Abstract
Background:
Information is needed on the national prevalence of zinc deficiency to guide program development and evaluation.
Objective:
To summarize results of national surveys that assessed plasma or serum zinc concentrations (PZC) and compare the prevalence of low PZC with other methods used to estimate countries’ risk of zinc deficiency.
Methods:
National surveys that included PZC were identified through Internet searches and personal contacts. A survey was eligible if a nationally representative sampling scheme was used, PZC was analyzed, and the survey was implemented in a low- or middle-income country. Twenty surveys were eligible; 19 countries assessed PZC in young children and 14 in women of reproductive age.
Results:
In 13 of the 19 surveys, the prevalence of low PZC in children was >20%. Only Afghanistan, Azerbaijan, Nigeria, the Republic of Maldives, Sri Lanka, and China found a low prevalence of inadequate PZC among children. Some of these conclusions may be due to the lower than recommended cutoff that was used. Similarly, in 13 of 14 surveys, the prevalence of low PZC in women was >20%. Estimates of percentage population with inadequate dietary zinc intake based on food balance sheets underestimate the risk of zinc deficiency. The national stunting prevalence seems to be a useful proxy, as both indicators categorized countries similarly into high versus low risk of zinc deficiency, with some exceptions.
Conclusions:
Results from 20 countries suggest that zinc deficiency is a public health concern in the majority of these countries and zinc intervention strategies should be considered.
Keywords
Introduction
Zinc deficiency increases the risk and severity of diarrhea and other infections, restricts physical growth, and increases the risk of adverse pregnancy outcomes, such as preterm birth. 1,2 Based on available evidence, 116 000 child deaths were attributable to zinc deficiency in 2011. 3 Because of the adverse health consequences of zinc deficiency, better information is needed on the national prevalence of zinc deficiency to guide program development and evaluation.
Guidelines on the assessment of population zinc status were published by the World Health Organization (WHO), the United Nation Children’s Fund (UNICEF), the International Atomic Energy Agency, and the International Zinc Nutrition Consultative Group (IZiNCG) in 2007. 4 Recommended indicators included plasma or serum zinc concentration (PCZ) and dietary zinc intake through the use of 24-hour recalls or weighed food records. The stunting prevalence among children less than 5 years of age was suggested as a proxy of the likely risk of zinc deficiency in a population. 4 The national stunting prevalence is regularly assessed in many countries during surveys such as the Demographic and Health Surveys, and Multiple Indicator Cluster Surveys, 5 and the national stunting prevalence is well documented in the WHO Global Database on Child Growth and Malnutrition and UNICEF’s annual report on the State of the World’s Children (SWOC), 6,7 so the data are readily available. Finally, the amount of zinc in national food balance supplies has been suggested as another indicator of risk of inadequate dietary zinc intake.
Wessells and Brown estimated the national risk of inadequate zinc intake based on the estimated absorbable zinc content of the national food supplies derived from the Food and Agriculture Organization of the United Nations’ (FAO) food balance sheets (FBS). These analyses suggest that at least 17% of the world’s population are at risk of inadequate zinc intake, 8 with the highest risk occurring in sub-Saharan Africa and South Asia.
In recent years, more information has become available on the distribution of PZC in nationally representative population samples. The purposes of the present article are to (1) summarize information available on the prevalence of zinc deficiency, based on the assessment of PZC from national surveys in low- and middle-income countries and (2) compare the prevalence of zinc deficiency from these surveys with the above-mentioned estimates of risk of zinc deficiency from FBS and with the national stunting prevalence.
Methods
National surveys with available zinc data were identified (1) through Internet searches (PubMed and Google using key words “zinc” and “survey” or “national” or “national survey”) and (2) by contacting experts known to be actively involved in conducting national nutrition surveys. National surveys were eligible for inclusion in the present analyses if (1) a nationally representative sampling scheme was used, (2) PZC were analyzed, and (3) the survey was implemented in a low- or middle-income country. Copies of the identified survey reports, conference abstracts, or published articles were obtained and reviewed. A total of 20 surveys were eligible. These reports included 5 surveys from Africa, 9 –14 4 surveys from the Americas, 15 –20 3 surveys from the Eastern Mediterranean and European regions, 21 –24 3 surveys from South-East Asia, 25 –27 and 5 surveys from the Western Pacific region 28 –32 (Table 1). Kenya, South Africa, and Mexico had implemented several national nutrition surveys, but not all included the assessment of PZC. In particular, because the most recent surveys did not include the assessment of zinc status or the report has not yet become available, results of previous national surveys are summarized. 10,14,19,20 Seventeen surveys collected venous blood samples for assessment of PZC in preschool children, and Mexico and the Philippines collected blood from 1- to 11-year-old and 6- to 12-year-old children, respectively. 19,31 Thirteen surveys also collected blood samples from adult women, and Fiji collected blood only from women. 30 The sample size varied greatly between the different surveys, as some surveys aimed to assess regional differences within the country. For the purpose of the present article, only national-level information will be considered.
Population Groups, Sampling Scheme, Analyses, and Interpretation of Plasma or Serum Zinc Concentration From 20 Nationally Representative Surveys.
