Abstract
Background:
The biological healing of the bone tunnel is crucial for success in anterior cruciate ligament reconstruction (ACLR). Nonetheless, conventional imaging cannot assess the key microarchitectural properties of the healing bone, resulting in a gap in our understanding of early recovery.
Purpose/Hypothesis:
This study aimed to identify the specific bone structure and density measurements most closely associated with successful recovery during the first 6 months. It was hypothesized that the microarchitectural properties of the bone tunnel, quantified by high-resolution peripheral quantitative computed tomography (HR-pQCT), would be directly correlated with functional and patient-reported outcomes in the early postoperative phase.
Study Design:
Cohort study; Level of evidence, 2.
Methods:
In a prospective cohort study, 50 patients undergoing primary hamstring ACLR were assessed at 4 and 6 months postoperatively. HR-pQCT was used to quantify bone tunnel parameters—including bone volume/total volume (BV/TV), trabecular thickness (Tb.Th), trabecular number (Tb.N), trabecular separation (Tb.Sp), and tunnel enlargement ratio. The isokinetic muscle strength test, single-leg hop test, ultrasound muscle thickness assessment, and patient-reported outcomes were assessed for clinical performance. The Spearman correlation was used to examine the relationship between knee joint clinical performance and bone tunnel conditions. Receiver operating characteristic (ROC) analysis was performed to identify predictive thresholds for good functional recovery.
Results:
HR-pQCT data revealed that at 4 months, femoral BV/TV (r = 0.361) and Tb.Th (r = 0.333) were positively correlated with knee extension strength, while the tibial tunnel enlargement ratio was negatively correlated (r = −0.380). Tibial Tb.N (r = 0.339) correlated positively with the International Knee Documentation Committee score, while tibial Tb.Sp (r = −0.367) correlated negatively. Femoral BV/TV at 4 months emerged as a significant predictor of early functional recovery (area under the ROC curve = 0.680; P = .027), with an optimal cutoff value of 0.2607.
Conclusion:
The microarchitectural quality of the bone tunnel, as assessed by HR-pQCT, is modestly correlated with the early functional recovery after ACLR, particularly at the 4-month postoperative time period.
Keywords
Anterior cruciate ligament (ACL) rupture is a common and serious sports injury. There are over 200,000 cases each year in the United States. 37 This injury causes ongoing knee instability, raises the risk of more damage to the knee, and leads to lower activity and quality of life.16,18 To restore knee movement and help people return to sports, ACL reconstruction (ACLR) is the standard surgery. Over 100,000 of these procedures are performed each year. 31 Even with improved surgery and recovery methods, many patients do not have good results. 28 Graft failure rates remain high,24,28,48 ranging from 3% to 14%. This is especially true for young, active people returning to sports. 48 A key reason for long-term success is the biological process of graft healing inside the bone tunnels. This is called tunnel healing. 45 Unsatisfactory graft healing, characterized by poor tendon-to-bone integration, is a primary contributor to graft laxity, failure, and subsequent revision surgery.9,13
Clinical evidence favors bone-patellar tendon-bone (BPTB) grafts over soft tissue grafts, showing superior return to sports and lower failure rates.11,12,52 This advantage likely stems from the robust bone-to-bone healing interface, in contrast to the slower, less reliable healing of soft tissue grafts to the tunnel wall.35,41 Therefore, the quality of the bone tunnel is an important factor in graft healing.5,32,50 However, assessing this biological healing remains a challenge. Conventional imaging (radiograph, computed tomography, and magnetic resonance imaging) focuses on gross geometry, such as tunnel widening, which correlates poorly with patient outcomes.15,19,39 These modalities fail to capture the microscopic details, such as bone density and volume, essential for graft anchorage.21,23,42
High-resolution peripheral quantitative computed tomography (HR-pQCT) is a powerful solution. This modality can produce detailed imaging, with resolutions as low as 61 µm. It allows for the noninvasive measurement of bone mineral density and detailed bone structure with high accuracy.4,30 Its application in orthopaedics is growing, proving valuable in assessing fracture healing and osteoporosis, yet its potential for evaluating tunnel healing after ACLR remains largely unexplored.8,42
Therefore, this study used HR-pQCT to examine how bone tunnels changed in structure during the crucial early healing phase after ACLR. We aimed to identify the specific bone structure and density measurements most closely associated with successful recovery during the first 6 months. We hypothesized that HR-pQCT–derived microarchitectural parameters of the femoral and tibial bone tunnels would be significantly associated with early functional performance and patient-reported outcomes within the first 6 months after ACLR.
Methods
Design and Setting
This study was conducted as a prospective observational cohort study in accordance with the principles of the Declaration of Helsinki. Informed consent was obtained from the patients after the surgery. The study was approved by the Joint CUHK NTEC-CREC (2021.332). The study was conducted at Prince of Wales Hospital. From October 2022 to October 2024, all participants underwent a standardized assessment protocol at 4 and 6 months postoperatively, which included HR-pQCT to evaluate bone tunnel characteristics, isokinetic dynamometry for muscle strength, ultrasonography for muscle thickness, functional performance tests, and patient-reported outcome measures—including the Lysholm score, International Knee Documentation Committee (IKDC), and International Physical Activity Questionnaire (IPAQ).
Inclusion and Exclusion Criteria
The inclusion criteria were (1) age between 18 and 50 years; and (2) primary ACLR (single bundle, 4-stranded hamstring autograft, tibial tunnel interference screw, fixed loop femoral endo-button).
The exclusion criteria were (1) concomitant multiple ligament injuries requiring additional surgical procedures; (2) preoperative radiographic signs of arthritis; (3) revision ACLR; (4) medical comorbidities; (5) long-term steroid intake; and (6) noncompliance with our rehabilitation protocol (Appendix 1, available in the online version of this article).
