children admitted to the inpatient treatment were included in the trial only when the child was ready to get discharged from inpatient treatment to continue an 8-week outpatient treatment. During the outpatient treatment, the child was provided with an RUTF for consumption at home. Children with SAM but without complications who did not require inpatient treatment started directly with the 8-week outpatient SAM treatment. Each child consumed the RUTF at home; however, they were required to come to the hospital fortnightly for a follow-up visit. At each follow-up visits, the food ration was calculated and counted before it was provided to the caregiver. The food ration was based on the weight and it was calculated to between 160 and 180 kcal/kg for both RUTFs (Table 2). For the NumTrey-RUTF, the food ration calculation is based on the paste inside the wafer not including the wafer, which was seen as additional calories since the content of the wafer is of minor importance. The caregivers were asked to bring back unused packages of RUTF to assess utilization of the RUTFs during the whole trial period.
Information Sheet on NumTrey-RUTF and BP-100 Food Ration Based on the Child’s Weight.
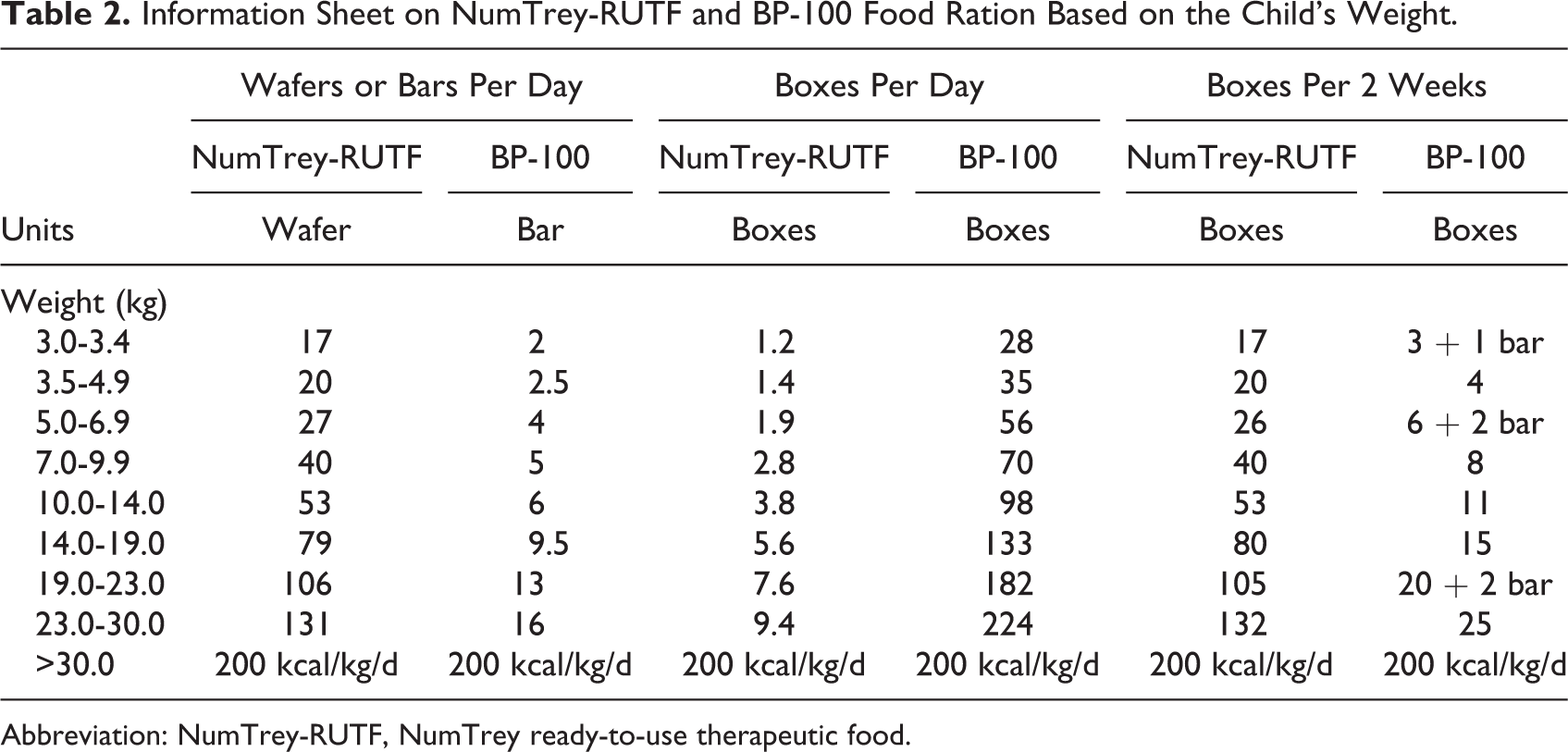
Abbreviation: NumTrey-RUTF, NumTrey ready-to-use therapeutic food.
