Abstract
Background:
There is no consensus over best approaches to reliably prevent malnutrition in rural communities in low-income countries.
Objective:
We compared the effectiveness of 2 lipid-based ready-to-use supplementary foods (RUSFs) differing in dairy protein content to improve the nutritional status of mothers and at-risk infants and young children in rural Guinea-Bissau.
Methods:
A 3-month cluster-randomized controlled pilot trial of 2 RUSFs was conducted with 692 mothers and 580 mildly or moderately malnourished infants (6-23 months) and children (24-59 months) from 13 villages. The RUSFs contained either 478 (mothers, children) or 239 kcal/d (infants) with 15% or 33% of protein from dairy and were distributed at community health centers 5 d/wk. Controls were wait-listed to receive RUSF. Primary outcomes were mid-upper arm circumference (MUAC) in mothers, and weight-for-age and height-for-age z-scores (WAZ and HAZ) in infants and children.
Results:
There was a significant effect of the RUSF-33% on MUAC in mothers (P = .03). The WAZ and HAZ increased substantially, by ≈1 z-score, in infants and children (P < .01) independent of group randomization. In children, but not infants, baseline WAZ and change in maternal MUAC were associated with change in WAZ (β = .07, P = .02).
Conclusion:
Ready-to-use supplementary foods with higher dairy protein content had a significant benefit in village mothers, supporting a comparable recent finding in preschool children. In addition, supplementation of children <2 years resulted in improved growth independent of family nutritional status, whereas success in older children was associated with change in maternal nutrition, suggesting the need for community-level education about preventing malnutrition in older, as well as younger, children.
Introduction
Childhood undernutrition and micronutrient deficiencies remain prevalent in low-income countries, especially those in Sub-Saharan Africa, 1 and different approaches to prevention have included community-based supplementary feeding programs for mothers, infants, and children at the village level, school lunch programs for older children, and twice-yearly vitamin A supplementation. In addition, some countries are performing surveillance for moderate acute malnutrition and severe acute malnutrition (SAM). However, to date, most community prevention interventions have had at best only a small effect, with population weight-for-age and height-for-age z-scores (WAZ and HAZ) typically improving by only 0 to 0.2 z-scores. 2 –5 Further work is needed to identify more effective approaches.
Several factors may influence the effectiveness of community supplementary nutrition programs. One of these is the nutritional composition of the supplement, in particular the relative proportion of protein from vegetable and animal sources. Higher relative dairy protein (50% of total protein intake) is now recommended for formulations of ready-to-use therapeutic foods treating SAM, 6 recognizing potential benefits of animal protein for prevention of relapse as well as regeneration of lean tissue. 7 However, there is no general agreement over whether dairy protein can improve the effectiveness of supplementary feeding programs for prevention of malnutrition. To our knowledge, the only study directly comparing supplementary feeding with different levels of animal protein has been by our group recently in preschools in Guinea-Bissau, and that study identified a potential benefit of higher dairy supplements for prevention of wasting as measured by mid-upper arm circumference (MUAC). 8 Since the benefits of higher animal protein supplements will depend on the amino composition of other food in the recipients’ diet, studies in community-based interventions are now needed.
Household food redistribution is another factor that is thought to be an important determinant of whether supplementary feeding initiatives are effective, because if supplemented individuals receive less than their usual share of family food the result is little or no net improvement in growth or weight gain 9,10 even if the quality of the food supplement increases some micronutrient benchmarks. 10 Multiple reports from different countries indicate that supplementary foods intended for designated infants and children are typically shared with other family members, 11,12 although RUSF may be less shared than general-use products such as corn-soy blend. 13 In addition, there are some suggestions that children in wealthier families benefit more from supplementation, presumably because family sharing is less necessary. 14 However, there is relatively little research exploring this topic, because measurements of household food consumptions are difficult and subject to substantial bias. 15 One factor that has received almost no attention is the age of the supplement beneficiary, and a better understanding of family food dynamics during supplemental feeding programs is needed.
We conducted a pilot randomized controlled trial to examine the effects of a 3-month supplementary feeding program delivered by community health workers on the nutritional status of mothers, and infants and children aged 6 to 59 months in subsistence farming villages in Guinea-Bissau. Two different lipid-based ready-to-use supplementary foods (RUSFs) differing only in the percentage of protein provided by dairy sources were tested against a wait-listed control for effects on growth and biochemical parameters in at-risk infants and children, their mothers, and other pregnant and lactating women living in the same villages. To our knowledge, this is also the first study to assess the effects of simultaneous use of RUSF in at-risk infants/children and their mothers, thus providing a unique opportunity to evaluate the effects of supplementation programs among multiple family members.
Methods
Study Location and Participants
As described previously, 8 Guinea-Bissau is a small country in West Africa with a population of 1.7 million that is currently ranked as the 11th poorest nation in the world. 16 The economy of Guinea-Bissau depends mainly on farming and fishing, and foods grown for family consumption include rice (the staple food), millet, groundnuts, sweet potatoes, mangoes, oranges, and (less frequently) domestic animals (chickens, pigs, goats, and cows), and fish. Families typically combine subsistence farming with growing cashews, which are sold for basic supplies including additional rice.
The study was a cluster-randomized controlled trial (clusters being defined as villages) in 13 villages in the Oio and Cacheu districts of Guinea-Bissau, 32 to 108 km from the capital city of Bissau. In the selection process, we identified 25 villages that were potentially suitable based on considerations of literacy of the community health workers, sufficient infants, children and pregnant women in the village to make research implementation feasible, and anticipated poor nutrition status of villagers. The first 13 villages to be invited to participate accepted the invitation and were enrolled. Participants were 681 village infants and toddlers aged 6 to 23 months, and young children (24-59 months), who had mild to moderate acute malnutrition at baseline based on predefined criteria created for this study (WAZ <1.0, or HAZ <2.0, or weight-for-height z-score [WHZ] <2.0 at screening), along with 807 women who were either the mothers of the enrolled infants and children or other women in the same villages who reported being pregnant or lactating. Most of the woman in the pregnancy group proved to be late in the third trimester, since they delivered during the study period. The schema for participant enrollment and dropout is summarized in Figure 1.