Data Collection
Eligible patients presenting to the outpatient clinic were screened consecutively for enrollment. All patients meeting the inclusion criteria were approached for participation 4 and 6 months after ACLR. During the patient's postoperative follow-up visit, anthropometric and descriptive data were collected via an assessment form and uploaded to the clinical management system by collaborating physicians and research assistants. HR-pQCT was conducted using XtremeCT II (SCANCO Medical AG). Knee strength was assessed using the dynamometer (Cybex NORM; Cybex International, Inc). Muscle thickness was assessed using the Aixplorer ultrasound system (SuperSonic Imagine). The single-leg hop test assessed clinical performance, and the patient self-reported condition using the Lysholm knee scoring system, IKDC, and IPAQ. The evaluators were 2 experienced, trained assessors under standardized conditions: 1 licensed physical therapist with expertise in postoperative rehabilitation (T.Y.Z.) and 1 board-certified orthopaedic surgeon (M.D.C.) specializing in knee surgery.
Radiological Assessment
Patients underwent HR-pQCT imaging at 4 and 6 months postoperatively. The Image Sequence data were analyzed using the micro-computed tomography (CT) 40 built-in software by 2 assessors (T.Y.Z. and M.D.C.). The analysis protocol begins with reorienting the CT dataset by defining the planar coordinates of the bone tunnel, ensuring the imaging plane is perpendicular to its long axis, and yielding true cross-sectional views. 7 Regions of interest (ROIs) encompassing the dense bone shell surrounding the tunnel are then manually delineated at 20-slice intervals according to the established criteria. 49 Subsequently, the software's overlay function is employed to intelligently interpolate these manual selections, automatically propagating the ROIs across the intervening slices. After this segmentation, a comprehensive quantitative analysis is conducted to generate a bone quality report detailing key morphometric parameters—including bone volume fraction (BV/TV), trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), and trabecular number (Tb.N) 47 (Figure 1).
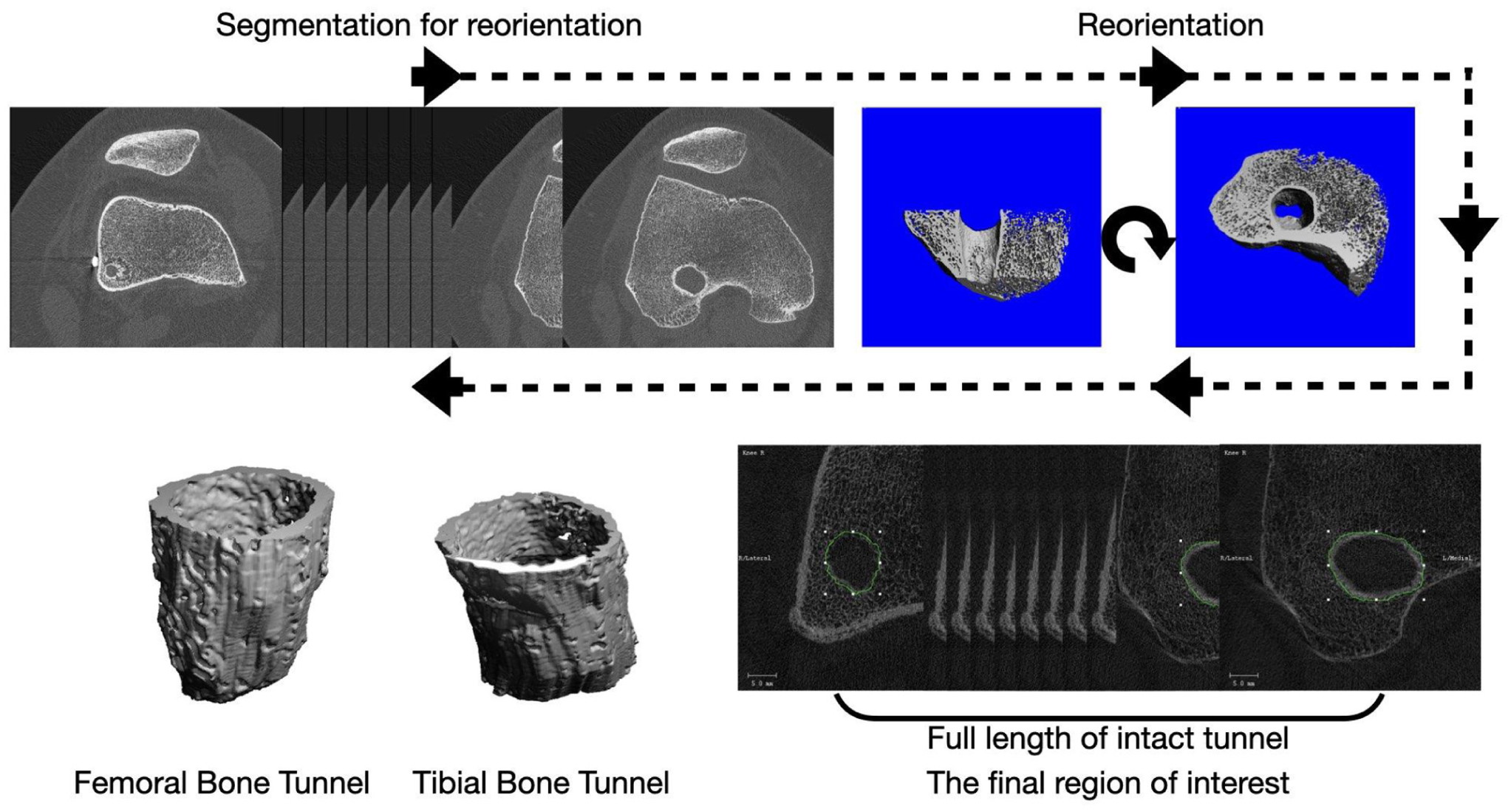
Method of HR-pQCT-based bone tunnel evaluation. HR-pQCT, high-resolution peripheral quantitative computed tomography.
The maximum bone tunnel cross-sectional area was quantified by first defining the coordinate plane of the largest cross-section following 3-dimensional (3D) reconstruction. The corresponding 2D CT slice was then identified, and the area was manually traced. Tunnel enlargement was expressed as a ratio, calculated by dividing the maximum measured cross-sectional area by the cross-sectional area of the original surgical tunnel.
