Abstract
Background:
Vitamin A supplementation (VAS) among children 6 to 59 months of age reduces vitamin A deficiency (VAD)-related mortality. Child health days (CHDs) only reach an estimated 16.7% of children at exactly 6 months, leaving uncovered children at risk of VAD-related mortality; similarly, VAS provided at 9 months of age with measles-containing vaccine leaves infants unprotected for 3 months.
Objective:
Using data from sub-Saharan Africa, we estimated the mortality benefits and safety of providing VAS at age 6 months, compared to delivery through CHDs and at 9 months.
Methods:
We modeled VAS-preventable mortality benefits at 6 months as a function of published VAS effect sizes, intervention coverage, and proportion of infant deaths occurring between 6 and 11 months. To evaluate safety, we modeled the effect of different VAS coverage scenarios on maximum hepatic vitamin A concentrations (HVACs).
Results:
VAS linked to a 6-month visit could reduce infant mortality by an additional 1.95 (95% confidence interval [CI]: 1.38-2.52) and 1.63 (95% CI: 1.15-2.11) percentage points compared to VAS through CHDs and at 9 months, respectively. The HVAC models indicate that VAS at 6 months is safe even in the presence of a second VAS dose 1 month later and other food-based vitamin A control strategies.
Conclusion:
Advancing the first VAS dose to 6 months should be considered in settings where VAS is currently given first at 9 months. A 6-month VAS dose should also be considered in settings where VAS is delivered through CHDs. VAS delivery at 6 months could also serve as a platform to deliver other high-impact interventions.
Introduction
Vitamin A deficiency (VAD) is a major public health problem and an important contributor to morbidity and mortality among preschool age children worldwide. 1 According to most recent global estimates, VAD (defined as serum retinol < 0.70 µmol/L) affects 29% of preschool age children in low- and middle-income settings, mostly in sub-Saharan Africa and South Asia. 2
Newborns have very low hepatic vitamin A reserves, regardless of maternal nutritional status, but healthy and exclusively breast-fed infants of vitamin A-replete mothers are able to increase their vitamin stores by 6 months. 3 However, only 37% of children younger than 6 months in low- and middle-income countries are exclusively breast-fed. 4 Other risk factors for low infant vitamin A stores at 6 months are poor maternal vitamin A status (leading to low breast milk vitamin A concentrations) and high infant infectious disease burden. 3,5 -7
In later infancy, continued breast-feeding provides an important source of vitamin A, even in the presence of maternal VAD. 8 Nutrition education, nutrition-focused agricultural programs, and novel biofortification approaches can also increase child vitamin A intakes. 9 -11 The preformed vitamin A contained in animal-source foods (ASFs) is more bioavailable compared to the provitamin A found in plant-based foods, but consumption of ASFs is often limited by their high price. 12,13 The industrial fortification of staple foods with vitamin A plays an increasingly important role in the control of VAD, but it is unclear whether young children consume sufficient amounts of these staples to affect their status. 14,15 Specially designed, fortified complementary foods may have a greater impact, but economically disadvantaged groups usually have low access to these products. 16 The home fortification of complementary foods with small-quantity lipid-based nutritional supplements or multiple micronutrient powders is promising, but the impact of current formulations on vitamin A status remains uncertain. 16
Until such dietary strategies are optimized, twice-yearly vitamin A supplementation (VAS) among children from 6 to 59 months is the most effective programmatic approach to reduce VAD-related mortality. 17 High dosages of VAS are adequately absorbed and retained, replenish liver stores, and are released to meet tissue requirements. Even though high-dose VAS only transiently improves vitamin A status (as measured by serum retinol concentration), 17 its benefits for child survival have been well documented. 18 -21 The summary estimate from 17 trials indicates that VAS reduces all-cause mortality by 24% (95% confidence interval [CI]: 0.69-0.83) among children of age 6 to 59 months. 1 The estimate was lowered to 12% when a large, loosely supervised effectiveness study from India was included, 22 but the mortality reduction remained highly significant (95% CI: 0.84-0.94). On the other hand, VAS for pregnant 23 and postpartum women 24 has shown no effect on maternal and infant mortality, and VAS given to neonates 25 -27 and infants 1 to 5 months 25 has shown inconsistent effect on infant mortality. Based on these findings, World Health Organization (WHO) recommends 1 high-dose supplement in the form of 100 000 IU retinol to infants aged 6 to 11 months, and 200 000 IU retinol every 4 to 6 months to children aged 12 to 59 months in settings where VAD is a public health problem (defined as ≥1% prevalence of night blindness in children 24 to 59 months of age or ≥20% prevalence of serum retinol concentration ≤0.70 μmol/L in children 6 to 59 months of age). VAS should be delivered during routine health system contacts, such as for vaccinations, and should be integrated into national immunization days (NIDs) or other vertical child health and survival programs. In this article, we define such twice-yearly vertical events delivering VAS within a package of child-survival interventions as child health days (CHDs). 28