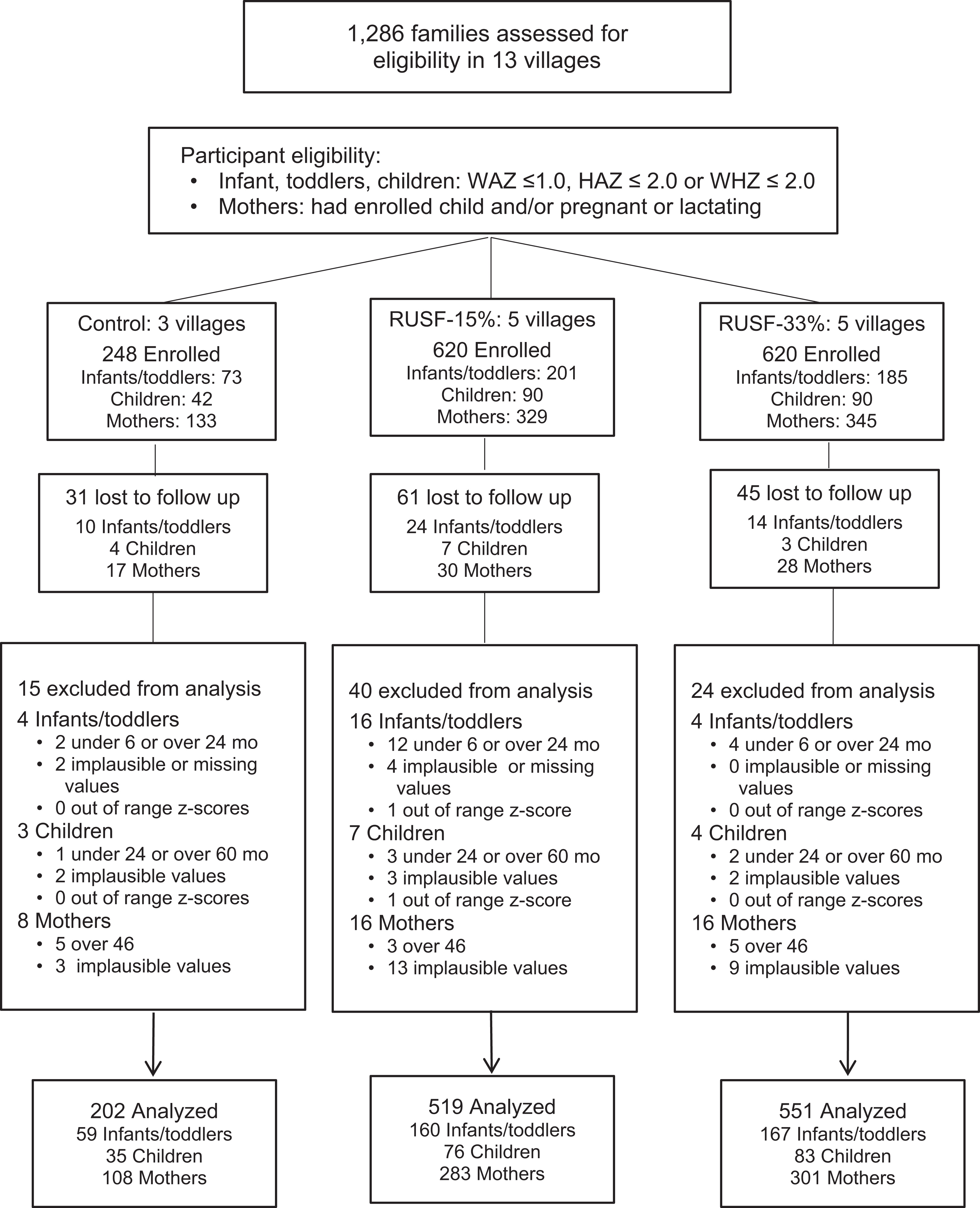
Consort diagram of randomized controlled trial of 2 ready-to-use supplementary foods (RUSFs) and control group in Guinea-Bissau villages.
Institutional review board (IRB) permission to conduct the study was provided by the ethical committee of the government of Guinea-Bissau. Following an explanation of the study protocol, all mothers and parents or legal guardians of children and infants agreed to participation and provided their informed consent with a signature or thumbprint in the presence of a member of the research team and a community health worker.
Study Protocol
The study was a cluster-randomized controlled pilot trial comparing 2 RUSFs to a wait-listed control in mothers, infants, toddlers, and children in 13 villages. The RUSFs were provided 5 d/wk for 3 months, while the wait-listed controls received no RUSF during the study but got an allotment of RUSF after final study measurements. Fieldwork took place between January 2013 and June 2013. The predefined primary outcomes were changes in infant and child WAZ and HAZ analyzed in an intention to treat (ITT) model, along with change in maternal body mass index (BMI, kg/m2). Because we were unable to determine pregnancy status in the mothers (they did not know their estimated delivery date, and only mothers who appeared to be in the last trimester acknowledged being pregnant), we retrospectively redefined the primary variable in mothers as MUAC, which was less influenced by unknown pregnancy status than weight and is known to be less variable than weight in populations where diarrhea is common. 17
Baseline assessments included anthropometry (weight, height, and MUAC) and biochemical parameters (hemoglobin [Hb] and retinol binding protein [RBP]). After baseline, villages were assigned a number and their treatment was based on a randomization schema generated in SAS with 3 of the 13 villages designated as controls and an equal number of remaining villages allocated to each supplement. Participants were blind to the different RUSFs, but it was not possible to blind to intervention versus control assignments. The villagers randomized to receive an RUSF were provided with their supplement at the community health center 5 mornings per week. Community health center workers distributed the supplement after training by the research staff from the International Partnership for Human Development (who were trained by the US team), and also recorded attendance and reasons for absences. When a child’s caregiver was routinely someone other than the mother, that person was allowed to collect supplement for her charge, in which case the mother came separately to receive her own RUSF. If mothers reported that they (with or without their children) would be out of town on planned distribution days, they were able to collect a supplement allowance for up to a week in advance of the trip. Outcome assessments (the same as baseline) were repeated at the end of the study period.
The Ready-to-Use-Supplementary Foods
The 2 RUSFs were the same as recently used by our team in a study in preschools. 8 They were shelf-stable lipid-based nutritional pastes manufactured by Challenge Dairy Products, Inc (Dublin, California) and had a similar composition to other products made by this manufacturer for United Nations International Children’s Emergency Fund. Two sachet sizes were used here: 92 g (478 kcal) for the children aged 24 to 39 months and the mothers and half that size for infants aged 6 to 23 months. The composition of the RUSF is given in Table 1. The 2 products were identical in terms of total energy, energy density (5.2 kcal/g), and total protein and micronutrients, and differed only in percentage of total protein provided by dairy ingredients. Specifically, both products contained protein in a combination of soy protein isolate and dairy protein (no peanuts); one had 15% of total protein from dairy ingredients (RUSF-15%) while the other had 33% (RUSF-33%), resulting in a calculated Protein Digestibility Corrected Amino Acid Score values of ≈1.0 for both products. 18,19 The specific levels of dairy protein were chosen with consideration of the cost of ingredients, product acceptability, and the limitation of needing other nutrients to be equivalent. The RUSF-15% had similar dairy protein content to commercially available peanut-based RUSF and met World Food Programme RUSF specifications at the time of the study for treatment of moderate acute malnutrition. It should be noted that a subsequent World Health Organization (WHO) Technical Note for the formulation of RUSF recommended higher levels of some micronutrients including iron. 20 Community health workers received a modest allowance of locally purchased rice in compensation for distributing supplements and making study records, and quality control for distribution and recordkeeping was assessed in regular visits by the study team.
