Abstract
Background:
Industrial food fortification is a major strategy to improve dietary micronutrient intakes and prevent deficiencies. Zambia introduced mandatory sugar fortification with vitamin A, at a target of 10 mg/kg, in 1998. Representative surveys conducted since that time do not support marked improvement in vitamin A status.
Objective:
To describe vitamin A concentrations in retail sugar, as well as vendor practices, perceptions of fortified foods, and sugar use practices.
Methods:
We conducted a census of sugar vendors in one Zambian community, capturing information on vendors, available brands and packaging options, and storage conditions. We purchased all brands and package types of sugar available at each vendor. In a 15% subsample, we conducted semi-structured interviews with vendor–consumer pairs. We tested 50% of sugar samples at random for vitamin A using an iCheck portable fluorimeter.
Results:
The distribution of vitamin A in sugar in market samples was highly skewed, with a median of 3.1 mg/kg (25th-75th percentiles: 1.8-5.5) and a range from 0.2 to 29.9 mg/kg. Only 11.3% of samples met the 10 mg/kg statutory requirement. Sugar was primarily repackaged and sold in small quantities, with rapid turnover of stocks. Perceptions of fortification by vendors and consumers were generally positive.
Conclusions:
Vitamin A in fortified sugar fell well below statutory requirements. Our data point to challenges at regional depot and/or poor adherence to fortification standards at the factory level. A renewed commitment to monitoring and enforcement will be required for Zambia to benefit from a food fortification strategy.
Introduction
Vitamin A (VA) deficiency remains a significant public health problem. Roughly 30% of children aged 6 to 59 months in low- and middle-income countries are classified as VA deficient. 1 Vitamin A supplementation of children from 6 to 59 months of age has been broadly adopted as a strategy to prevent the consequences of deficiency, 2 but sustained efforts to improve dietary VA intakes and status have been more limited. Fortification of sugar with VA has been successful in Guatemala. Evaluations found substantial improvements in VA status after only 2 years of implementation, 3 and recent data suggest that VA deficiency is no longer a problem of public health concern. 4 Other countries in Central America can point to similar achievements. 5
Zambia began fortifying sugar with VA in 1998. Representative surveys have shown some success in reducing the prevalence of VA deficiency, from 54% nationally in 2003 and in 2 districts in 2009 (40% adjusted for inflammation) 6,7 to 35% in Northern and Luapula Provinces in 2014 (26% adjusted for inflammation). 8 Cross-sectional assessments have shown poor compliance to the fortification statute. 6,7 A national survey in 2003 found that only 18% of sugar samples met the minimum statutory requirement of 10 mg VA (as retinyl palmitate) per kilogram sugar. 6 This proportion was somewhat higher in a 2009 survey, with 59% of samples meeting requirements in 2 districts. 7 However, estimates of adequate coverage with fortified sugar from both surveys were based on sugar available in households at the time of the survey visits. This was the case for only 49.5% of households in the 2003 study and 14.2% of households in the 2009 study.
We conducted a market-level assessment of sugar fortification in one Zambian community. Our objective was to describe the distribution of VA concentrations in sugar. As the stability of VA varies with exposure to sunlight or oxygen, 9 and over time, 5 we further investigated market-level factors that may influence sugar VA concentrations. Finally, we sought to better understand sugar use practices in this population.
Methods
This study was carried out in Mkushi District, where marginal VA status is a public health concern. 10,11 We conducted a census of sugar vendors in the District’s commercial center, capturing information on neighborhood, vendor type (kiosk, household, grocery, storefront, or wholesale), sun exposure, and availability of various brands and package types (manufacturer’s paper or plastic, repackaged plastic). We purchased all brands and package types available from each vendor. In a 15% subsample of vendors, we obtained written consent and conducted semi-structured interviews with both the shop owner and their next available customer.
We randomly selected 50% of sugar packages for testing, oversampling sugar that had been stored in the sun to achieve a sufficient sample to describe the distribution of VA in that category. For large packages, we mixed sugar in the package and removed 50 g for testing. Small repackaged bags were mixed and tested in their entirety. This ensured even distribution of VA particles such that settling would not affect results. We dissolved 50-g sugar samples in 250 mL water to produce a 1:5 dilution. An iCheck portable fluorimeter (BioAnalyt, Teltow, Germany) was used to assess the VA concentration in samples. If the result exceeded the iCheck’s range of detection (>3000 µg Retinol Equivalents (RE)/L), the solution was further diluted to a 1:10 ratio and retested.