A total of 121 children were randomized to receive either NumTrey-RUTF or BP-100 during this period. Randomization was done according to a computer-generated randomization list. At each fortnightly follow-up visit, the acceptability, sharing, and loss or waste of the products were assessed.
Evaluation of the RUTF in SAM treatment
At each fortnightly visit, caregivers were asked to evaluate the RUTF the child was randomized to (either NumTrey-RUTF or BP-100) using a similar 5-point hedonic scale with smiley faces. The caregiver evaluated on the child’s behalf how they thought their child liked the RUTF they were randomized to. The evaluation was as either 1 = liked a lot, 2 = liked a little, 3 = neither liked nor disliked, 4 = disliked a little, or 5 = disliked a lot. Acceptability of the products was considered to be a score of 1 to 3 (liked a lot to neither liked or disliked). Caregivers were also asked questions concerning sharing of the RUTFs and waste of the RUTF during the trial.
Sample Size
Due to limited resources, the sample size in the taste trial was based on available cases at the National Pediatric Hospital in Phnom Penh within the trial period. The main objective of the SAM intervention trials was to compare the superiority of any of the 2 RUTFs (NumTrey-RUTF and BP-100) with regard to weight gain (g/kg/d). An average increase in body weight of 4 g/kg/d was aimed for. 32,33 To detect a 10% difference, which was regarded as biological relevant (ie, a weight gain of 3.6 g/kg/d) and with an SD of 0.7 g/kg/d, a sample size of 49 patients per intervention group was required (80% power; α = .05; 2 sided). To allow for dropout, it was planned to recruit 60 patients in each intervention group, thus in total 120 children. The hedonic rating, sharing, waste, and utilization of the RUTF were secondary outcomes of the intervention trial.
Statistical Analyses
For the taste trial, all data were double entered into EpiData 3.1 software (The EpiData Association, Odense, Denmark). The data from the SAM intervention trial were entered directly into tablets using an online application tool. All data were analyzed using SPSS 23.0 and 25.0 for Windows (IBM Corporation, Armonk, New York). For each organoleptic quality, the corresponding categories (1 through 5) were compared between products using a Pearson χ2 test.
Acceptability of the organoleptic qualities in the taste trial was categorized as hedonic points from 1 to 3 (1 = very good to 3 = neutral) and unacceptable from 4 to 5 (4 = bad to 5 = very bad; not reported). Ranking of the products was analyzed using the 3 categories stated in the Materials and Methods section.
For categorical outcomes in the taste trial and SAM intervention trial (acceptability of organoleptic qualities, a ranking of the products, fortnightly rating, sharing and the waste of RUTF), χ2 test was used. Median Interquartile range (IQR) is reported for all organoleptic qualities and ranking of the products assessed using the hedonic and Likert scales.
The percentage of RUTF utilized for the whole SAM intervention trial was calculated for each child:
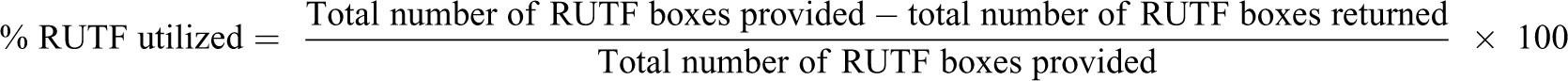
The percentage of RUTF utilized for each patient was compared between groups by using Analysis of Covariance (ANCOVA) adjusted for age and gender. For the categories liked the most, liked second best, and liked least product difference between age and gender were evaluated by means of ordered logistic regression.
P values for comparisons across NumTrey-Paste, NumTrey-RUSF, and BP-100 were adjusted using the Bonferroni method. P value of < .05 was considered statistically significant.
Ethical Approval and Informed Consent
The trials were conducted in accordance with the ethical standards written in the Declaration of Helsinki. Ethical approval was permitted on the 24th of April by the National Ethical Committee for Health Research, Ministry of Health, The Royal Government of Cambodia (120 NEHR).