The WHO recommendation for VAS doses every 4 to 6 months among children 6 to 59 months of age has been implemented successfully in many countries. Globally, the coverage of CHDs increased from 16% in 1999 to 69% to 75% in recent years. 29 The first VAS events, initiated in the 1990s, linked VAS with NIDs for poliomyelitis eradication. Currently, in many countries CHDs deliver 2 annual dosages of VAS, often alongside other essential child-survival interventions, such as deworming and vaccinations. 30
CHDs draw on available human and physical resources of the health sector, often with financial and technical support from development partners. 31 When appropriately planned and targeted, they achieve far greater coverage than the routine primary health–care system, which is often too weak to reach children reliably with VAS and other preventive interventions, especially those who have completed the routine vaccination schedule in late infancy (Supplementary Table 1). 32 However, owing to their standard implementation during 2 prespecified periods twice a year, CHDs often fail to reach the majority of children during their 6 months of life. Data from postevent coverage surveys from 7 sub-Saharan countries indicate that infants are reached during these events on average at 9.1 months of age (standard deviation 1.53; Helen Keller International, unpublished data, 2012). Similarly, the routine codelivery of VAS with the first dose of a measles-containing vaccine (MCV1) as recommended by WHO, 33 which generally occurs at age 9 months in countries implementing VAS programs, also creates an unnecessary 3-month delay (or longer, if the visit is missed) without protection against vitamin A-preventable mortality. 33,34
It is unclear how many children currently receive VAS alongside routine measles vaccination or through other routine contacts within the health system. According to WHO, 28 countries report distributing VAS through routine immunization services, 42 through routine services as well as CHDs, and 13 only through CHDs. 35 Numerous countries report the provision of VAS to children at 6 months, 36 but currently only a few countries are rigorously assessing the feasibility of this approach through pilot programs, 37 -39 given the lack of a 6-month visit in most routine health-care systems. Even though an estimated 84% of children in developing countries receive MCV1 through the routine health-care system, 40 VAS coverage is likely to be substantially lower, given that the provision of VAS through the routine health-care system has received little programmatic support.
The delays associated with reaching children through CHDs and through the MCV1 contact may represent an important missed opportunity for saving lives, where VAD is common and mortality rates are high during the second half of infancy. A solution for addressing this critical gap in VAS coverage would be to include a 6-month visit for the delivery of VAS and possibly other services in the routine vaccination schedule. 41 Given that the administration of repeat VAS dosages through multiple delivery mechanisms is possible owing to the poor child-centered VAS monitoring systems in many settings, potential harms related to vitamin A overdosing need to be assessed, however. We therefore examined the potential benefit, safety, and programmatic implications of including a 6-month visit for the delivery of VAS in the routine vaccination schedule. We used data from sub-Saharan Africa, given that this is the geographic region with most VAS programs. 42
Methods
Mortality Impact
We modeled the additional mortality benefit of a VAS dose delivered at 6 months for children 6 to 11 months, as compared to delivery through CHDs only and delivery at 9 months (as observed during MCV1), using available age-specific mortality rates and the summary effect size of VAS on mortality. We drew on analyses based on Demographic and Health Surveys conducted between 1985 and 2004 to determine the proportion of infant deaths that occur during the period from 6 to 11 months in sub-Saharan Africa (J. Ross and B. Burkhalter, unpublished results).
We estimated the average proportion of children reached by CHDs per monthly age interval. We estimated that there is a 1 (16.7%) in 6 probability that an infant receives VAS through a twice-yearly CHDs at age 6 months, whereas the probably of reaching an infant aged 6 to 11 months through a twice-yearly CHDs is 6 (100%) in 6 (assuming that the CHDs achieve universal coverage). We subsequently applied different VAS coverage scenarios for the 6- and 9-month facility-based contacts as well as CHDs. There is no evidence that the mortality effect of VAS is modified by child age within the age range 6 to 59 months 43 ; therefore, we applied the effect observed among children 6 to 59 months to the 6- to 11-month age-group and assumed that VAS provides a constant mortality benefit for a duration of 6 months. In models for the 6- and 9-month contacts, we assumed that CHDs were implemented with 100% VAS coverage. We modeled the VAS effect using the generally accepted 24% mortality effect; to obtain 95% CI around these summary estimate, we used the 95% CI published in 28 as a basis for our calculations. Finally, we also applied a conservative 12% mortality effect that is reflective of all published trials. 1 The percentage of all deaths occurring among the 6- to 11-month age-group without VAS served as the reference category.