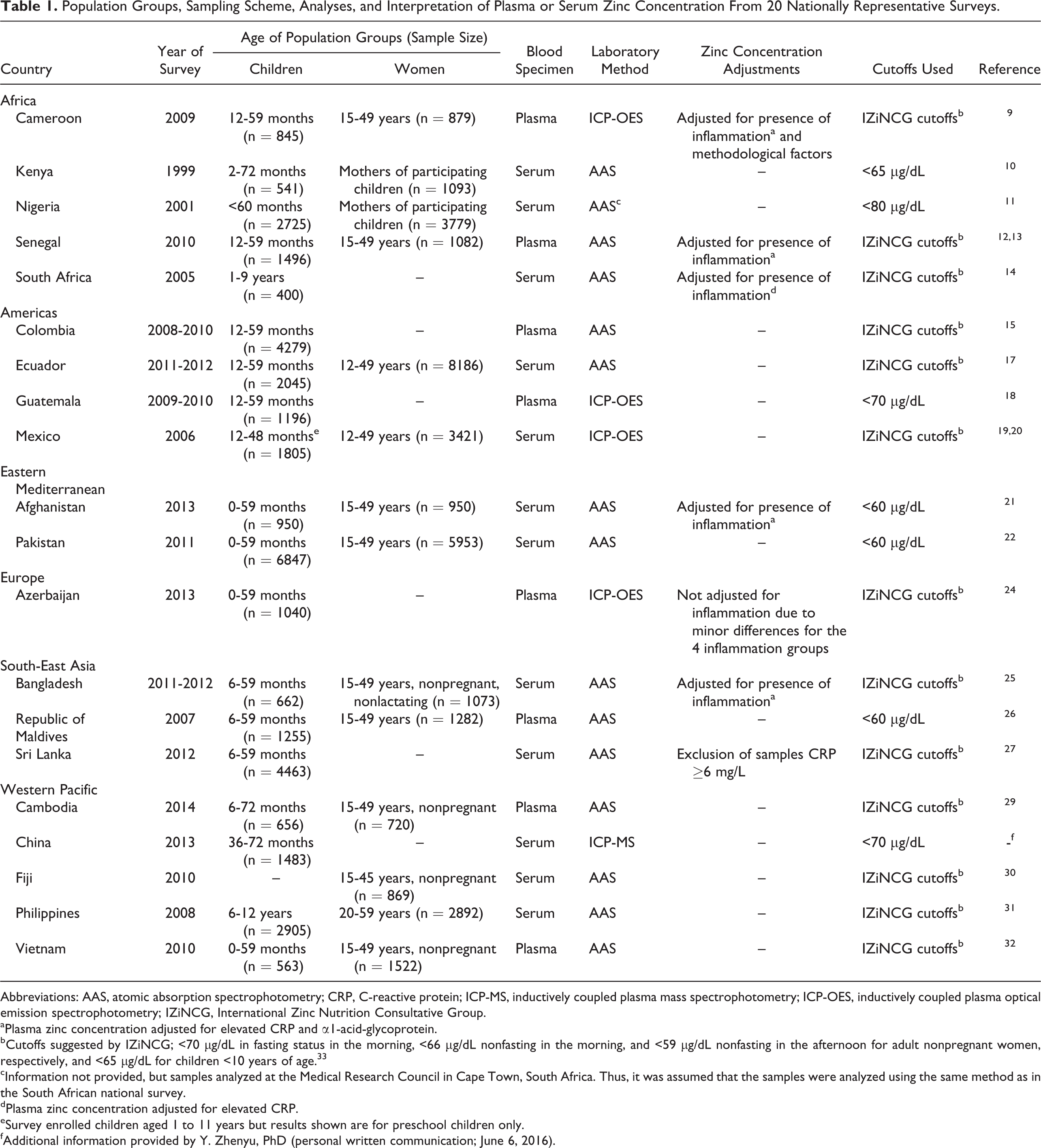
Abbreviations: AAS, atomic absorption spectrophotometry; CRP, C-reactive protein; ICP-MS, inductively coupled plasma mass spectrophotometry; ICP-OES, inductively coupled plasma optical emission spectrophotometry; IZiNCG, International Zinc Nutrition Consultative Group.
aPlasma zinc concentration adjusted for elevated CRP and α1-acid-glycoprotein.
bCutoffs suggested by IZiNCG; <70 µg/dL in fasting status in the morning, <66 µg/dL nonfasting in the morning, and <59 µg/dL nonfasting in the afternoon for adult nonpregnant women, respectively, and <65 µg/dL for children <10 years of age. 33
cInformation not provided, but samples analyzed at the Medical Research Council in Cape Town, South Africa. Thus, it was assumed that the samples were analyzed using the same method as in the South African national survey.
dPlasma zinc concentration adjusted for elevated CRP.
eSurvey enrolled children aged 1 to 11 years but results shown are for preschool children only.
fAdditional information provided by Y. Zhenyu, PhD (personal written communication; June 6, 2016).
Laboratory analyses and interpretations of data differed somewhat in the different surveys (Table 1). The PZC was determined by atomic absorption spectrophotometry (AAS), inductively coupled plasma optical emission spectrophotometry (ICP-OES), or inductively coupled plasma mass spectrophotometry (ICP-MS). Most surveys used the cutoffs suggested by IZiNCG 33,34 ; namely, <65 µg/dL for nonfasting children <10 years of age and <70 µg/dL for fasting adults in the morning and <66 µg/dL for nonfasting adults in the morning, and <59 µg/dL for nonfasting adults in the afternoon. The surveys in Afghanistan, Pakistan, and the Republic of Maldives used <60 µg/dL to define low PZC, Guatemala and China <70 µg/dL, and Nigeria <80 µg/dL. 11,18,21,22,26 Not many reports included information on the mean PZC and only reported the prevalence of low PZC. Because underlying inflammation reduces the PZC, some surveys, 9,10,12 –14,25 but not all, adjusted PZC for elevated C-reactive protein (CRP), and α1-acid-glycoprotein (AGP). In addition to the adjustments for CRP and AGP, Engle-Stone et al 9 also adjusted for time of day of sample collection, time since previous meal, and time from blood collection to centrifugation.