Written and oral informed consent was obtained from the child’s parent or legal guardian. No additional consent was obtained from any of the children. All participants were compensated for their time with gifts suitable in the context. The SAM intervention trial is registered at http://ClinicalTrials.gov (NCT02907424).
Results
Taste Trial
In total, 62 children and caregivers were invited to participate in the trial. Of those, 2 children were excluded due to their age (< 6 months old) and 2 children > 6 months were still exclusively breastfed. Three children refused to taste the products, 2 children had to leave during the interview, and 1 child was too sick and was therefore excluded. Two caregivers had to leave after the children had performed the sensory test and ranking. Three caregivers had 2 children (siblings) participating in the trial. Hence, in total, 52 children and 57 caregivers were included in the taste trial. Characteristic of the participants are shown in Table 3.
Demographic Characteristics of Children and Their Caregivers Participating in the Taste Trial of the Locally Produced RUTF and RUSFa.
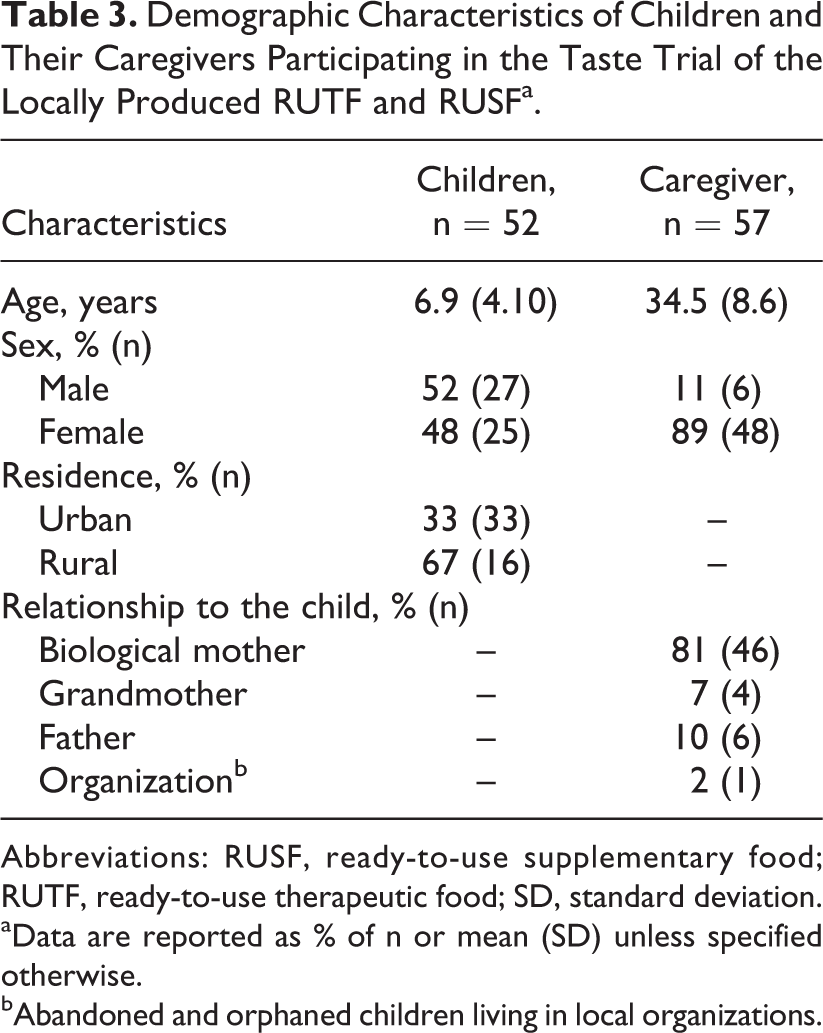
Abbreviations: RUSF, ready-to-use supplementary food; RUTF, ready-to-use therapeutic food; SD, standard deviation.
aData are reported as % of n or mean (SD) unless specified otherwise.
bAbandoned and orphaned children living in local organizations.
The mean (SD) age (years) of the participating children and caregivers was 6.9 (4.10) and 34.5 (8.6), respectively. Of the caregivers, 81% were the child’s biological mother, 10% were the child’s biological father, and 7% were the grandmother of the participating child. The clinical reasons for the children visiting the hospital were multiple, not restricted to acute malnutrition.