Nutritional Composition of the 2 RUSFs Compared to WHO Guidelines.a,b
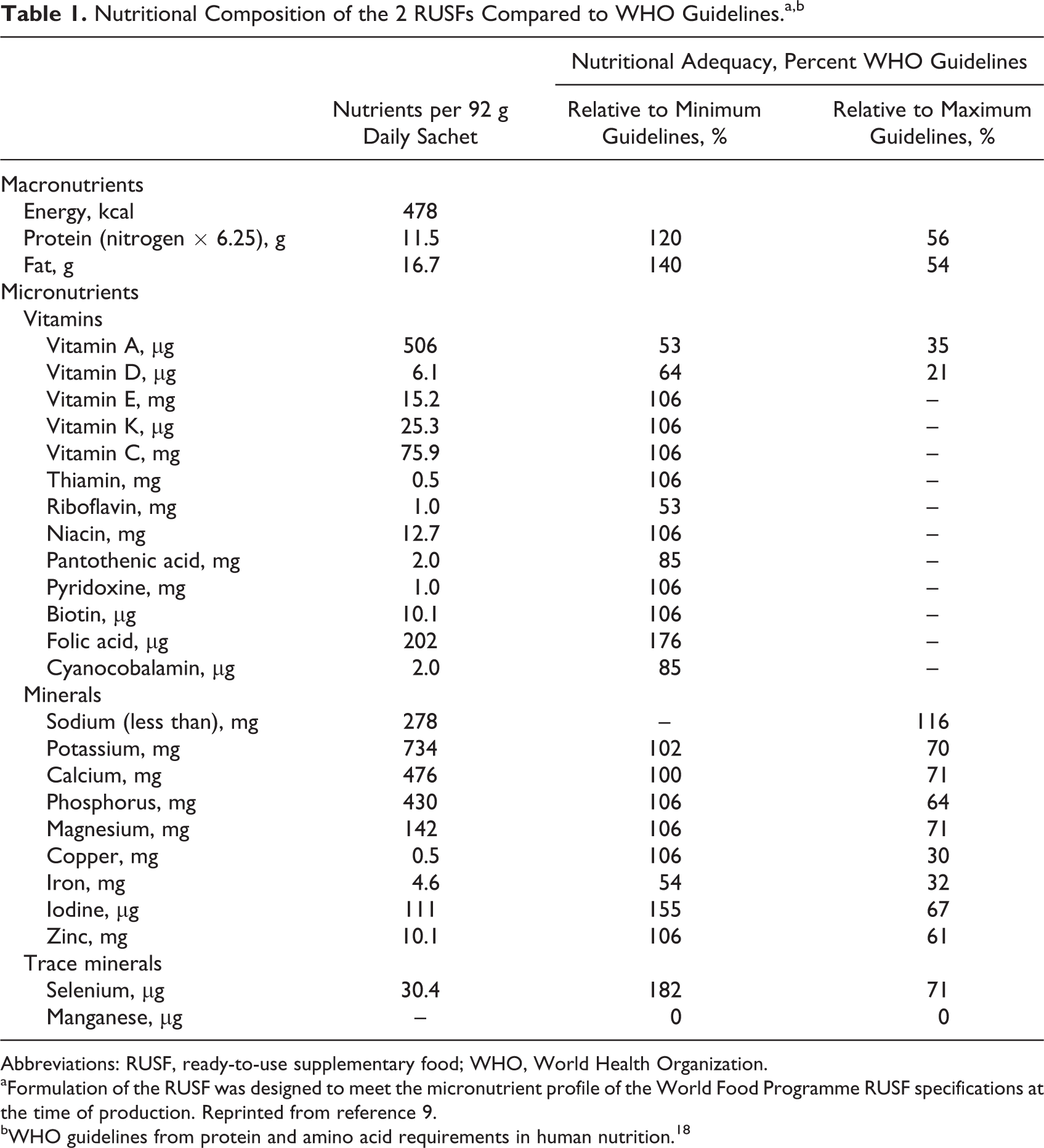
Abbreviations: RUSF, ready-to-use supplementary food; WHO, World Health Organization.
aFormulation of the RUSF was designed to meet the micronutrient profile of the World Food Programme RUSF specifications at the time of production. Reprinted from reference 9.
bWHO guidelines from protein and amino acid requirements in human nutrition. 18
Anthropometric and Biochemical Measurements
Nonfasting weight was measured at baseline and 3 months using a digital calibrated scale weighing to ±0.1 kg (floor scale model 813 for children and mothers, and recumbent model 453 for infants; Seca, Chino, California). Height was assessed using an upright stadiometer measuring to 0.1 cm for children and mothers and a recumbent model for infants (models 213 and 453; Seca). Mid-upper arm circumference was taken at the midpoint between the acromion process of scapula and olecranon process with a paper tape appropriately sized for the participant using standardized WHO methods as an index of waisting. 21,22 In addition, a finger stick blood sample was taken at both time points in accordance with WHO guidelines from those participants who were willing. Hemoglobin was measured with a point of care Hb meter (model 121721; Hemocue, Brea, California), and 3 blood spots were taken onto a protein saver card for analysis of RBP (a proxy for vitamin A) by Craft Technologies, Inc (Wilson, North Carolina). Following blood collection, the spots were protected from light and shipped by air to the United States and stored at −20°C prior to RBP being measured in all good-quality spots by sandwich enzyme-linked immunoabsorbent assay in a 96-well format employing polyclonal antibodies. 23 Outcome assessments were performed by a group of trained medical/nursing staff seconded from the Guinea-Bissau Ministry of Health who had no role in study design or supplement distribution.
Absenteeism and Morbidity
For intervention families, community health workers asked whether each participant was well or sick during visits to the community health center to receive the RUSF. If an illness was reported, it was categorized by the caregiver as fever, diarrhea, respiratory, or other. Nonillness reasons for absence were also recorded. For control families, mothers were asked to report particular illnesses at their local health center, and in addition community health workers visited each family weekly to question the mothers. Forms were checked by the research team when they visited the villages for quality control assessments.
Product Acceptability
An acceptability evaluation was administered during the first and last study weeks by community health workers, consisting of 3 hedonic scales (ratings 1-5) assessing participant’s perception of the supplement’s taste, texture, and how much they liked the product overall.
