Abstract
Background:
Cow’s milk is a core ingredient in foods for prevention and treatment of undernutrition in children. It promotes growth, but there is limited knowledge of which components in milk have growth-stimulating effects.
Objective:
To discuss the growth-stimulating effects of milk in undernourished children with emphasis on protein, lactose, and minerals.
Methods:
We reviewed literature on dairy and growth in undernourished children.
Results:
Cow’s milk has a specific stimulating effect on linear growth, even in well-nourished children, and it may stimulate weight gain and muscle accretion in wasted children. Dairy protein has high protein quality scores with no major differences between whey protein concentrate and dried skimmed milk. Lactose has potential growth-stimulating effects, and the high content of potassium, phosphorus, magnesium, and zinc in milk is also likely to stimulate growth. Furthermore, a high dairy content reduces the amount of antinutrients from plant protein.
Conclusions:
Dairy protein, lactose, and bioavailable minerals all have potential growth-stimulating effects, which make cow’s milk an important ingredient in foods for undernourished children. Different dairy ingredients vary with regard to protein amount and quality, content of lactose, and minerals, and thereby growth stimulating effects. This should be taken into consideration when deciding which dairy ingredient to use. Challenges include the relatively high cost of dairy. Therefore, future studies should include cost-effectiveness analysis and assessment of the desirable content of each growth-promoting milk component.
Introduction
Undernutrition is a major global problem. Based on estimates of undernutrition among children below 5 years, 165 million are stunted, 33 million have moderate acute malnutrition (MAM), and 19 million have severe acute malnutrition (SAM). 1 Furthermore, undernutrition is estimated to contribute to 3.1 million deaths per year of children below 5 years of age. It is also estimated that more than 450 000 deaths can be saved if treatment of MAM and SAM is optimized. 2
Milk contains all the nutrients needed to support growth in the offspring and therefore contains ingredients with growth-stimulating effects. Cow’s milk has been used as a key ingredient in foods aimed at preventing and treating undernutrition in children. 3 -5 The aim of this short paper is to discuss the components in cow’s milk which have potential growth-stimulating effects with emphasis on protein, lactose, and minerals.
Specifications for Using Dairy in Foods for Undernourished Children
Standard therapeutic foods used for treating SAM contain dairy ingredients (Table 1). In F-75 and F-100, which are used for inpatient treatment of SAM, 100% of the protein comes from dairy. Ready-to-use therapeutic foods (RUTFs) are used for community-based treatment of SAM, and according to specifications from United Nations International Children’s Emergency Fund (UNICEF) and United States Agency for International Development (USAID), they should contain at least 50% of protein from dairy. 6 For supplementary foods used for treatment of MAM, there is no agreement on the content of dairy. In a technical note from World Health Organization from 2012 on supplementary foods for MAM, it was stated that the protein quality is sufficient if the protein digestibility-corrected amino acid score (PDCAAS) is equal to or above 70%. This can be obtained with a cereal/legume mixture with or without milk or other animal proteins. 7 In a note, the authors wrote that milk improves the PDCAAS, has a specific stimulating effect on insulin-like growth factor 1 (IGF-1) and linear growth, and does not contain antinutrients. They also called for further research. Specifications for ready-to-use supplementary foods (RUSFs) for treatment of MAM and lipid-based products distributed for prevention of acute malnutrition lipid-based nutrient supplement medium quantity (LNS-MQ ) from World Food Programme (WFP) and USAID state that one-third of protein should come from milk powder. 8 The composition of RUSF and LNS-MQ is exactly the same, but the quantity supplied per day differ with 100 g RUSF/day for treatment of MAM and 50 g LNS-MQ/day for prevention of acute malnutrition There is also dairy in some newly developed fortified blended foods (FBFs). The WFP has developed Super Cereal plus which includes 8 g of skimmed milk powder (SMP) per 100 g, which is equivalent to approximately 25% of the protein. 9 The USAID has developed and is at present testing CSB14, which contains 3 g whey protein concentrate with 80 % protein per 100 g, which is approximately 17% of the protein content. 10
Dairy in Food Products for Treatment of SAM and MAM and Prevention of Acute Malnutrition.