Acceptability by children and their caregivers
The NumTrey-Paste was found to be considerable less “acceptable” by children and caregivers, compared to BP-100 and NumTrey-RUSF in appearance, color, and smell (Table 4, P < .05). With regard to the organoleptic qualities, texture, and ability to swallow the product, no statistically significant differences were found among the products. For all 8 organoleptic qualities, median Interquartile range (IQR) on the 5-point hedonic scale can be found in Table 4.
Taste Trial: Proportion of Children (n = 52) and Their Caregivers (n = 57) Assessing Various Organoleptic Qualities as Acceptablec and the Median Hedonic Scores (Scale 1-5).a
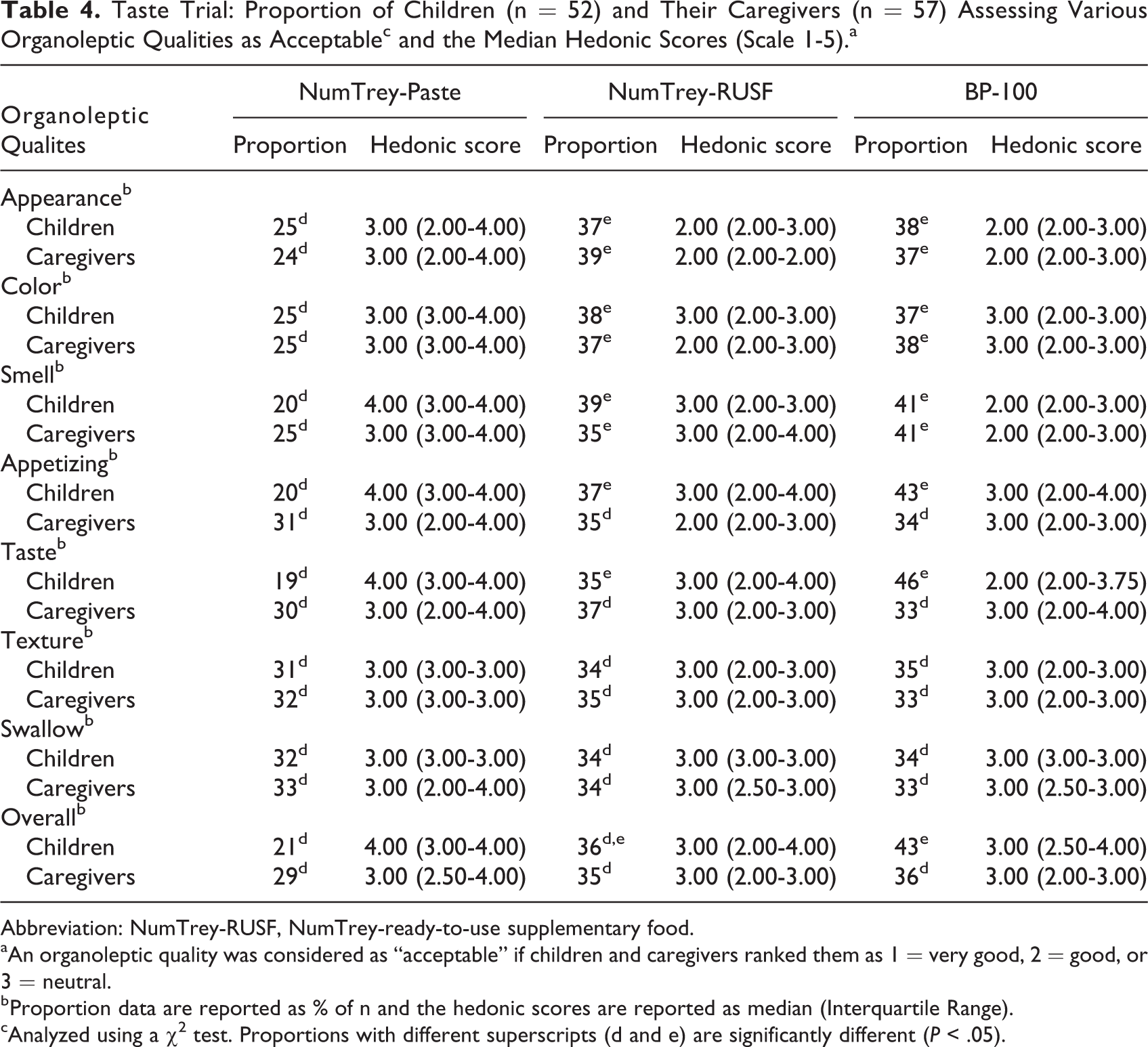
Abbreviation: NumTrey-RUSF, NumTrey-ready-to-use supplementary food.