Data Analyses
Z-scores for WAZ, HAZ, and WHZ were calculated with macros based on WHO child growth standards. 20 All analyses were stratified by infants, children, and mothers (including with and without self-diagnosed pregnancy for descriptive statistics). Infants and children with baseline ages outside the defined age ranges, mothers older than 45, and those identified as nonmother caretakers were excluded. Participants with missing or implausible values were also excluded. Details of these exclusion criteria are outlined in Figure 1. Mothers with a change in MUAC greater or less than 3 times the standard deviation (SD) were excluded only from analyses with that variable. Primary analyses were ITT based on the initial study group randomization. A per protocol (PP) analysis restricting the population to those in the treatment groups with a minimum of 50 days of RUSF consumption was also performed; however, only 5% of mothers, 4% of infants, and less than 1% of children had fewer than 50 RUSF consumption days and removal of these participants from the analysis did not impact any findings and data are not presented.
Differences in baseline variables between the 2 supplement groups and control group were compared using χ2) for categorical variables, 1-way analysis of variance for continuous variables and the Kruskal-Wallis test for nonnormal continuous variables. A logistic regression model was used to assess differences in supplement acceptability rating between RUSF-15% and RUSF-33%, with a rating of 4 or 5 considered acceptable. Analysis of covariance models adjusting for baseline acceptably were used to assess change in acceptability from baseline.
Analysis of covariance models controlling for age, sex, region, and baseline values with the cluster-randomized effect of village were used to compare mean changes in anthropometric variables between the intervention groups and control, with Tukey honest significant difference used to adjust for multiple comparisons. Age, sex, and region were included as covariates in these models to adjust for any imbalances due to randomization at the village level and also to reduce any underlying variability attributable to these variables. Infants and children were analyzed separately. Change in MUAC, Hb, and RBP for mothers were analyzed together, regardless of pregnancy status. A regression model of change in MUAC on change in BMI for all mothers not reporting pregnancy was used to assess the relationship between MUAC and BMI. Poisson regression models adjusting for age, sex, region, and the cluster-randomized effect of village were used to assess differences in rates of reported sicknesses and absences. Pearson correlation coefficients between changes in MUAC of mothers and change in infant WAZ and change in child WAZ were calculated to explore the effect of family unit on change in anthropometric variables. Multiple regression models regressing change in mother MUAC, treatment (control vs RUSF-15% vs RUSF-33%), and baseline values on change in infant WAZ and child WAZ were also computed. Analyses were performed using SAS version 9.3 (SAS Institute, Inc, North Carolina), and statistical significance for all variables was set at a 2-sided P value of .05.
Results
Baseline characteristics for each age group are shown in Table 2. There were no significant differences between groups for most of the anthropometry variables, and 57% of infants and 66% of children had at least 1 z-score below −2.0 (data not shown). However, baseline Hb was higher and RBP was lower in controls compared to RUSF groups. In addition, the MUAC of pregnant mothers was higher in controls. In a regression of change in MUAC on change in BMI for nonpregnant mothers in both control and treatment groups, a 1 cm difference in MUAC was associated with an 0.68 significant difference in BMI (P < .0001, data not shown), a relationship that will be discussed further below.
Baseline Characteristics of Infants and Toddlers, Children, and Mothers in Rural Guinea-Bissau Villages by Randomly Assigned Groups, Mean (SD).
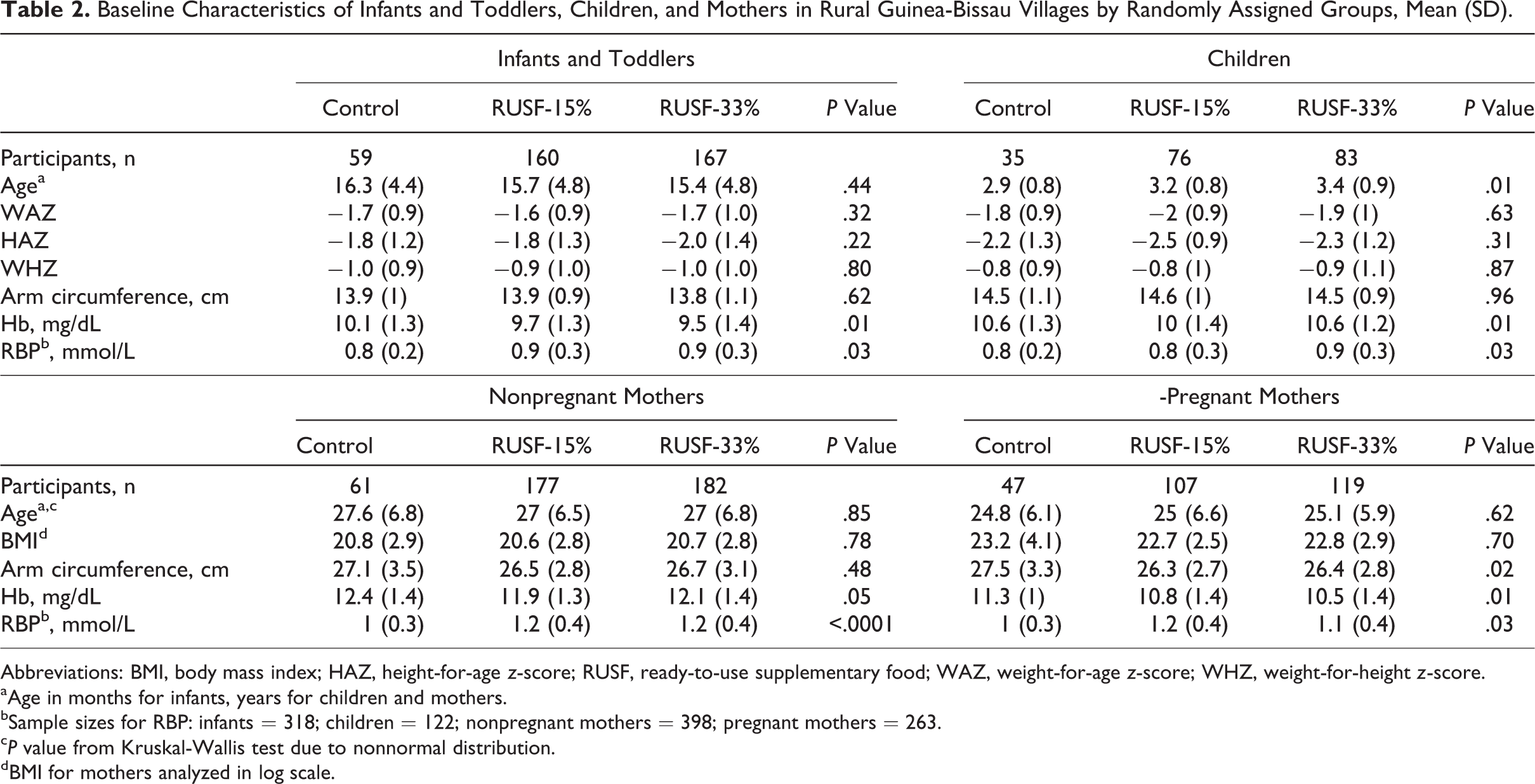
Abbreviations: BMI, body mass index; HAZ, height-for-age z-score; RUSF, ready-to-use supplementary food; WAZ, weight-for-age z-score; WHZ, weight-for-height z-score.
aAge in months for infants, years for children and mothers.
bSample sizes for RBP: infants = 318; children = 122; nonpregnant mothers = 398; pregnant mothers = 263.
c P value from Kruskal-Wallis test due to nonnormal distribution.
dBMI for mothers analyzed in log scale.