The estimated proportion of population with inadequate dietary zinc intake was retrieved from the online supplementary table provided by Wessells and Brown. 8 Information on the national stunting prevalence was collected using 1 of 2 approaches. If stunting prevalence was assessed during the same survey as the PZC survey, that value was used preferentially. Otherwise the national stunting prevalence assessed during the same time period as the PZC survey was retrieved from the UNICEF SWOC report or the WHO’s global database on child growth and malnutrition. 6,7
Countries were categorized as having a high risk of zinc deficiency using the following cutoffs: prevalence of low PZC is >20%, 4 proportion of population with estimated prevalence of inadequate dietary zinc intake is >25% based on FBS, 4,8 and prevalence of stunting among children younger than 5 years of age is >20%. 4,35
Results
In the past 2 decades, 20 low- and middle-income countries included the assessment of PZC in their national nutrition surveys (Table 2). Cameroon found the highest prevalence of low PZC in young children (83%), 9 followed by Cambodia (68%), 29 and about half of the young children in Kenya, Senegal, South Africa, and Vietnam had low PZC. 10,13,14,32 Similarly, the prevalence of low PZC in women of reproductive age was highest in Cameroon (83%), 9 Vietnam (67%), 32 and Cambodia (63%). 29 In 4 Latin American countries, 28% to 43% of young children had low PZC. 15,17 –19 Only Afghanistan, Azerbaijan, Nigeria, the Republic of Maldives, Sri Lanka, and China found a prevalence of low PZC among young children ≤20%, 11,21,24,26,27 and Fiji among women. 30 Of these, Afghanistan and the Republic of Maldives used a lower cutoff to define low PZC (<60 µg/dL), 21,26 which may partly account for the low reported prevalence. By contrast, Nigeria used a higher cutoff (<80 µg/dL). 11 In countries that assessed PZC in both young children and women, the prevalence of low PZC was generally similar for the 2 population subgroups. An exception was Ecuador, where the prevalence of low PZC of young children was only half of that of women (28% vs 56%). 17
Mean Plasma or Serum Zinc Concentration (PZC) and Prevalence of Zinc Deficiency From Nationally Representative Surveys.
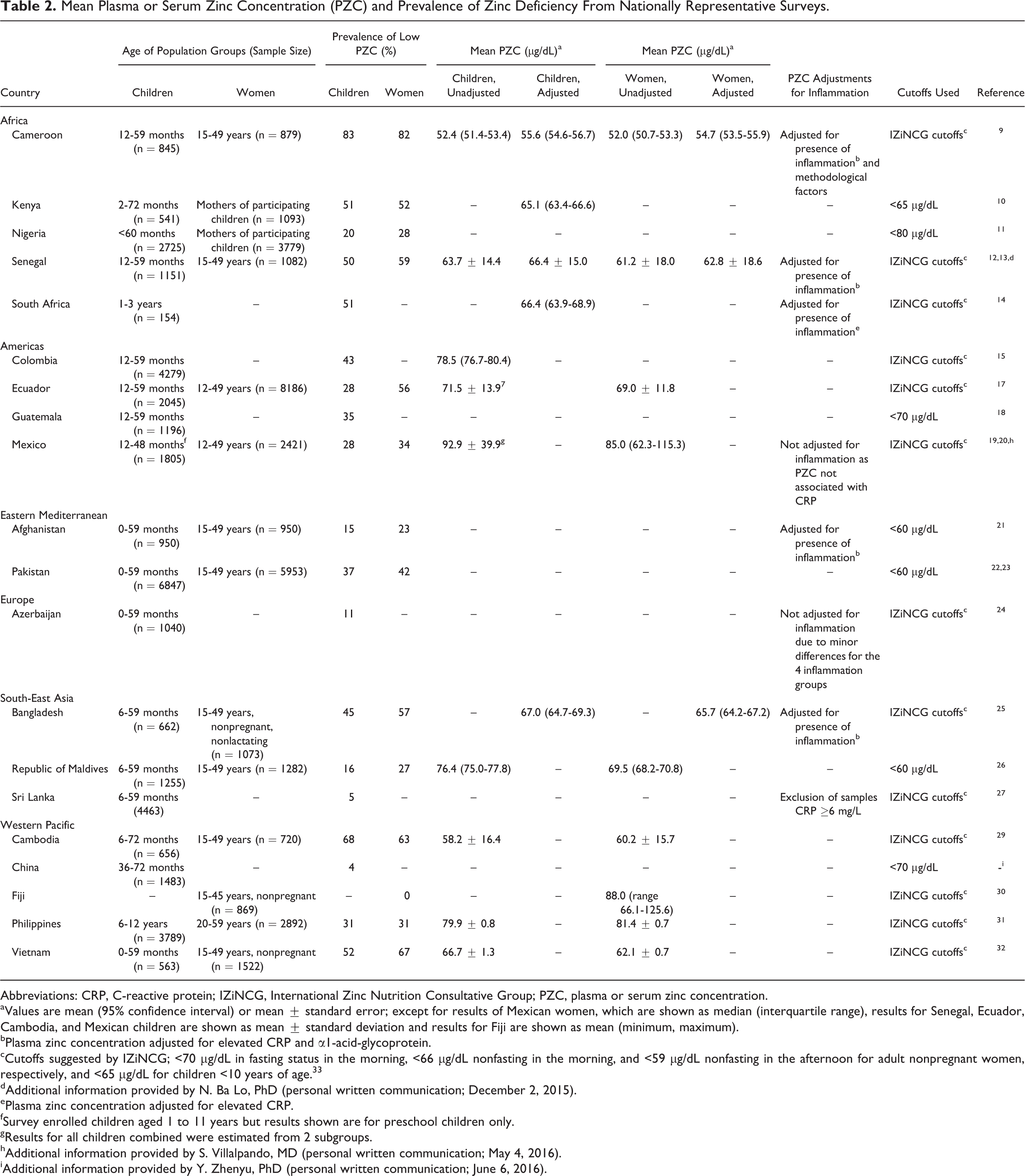
Abbreviations: CRP, C-reactive protein; IZiNCG, International Zinc Nutrition Consultative Group; PZC, plasma or serum zinc concentration.
aValues are mean (95% confidence interval) or mean ± standard error; except for results of Mexican women, which are shown as median (interquartile range), results for Senegal, Ecuador, Cambodia, and Mexican children are shown as mean ± standard deviation and results for Fiji are shown as mean (minimum, maximum).
bPlasma zinc concentration adjusted for elevated CRP and α1-acid-glycoprotein.
cCutoffs suggested by IZiNCG; <70 µg/dL in fasting status in the morning, <66 µg/dL nonfasting in the morning, and <59 µg/dL nonfasting in the afternoon for adult nonpregnant women, respectively, and <65 µg/dL for children <10 years of age. 33
dAdditional information provided by N. Ba Lo, PhD (personal written communication; December 2, 2015).
ePlasma zinc concentration adjusted for elevated CRP.
fSurvey enrolled children aged 1 to 11 years but results shown are for preschool children only.
gResults for all children combined were estimated from 2 subgroups.
hAdditional information provided by S. Villalpando, MD (personal written communication; May 4, 2016).
iAdditional information provided by Y. Zhenyu, PhD (personal written communication; June 6, 2016).