We determined the mortality benefit for VAS delivered at 6- and 9-month contacts as well as CHDs as the sum of the age-specific mortality reductions (expressed as percentage points) for each delivery mode. For the additional mortality benefit for VAS delivered at 6 months, we calculated the difference (in percentage points) between VAS delivered at 6 months with VAS delivered at 9 months and with VAS delivered through CHDs. We derived mortality benefits expressed as infant deaths averted per 1000 live births by applying the mortality benefits for VAS delivered at 6- and 9-month contacts as well as CHDs (assuming a VAS-preventable 24% mortality reduction and 100% intervention coverage) to the infant mortality rate of 69 deaths per 1000 live births listed for sub-Saharan Africa. 44 We obtained mortality benefits expressed as absolute number of infant deaths averted by multiplying the infant deaths averted per 1000 live births by the annual number of births for sub-Saharan Africa. 44
Safety
The age-specific VAS dosages recommended by WHO are considered safe among children; adverse effects, if any, are usually mild and transient. 28 Although providing 2 separate dosages within less than 1 month of each other is not recommended, the risk of adverse effects is considered negligible. 41 To further examine the possibility of overdosing when VAS programs coexist with other VAD control strategies, we used a simulation model to estimate hepatic vitamin A concentrations (HVACs) under different scenarios of vitamin A intake and vitamin A intervention programs, as well as different estimations of vitamin A loss from the body. Details of this kinetic model have been described previously. 45
The modeled HVACs used to estimate total body vitamin A content was based on published results of the vitamin A concentration of liver samples obtained from children at autopsy, as approximately 90% of total body vitamin A is stored in the liver. These previously measured HVACs were multiplied by the estimated liver weight for children of different ages and divided by 0.9 to estimate the range of whole body vitamin A content of these children who presumably had access to adequate dietary vitamin A. One such autopsy study of US children found that the mean HVAC was ≈20 μg/g during the first 3 months of life, increasing to ≈80 μg/g by 2 years of age. 46 Among the 18 children 6 to 23 months included in that study, the HVAC ranged from 16 to 165 μg/g. We therefore defined 165 μg/g as the threshold for upper HVAC limit and defined HVAC <10 μg/g as deficient. 47 In the models presented, we assumed that an exclusively breast-fed infant has low, but presumably adequate, HVAC of 20 μg/g liver at 6 months of age, when, ideally, other foods are first introduced into the diet. Hepatic vitamin A concentration was modeled up to 24 months of age as a function of varying vitamin A intake through breast milk, complementary foods, fortified cooking oil, as well as of varying vitamin A disposal rate and VAS scenarios. All statistical analyses were performed using Microsoft Excel 2013 (Microsoft, Redmond, Washington). This research was judged to be exempt from ethical review.