Table 3 shows average number of days the supplements were consumed, as well as the acceptability ratings of the 2 products at the end of the study. The RUSF-33% was consumed for slightly fewer days than for the RUSF-15%, and this difference was significant for children and nonpregnant mothers (P < .001 and P = .01, respectively). However, days that supplement was consumed relative to distribution days was high for both groups, ranging from 92% to 97% of total days. The overall taste acceptability was scored on a scale of 1 to 5, with 5 being the most positive rating, and the average ranged from 4.8 to 4.9 for the RUSF-15% group, and from 4.6 to 4.8 for the RUSF-33% group. In addition, 87% to 98% of participants ranked the taste acceptability as a 4 or 5, indicating general acceptability of both products. There were no significant difference in taste acceptability of the RUSF-15% and the RUSF-33% at the end of the study, and there were no differences in changes in taste acceptability over time between the 2 products (data not shown).
RUSF Consumption and Taste Ratings for Infants, Children, and Mothers in Rural Guinea-Bissau Villages Randomly Assigned to Receive RUSF-15% or RUSF-33% for 3 Months.a
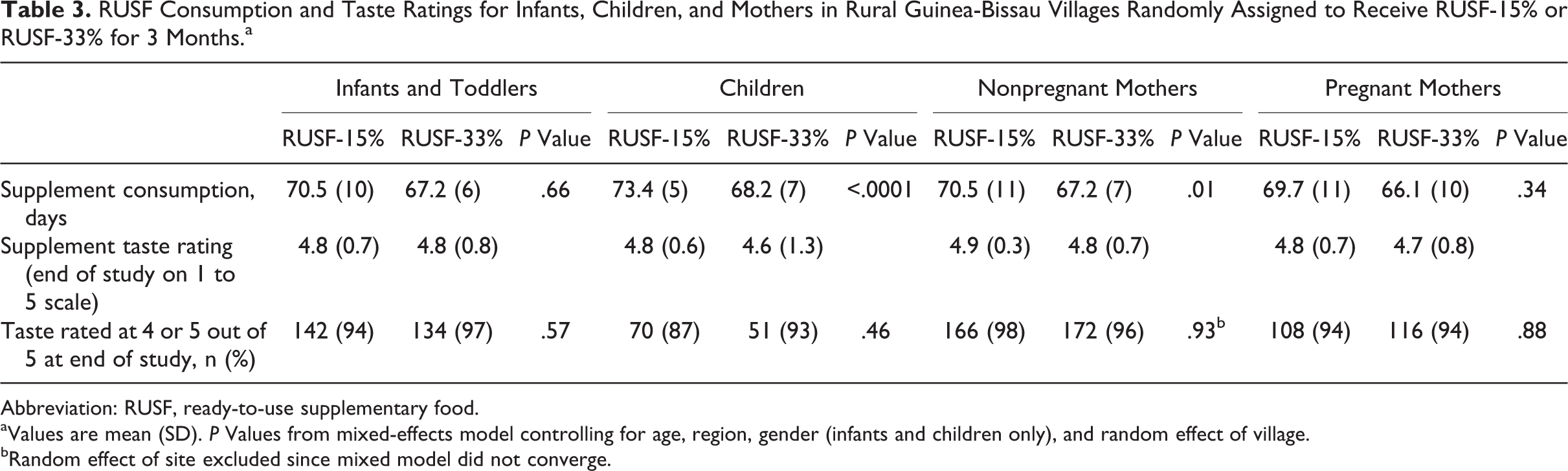
Abbreviation: RUSF, ready-to-use supplementary food.
aValues are mean (SD). P Values from mixed-effects model controlling for age, region, gender (infants and children only), and random effect of village.
bRandom effect of site excluded since mixed model did not converge.
Mean (SD) values for changes from baseline to 3 months for all outcome variables are shown for each participant group in Table 4 and illustrated in Figure 2. Concerning infants and children, WAZ, HAZ, and MUAC increased substantially and significantly in both supplement groups and the control group. There were no significant differences among the 3 groups for any change over time in either infants or children, and the same lack of significance was seen in a PP analysis of those individuals who received the supplement for at least 50 days (data not shown). Mean Hb levels increased significantly from baseline to 3 months in infants and children in both RUSF groups (which had similar mean changes), and there were no significant changes in control infants and children. However, the changes over time in the RUSF groups did not differ significantly from the changes in the controls except for the Hb change in infants receiving the RUSF-15% (P = .01). Concerning mothers, it proved to be impossible to know which mothers were pregnant, other than the ones who admitted pregnancy and were clearly close to delivery, which led us to combine all mothers into a single group for most analyses. Furthermore, because of the unknown number of pregnant women at different stages of pregnancy in each group, we considered BMI data unreliable and redefined the primary outcome for mothers as MUAC. Mothers randomized to receive the RUSF-33% had an increase in MUAC compared to controls (P = .03). The change in MUAC in the RUSF-15% group was not significantly different from controls or from the RUSF-33% group (P = .10 and P = .81, respectively). There were no significant changes in RBP in any group of mothers and no differences in changes over time between groups, and similarly no significant changes in Hb or differences in changes between groups, except that Hb levels fell significantly over time in control mothers (P < .05). There were also no significant differences in reported rates of any specific disease between control and treatment groups for any patient type (data not shown).
Effect of 2 RUSFs Consumed Over 3 Months by Infants, Children, and Mothers in Rural Guinea-Bissau Villages on Anthropometric and Biochemical Changes, Mean (SD).