aAn organoleptic quality was considered as “acceptable” if children and caregivers ranked them as 1 = very good, 2 = good, or 3 = neutral.
b Proportion data are reported as % of n and the hedonic scores are reported as median (Interquartile Range).
cAnalyzed using a χ2 test. Proportions with different superscripts (d and e) are significantly different (P < .05).
Overall, a lower proportion of the participants found NumTrey-Paste acceptable compared to the NumTrey-RUSF and BP-100 (Table 4). However, the median score for most caregivers and children ranked the NumTrey-Paste as neutral on most organoleptic qualities (Table 4). No statistically significant differences were found between BP-100 and NumTrey-RUSF in any of the organoleptic qualities in acceptability by children.
There were no significant differences (Table 5, P < .05) in the proportions of either children or caregivers ranking BP-100 and NumTrey-RUSF as the products they liked the most. NumTrey-RUSF scored a median Interquartile range (IQR) of 2.00 (1.00-2.00) and 1.00 (1.00-2.00) among children and their caregivers, respectively. In addition, BP-100 scored a median Interquartile range (IQR) of 2.00 (1.00-2.00) and 2.00 (1.00-2.00) among children and their caregivers, respectively (data not shown). Among both caregivers and children, NumTrey-Paste was ranked as the product they liked least of the 3. For the 3-point hedonic scale, NumTrey-Paste scored a median Interquartile range (IQR) of 3.00 (2.00-3.00) and 3.00 (2.00-3.00) for children and caregiver, respectively (data not shown). No statistically significant difference in the children’s preference of the products between gender or age and ranking was found in a multinomial logistic regression model (data not shown).
Taste Trial: Product Ranking by Children (n = 52) and Their Caregivers (n = 57).a
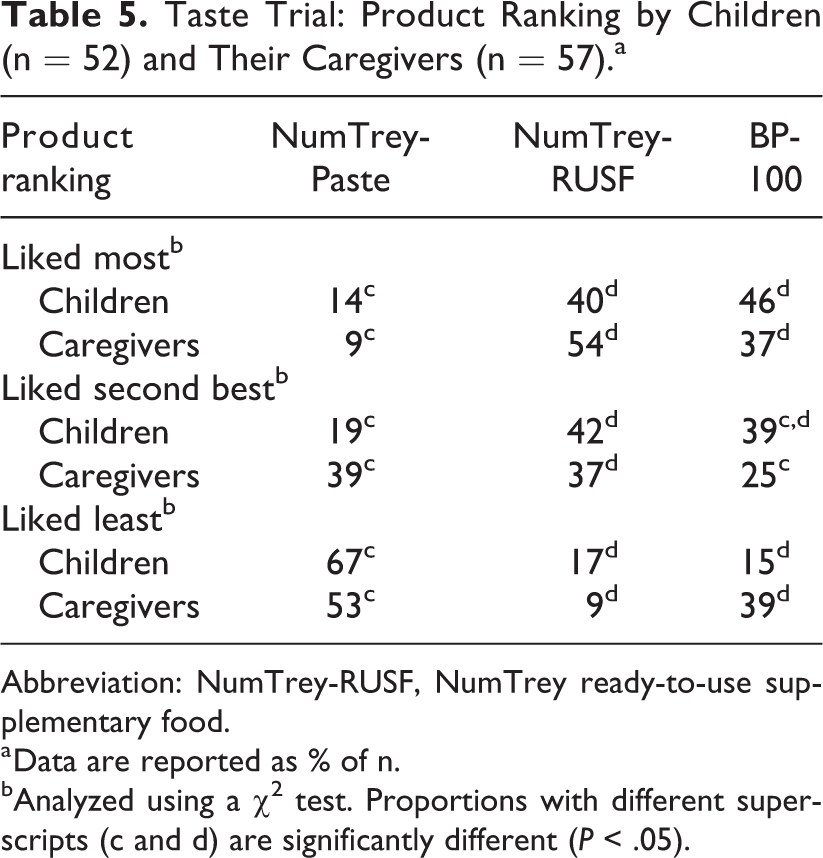
Abbreviation: NumTrey-RUSF, NumTrey ready-to-use supplementary food.
a Data are reported as % of n.
bAnalyzed using a χ2 test. Proportions with different superscripts (c and d) are significantly different (P < .05).