Clinical Assessments
Muscle Strength
The dynamometer (Cybex NORM) was performed at 4 and 6 months after ACLR by 1 physical therapist (T.Y.Z.) and 1 board-certified orthopaedic surgeon (M.D.C). The participant was positioned in the machine as per the guidelines. The machine's axis was lined up with the knee joint. The participant did a warm-up of 10 practice repetitions. They then rested for 30 seconds. Next, the participant performed 5 maximum effort repetitions for both leg extension and flexion. The tester gave strong oral encouragement. After testing 1 leg, the participant rested for 30 seconds. The other leg was then tested the same way. Testing was performed at an angular velocity of 60° per second (60 deg/s). Peak torque (Nm) was recorded as the primary outcome measure for both knee extension and flexion. To account for individual differences in body size, peak torque values were normalized to body weight (Nm/kg).
Muscle Thickness
As part of the standardized care at our center, muscle thickness measurement was performed at 4 and 6 months after ACLR by a single trained investigator (T.Y.Z.). Muscle thickness of the vastus medialis (VM), vastus lateralis (VL), and rectus femoris (RF) was bilaterally assessed using an Aixplorer ultrasound system (SuperSonic Imagine), with participants in a supine position. Measurement sites were defined using anatomic landmarks. The RF site was halfway between the anterior inferior iliac spine and the patella. The VM site was one-fifth of this distance. The VL site was one-third of this distance. After marking the sites, a transducer took images with minimal pressure. Muscle thickness was calculated from the mean of 3 measurements at each site.
Single-Leg Hop Test
The single-leg hop test measured lower limb power and function. Participants hopped as far as they could on 1 leg. They had to land and keep their balance. The distance was measured from the point of push-off to the point of landing. Each leg was tested 3 times. The order of testing was random. The mean distance of the 3 hops for each leg was used for analysis.
Patient-Reported Outcome Measurement
Subjective functional outcomes were assessed using 3 validated patient-reported outcome measures: the Lysholm Knee Scoring Scale, the IKDC Subjective Knee Form, and the IPAQ. The Lysholm scale ranges from 0 to 10, with higher scores indicating better knee function. 26 The IKDC form also ranges from 0 to 100, with a score of 100 indicating no limitations. 22 The IPAQ measured general physical activity over the past week. The validated Chinese version was used. 2
Statistical Analysis
Sample size was calculated using G*Power Version 3.1.9.7 (Franz Faul, Universität Kiel) based on our pilot result on an effect size of 0.91 in terms of femur tunnel bone shell, a sample size of 48 was found to achieve 88.3% power to detect the correlation between the bone density and clinical outcomes, with a significance level (alpha) of .05 and using a 2-tailed exact-test. 14 Hence, 60 participants (allowing for a 20% dropout rate) were recruited. Complete case analysis (Listwise Deletion) was used for the missing data. Only participants with no missing data in the variables of interest were analyzed. 29
The Kolmogorov-Smirnov test was used to check whether the data were normally distributed. Depending on the result, either the Pearson or Spearman correlation was used to check the strength of the association between the bone tunnel parameters (BV/TV, Tb.Th, Tb.Sp, Tb.N, and Enlargement ratio) and the clinical outcomes (quadriceps and hamstrings strength at 60 deg/s, VM, RF, VL muscle thickness, single leg hop test, Lysholm, IKDC, and IPAQ). The strength of the correlation was interpreted using guidelines, where coefficients between 0 and ±0.19 were considered none to slight, ±0.20 to ±0.39 low, ±0.40 to ±0.69 moderate, ±0.70 to ±0.89 high, and ±0.90 to ±1 very high. 43
Receiver operating characteristic (ROC) curve analysis was employed to determine optimal bone tunnel parameter thresholds for discriminating between good and poor clinical performance, as defined by limb symmetry indices (LSI) at 2 postoperative time points. For the 4-month assessment, good performance was classified 1 as knee extension strength LSI of ≥0.7, while at 6 months, it required both knee extension strength LSI of ≥0.8 and single-leg hop test LSI of ≥0.8.27,33 The Youden index method was used to identify cutoff values that maximize diagnostic accuracy. The area under the ROC curve (AUC) was computed to evaluate the overall discriminative ability of the parameters. Following Nahm's guidelines, the AUC predictive accuracy was interpreted as follows: 0.9-1.0 indicated excellent accuracy, 0.8-0.9 very good, 0.7-0.8 good, 0.6-0.7 sufficient, and 0.5-0.6 bad. 34 An AUC of <0.5 indicated performance worse than random chance.
A 2-sided P < .05 was considered statistically significant. All analyses were conducted using SPSS Version 29.0 (IBM).
Bias and Efforts to Address It
To minimize bias, this study implemented several methodological controls. The included patient was undergoing isolated hamstring ACLR without significant concomitant injuries. Performance and detection bias were addressed through standardized surgical technique performed by a single team and consistent measurement protocols using trained personnel. Intra- and interobserver reliability of imaging metrics were assessed via intraclass correlation coefficients (ICCs). All ICCs exceeded 0.70, indicating excellent agreement (Table 1). Selection of validated patient-reported outcome measures and objective functional tests further reduced reporting bias.