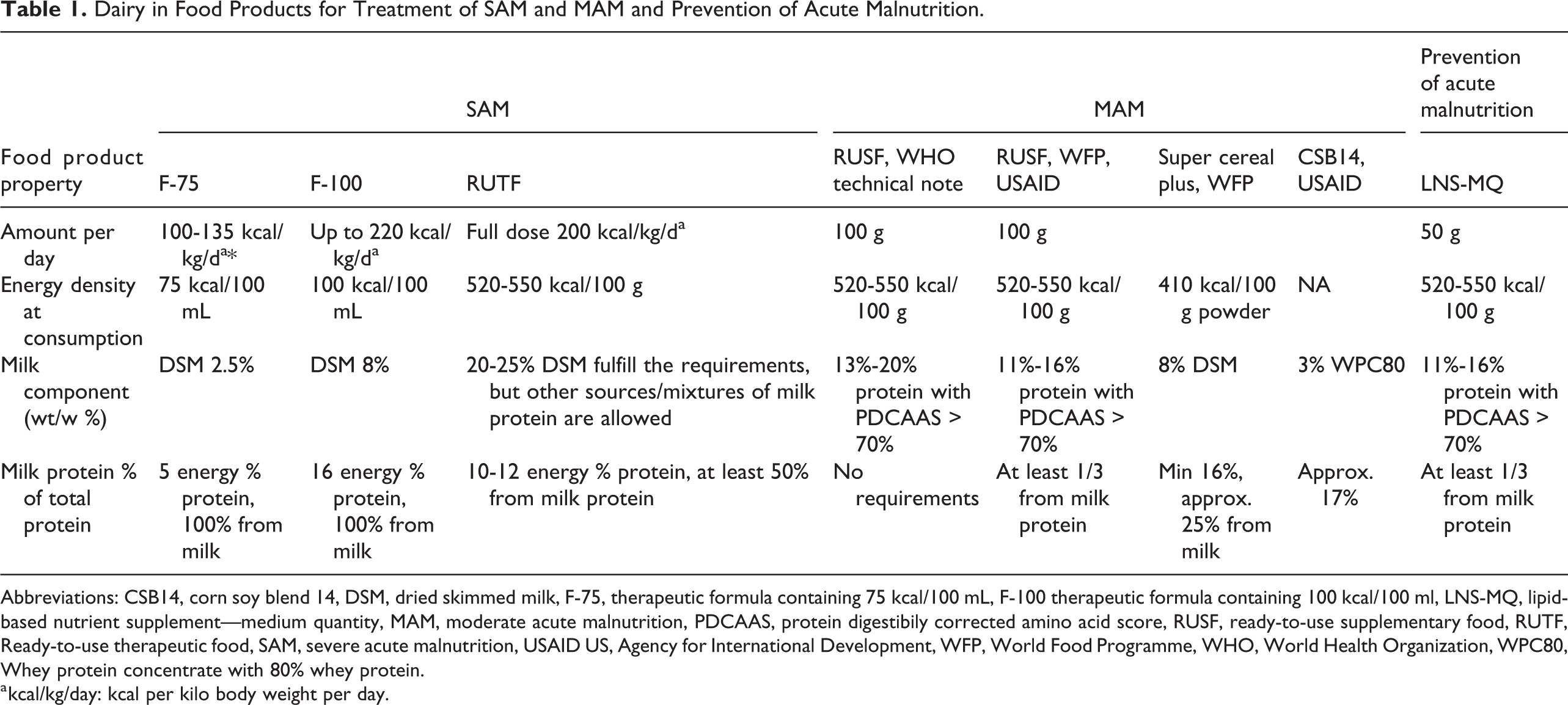
Abbreviations: CSB14, corn soy blend 14, DSM, dried skimmed milk, F-75, therapeutic formula containing 75 kcal/100 mL, F-100 therapeutic formula containing 100 kcal/100 ml, LNS-MQ, lipid-based nutrient supplement—medium quantity, MAM, moderate acute malnutrition, PDCAAS, protein digestibily corrected amino acid score, RUSF, ready-to-use supplementary food, RUTF, Ready-to-use therapeutic food, SAM, severe acute malnutrition, USAID US, Agency for International Development, WFP, World Food Programme, WHO, World Health Organization, WPC80, Whey protein concentrate with 80% whey protein.
a kcal/kg/day: kcal per kilo body weight per day.
Dairy and Linear Growth
Many studies have examined the association between milk intake and linear growth. 11,12 The strongest evidence comes from intervention studies in low-income countries; however, many observational studies from high-income countries suggest that there is also an effect in well-nourished populations. In a Dutch study analyzing the secular trend in adult stature from 1955 to 1997, it was suggested that an explanation for the Dutch being among the tallest in the world is that their milk intake is very high. 13 One of the mechanisms behind the stimulating effect of dairy products on linear growth is likely to be a direct stimulation of IGF-1 and insulin. In short-term intervention studies in well-nourished Danish boys, a high intake of milk but not meat increased the levels of serum IGF-1 and insulin. 14 A similar intervention study tested the effects of whey and casein and found results suggesting that casein mainly stimulates IGF-1 and whey mainly stimulates insulin. 15 Thus, both casein and whey are likely to have a role in the stimulating effect of dairy on linear growth.