The estimated proportion of the population with inadequate dietary zinc intake based on FAO’s FBS ranged from 8% in Ecuador and China to 46% in Sri Lanka. 8 In the majority of countries, the prevalence of low PZC among young children was much higher than the estimated percentage of population with inadequate dietary zinc intake based on FBS (Figure 1). In fact, in Cameroon, Senegal, South Africa, Colombia, Ecuador, Cambodia, the Philippines, and Vietnam, the estimated prevalence of inadequate dietary zinc was less than half of the prevalence of low PZC among children. In contrast, Afghanistan, Azerbaijan, and Sri Lanka found a lower prevalence of inadequate PZC among young children than of inadequate dietary zinc intake. Only China found both a low prevalence of low PZC (4%) and a low prevalence of inadequate dietary zinc intake (8%; personal written communication; June 6, 2016; Y. Zhenyu, PhD).
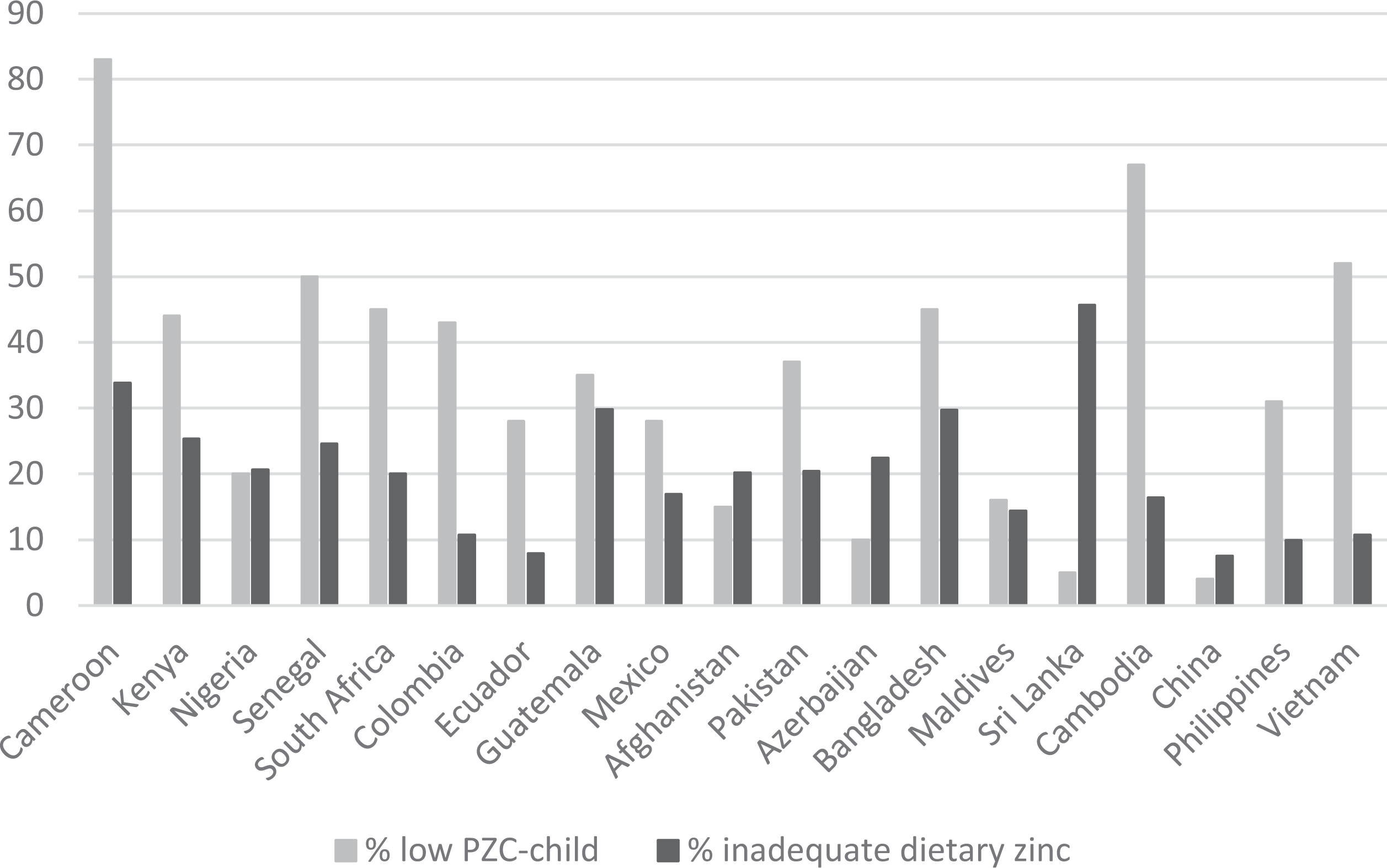
Percentage of children with low plasma or serum zinc concentration from 19 national nutrition surveys compared with the estimated percentage of population with inadequate dietary zinc intake.a,b aEstimated percentage of population with inadequate dietary zinc intake based on Food and Agriculture Organization of the United Nations’ (FAO’s) Food Balance Sheets. 8 bMost surveys used <65 µg/dL to define low plasma or serum zinc concentration (PZC). 33 The surveys in Afghanistan, Pakistan, and the Republic of Maldives used <60 µg/dL, Guatemala and China <70 µg/dL, and Nigeria <80 µg/dL.