Intra- and Interobserver Reliability Test Results a
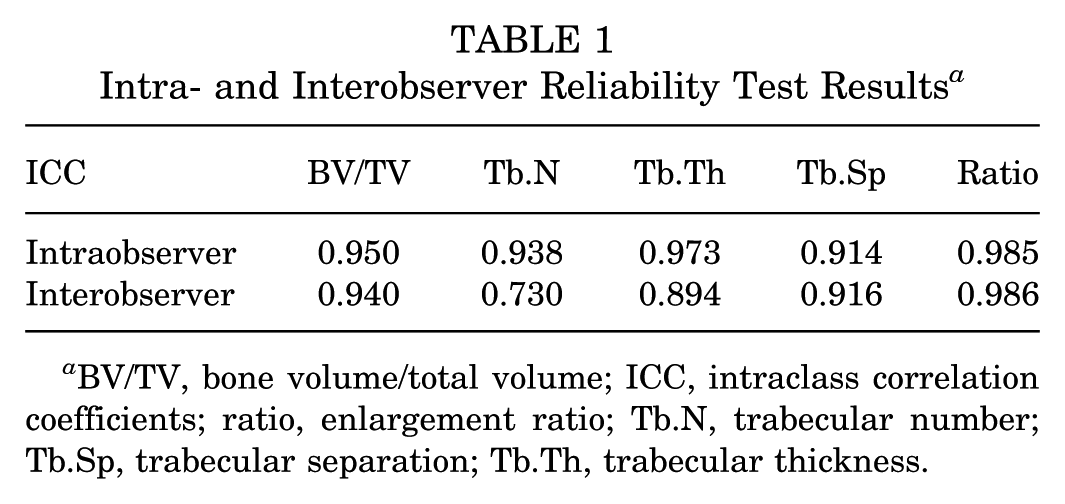
BV/TV, bone volume/total volume; ICC, intraclass correlation coefficients; ratio, enlargement ratio; Tb.N, trabecular number; Tb.Sp, trabecular separation; Tb.Th, trabecular thickness.
Results
A total of 60 patients met the inclusion criteria. Of these, 50 patients who underwent all evaluations at 6 months postoperatively were ultimately enrolled in this study (Figure 2).
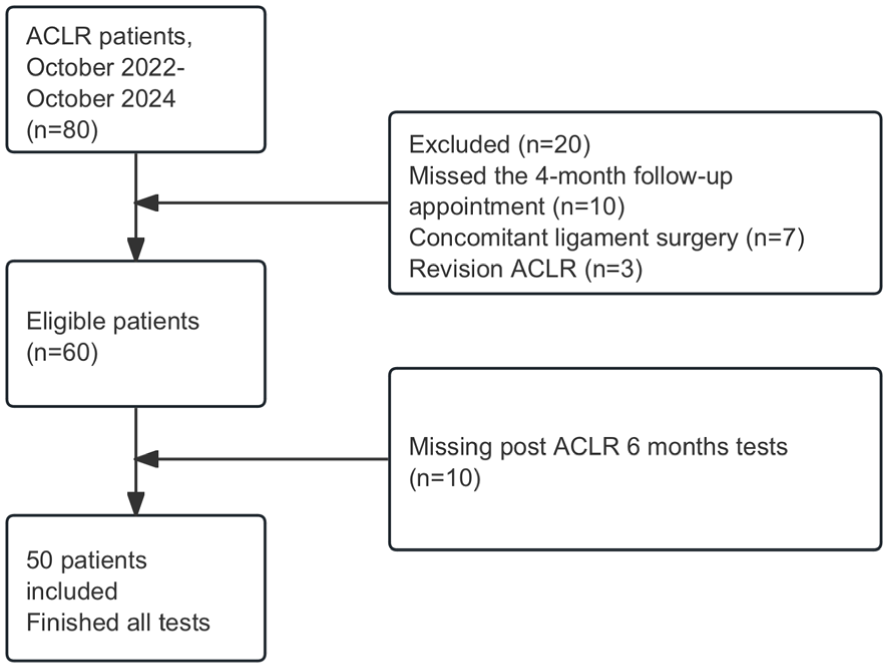
Study flowchart. ACLR, anterior cruciate ligament reconstruction.
Clinical Characteristics and Bone Tunnel Parameter Distributions
Between October 2022 and October 2024, we enrolled 60 patients who met the study inclusion criteria. Of these, 50 patients finished the 6-month follow-up and formed the final study group (Figure 2). The group consisted of amateur athletes and sports enthusiasts. The mean age at the time of surgery was 25.60 ± 7.04 years (56% men) (Table 2). Analysis also yielded results regarding the bony microstructure of the tibial and femoral tunnels at 4 and 6 months postoperatively (Table 3). Representative HR-pQCT images and 3D reconstructed models, demonstrating normal and poor healing after ACLR (Appendix 2, available online).
Clinical Characteristics a
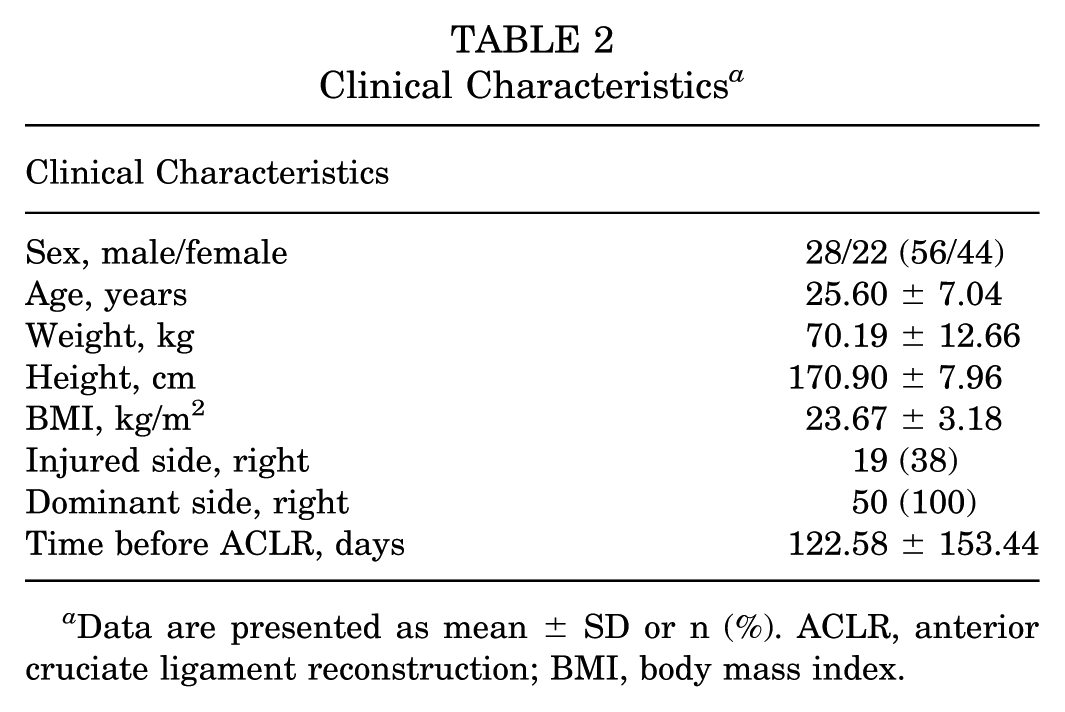
Data are presented as mean ± SD or n (%). ACLR, anterior cruciate ligament reconstruction; BMI, body mass index.