Dairy in Treatment of Wasting
Several studies, reviews, and comments have tested and discussed the effects and implications of using dairy in treating SAM and MAM. 3 -5 Dairy ingredients are generally recognized as good for catch-up growth; however, they are also quite expensive. To reduce costs of RUTF for treatment of SAM, researchers have tried to substitute more expensive milk ingredients with cheaper milk ingredients, 16 to reduce the amount of milk, 17 to exchange milk protein with plant protein, 18 and to divide SAM children into age groups below and above 2 years of age to see if they respond differently to therapeutic food with/without milk. 19 The results of these studies showed that whey protein concentrate was noninferior to SMP regarding weight gain and recovery rate, 16 that 25% SMP resulted in higher recovery rate, weight, height, and mid-upper arm circumference (MUAC) gain than 10% SMP + soya, 17 and that milk-free RUTF resulted in lower recovery rates than standard RUTF, mainly due to lower recovery in children below 2 years of age. 18 A subsequent study testing a modified version of the milk-free RUTF in children below and above 2 years found inferior recovery rates of the milk-free RUTF compared to standard RUTF in children below but not above 2 years. 19
In a recent nonblinded study, free amino acids were added to milk-free RUTF and compared to standard RUTF with milk and RUTF with both free amino acids and a low content of milk (9.3%). 20 The RUTFs with free amino acids had the same recovery rate as the standard RUTF with milk in children with SAM both below and above 2 years. A limitation is that there was no information of which free amino acids were added and the cost of adding free amino acids to RUTF. Other factors than the content of milk differed between the studies and are likely to have influenced the results.
The effect of dairy in the treatment of MAM was discussed in a narrative review by Suri et al. 21 Although the included studies were quite heterogeneous, the authors concluded that in FBF there was a benefit of using dairy while they found that the benefits of using dairy in RUSF required further evidence. Two recent studies which were not included in the review are summarized here. Ready-to-use supplementary food with whey protein combined with whey permeate was compared with soy RUSF in children with MAM. 22 Recovery rates and growth was better among those getting the milk-based RUSF, but the differences were small. An intervention study in Burkina Faso with 1609 children between 6 and 24 months with MAM assessed the effects of 12 supplementary foods in a 2 × 2 × 3 factorial design. Supplements with 20% or 50% of protein from milk was compared with 0% milk. 23 The primary outcome was lean body mass. There was no overall effect of milk on accretion of lean body mass, but there were borderline effects on linear growth. It was speculated that a reason for no effect of milk could be that 94% of the children were breastfed at the start of the study.
In a comment on food supplements for treatment of MAM, Briend et al concluded that the high content of milk in RUTF for treatment of SAM makes it an expensive product, and it would be desirable to use lower quantities in RUSF for MAM. 4 However, they also stated that if less dairy is used, there are uncertainties regarding the effects in terms of protein quality on growth and the effects of antinutrients if more plant-based sources of protein are used.
Milk Protein and Growth
Protein quality in foods for prevention or treatment of undernutrition is important in order to optimize growth. In a recent review by Manary et al, the importance of protein quality in treating children with undernutrition was discussed. 24 They analyzed studies of children with SAM treated with different therapeutic foods and compared weight gain with different protein quality scores. All the scores were highly correlated with weight gain, but the digestible indispensable amino acid score (DIAAS) adjusted for the expected higher weight gain in SAM children during catch-up growth had the strongest correlation. They also concluded that dairy protein is likely to be associated with better growth because of the specific amino acid requirement of children with undernutrition.
The protein quality of whey compared to SMP measured by protein score is not very different. When protein quality is measured by PDCAAS, whey is slightly higher than SMP. 25 However, using DIAAS there are no major systematic differences in protein scores between whey products, SMP, and milk protein concentrate. 26,27
Whey protein has an amino acid composition including branched chain amino acids, especially leucine, which is important for muscle protein synthesis. 3,25 It has been shown that intake of whey can increase muscle mass, particularly in connection to resistance training. 28 Whether there is an effect of whey protein on muscle synthesis in wasted children greater than the effect of protein from SMP is not known. Skimmed milk powder might have comparable effects on muscle mass in treating malnutrition, but more research is needed. Encouraging physical activity should be part of treating undernutrition in children for many reasons. It might also facilitate an effect of dairy protein on muscle synthesis.