A similar tendency is evident when comparing the prevalence of low PCZ of women of reproductive age with the estimated percentage of the population with inadequate dietary zinc based on FBS (Figure 2). In 11 of the 14 countries with information on women’s PZC, the prevalence of low PZC among women is at least about twice the estimated percentage of population with inadequate dietary zinc intake. Only in Afghanistan and Nigeria, the 2 estimates indicate a similar proportion of the population as being at risk of zinc deficiency (23% low PZC vs 20% inadequate dietary zinc intake in Afghanistan and 28% low PZC vs 21% inadequate dietary zinc intake in Nigeria). 8,11,21
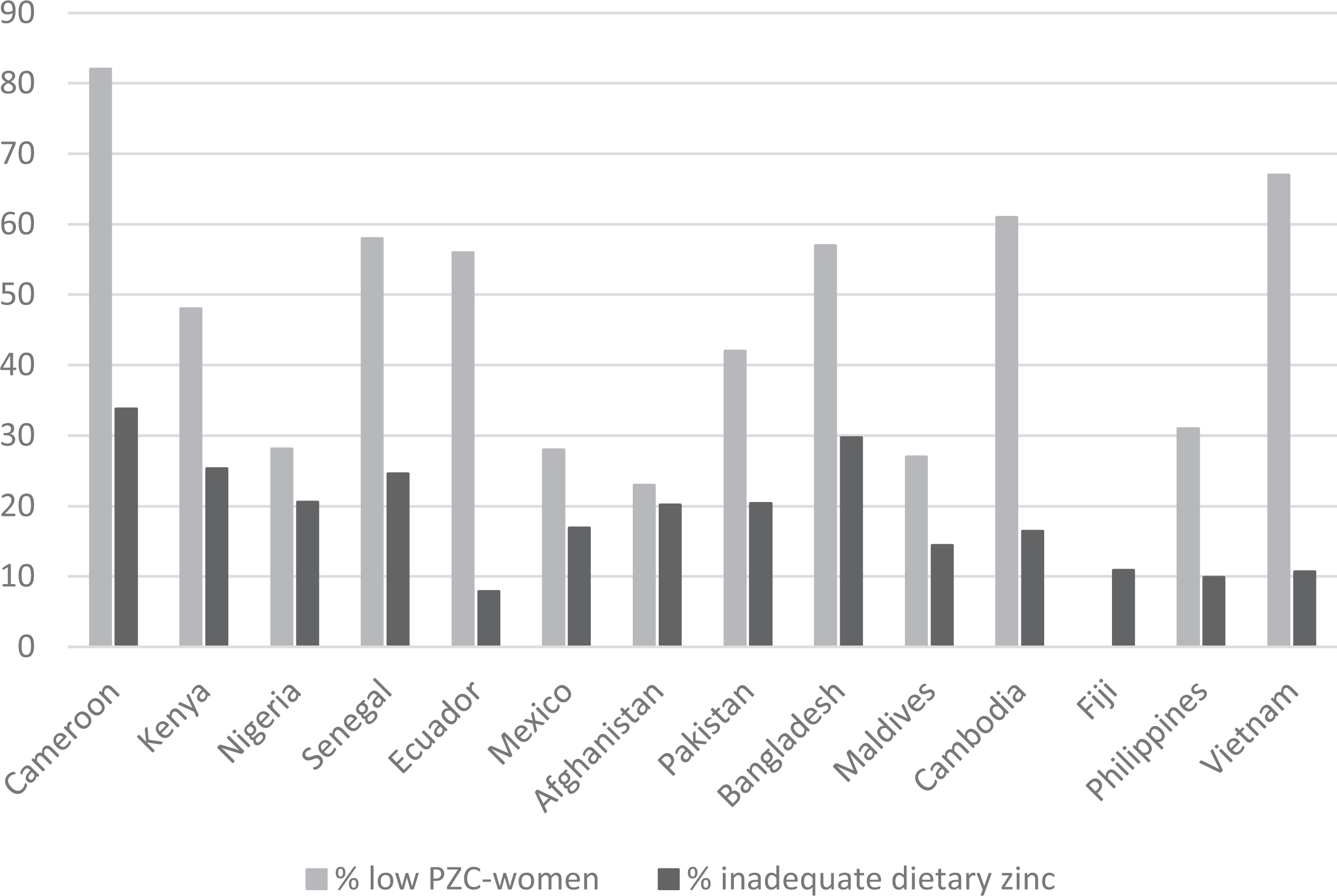
Percentage of women of reproductive age with low plasma zinc concentration from 14 national nutrition surveys compared with the estimated percentage of population with inadequate dietary zinc intake.a,b aEstimated percentage of population with inadequate dietary zinc intake based on Food and Agriculture Organization of the United Nations’ (FAO’s) Food Balance Sheets. 8 b Most surveys used the International Zinc Nutrition Consultative Group (IZiNCG) cutoffs to define low plasma or serum zinc concentration. 33 The surveys in Afghanistan, Pakistan, and the Republic of Maldives used <60 µg/dL and Nigeria <80 µg/dL.
The stunting prevalence ranged from 8% in Fiji to 48% in Guatemala. 6,7 There is some inconsistency when comparing the prevalence of low PZC among young children with national stunting rates of young children (Figure 3). Cameroon, Senegal, South Africa, Colombia, Mexico, and Vietnam found a much higher prevalence of low PZC among young children than the prevalence of stunting. In contrast, Nigeria, Afghanistan, and Sri Lanka found a higher stunting prevalence than the prevalence of low PZC among young children. In Ecuador, Pakistan, Bangladesh, the Republic of Maldives, China, and the Philippines, the 2 prevalence were comparable.