Descriptive Statistics of Bone Microarchitecture After ACLR a
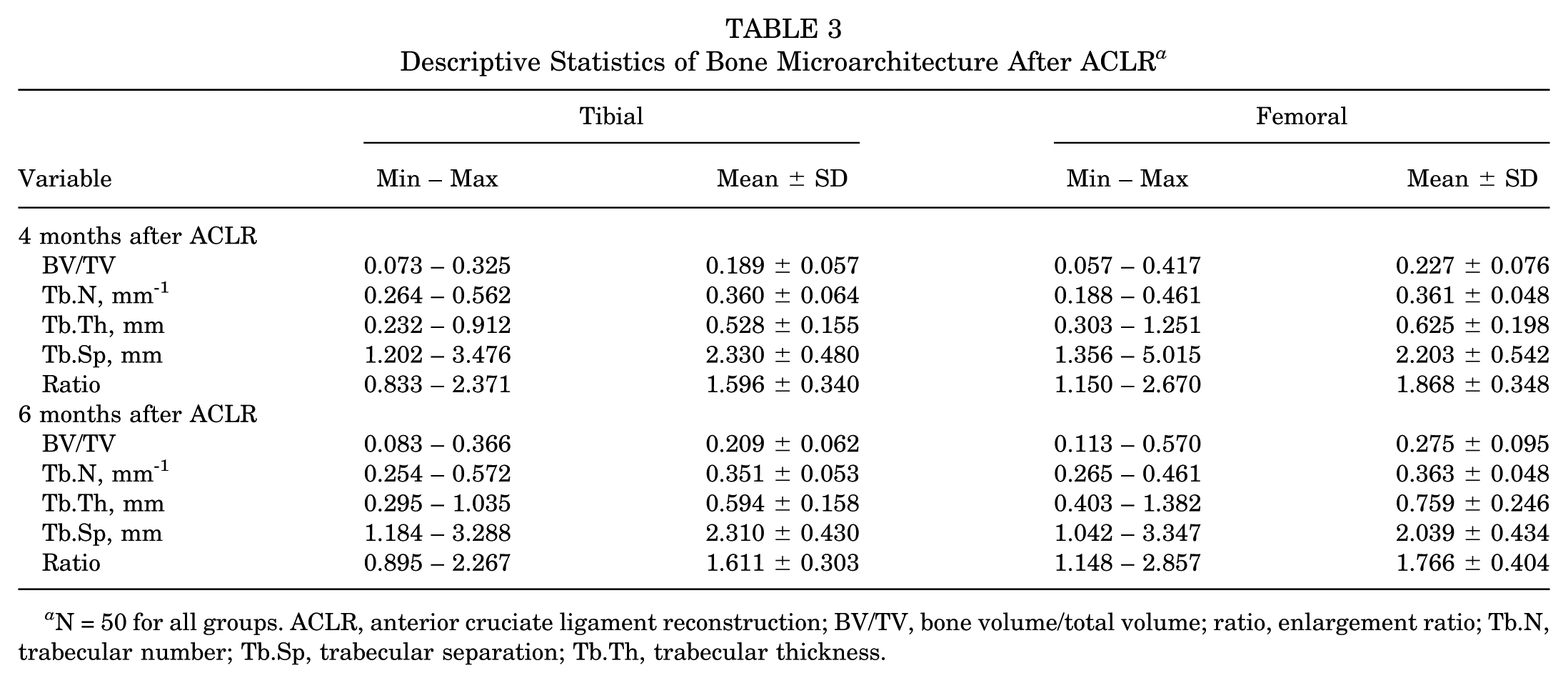
N = 50 for all groups. ACLR, anterior cruciate ligament reconstruction; BV/TV, bone volume/total volume; ratio, enlargement ratio; Tb.N, trabecular number; Tb.Sp, trabecular separation; Tb.Th, trabecular thickness.