Acceptability of NumTrey-RUTF During SAM Treatment
The acceptability of NumTrey-RUTF and BP-100 used in the home-based treatment of children with SAM was evaluated fortnightly based on caregivers’ perception on how the child liked the RUTF, as shown in Table 6. At the first fortnightly visit, 72% and 92% of the caregivers to those children receiving NumTrey-RUTF and BP-100, respectively, found their RUTF to be acceptable (P = .036). A tendency toward increased acceptability over the treatment period was seen for NumTrey-RUTF (from 72% to 86%), whereas a variance was seen in the acceptability of BP-100 over the treatment period. For the last 3 fortnightly visits, no statistically significant difference in acceptability was found between the 2 products (P = .790, P = .213, and P = .710, respectively). The median Interquartile range (IQR) score on the 5-point hedonic scale was 2.00 (2.00-4.00) and 2.00 (2.00-3.00) for NumTrey-RUTF and BP-100, respectively, for the first follow-up visit (data not shown). During the last 3 fortnightly visit, a trend toward NumTrey-RUTF ranked higher on the 5-point hedonic scale compared to BP-100, for example, at end line NumTrey-RUTF ranked 2.00 (2.00-3.00) compared to 2.00 (1.00-2.00) for BP-100 (data not shown).
Fortnightly Hedonic Rating of the NumTrey-RUTF and BP-100 by Children Randomized to One of the Products for 8-Week Home-Based Treatment of Uncomplicated SAM.a
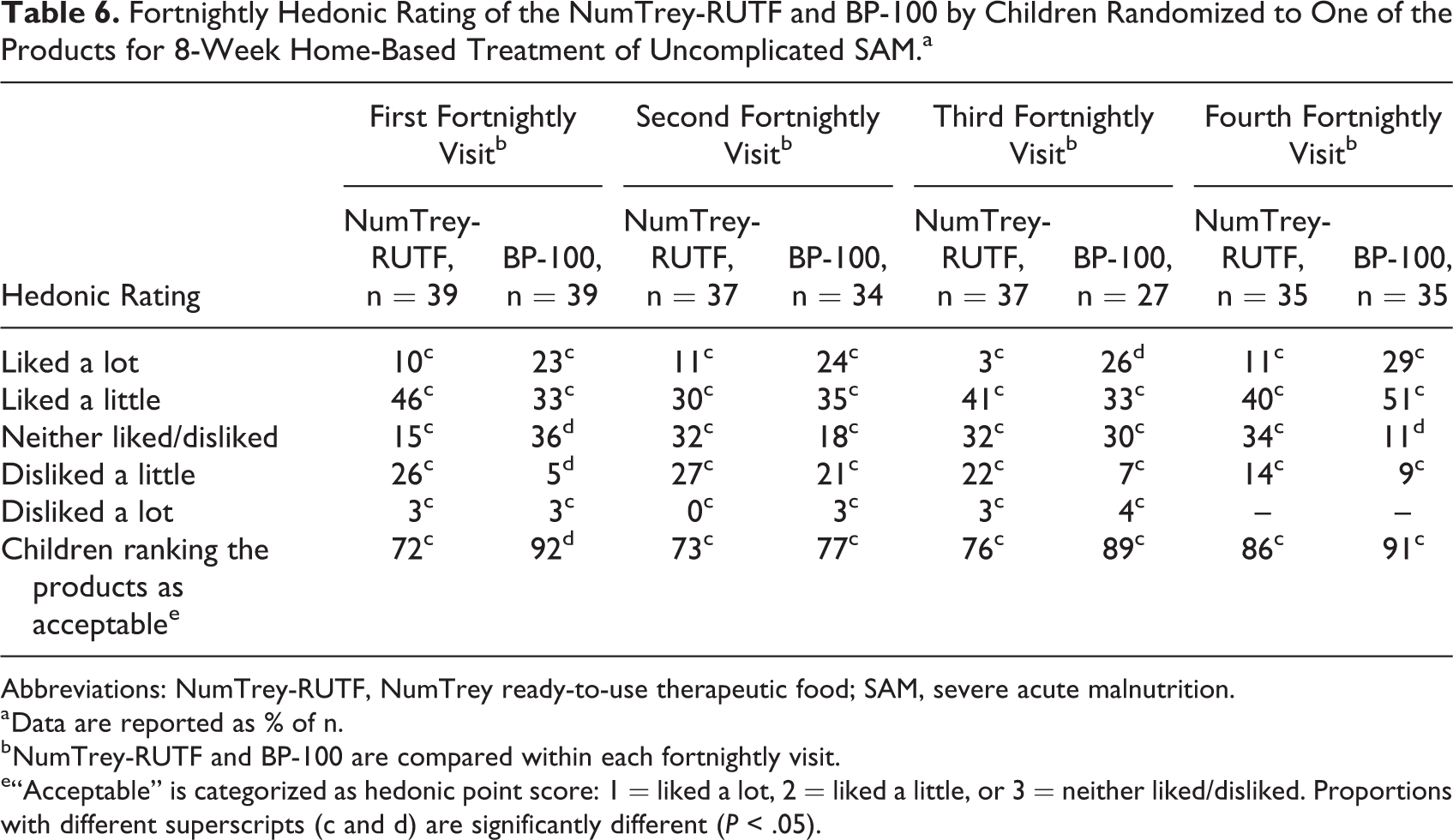
Abbreviations: NumTrey-RUTF, NumTrey ready-to-use therapeutic food; SAM, severe acute malnutrition.
a Data are reported as % of n.
b NumTrey-RUTF and BP-100 are compared within each fortnightly visit.
e“Acceptable” is categorized as hedonic point score: 1 = liked a lot, 2 = liked a little, or 3 = neither liked/disliked. Proportions with different superscripts (c and d) are significantly different (P < .05).
The children returned on average for 2.6 of 4 scheduled fortnightly visits during treatment. At each visit, the caregiver returned unused packages of the RUTF the child had received as treatment and received a ratio for the next 2 weeks. Based on counts of returned RUTF packages, approximately half of the distributed RUTF was utilized.