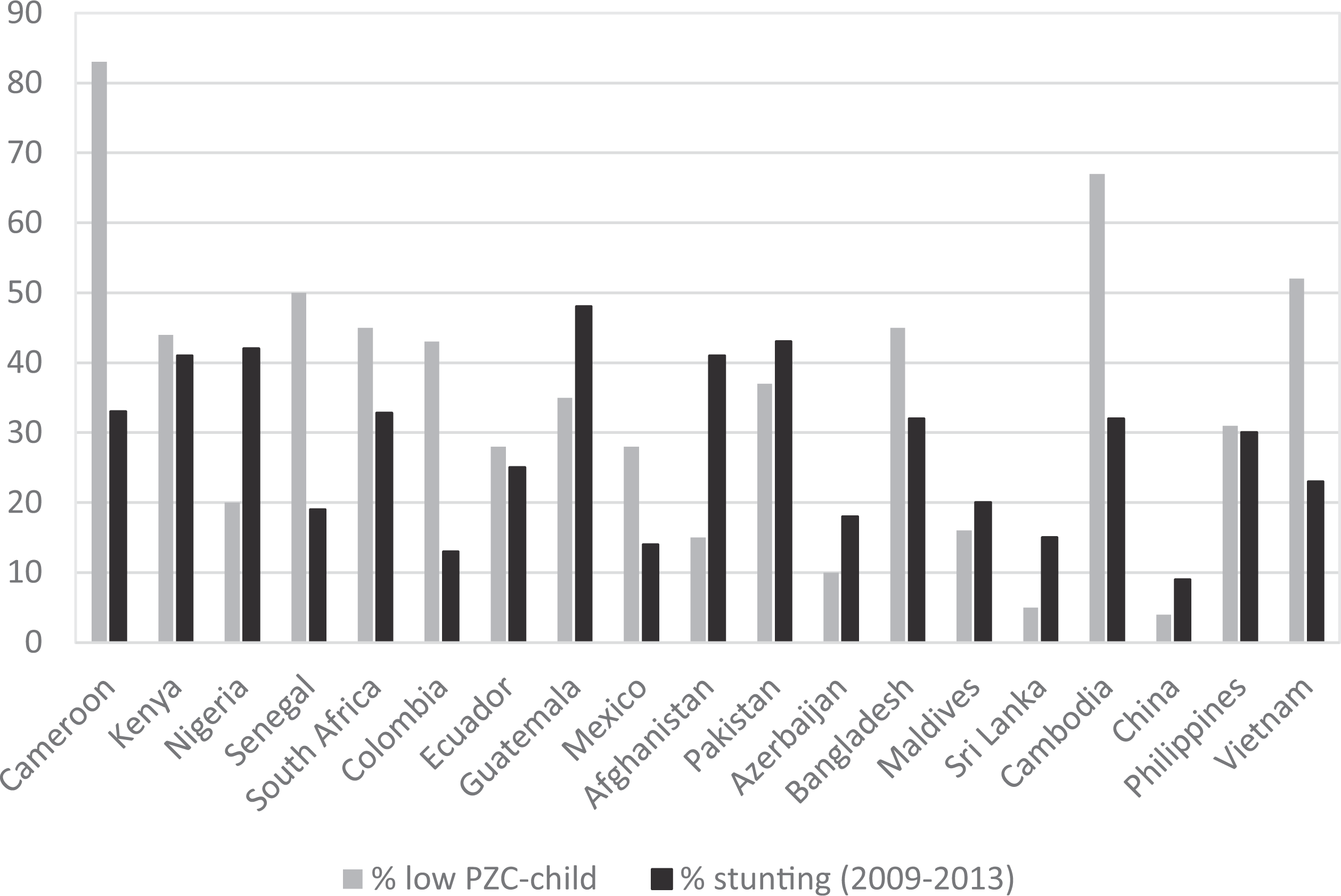
Categorizing countries into low or high risk of zinc deficiency based on the prevalence of low PZC in young children and women of reproductive age identified 13 countries as being at risk of zinc deficiency (Table 3). This is in strong contrast to only 5 countries that would be considered at risk of zinc deficiency based on the FBS-derived estimates. 8 The number of countries identified as being at risk of zinc deficiency was more comparable between low PZC and the stunting prevalence (n = 12). However, the results were inconsistent in a few individual countries. For example, Afghanistan and Nigeria would be considered at low risk of zinc deficiency based on PZC of young children, but at high risk of zinc deficiency based on the stunting prevalence. 11,21 In contrast, Colombia, Mexico, and Senegal were identified as low risk of zinc deficiency based on the stunting prevalence but at high risk of zinc deficiency based on PZC among young children. 6,13,15,19
Categorization of Risk of Zinc Deficiency Based on PZC Assessed in National Surveys, Estimated Percentage of Population With Inadequate Dietary Zinc Intake Based on FAO’s Food Balance Sheet and National Stunting Prevalence.