Correlation Coefficients
Statistical analyses at 4 and 6 months after surgery showed a clear pattern. Measurements from bone scans were consistently weakly correlated with patient recovery (Figure 3). At 4 months, knee extension strength was positively correlated with tibial BV/TV (r = 0.343; P = .015), tibial Tb.Th (r = 0.298; P = .036), and femoral BV/TV (r = 0.361; P = .010) and Tb.Th (r = 0.333; P = .018) (Figure 3A). Strength was negatively correlated with the tibial tunnel enlargement ratio (r = −0.380; P = .007) and femoral Tb.Sp (r = −0.285; P = .045). The IKDC score also showed a relationship with bone quality. It showed positive correlations with tibial Tb.N (r = 0.339; P = .016) and femoral BV/TV (r = 0.301; P = .034). It had negative correlations with tibial Tb.Sp (r = −0.367; P = .009) and femoral Tb.Sp (r = −0.300; P = .034). These links continued at 6 months (Figure 3B). Knee extension strength correlated with tibial Tb.Th (r = 0.299; P = .035) and femoral BV/TV (r = 0.279; P = .050). It was negatively correlated with the tibial enlargement ratio (r = −0.296; P = .037). Knee flexion strength was linked to femoral BV/TV (r = 0.308; P = 0.030) and Tb.Sp (r = −0.297; P = .036). Muscle measurements also showed connections. VM thickness was negatively correlated with the tibial enlargement ratio (r = −0.372; P = .008). VL thickness correlated with femoral Tb.N (r = .308; P = .029) and Tb.Sp (r = −0.325; P = .021). Functional performance, from the single-leg hop test, correlated with tibial BV/TV (r = 0.281; P = .048). The IKDC score remained associated with femoral BV/TV (r = 0.280; P = .049). All the correlation coefficients were considered weak. 43
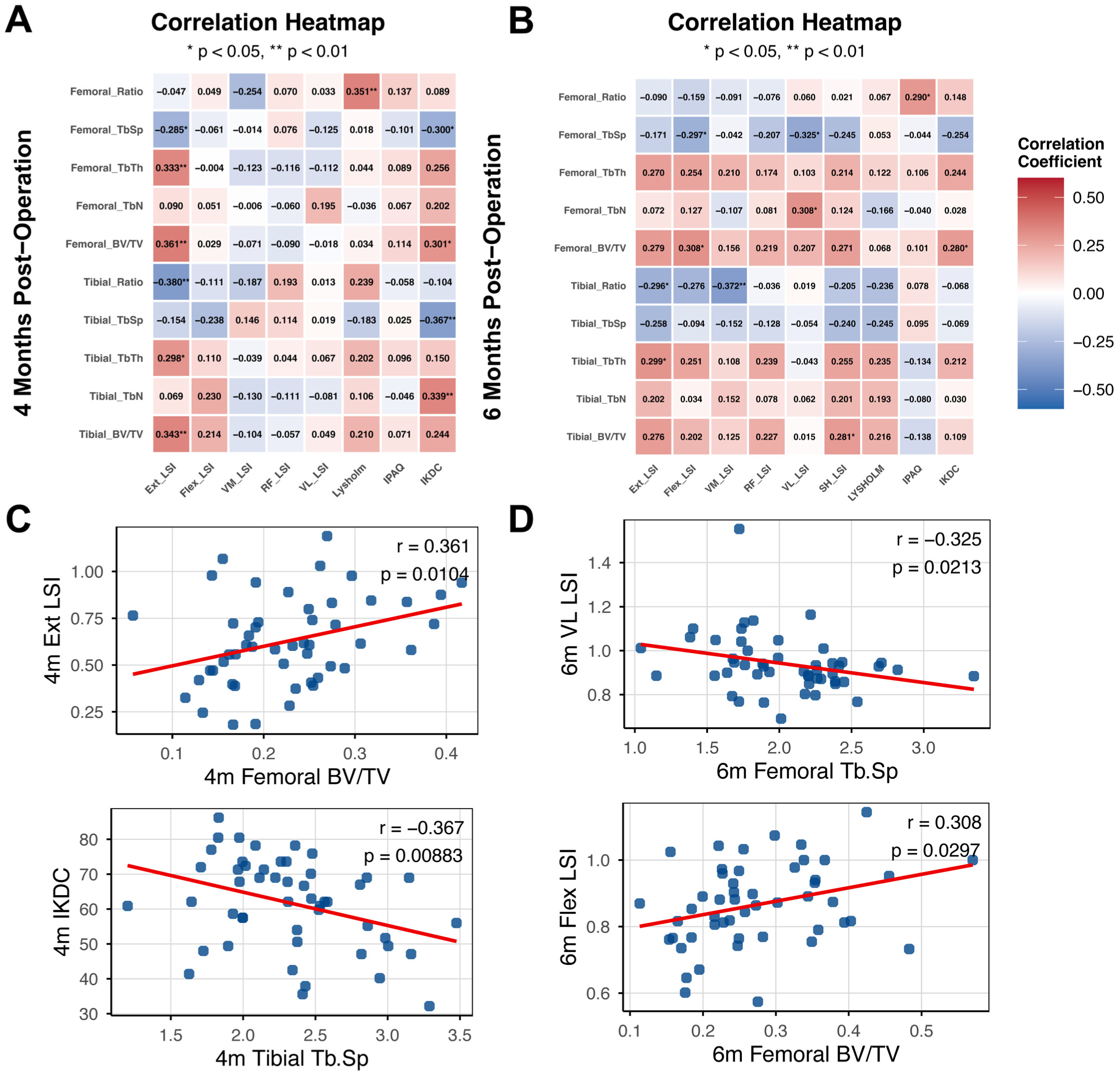
The correlation heatmap for 4 and 6 months after ACLR. (A) Correlations between bone tunnel parameters and clinical outcomes at 4 months after ACLR. (B) Correlations between bone tunnel parameters and clinical outcomes at 6 months after ACLR. (C) Representative scatter plots for correlations at 4 months after ACLR. (D) Representative scatter plots for correlations at 6 months after ACLR. *Correlation is significant at the .05 level (2-tailed). **Correlation is significant at the .01 level (2-tailed). BV/TV, bone volume/total volume; EXT, extension muscle strength; FLEX, flexion muscle strength; IKDC, International Knee Documentation Committee; IPAQ, International Physical Activity Questionnaire; LSI, limb symmetry index; LYSHOLM, Lysholm Knee Scoring Scale; ratio, enlargement ratio; RF, rectus femoris muscle thickness; Tb.N, trabecular number; Tb.Sp, trabecular separation; Tb.Th, trabecular thickness; VL, vastus lateralis muscle thickness; VM, vastus medialis muscle thickness.