Data were analyzed using Stata/IC 13.1 (StataCorp LP, College Station, Texas). Our a priori hypothesis was that repackaged sugar, sun-exposed sugar, and sugar from household or kiosk-type vendors would have lower VA concentrations. We first described the distribution of VA concentrations. Samples ≥10 mg VA/kg were classified as meeting the statutory requirement. Samples <2.5 mg/kg were classified as having no VA. 6 We created categorical variables for package material, storage condition, neighborhood where sugar was collected, and vendor type. Bivariate associations between these variables and VA concentrations were examined using an analysis of variance test with log-transformed concentrations.
The protocol for this study was exempted by the Institutional Review Board of the Johns Hopkins Bloomberg School of Public Health and approved by the Institutional Review Board of Eres Converge in Lusaka, Zambia.
Results
We identified 145 vendors, from whom we purchased a median of 2 sugar packages each (range: 1-4). Of 291 packages purchased, we tested 150 samples. The distribution of VA was positively skewed. The median concentration of VA in sugar was 3.1 mg/kg (25th-75th percentiles: 1.8-5.5), ranging from 0.2 to 29.9 mg/kg; 11.3% (n = 17) of samples met the statutory requirement; 38.7% (n = 58) had no VA. There were minor differences in VA concentration by neighborhood (P = .02) and package type (P = .07), but no differences by vendor type (Table 1). There were no apparent differences by sun exposure, although only one-fifth of sugar packages were exposed to sunlight in this setting.
Retinyl Palmitate in Sugar Samples Collected in Mkushi District, Central Province, Zambia.a
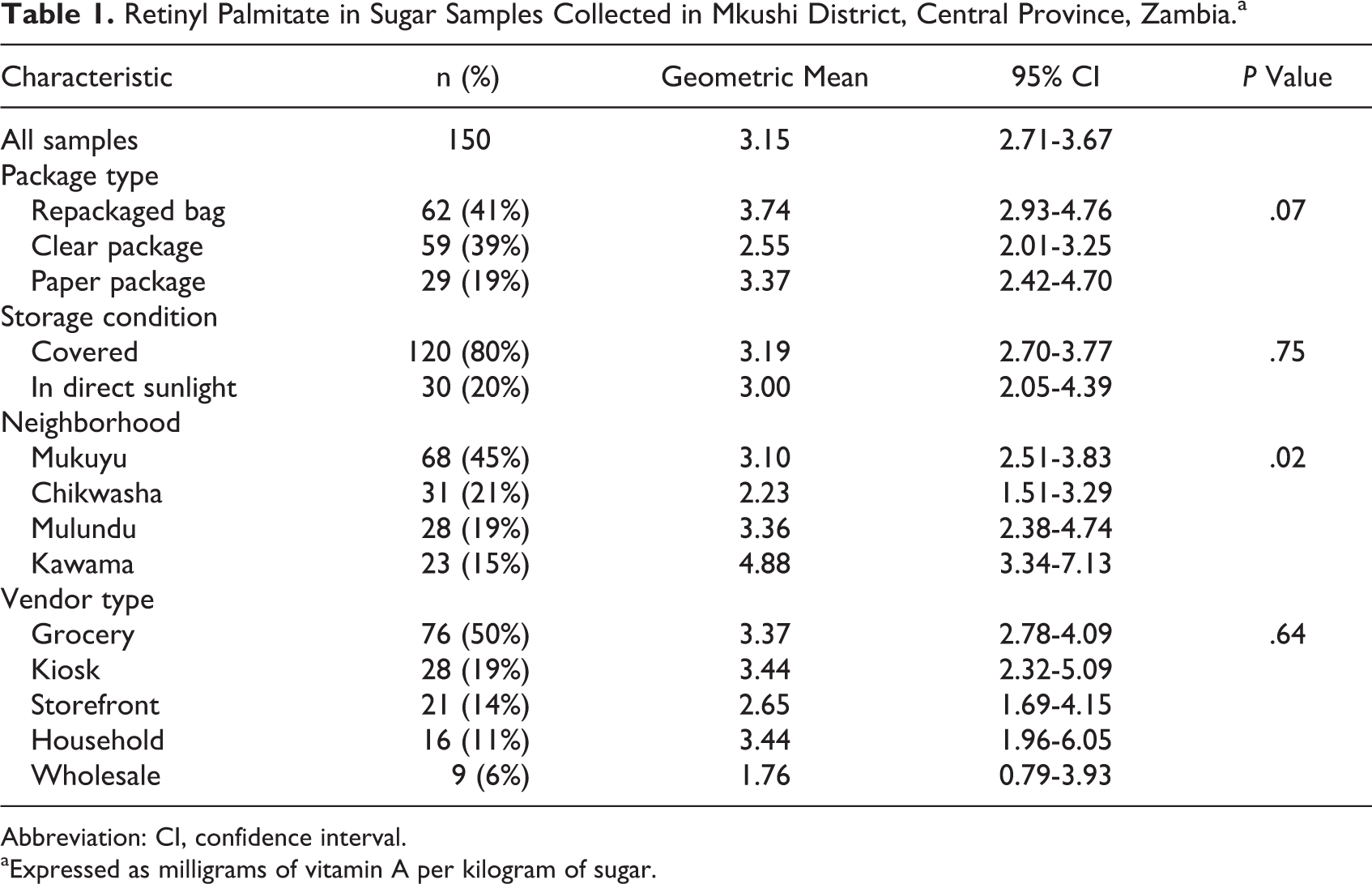
Abbreviation: CI, confidence interval.