Results
Mortality Impact
Demographic and Health Surveys conducted between 1985 and 2004 indicated that approximately 17.1% of all infant deaths occur during the period from 6 to 11 months in sub-Saharan Africa, with 3.7% of all infant deaths occurring among infants 6 months of age and 2.0% occurring among those 11 months of age (Table 1; J. Ross and B. Burkhalter, unpublished results).
Effect of Delivering High-Dose Vitamin A Supplements (VAS) During Twice-Yearly Child Health Days (CHDs) or When Infants Attain 6 or 9 Months of Age.a
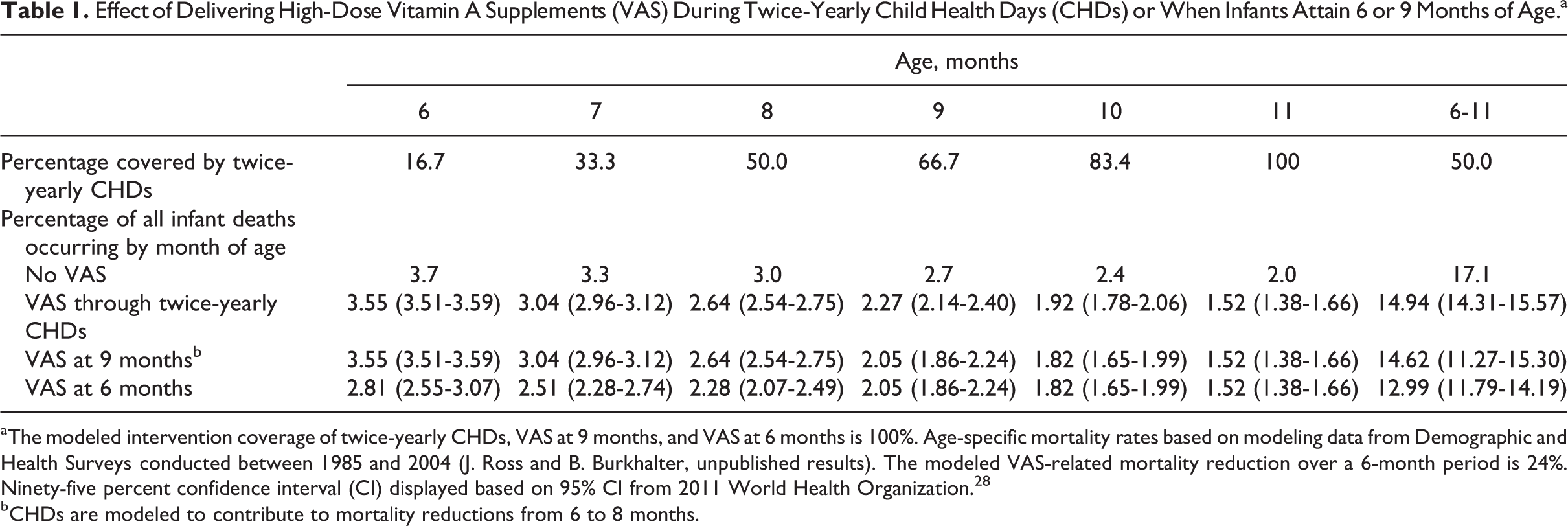
aThe modeled intervention coverage of twice-yearly CHDs, VAS at 9 months, and VAS at 6 months is 100%. Age-specific mortality rates based on modeling data from Demographic and Health Surveys conducted between 1985 and 2004 (J. Ross and B. Burkhalter, unpublished results). The modeled VAS-related mortality reduction over a 6-month period is 24%. Ninety-five percent confidence interval (CI) displayed based on 95% CI from 2011 World Health Organization. 28
bCHDs are modeled to contribute to mortality reductions from 6 to 8 months.
The probability that a child would receive VAS at exactly 6 months through a twice-yearly CHDs with 100% coverage is 16.7%, and the probability that an infant would receive VAS during the 6- to 11-month age window increases to 100% by 11 months, for an average probability of 50% among all children 6 to 11 months of age.
Assuming a 24% all-cause mortality reduction and 100% coverage, VAS delivered by CHDs would reduce infant deaths by 2.16 (95% CI: 1.53-2.79) percentage points, and VAS provided at 9 months would reduce infant deaths by 2.48 (95% CI: 1.76-3.20) percentage points. By contrast, VAS at 6 months would reduce infant deaths by 4.11 (95% CI: 2.91-5.31) percentage points, representing a mortality benefit of 1.95 (95% CI: 1.38-2.52) percentage points over delivering VAS through CHDs, and 1.63 (95% CI: 1.15-2.11) percentage points over VAS at 9 months. Compared to CHDs and VAS at 9 months, the benefits of VAS at 6 months would translate into annual additional reductions in infant mortality of 1.34 (95% CI: 0.95-1.74) and 1.13 (95% CI: 0.77-1.71) deaths per 1000 live births, respectively (Figure 1). These reductions would translate into absolute annual reductions of 43 700 (95% CI: 31 000-56 700) and 36 700 (95% CI: 25 000-55 800) deaths, respectively (data not shown).