ROC Curve Analysis
ROC analyses tested whether bone tunnel scans could predict patient recovery after ACL surgery. The results are shown in Figure 4. At the 4-month check, femoral bone tunnel density was the most predictive factor (Figure 4A). It had an AUC of 0.680 (P = .027) (Figure 4B), a Gini index of 0.360, and a cutoff value of 0.2607 (sensitivity, 50%; specificity, 86.7%). Femoral Tb.Th also showed a predictive value (AUC = 0.644; P = .091). For tibial parameters at 4 months, the tunnel volume fraction had an AUC of 0.635 (P = .109), with a cutoff of 0.202 (sensitivity, 65%; specificity, 73.3%). Trabecular thickness had an AUC of 0.622 (P = .153), with a cutoff of 0.554 mm (sensitivity, 60%; specificity, 73.3%). At the 6-month check, femoral Tb.N was the strongest predictor (AUC = 0.645; P = .115), with a cutoff of 0.382 mm (sensitivity, 52.9%; specificity, 81.8%) (Figure 4A). Femoral tunnel BV/TV was a weaker predictor at this time (AUC = 0.585; P = .311). Tibial parameters at 6 months were generally less accurate than at 4 months. Tb.N had an AUC of 0.630, with a cutoff of 0.330 mm (sensitivity, 82.4%; specificity, 45.5%) (Figure 4B). BV/TV had an AUC of 0.575, with a cutoff of 0.241 (sensitivity, 47.1%; specificity, 75.8%). According to the classification proposed by Hosmer et al, 20 all observed AUC values fell within the poor discriminative range (<0.70), indicating that although certain femoral tunnel parameters showed statistically or clinically suggestive associations with recovery, their overall predictive accuracy was limited.
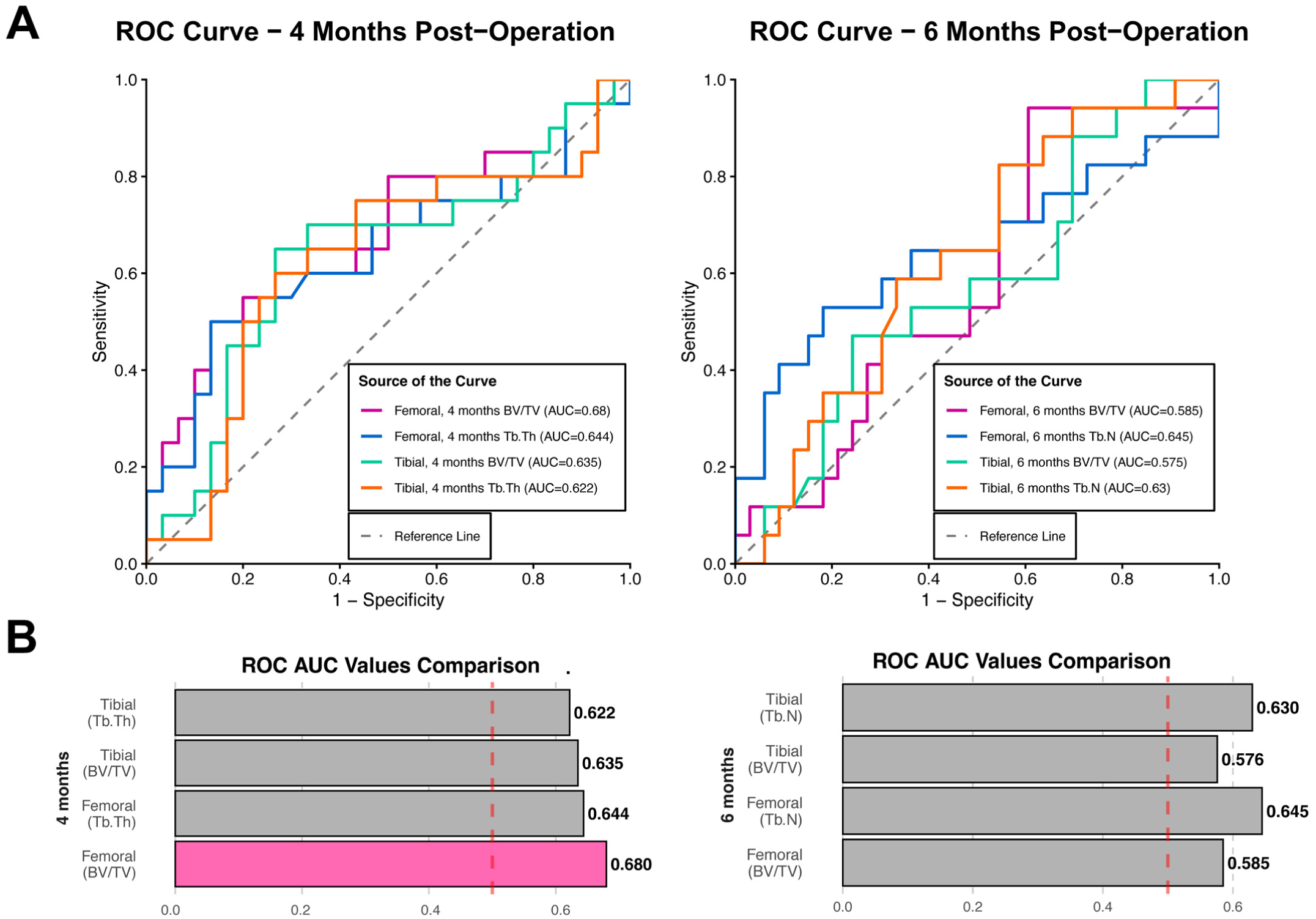
The results of the ROC test. (A) The ROC curve at 4 and 6 months after ACLR. (B) The bar chart of the AUC at 4 and 6 months after ACLR. Null hypothesis: true area = 0.5. ACLR, anterior cruciate ligament reconstruction; AUC, area under the ROC curve; BV/TV, bone volume/total volume; ROC, receiver operating characteristic; Tb.Th, trabecular thickness.
Discussion
The main finding is a moderate correlation between specific bone structure measurements and patient recovery at 4 and 6 months after surgery.
Our data suggest that the microarchitectural properties of the bone tunnel may play a role in successful early functional recovery. By quantifying parameters—such as BV/TV, Tb.Th, and Tb.N—we observed a direct structure-function relationship that is difficult to quantify with radiograph and conventional CT.