The mean utilized percentage of the RUTF for the treatment period was 48.1% and 51.7% of NumTrey-RUTF and BP-100, respectively (P = .525, 95% confidence interval: 7.6-15.3), after adjusting for age and gender. Upon questioning, approximately one-fourth (27%) of the caregivers whose children received NumTrey-RUTF and 41% of the caregivers receiving BP-100 reported they had either sold, given away, thrown away, lost, or the RUTF was eaten by animals. Sharing (reported sharing minimum 1 time during the trial) of the RUTF during the whole trial, the percent of all participants was 64.6% for NumTrey-RUTF and 46.7% for BP-100. Of these, the majority was shared with siblings (33.3% and 25.0% for NumTrey and BP-100, respectively). The leftovers of RUTF during each feeding session was indicated in > 80% of the cases as being “a small bite.”
Discussion
These trials demonstrated that food supplements suitable for prevention (RUSF products) and treatment (RUTF products) of SAM can be produced based on locally available ingredients including small fish species in Cambodia. It also showed that a pure paste product is less acceptable in terms of organoleptic properties to children and caregivers in Cambodia than other products, a finding earlier reported from Vietnam. 12 The lower preference for the paste can be attributed to the characteristic of the product being a paste. A similar dislike toward a paste-based RUTF was found in Vietnam. 34 Also, an earlier report on the low acceptability of Plumpy’Nut in Cambodia attributed this to the fact that the product was a paste. 18 The results from Vietnam and Cambodia might suggest that a specific regional unfamiliarity with a sweet and fat paste-like food might explain the limited success of the global most used RUTF paste: Plumpy’Nut.
When we filled the paste in a crispy wafer and turned the product into a more familiar “snack,” this barrier appeared to be less pronounced. The subsequent NumTrey-RUTF resulted in a higher acceptability, not different from the acceptability of the milk-based BP-100 RUTF bar, which is the standard product currently used for SAM treatment in Cambodia. The current nutrition composition was made for trial and may undergo further development. For example, optimizing the recipe and the ratio between the wafer and paste is needed to ensure that the NumTrey-RUTF fulfills international nutrition composition guidelines for RUTFs.