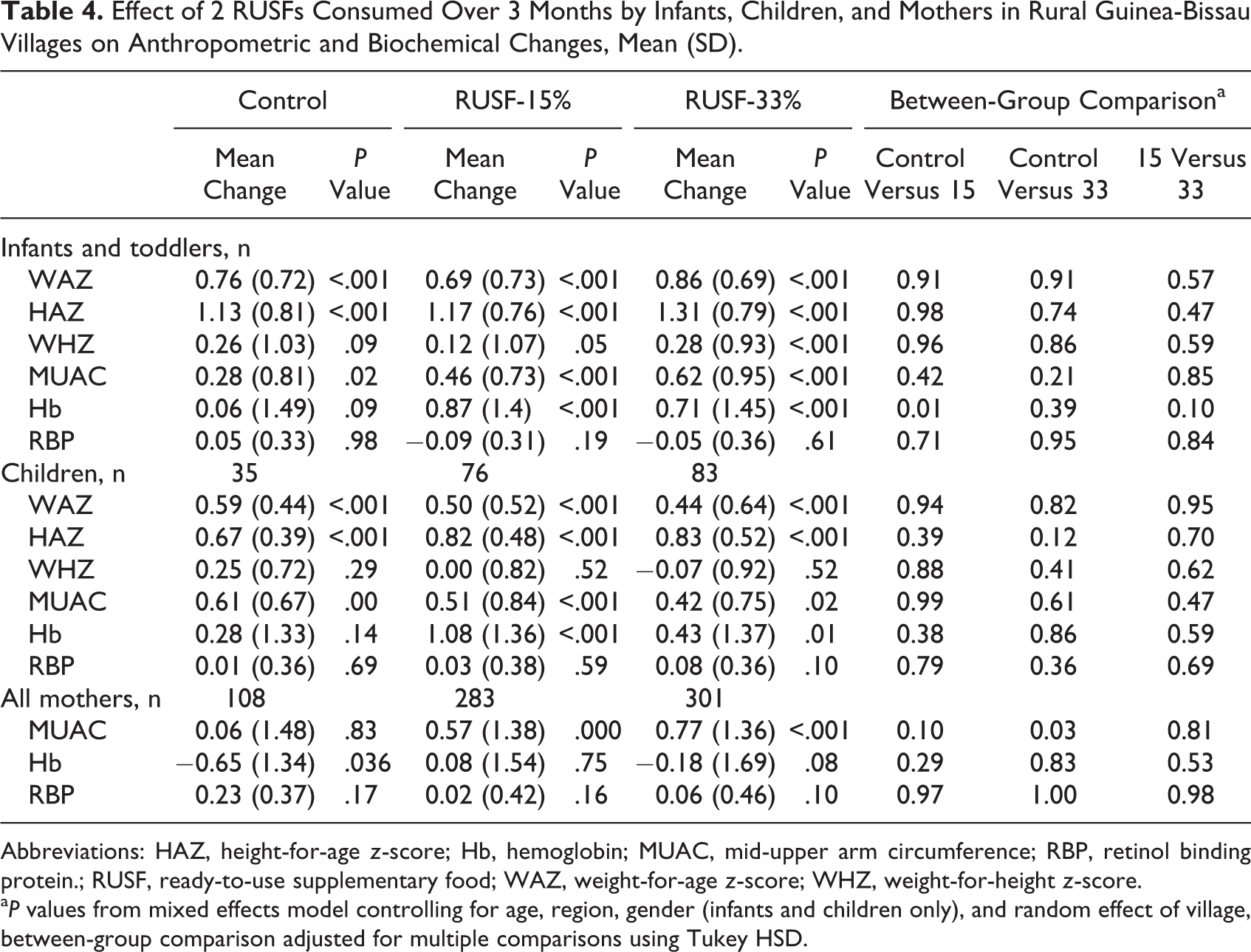
Abbreviations: HAZ, height-for-age z-score; Hb, hemoglobin; MUAC, mid-upper arm circumference; RBP, retinol binding protein.; RUSF, ready-to-use supplementary food; WAZ, weight-for-age z-score; WHZ, weight-for-height z-score.
a P values from mixed effects model controlling for age, region, gender (infants and children only), and random effect of village, between-group comparison adjusted for multiple comparisons using Tukey HSD.
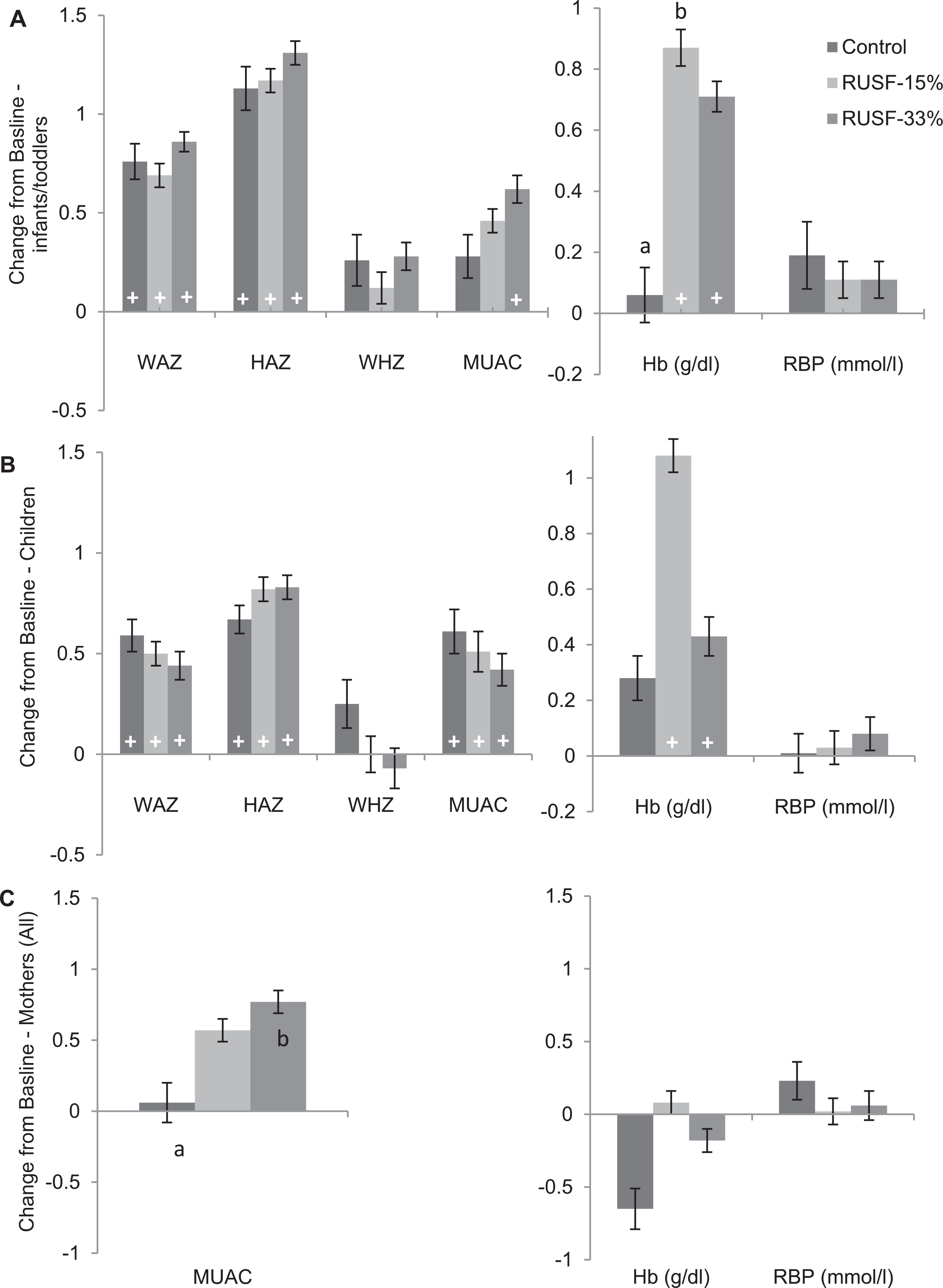
Mean and standard error for change from baseline for anthropomorphic variables for (A) infants and toddlers, (B) children, and (C) mothers. a,b Significant difference between groups at P < .05; + within-group significant change from baseline at P < .05.