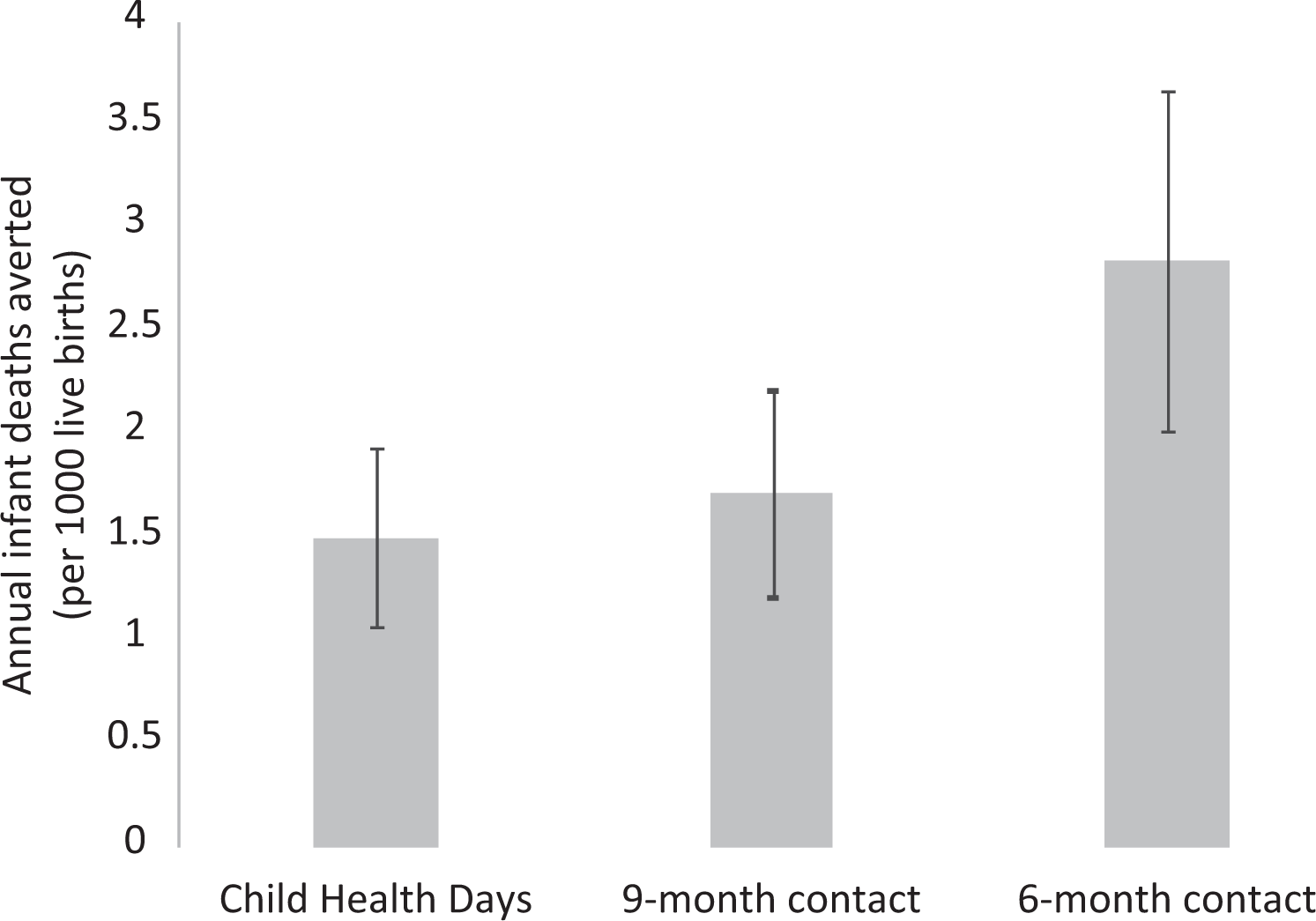
Effect of vitamin A supplementation delivered through Child Health Days or as part of a 9- or 6-month visit in the routine vaccination schedule on infant mortality. The model assumes a 100% VAS coverage of the three delivery mechanisms, a 24% VAS-related reduction in the current infant mortality in sub-Saharan Africa (69 deaths per 1000 live births). 95% confidence intervals (CI) displayed based on 95% CI from World Health Organization. 28
The additional mortality benefit of providing VAS at 6 months would drop to 1.90 (95% CI: 1.34-2.45) and 1.51 (95% CI: 1.06-1.95) percentage points if coverage of VAS at 6 months and during CHDs dropped to 80% and 50%, respectively (Table 2). The additional mortality benefit would be 1.19 (95% CI: 0.84-1.54) percentage points if CHDs reached a VAS coverage of 80% (the approximate VAS coverage observed in sub-Saharan Africa) and VAS at 6 months a 50% coverage (the approximate coverage noted in weak routine vaccination programs). The additional mortality benefit of providing VAS at 6 months compared to VAS at 9 months was 1.65 (95% CI: 1.17-2.13) and 1.35 (95% CI: 0.95-1.75) percentage points if the VAS coverage dropped to 80% and 50% at both time points, respectively. For all calculations, using a more conservative VAS-preventable all-cause mortality reduction of 12% 28 reduces all mortality benefits by half (data not shown).
Mortality Effect of Vitamin A Supplementation (VAS) Delivery at 6 Months Compared to Twice-Yearly Child Health Days (CHDs) or to Delivery at 9 Months.a

aAge-specific mortality rates based on modeling data from Demographic and Health Surveys conducted between 1985 and 2004 (J. Ross and B. Burkhalter, unpublished results). The modeled VAS-related mortality reduction over a 6-month period is 24%. Ninety-five percent confidence interval (CI) displayed based on 95% CI from 2011 World Health Organization. 28
bIncludes the mortality benefit of VAS given through twice-yearly CHDs (modeled at equivalent coverage) for 6- to 11-month-old children who were missed at the 6-month visit.
cIncludes the mortality benefit of VAS given through twice-yearly CHDs (modeled at equivalent coverage) for 6- to 8-month-old children and for 9- to 11-month-old children who were missed at the 9-month visit.