aExpressed as milligrams of vitamin A per kilogram of sugar.
Nearly all vendors reported purchasing sugar from wholesale shops within Mkushi District (86%), and a third (32%) of vendors supplemented their inventories with trips to larger cities. The majority (68%) reported purchasing less than 4, 26-kg bales of sugar on a twice-monthly schedule, or more frequently. More than half of vendors (55%) sold their entire inventory of sugar within 2 weeks of purchase. Small (∼50 g) repackaged bags of sugar were sold by 73% of vendors. This package type was the most frequently purchased based on reports from both vendors and consumers.
Perceptions of fortification by vendors and consumers were generally positive (Figure 1). Mothers in Mkushi reportedly seek out products with added vitamins: “We do sell a lot of those [fortified] foods because mothers want them for their babies.” Consumer interviews highlighted the use of added sugar for tea and traditional beverages (100%), as well as grain-based foods such as porridge (95%), rice (95%), and samp (79%). Consumption of sugar water, locally referred to as zigolo, was reported by 82% of participants, with 63% consuming zigolo ≥2 to 4 times per week. Zigolo is believed to be a source of energy and is taken before meals if meal preparation is lengthy: “maybe you haven’t eaten in a long time, you take zigolo and at least you feel energized.”
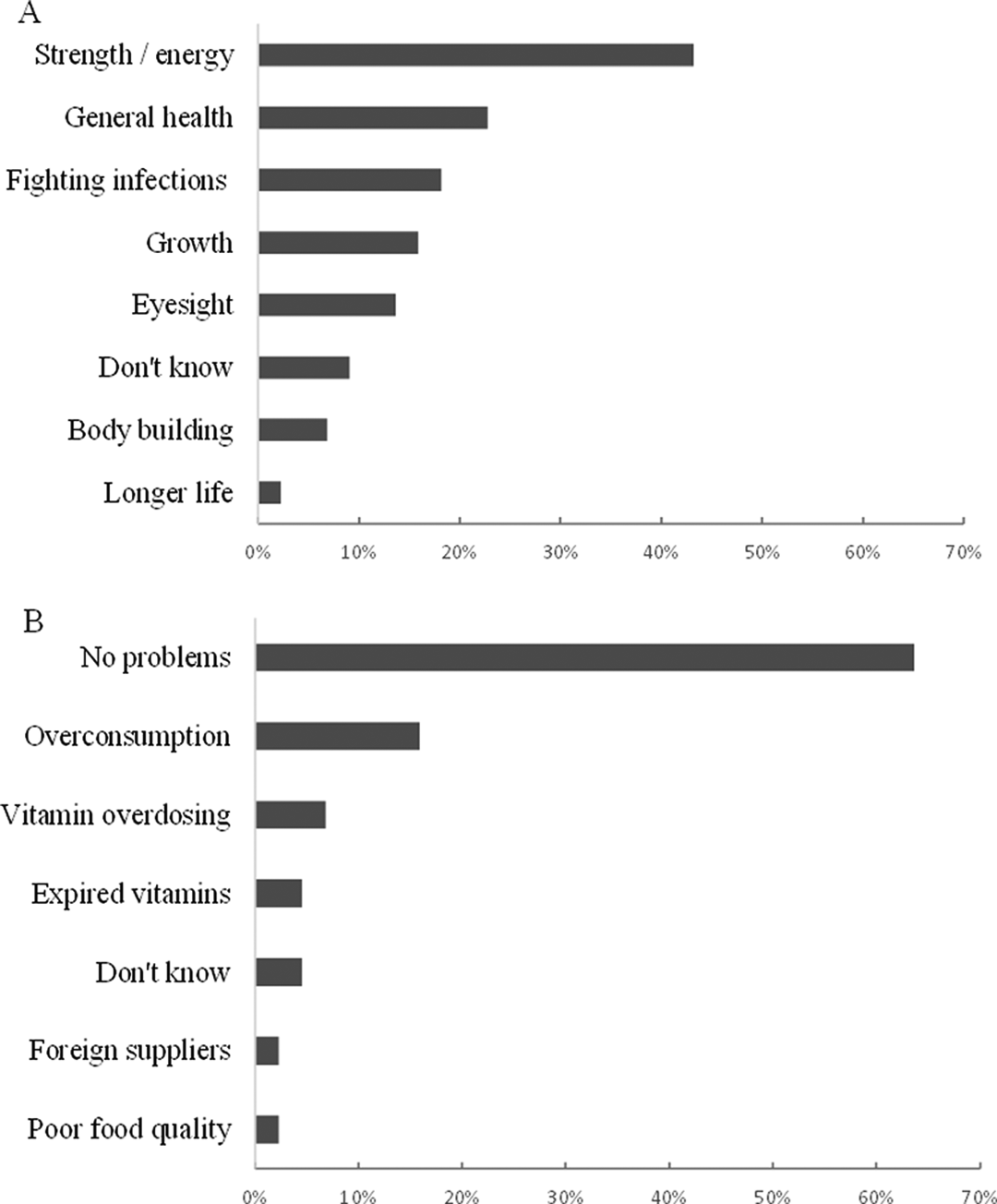
Perceived benefits (A) and potential problems (B) with fortified foods identified through semi-structured interviews with vendors and consumers (n = 44) in Mkushi District, Central Province, Zambia.
Discussion
We found that the median concentration of VA in retail sugar in Mkushi District was well below the statutory requirement, with only ∼10% providing the mandated 10 mg/kg. Contrary to our original hypothesis, neither sun exposure nor vendor type was associated with VA concentration. Small repackaged bags of sugar had higher concentrations than sugar in manufacturer’s packaging. These findings, combined with vendor and consumer reports, indicate high turnover of repackaged sugar, such that VA degradation over time or with exposure to sunlight is less of a concern. This contrasts with 1-kg bags of sugar in manufacturer’s packaging, which may sit on store shelves for a longer period of time. Slow turnover may similarly explain low VA concentrations in sugar from Chikwasha, where there is a high density of larger shops and likely greater competition for customers. Although fortified sugar is not providing expected amounts of VA in this setting, vendor and consumer perceptions and reported sugar use practices suggest that this is still a viable strategy for improving status.