Intrafamily associations between anthropometric changes over time were explored in the 340 mother–infant and 160 mother–child dyads within the overall data set. In multiple regression models, change in mother MUAC and baseline child WAZ, but not treatment group or baseline mother MUAC, predicted change in WAZ in children. In contrast, baseline child WAZ predicted change in WAZ in children but there was no association between change in mother MUAC and change in child WAZ. Scatterplots showing the relationships between change in WAZ and both baseline WAZ and change in mother MUAC for infants and children are shown in Figure 3.
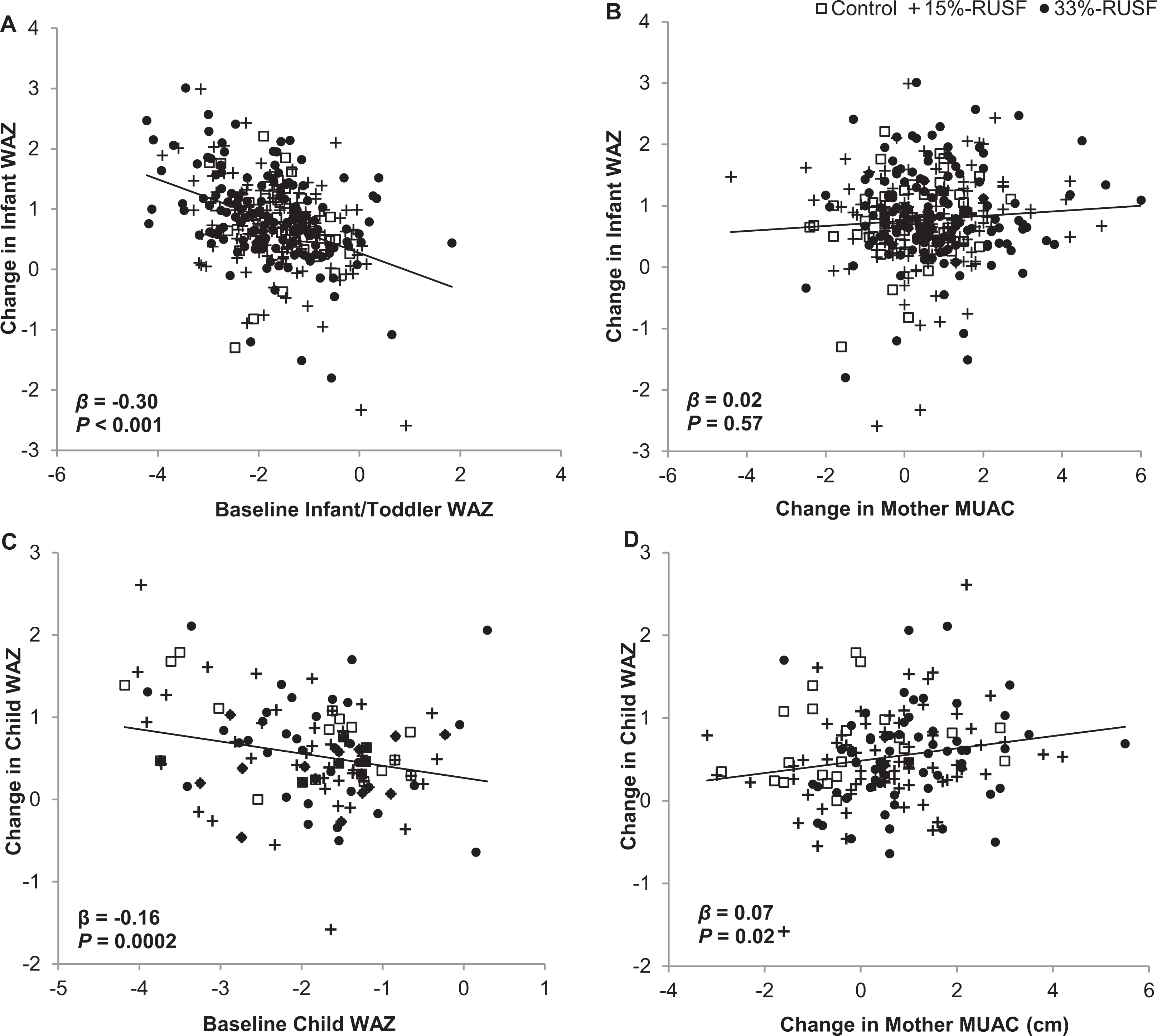
Correlations between family pairs. A, Infant baseline WAZ versus change in WAZ for infants. B, Mother change in MAUC versus change in WAZ for infants. C, Child baseline WAZ versus change in WAZ for children. D, Mother change in MUAC versus change in WAZ for children. β-Coefficient and P value from multiple linear regression model adjusted for region, gender, and random effect of village. For children/mother pairs: control n = 26, RUSF-15% n = 69, and RUSF-33% n = 68; for infant/mother pairs: control n = 47, RUSF-15% n = 146, and RUSF-33% n = 152. MUAC indicates mid-upper arm circumference; RUSF, ready-to-use supplementary food; WAZ, weight-for-age z-score.