Safety
To address concerns about the safety of 6-month VAS delivery in the context of other vitamin A intervention programs, we modeled the effects of increasing vitamin A intake by means of different interventions, as presented in Figure 2. For the analysis presented in Part 1 of Figure 2, we assumed that an exclusively breast-fed infant has low, but presumably adequate, HVAC of 20 μg/g liver at 6 months of age, when, ideally, other foods are first introduced into the diet. We then assumed that breast milk vitamin A concentrations are nondeficient (30 μg/dL) 48 and that mean breast milk intake ranges from 549 to 660 g/d between 6 and 24 months. 49 Under these conditions, breast milk provides 165 to 198 μg RAE/d. To allow the infant to meet the Institute of Medicine Recommended Dietary Allowance of 300 μg RAE/d, 50 we modeled that complementary foods provide an additional 102 to 135 μg RAE/d depending on age. Assuming that 50% of the ingested vitamin A is retained and the subsequent vitamin A disposal rate is 2.2% of body pool/d, as has been observed in studies among children 12 to 24 months of age in Peru, 51 we estimated that the mean daily HVAC from 6 to 11 months ranges from 17 to 22 μg/g over this age range. We added to this scenario, a dose of 100 000 IU retinol given at 6 months of age and doses of 200 000 IU retinol at age 12 and 18 months (part 2 of Figure 2). The observed HVAC peaks of 75, 116, and 102 μg/g at 6, 12, and 18 months, respectively, are considerably below the upper limit of HVAC set at 165 μg/g. Adding to the previous scenario a dose of 100 000 IU retinol at 7 months, as could occur during a subsequent CHDs (Part 3 of Figure 2), led to HVAC peaks between 75 and 118 μg/g.
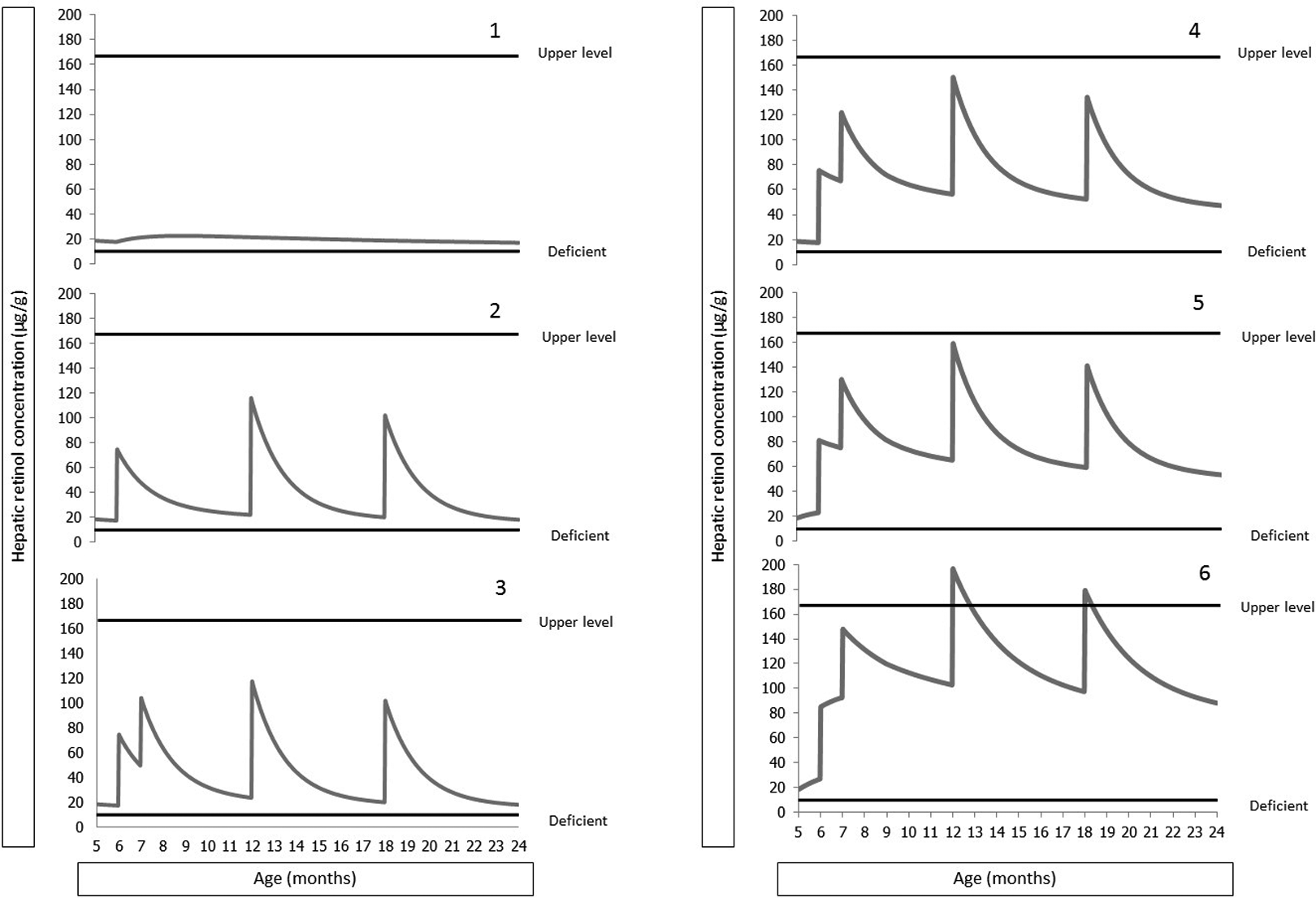
Simulation models to estimate hepatic vitamin A (VA). Part 1: Baseline scenario with hepatic VA concentrations of 20 μg/g liver at 6 months of age, breast milk intake 549 to 660 g/d (depending on age) and VA concentration of 30 μg/dL, complementary food VA intake 102 to 135 μg RAE/d (depending on age), 50% retention of VA ingested, and VA disposal rate 2.2% of body pool/d. Part 2: Previous scenario with dose of 100 000 IU retinol given at 6 months of age, and doses of 200 000 IU retinol at age 12 and 18 months. Part 3: Previous scenario with 100 000 IU retinol at 7 months. Part 4: Previous scenario with daily dose of 400 μg RAE/d, 30 to 122 μg RAE/d from a cooking oil fortification program. Part 5: Previous scenario with breast milk VA intake 50 μg/dL. Part 6: Previous scenario with VA disposal rate 1.4% of body pool/d.