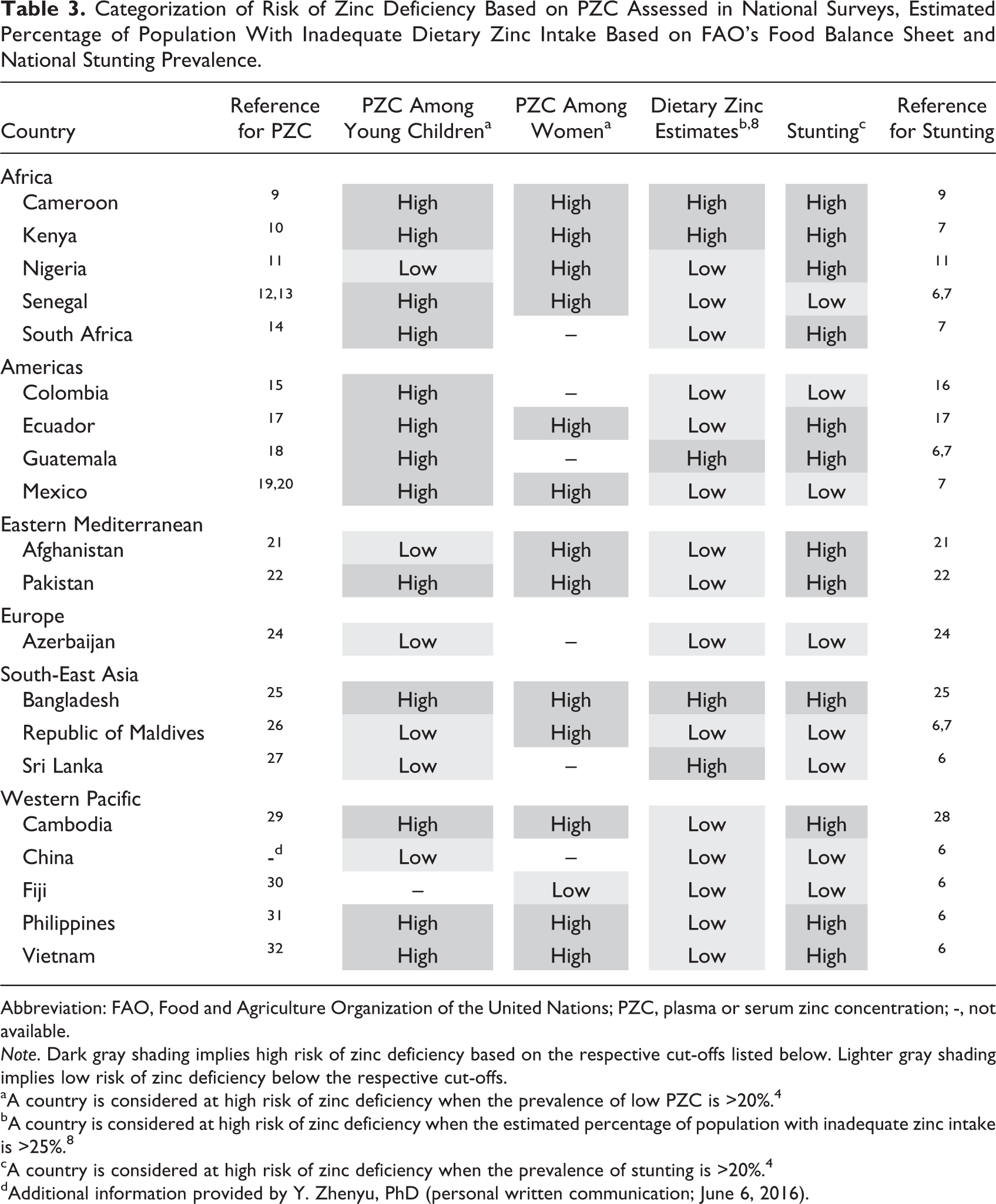
Abbreviation: FAO, Food and Agriculture Organization of the United Nations; PZC, plasma or serum zinc concentration; -, not available.
Note. Dark gray shading implies high risk of zinc deficiency based on the respective cut-offs listed below. Lighter gray shading implies low risk of zinc deficiency below the respective cut-offs.
aA country is considered at high risk of zinc deficiency when the prevalence of low PZC is >20%. 4
bA country is considered at high risk of zinc deficiency when the estimated percentage of population with inadequate zinc intake is >25%. 8
cA country is considered at high risk of zinc deficiency when the prevalence of stunting is >20%. 4
dAdditional information provided by Y. Zhenyu, PhD (personal written communication; June 6, 2016).
Discussion
The objectives of the present article were to summarize the results of nationally representative surveys that assessed PZC and compare the prevalence of low PZC with other methods used to estimate the countries’ risk of zinc deficiency. These latter methods include the estimated percentage of population with inadequate dietary zinc intake and the national stunting prevalence, which have been suggested previously as potential proxies for the risk of zinc deficiency. 4,8 Nineteen countries assessed PZC in young children and 14 countries assessed PZC in women of reproductive age. In 13 of the 19 surveys, the prevalence of low PZC in young children is >20%. Similarly, in 13 of 14 surveys, the prevalence of low PZC in women of reproductive age is >20%, implying that zinc deficiency is a public health concern in the majority of these countries and zinc intervention strategies should be considered. 4
Zinc homeostasis in the cell, tissue, and whole blood is tightly controlled to sustain metabolic functions over a wide range of zinc intakes, which makes assessing zinc status challenging. Nevertheless, several expert committees have endorsed PZC as a useful biomarker of zinc status, especially for assessing the risk of zinc deficiency in populations. 4,36 –38 Thus, among the reviewed assessment methods, national prevalence of low PZC should be considered the most reliable information, whereas the estimated percentage of population with inadequate dietary zinc intake based on FBS and the national stunting prevalence should only be considered a proxy of risk of zinc deficiency. Indeed, there was some discrepancy between the countries identified as at high risk of zinc deficiency depending on the method used.
Only Afghanistan, Azerbaijan, Nigeria, the Republic of Maldives, Sri Lanka, and China found a low prevalence of inadequate PZC among young children. These findings may be due to the cutoff used in some of the surveys. Surveys in Afghanistan, the Republic of Maldives, and Pakistan used a cutoff of <60 µg/dL, whereas most others used <65 µg/dL as was recommended by IZiNCG for nonfasting children <10 years of age. 33,34 In contrast, the survey in Nigeria used a cutoff <80 µg/dL and found that only 20% of children and 28% of women had low PZC, which is not surprising considering the high PZC cutoff that was applied. All other surveys in Africa, which used <65 µg/dL as cutoff to define low PZC, found that at least half of the children had low PZC. A similarly high prevalence of low PZC among young children was also found in recent studies in Burkina Faso, 39 –41 albeit in population samples that were not nationally representative.