Discussion
Identifying reliable ways to prevent the progression from mild to severe malnutrition in at-risk infants and young children in low-income nations is essential for reducing mortality and improving long-term health and cognition. 6 To our knowledge, this is the first study examining the importance of animal protein in a controlled study of supplementation programs to prevent malnutrition at the village level, and we demonstrate a significant benefit of using higher dairy supplements to improving the nutritional status of mothers. We also document substantial improvements in infant and child z-scores (≈1 z-scores in both WAZ and HAZ) independent of group randomization, perhaps attributable to both the supplements and (in the case of control families) the use of traditional malnutrition remedies as recommended by community health workers. We also show that infant growth is independent of change in maternal nutrition, but that weight change in children parallels anthropometric changes in their mothers, indicating that there is an effect of age on the extent to which childhood malnutrition can be prevented in village supplement programs without consideration of family nutrition dynamics.
To our knowledge, this study provides the first village-level test of RUSFs that are identical except for higher and lower percentages of protein provided by dairy sources to prevent malnutrition in mothers, infants, and children. We used change in MUAC as a surrogate for changes in weight and wasting in mothers, because for cultural reasons it proved impossible to ascertain which mothers were pregnant, and because MUAC is a proxy for nutritional status that is less sensitive to pregnancy than weight. 24 Only randomization to the RUSF with relatively higher dairy protein caused a significant increase in MUAC in the mothers, and based on a correlation between MUAC and BMI in baseline data the change was equivalent to 0.5-unit increase in BMI (about 1.5 kg weight). There was also a positive but nonsignificant change in MUAC with the lower dairy protein supplement indicating that a smaller effect might have been observed in a study with more villages. Combined, our previous report on preschool pupils and these new data in village mothers, provides strong evidence that a relatively small increase in the dairy protein content of food supplements has measurable benefits in prevention of wasting in low-income Sub-Saharan African communities, and also provide further support for the increased dairy protein recommendations for SAM. 7 Moreover, the cost of the additional dairy protein in this study was only $0.0017/92g ration (April 2015 cost basis), indicating that the benefits of the higher dairy protein RUSF would be achieved for a very small cost increment.
In contrast to the significant beneficial effect of the higher dairy RUSF versus control on nutritional status of mothers, no significant benefit of randomization to a RUSF was seen in infants and children in either the primary ITT analysis or a PP analysis of those patients who had >50 days of supplement provision. Superficially, these results appear consistent with those of other groups who reported no benefit of both nondairy and higher protein dairy supplements in village infants and children spanning a similar age range. 2 –5 However, in contrast to those studies, all groups in this project had extremely rapid growth, with HAZ increasing by 0.82 to 1.31 and WAZ increasing by 0.44 to 0.86. The rapid growth of infants and children during the preharvest season was surprising because food is typically in short supply at this time of year, and indeed there was no weight gain in control mothers’ which provides objective data that food was not in plentiful supply. Postprotocol discussions in control villages revealed that, because being at risk of malnutrition was a criteria for study entry, community health workers in control groups had instructed mothers to use a traditional remedy to alleviate malnutrition (a paste of boiled small fish, green leaves, and cashews) in addition to other food given to the children. Based on these findings and observations, it seems likely that the lack of significant difference in growth among groups was due to comparable effectiveness of the RUSFs and the local remedies, rather than comparable lack of effectiveness. Because community health workers are respected senior individuals living in the same villages, their recommendations may carry particular weight and further studies are needed to examine their utility in interventions to prevent malnutrition at the village level.
To our knowledge, this is also the only study to report on within-family associations of anthropometric changes during RUSF supplementation, providing a new approach to explore the impact of interfamily food dynamics on interventions to prevent malnutrition. We found that baseline child WAZ and change in maternal MUAC, but not baseline maternal MUAC, predicted gain in child WAZ independent of study randomization, whereas such correlations were not observed in infants, indicating that family food dynamics impacted relative food redistribution between family members. One implication of these observations is that the families prioritized preventing malnutrition in infants up to age 2 years, perhaps reflecting the successful public health emphasis on nutrition in the first 1000 days, 17 whereas the success of treatment in older children depended to a greater extent on the adequacy of food supplies within the household. Previous studies have suggested that food sharing frequently negates the impact of food supplementation programs in low-income countries 11 –13 and this study now indicates that food sharing may impact children older than 2 years more than children younger than 2 years, and such culture-specific influences could potentially be addressed through education at the time of supplement distribution.
Concerning study limitations, although the enrolling study population was 1488 infants, children and mothers, and there was only a moderate number lost to follow-up, the study was considered a pilot because no relevant data were available from the local area for power calculations, and retrospective calculations indicated the study was underpowered to detect significant effects of the RUSF because of high measurement variability and cluster randomization at the village level. In addition, although there was a significant benefit of the higher protein RUSF in mothers which was higher than typically seen in such studies, standard relationships between weight change and energy intake 25 indicate a net increase in energy intake of only 142 kcal/d (ie, about one-third of the daily supplement intake averaged over 7 d/wk) for the amount of weight gain expected for the change in MUAC, suggesting that there was some food redistribution that occurred and reduced the gain in net maternal energy intake. An additional and very significant limitation was that the wait-listed control families apparently changed their usual feeding practices for infants and children following recommendations from community health workers. Future studies could potentially avoid the latter problem by enrolling all children within the age group but conducting analyses only on the subset with malnutrition at baseline.
In conclusion, this pilot study provides the first evidence from a randomized controlled trial in villages that RUSFs with 33% of protein provided by dairy sources can have benefits for the nutritional status of mothers that are not seen with RUSF providing only 15% of protein from dairy. This finding is consistent with our recent report of beneficial effects of higher dairy RUSF in preschool children and, given the small impact of the differences in dairy content in this study on ingredient cost, suggests that higher dairy protein RUSFs have the potential to achieve broad benefits. The study also showed that the extent to which malnutrition prevention is successful in children >2 years is related to parallel changes occurring in their mothers, whereas such interrelationships do not exist in younger children, suggesting the need to educate parents of older children about the importance of malnutrition prevention.
Footnotes
Authors’ Note
N.S. was responsible for study design. A.B.S., A.B., and I.B. provided oversight of the local International Partnership for Human Development team and were responsible for obtaining IRB permission and recruiting staff to conduct the study. P.B. implemented the training and quality control oversight at the sites. A.B.S. planned recruitment strategy recruited villages, and prepared IRB documentation. W.P. provided implementation personnel and oversight for supplement supply. S.B.R. provided consultation on protocol development and led development of a plan for staff training and quality control, and oversaw statistical analyses and manuscript preparation. M.G. performed literature reviews and reviewed the manuscript revisions. C. Brown conducted all statistical analyses. E.S. provided consultations on protocol safety and discussion of micronutrient data, and C. Bale provided local safety oversight and interpretation of results. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the US Department of Agriculture as part of a pilot project to improve nutrition under the McGovern-Dole International Food for Education and Child Nutrition Program (agreement no FFE-657-2012/043-00) and conducted with local implementation by International Partnership for Human Development.