In part 4 of Figure 2, we further added a daily dose of 400 μg RAE/d (such as by means of a single sachet of multiple micronutrient powder or of small quantity, lipid-based micronutrient supplement). Furthermore, we added 30 to 122 μg RAE/d from a cooking oil fortification program that uses a retinol fortification level of 23 μg/g oil and that contributes 10% of the non–breast milk energy. In this scenario, the HVAC reaches a maximum of 150 μg/g at 12 months. Increasing breast milk vitamin A concentration from the 30 μg/dL used in previous scenarios to 50 μg/dL, a level that has been reported in some studies conducted in developing countries, 3 HVACs reached 159 μg/g at 12 months and thus approached the upper limit (Part 5 of Figure 2). Using a more conservative estimate of vitamin A disposal at a rate of 1.4% of body pool/d, which was the lower end of the confidence limit in the study of Peruvian children and similar to the disposal rate measured in a single healthy US child, 52 the maximum HVAC transiently reaches 196 μg/g at 12 months and 179 μg/g at 18 months (Part 6 of Figure 2), which exceed the upper limit. Even though these HVACs dropped below 165 μg/g 12 to 27 days later, it would not be desirable to reach these levels in children until further information is available to confirm that they are not associated with any adverse effects. In sum, in all of the foregoing simulations the maximum HVACs did not exceed the maximum range observed in US children, except for a < 1 month period in the extreme sixth scenario.
Discussion
In this article, we demonstrate that providing VAS linked to a 6-month contact point in the routine immunization schedule offers additional mortality benefits as compared to delivering VAS through CHDs and during the 9-month MCV1 visit. Modeling exercises indicate that providing VAS at 6 months results in HVAC in the range reported for US children 6 to 23 months of age and which are likely to be safe, even when a second dose of VAS is given as soon as 1 month after the first dose and in the presence of other dietary strategies to improve vitamin A status. Nevertheless, caution is needed in settings with a wide array of initiatives to control VAD.