Many surveys did not report how the specimens were processed or whether trace-element-free laboratory supplies were used to reduce the risk of contamination. Zinc is present in serum in very low concentrations, so any contamination from exogenous sources of zinc can dramatically alter the results. 33 Another potential source of error is the leakage of intracellular zinc into serum or plasma if blood samples are not refrigerated or stored over ice prior to separation. 42 Therefore, IZiNCG recommends the use of trace-element-free needles, syringes, and other laboratory supplies, refrigerating blood samples after collection, rapid separation of plasma, and avoidance of possible contamination from water, dust, or other sources. 33,43
Other methodological differences among the surveys include the laboratory analyses and the adjustment of PZC for indicator inflammation. The ICP-OES has been shown to result in slightly higher PZC than AAS. 44 However, the difference was small among 77 test samples (95.1 µg/dL by ICP-OES vs 93.5 µg/dL by AAS) and would likely not result in substantial difference in the prevalence of low PZC. The effect of inflammation on reducing PZC is well documented. Thus, in settings of high prevalence of inflammation, the adjustment of PZC for indicators of inflammation such as CRP and AGP is recommended. 33,43 However, even in a setting of high burden of inflammation like Cameroon, the mean PZC remained low even after adjusting for inflammation. 9 In surveys, where inflammation was not considered, PZC may have been lower due to inflammation and the prevalence of zinc deficiency may therefore be overestimated. This is of particular concern in countries with a high morbidity burden. Not many survey reports presented the mean and standard error or 95% confidence interval (CI) of PZC, which would be useful for the interpretation of the results. Thus, it is impossible to judge whether there might have been any technical issues such as contamination in some of the surveys. For future surveys, implementers should consult with IZiNCG’s recommendations on blood collection and processing techniques. 43,45 It would also be useful if national nutrition survey reports would report not only the percentage population with low zinc status but also the mean PZC concentration along with the standard deviation, standard error, or 95% CI. In fact, it would be useful if this was done for all micronutrient indicators not just PZC.
The cutoffs to define low PZC are derived from a large sample of a presumably healthy population assessed during the second National Health and Nutrition Examination Survey (NHANES II) using the 2.5th percentile of the population’s serum zinc concentration to set the PZC for subclinical zinc deficiency. 33,34 Considering that these cutoffs derive from a national survey in the United States, it is possible that they overestimate the risk of subclinical zinc deficiency in lower- and middle-income countries, and further research is required to explore the usefulness of these cutoffs. To identify an increased risk of clinical zinc deficiency, as evidenced by dermatitis and diarrhea among other symptoms, a cutoff of 50 µg/dL has been suggested recently. 46 In Cambodia, Wieringa et al 29 reported that 34% of children and 26% of women had PZC below this cutoff, which has been shown to be associated with signs of clinical zinc deficiency. It may be useful for future surveys to present the prevalence of both the subclinical and clinical zinc deficiency as together they provide relevant information about the severity of zinc deficiency as a public health problem.
The rationale for using stunting prevalence as a proxy for the prevalence of zinc deficiency is that previous randomized controlled trials indicate that children respond to zinc supplementation with increased linear growth. 47 –49 However, it is well recognized that stunting is caused by many other risk factors in addition to zinc deficiency. 50 Thus, it is not surprising that the stunting rates are not directly reflective of the prevalence of low PZC. Nevertheless, with few exceptions, both indicators categorized countries similarly into high versus low risk of zinc deficiency. It may be interesting to redo this comparison in a few years as global efforts for reducing childhood stunting continue and as more countries include PZC in their national nutrition surveys. Although there is consistent evidence that zinc supplementation reduces the incidence of diarrhea, 51 diarrhea prevalence currently is not recommended as a proxy indicator for zinc deficiency, 48 as there is considerable variation in reported diarrhea prevalence within and between countries, and many other factors likely explain these differences.
The present analyses suggest that the estimated percentage of population with inadequate dietary zinc intakes based on national FBS underestimates the risk of zinc deficiency in most countries. The FAO’s FBS provide annual national per capita food supply estimates, include all major countries, and are publically available. However, as discussed by Wessells et al, 52 there is substantial variation in the prevalence estimates of populations with inadequate dietary zinc, depending on the assumptions used in the model to calculate the dietary zinc intake from FBS. Moreover, a recent comparison of FBS with the Global Dietary Database found that FBS overestimated individual-based dietary intakes for red and processed meats by 120% (an important source of bioavailable zinc), for fish and seafood by 144%, and for whole grains by 270%. 53 Thus, this overestimation of food availability may have resulted in an overestimation of dietary zinc availability and thus an underestimate in the estimated percentage population with inadequate dietary zinc intakes. Wessells and Brown 8 previously compared estimated percentage of population with inadequate dietary zinc intakes with stunting prevalence and found that they were not correlated. Interestingly, they found that the prevalence of stunting in low- and middle-income countries decreased from 43% to 30% between 1990 and 2005, whereas the estimated prevalence of inadequate zinc intake in the same countries remained unchanged. Although the FBS are a useful tool to assess food availability at the national level, the information seems too crude to estimate the micronutrient status of a population and/or make any program or policy decisions regarding intervention strategies.
The present summary has several weaknesses. Although substantial efforts were made to identify and obtain available reports of national zinc status surveys, the present overview may not be complete. There is currently no global archive available for national zinc survey reports, which would be very useful to follow the future trends of zinc deficiency. Also, although attempts were made to provide as much detail about PZC-related information for the present article, this was not always possible as survey reports summarize many aspects of the survey and do not always include details regarding methods relevant to interpretation of PZC. Finally, some of the surveys included were implemented more than a decade ago and may no longer reflect the current status of the population, in particular, in those countries where major progress has been made to improve the population of zinc intake. However, to date no country has reported results from more than 1 national survey of zinc status. Thus, global trends cannot currently be assessed, and future analyses of changes over time would be of interest.
In summary, 20 national surveys in low- and middle-income countries have included the assessment of PZC in national micronutrient surveys over the past 2 decades and thus provide relevant information on national zinc status. The majority of countries found that zinc deficiency is a public health concern, and it is recommended that preventive zinc interventions should be considered in these settings. 54 Although the prevalence of stunting and the estimated percentage of the population with inadequate dietary zinc intake can be useful in identifying countries at risk of zinc deficiency, the assessment of PZC is recommended to confirm the presence of zinc deficiency and evaluate the extent of the problem.
Footnotes
Author’s Note
Lisa Rogers (World Health Organization, Switzerland) and Fabian Rohner (GroundWork, Switzerland) helped to identify the available national zinc surveys. Kenneth H. Brown (University of California, Davis, CA, USA) provided valuable comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by a grant from the International Zinc Association to the International Zinc Nutrition Consultative Group (IZiNCG).