In the taste trial of the paste versus the wafer (NumTrey-RUSF) and the bar (BP-100), the age range of the children was large, from 6 months old to 18 years. The large age range could help to identify age-specific differences in preferences of products. Infants and young children may, for example, have a preference for a soft paste. However, the trial did not find any age-specific preferences among the 3 products. Caregivers and children were very consistent in their preferences, showing a general reluctance toward the paste-based RUTF. Both the fish-based NumTrey-Paste and NumTrey-wafer had a pronounced smell and taste of fish. If the smell and taste of fish was a determinant for acceptability, it would have affected both products. Although children may detect “unpleasant” smells, they may not be able to categories these as “unpleasant” until they are approximately 5 years of age. 35 Actually, there was a tendency among caregivers to prefer the NumTrey-RUSF over the BP-100 bar.
A novel product may be met with some reluctance in the beginning. This may be borne out by the lower acceptability of NumTrey-RUTF as seen at the first fortnightly visit compared to BP-100. On the other hand, BP-100 has been a part of the SAM treatment protocol for years in Cambodia and most caregivers were familiar with the product beforehand. In general, exposure to the same food several times drives preferences among children, unless caregivers teach the child that it is unpleasant. 36 In this SAM intervention trial, from the second fortnightly visit onward, the acceptability of NumTrey-RUTF increased. Hence, this may indicate that the children were getting used to the new product and that caregivers were encouraging acceptance. The total treatment period was 8 weeks, in which time the children were randomized to only 1 of the 2 products. The large intake required and long period could have affected the acceptability of the products as the trial progressed, but this is not reflected in the results of the present trial, suggesting long-term acceptability of the products. To the authors’ knowledge, no studies have tested acceptability or changes in acceptability over a longer time period. In both treatment groups, sharing occurred, as well as wasting of a small amount of the products, as is to be expected in field settings such as the one in which the present trial was conducted.
Strengths and Weaknesses of These Trials
This trial contributes to the literature comparing a locally produced non-milk-based RUTF compared with a commercial milk-based RUTF on acceptability in Cambodia. The acceptability was both tested during a taste trial, thus short-term, and during an 8-week intervention trial measuring long-term acceptability. The taste trial was conducted among children and their caregivers visiting the outpatient department at a local pediatric hospital. Information on children sickness was not collected during the trial; however, severely sick children were excluded and the children or caregiver were not obligated to participate. The taste trial design, the target groups of the food, and the wide age range did not allow to measure the amount of food consumed as the indictor of acceptability of the products, though this would have been a more objective measure of acceptability. However, combining the crossover design, the use of acknowledge methods, comparing several foods including the use of ranking in the taste trial and including continues evaluation of the product during a longer time period, makes these trials robust.
We have shown that small dried fish can be used in the development of products for prevention and treatment of acute malnutrition in Cambodia. Fish powder can be substituted for milk in RUSF and RUTF products.
We confirmed earlier observation that lower proportions of children and caregiver ranked a paste-like product as acceptable compared to other ready-to-use products. The same paste wrapped in a crispy wafer is as acceptable as the milk-based bar currently used in the treatment of SAM in Cambodia.
The large number of severely wasted Cambodian children who are not receiving treatment adds to the burden of morbidity and mortality in children less than 5 years of age, and it is likely to limit the optimal growth of thousands of children. Due to limited budget available for supply through government and development partners in nonemergency, middle-income Cambodia, it is essential to develop affordable and well-accepted ready-to-use therapeutic food such as NumTrey.
Footnotes
Authors’ Note
C.C., A.L., N.R., S.S., M.A.D., and F.T.W. designed the trials. A.L., N.R., and F.T.W. secured the funding. F.T.W., C.C., D.S., B.B., A.L., N.R., and M.D. participated in the development of the questionnaires. S.S. collected the data, conducted the statistical analysis, and wrote the first draft of the manuscript. F.T.W. and N.R. contributed on the statistical analysis and write-up of the first draft. All authors have read, critically revised the manuscript, and approved the manuscript for submission.
Acknowledgments
The author(s) would like to thank the staff and patients at the National Pediatric Hospital in Phnom Penh, Cambodia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These trials were financially supported by UNICEF’s national committees (Australia, Republic of Korea and Hong Kong), University of Copenhagen, Denmark, and Institut de Recherche pour le Dévelopment (IRD). Danida MSc travel grant provided financial support to the corresponding authors travel to Cambodia, and Neys-Van Hoogstraten Foundation provided finical support for the effectiveness trial. The sources of funding had no influence on how the data were collected and analyzed, or in the publication of results and interpretation.