Whey contains bioactive proteins and peptides with antimicrobial or positive immune modulatory effects, for example, lactoferrin. 3,29 In children with HIV infection, supplementation with whey protein has resulted in improved immune function and decrease in coinfections. 30 The potential immune effects of whey might reduce the risk of infections in wasted children and thereby improve growth, but this is speculative. The quality and quantity of growth promoting milk components may be influenced by processing of the milk. Milder thermal treatment seem to preserve bioactive milk proteins. 31
Lactose and Growth
Lactose is a major constituent of mammal milk with about 47 and 69 g/L in cow’s and human milk, corresponding to approximately 30 and 40 energy%, respectively. 32 Despite being a major component of milk, the importance of lactose on growth and development of offspring is not very well understood. Two observational studies found a positive association between the lactose energy percentage of breastmilk and infant weight gain from 3 to 12 months 33 and up to 6 months, 34 respectively. However, in one of the studies, the association was only found in overweight mothers. 34 Another smaller study did not find any association of breastmilk lactose and infant weight gain at 6 months of age. 35 Studies in piglets have shown a growth-stimulating effect of lactose added to feed. 36 -38 In these studies, lactose was mostly added at the expense of more complex carbohydrate from corn or wheat. Both feed intake and daily weight gain increased, 36 -38 and in some studies, the feed conversion rate also increased. 36,37 The effect was higher in younger piglets 36 and was observed in piglets up to 5 to 9 weeks postnatally. 38 This resembles infants up to 6 months of age. 39 No studies were identified investigating whether lactose can stimulate growth in young children beyond 6 to 12 months. The growth stimulation of lactose in infants may not differ from other carbohydrates. Studies on lactose-free infant formulas with equivalent carbohydrate contents have resulted in similar weight and linear growth as standard formulas with lactose. 40,41
In addition to a potential direct effect on growth, lactose may have a prebiotic effect which could indirectly affect growth. Although most ingested lactose is hydrolyzed and absorbed in the small intestine, some lactose—especially in preterm infants—passes into the colon where it is fermented by specific gut microorganisms and thereby affecting the gut microbiota. 42 The importance of the gut microbiota for healthy growth is still speculative.
Lactose intolerance has often been highlighted as a concern, especially in the treatment of malnourished children or in preterm infants. Severely malnourished children often have a disrupted gut barrier which may lead to loss of lactase-producing epithelial cells. If large amounts of lactose is ingested without being hydrolyzed and absorbed in the small intestine, it continues to the large bowel where it may lead to osmotic diarrhea. Healthy children rarely develop lactose intolerance before the age of 2 to 6 years, 43 -45 and most individuals with lactose intolerance can ingest 1 cup of milk with no or minimal symptoms. 46 A more thorough review of the use of lactose in undernourished children has been described elsewhere. 47
Milk Minerals and Growth
When discussing nutrients and minerals in relation to growth, Golden has suggested to categorize nutrients in type I and type II nutrients. 48 Type I nutrients have late or no growth response, whereas type II nutrients show immediate growth response. Type I minerals are stored in the body, while type II nutrients are not. Type II minerals include potassium, sodium, magnesium, zinc and phosphorous.
In cow’s milk, there is a large quantity of bioavailable minerals, including the type II minerals potassium, phosphorus, magnesium, and zinc, as well as calcium which is a type I mineral. 32 Lactose might enhance mineral absorption, which could indirectly affect growth. However, most human studies have focused on calcium, a type I nutrient 32,49
Phosphorus is one of the important growth minerals. If phosphate content of foods used during early treatment of undernutrition is low, refeeding syndrome, which is characterized by low plasma phosphate, can develop. An observational study in Uganda with SAM children showed that low plasma phosphate early in treatment was strongly correlated with later death. 50
Whey permeate is a potential cheaper source of milk minerals and lactose. 51 Its contents of sodium, potassium, and magnesium are comparable to SMP; phosphorus and calcium are reduced, and the amount of zinc is almost negligible. The amount and bioavailability of minerals in whey permeate are regarded as adequate for foods for children with MAM. 51
Conclusion
The high-quality protein, lactose, and bioavailable minerals available in cow’s milk make it an important ingredient in products for treatment and prevention of undernutrition. Apart from direct growth-stimulating effects of milk components, the use of dairy protein also reduces the need for plant-based protein, which often contains antinutrients with a potential negative effect on growth.
The growth effects of dairy are likely to be impacted by the choice of dairy ingredients. There are only small differences in protein quality between different dairy products (SMP, whey or milk protein concentrates), but the content of lactose and minerals differ between different dairy ingredients (SMP, different milk and whey protein concentrates and whey permeate).
Challenges using dairy ingredients in food for preventing and treating undernutrition include the relatively high price of dairy ingredients. Prices for dairy products fluctuate considerably. From early 2014 to late 2015, prices decreased by more than 50%, and from late 2015, there has been a moderate increase. 52 However, the price of ingredients is only part of cost-effectiveness which also includes production, packaging, transportation, distribution, health care/hospitalization and caregiver time.
Future studies are needed investigating how different milk components affect growth and body composition in undernourished children. The effect of lactose and mineral content and how different milk components affect amino acid metabolism are of special interest. Finally, it is recommended to research to what degree plant protein and free amino acids combined with milk components can provide effective and affordable products.
Footnotes
Acknowledgments
The authors want to thank Saskia de Pee from World Food Programme for input regarding specifications of food aid products.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors have received research grants from US Dairy Research Council, Arla Foods for Health and the Danish Dairy Research Foundation.