Provision of VAS at 6 months would not only have direct survival benefits but also serve as a platform for the delivery of other high-impact interventions. These include counseling in family planning options, improvements in hygiene practices, cognitive stimulation, and optimization of infant and young child feeding practices (including, in appropriate settings, the provision of lipid-based nutrient supplements or multiple micronutrient powders). 16,53 -55 A 6-month VAS contact would fall between the 14-week diphtheria–tetanus–pertussis (DTP) and the 9-month MCV1 contact and thus shorten the gap between visits from approximately 6 months to 3 months, which may help reduce attrition rates and provide catch-up vaccinations. The 6-month contact could thereby help reach the international goal of attaining 90% national coverage and 80% coverage in every district for all vaccines in national programs by 2020. 56
The 6-month VAS contact may serve as the only VAS contact in the routine health system, as delivery at 9 months may no longer be necessary in case CHDs ensure the delivery of subsequent doses up to 59 months of age. In settings where a 6-month contact achieves high coverage, policy makers may be tempted to target CHDs to children 12 to 59 months. This would align programs with the target age-group for deworming programs and streamline procurement and supply management systems by requiring only vitamin A capsules of 200 000 IU for these events. 57 However, this strategy would fail to reach the majority of children with VAS at exactly 12 months and would therefore provide suboptimal coverage against vitamin A-preventable mortality until the next VAS round. Therefore, it is preferable to provide VAS at 6 months while continuing CHDs among the entire 6- to 59-month age range. The 6-month contact point could also serve as a platform to improve the coverage of routine immunization programs and other nutrition interventions and therefore provide further child-survival benefits.
Our study has several limitations. The kinetic model assumes that retention of vitamin A is 50%, which is a value taken from an estimate for adults 58 and which may differ in young children. The model uses a fractional catabolic rate for vitamin A that was estimated in children 1 to 2 years of age 51 and which may differ in infants <12 months of age. The determination of lower and upper HVAC limits is based on a small number of children from North America. However, the reported HVAC for Brazilian children 1 to 11 months of age, who died of various causes, is within a similar range (mean 58.7 ± 47.6 µg/g [0-221 µg/g]). 59 The reported HVAC for a 6-month-old Thai infant who died from pneumonia and severe malnutrition was as 5.4 µg/g, 60 which is below the our stated lower limit of 10 µg/g. Even though we believe that the assumptions used in the kinetic models and the stated HVAC limits apply to young children in low-resource settings, more data are needed to have more robust models for predicting the impact of vitamin A intake on HVAC. There is some controversy over the current mortality impact of VAS. 61,62 In our models, we used the estimates supported by a recent systematic review 43 and supported by WHO 28 ; in addition, we stated a potential lower bound obtained when a large evaluation from India was taken into account that was unpublished at the time of the systematic review. The Lives Saved Tool has been used to estimate the contributions of different interventions (including VAS) on child mortality 63 and has become a useful planning tool. However, the tool may underestimate VAS mortality benefits 64 because it relies on cause-specific mortality estimates of VAS, for which trial data were less robust, given that VAS mortality trials were generally designed to model overall mortality only. Even though postneonatal mortality has started to account for decreased proportions of under-5 mortality in sub-Saharan Africa, 65 the strategies outlined in this article are likely to remain valid in the context of persistently high under-5 mortality rates in sub-Saharan Africa.
Finally, there have been previous recommendations to deliver VAS as part of the routine Expanded Programme on Immunization schedule within the first 6 months of life, such as during DTP contacts, 66 but these recommendations were not implemented at scale. However, we believe that the current call for provision of VAS at 6 months is more promising given that the evidence base for provision at 6 months is stronger than providing VAS in the first 6 months, routine health-care systems already have experience to providing VAS to children in the 6 to 59 months period, routine health systems are being strengthened, and the potential to position a 6-month visit as a platform for other nutrition and health interventions may garner additional support. In this regard, pilots of this approach show promising results. 37 -39
In aggregate, the information presented indicates that adding a 6-month visit to the routine immunization schedule is an effective, safe, and feasible way to lower under-5 mortality rates in areas where VAD is a public health problem. In settings where VAS is currently first given at 9 months, advancing the first VAS dose to 6 months should be considered. Delivery of high-dose VAS at 6 months and during the remainder of the preschool age period (such as through CHDs) should be maintained until a population has reached stable and adequate dietary vitamin A intake and status. 17
Footnotes
Authors’ Note
R.K., J.N., C.N.D, J.B., and K.H.B. drafted the manuscript. M.J.H. and K.H.B. helped develop the hepatic vitamin A modeling tool. R.K. and K.H.B. conducted the mortality calculations. S.K.B. helped develop the methodological approach. All authors have read and approved the final manuscript and are responsible for the final content. Department of Foreign Affairs, Trade and Development (DFATD) had no role in the design, analysis, or writing of this article. Findings from this article were presented as an oral presentation at the 20th International Conference of Nutrition, Granada (Spain), 2013. Abstract available at Ann Nutr Metab 2013;63 (S1):83.
Acknowledgments
The authors thank David Brown, Tracey Goodman, Lisa Rogers for their comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants made available from the Department of Foreign Affairs, Trade and Development (DFATD), Canada to UNICEF (project number M013618-003) and HKI (project number M013618-001).