Sugar fortification has had a limited impact on VA status in Zambia. This could be attributed to: (1) inadequate monitoring at the factory level, (2) degradation of VA prior to sale, or (3) low intake of adequately fortified sugar by high-risk groups. Our data point to the first of these issues. Although degradation may be a challenge, it seems that sugar is arriving in this commercial center with a low VA concentration. This may be due to improper storage practices at regional depots or, more likely, poor adherence at the factory level. A renewed focus on program monitoring is clearly warranted. As an initial step, we recommend an audit at the point of production to assess the quality of fortificant and compliance with technical standards. Our study was limited to one centrally located district, which may not be representative of other parts of Zambia. Thus, we recommend a national-level assessment of fortification efforts to test VA concentrations in sugar at the factory level, at distribution depots, and within key markets.
The present study is also limited in that it cannot directly address the intake of fortified sugar by high-risk groups. Although interviews provided some information on sugar use practices, our methods and small sample size preclude any estimate of usual sugar intakes. We plan to link these market-level data on VA in fortified sugar with a parallel assessment of usual dietary intakes among lactating mothers in this community to inform estimates of dietary adequacy. Although we specifically call for a nationwide assessment of fortified sugar, an investment would ideally be made in a national survey to capture coverage of adequately fortified sugar, as well as dietary intakes and VA status. This information will be crucial as Zambia works to control and eventually eliminate VA deficiency as a public health problem.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Sight and Life, with support from the Christian Blind Mission, and HarvestPlus. Additional support and materials were provided by BioAnalyt, Inc.