The positive correlation between knee extension strength LSI and femoral BV/TV indicates a potential link between the femoral bone volume and quadriceps function. A higher bone volume means a denser, stronger anchor for the new ligament. 6 This strong base is needed to handle the forces of exercise and can allow patients to build muscle effectively. 17 This relationship is further evidenced by the correlation between the IKDC score and femoral BV/TV, demonstrating that the patient's subjective sense of knee stability and function is rooted in this underlying biological reality. 51
The positive correlation between the IKDC score and tibial Tb.N and the negative correlations of both IKDC and strength metrics with Tb.Sp in both the tibia and femur suggests that the trabecular network is associated with better outcomes. A healing tunnel characterized by numerous, closely spaced trabeculae (high Tb.N and low Tb.Sp) creates a complex, 3D lattice that facilitates superior biological fixation and mechanical interlocking of the graft. Conversely, a sparse, porous structure (low Tb.N and high Tb.Sp) represents a weak link, compromising the graft's anchorage and leading to inferior clinical outcomes. 36 This is supported by preclinical research indicating that a fine, well-connected trabecular structure is essential for effective osteointegration in bone-to-soft tissue healing. 46
The ROC analysis identified femoral BV/TV at 4 months as the strongest predictor (AUC = 0.680; P = .027) for early functional recovery, with a specific cutoff value of 0.2607 (sensitivity, 50%; specificity, 86.7%). While the AUC value indicates low to fair predictive accuracy, it may indicate that insufficient mineralized bone within the femoral tunnel at this stage may be a factor in delayed recovery. The femoral tunnel is subject to significant stress, making it a sensitive indicator of healing. 51
By 6 months, the predictive value of femoral BV/TV decreased, while femoral Tb.N showed a stronger prediction value (AUC = 0.645). Although the accuracy level is considered low, 34 this shift still may indicate a change in the healing process. Our findings may suggest that a layer of bone is created, which subsequently becomes more organized into a stronger, more effective structure.
Our focus on microstructure helps explain the conflicting results from past studies.25,44 These previous studies only measured tunnel width on radiographs. Many long-term studies have found that tunnel widening does not correlate with patient knee scores or stability. 40 This underscores a key point highlighted by our use of HR-pQCT, that a tunnel can maintain its diameter yet have poor biological quality if the surrounding bone is porous and structurally weak. Conventional imaging can measure the void, but it cannot assess the quality of the bone that defines the void's walls. 10 Our research moves beyond this limitation by quantifying the material properties of the bone itself, revealing a structure-function relationship that simpler measurements may miss.
The role of bone microarchitecture highlighted in our study may help explain the biological mechanisms underlying challenges in graft integration. Historically, BPTB grafts have demonstrated superior clinical outcomes—including higher return to sports rates and lower revision risks compared with soft tissue grafts.11,12,52
This advantage may be attributed to the bone-to-bone healing interface, which mimics fracture healing—a process that is inherently robust and rapid.3,38 In contrast, soft tissue grafts (eg, hamstring or quadriceps tendon) lack this bony interface and must integrate directly with the bone tunnel wall.35,41
Therefore, our findings are particularly relevant for the large number of patients receiving soft tissue grafts, as they offer a way to quantify the biological quality of the tunnel, which serves as the sole foundation for graft stability in the absence of a bone block.
This study has limitations—including its single-center design and a 6-month follow-up period, which, although capturing the early healing phase, is insufficient to correlate these microarchitectural findings with long-term outcomes, such as graft failure or posttraumatic osteoarthritis. Additionally, while we identified statistically significant modest correlations, the AUC values for the HR-pQCT parameters ranged from 0.6 to 0.7. This range is generally considered to have low predictive accuracy, suggesting that HR-pQCT metrics alone may not be sufficient for robust individual prognostication. However, these data remain valuable for guiding future research by establishing a baseline for the role of bone microarchitecture in recovery. Additionally, analyses were not adjusted for sex-related differences in bone microarchitecture, which may influence healing responses. Finally, the lack of long-term follow-up precluded correlation of microarchitectural findings with clinically meaningful outcomes. In the future, research should focus on longitudinal studies lasting 2 to 5 years that integrate HR-pQCT data with advanced functional biomechanics, patient-reported scales, and additional bone-healing–related outcomes, such as KT-1000 measurements. Furthermore, exploring the impact of surgical variables (eg, drilling technique and graft type) and patient-specific factors (eg, biology and nutrition) on these microarchitectural parameters will be essential for building a comprehensive model of ACLR recovery.
Conclusion
Bone tunnel microarchitectural parameters assessed by HR-pQCT were modestly associated with early functional recovery after ACLR, particularly at 4 months postoperatively. These findings suggest that HR-pQCT may provide complementary information to conventional assessments during early postoperative recovery.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465261426355 – Supplemental material for Bone Tunnel Microarchitecture and Early Functional Recovery After Anterior Cruciate Ligament Reconstruction: A Prospective HR-pQCT Cohort Study
Supplemental material, sj-pdf-1-ajs-10.1177_03635465261426355 for Bone Tunnel Microarchitecture and Early Functional Recovery After Anterior Cruciate Ligament Reconstruction: A Prospective HR-pQCT Cohort Study by Michael Tim-Yun Ong, Tian-Yi Zang, Mingde Cao, Pauline Po-Yee Lui, Bruma Sai-Chuen Fu, Dennis Cham-Kit WONG, Rex Wang-Fung Mak, Jonathan Patrick Ng and Patrick Shu-Hang Yung in The American Journal of Sports Medicine
Supplemental Material
sj-pdf-2-ajs-10.1177_03635465261426355 – Supplemental material for Bone Tunnel Microarchitecture and Early Functional Recovery After Anterior Cruciate Ligament Reconstruction: A Prospective HR-pQCT Cohort Study
Supplemental material, sj-pdf-2-ajs-10.1177_03635465261426355 for Bone Tunnel Microarchitecture and Early Functional Recovery After Anterior Cruciate Ligament Reconstruction: A Prospective HR-pQCT Cohort Study by Michael Tim-Yun Ong, Tian-Yi Zang, Mingde Cao, Pauline Po-Yee Lui, Bruma Sai-Chuen Fu, Dennis Cham-Kit WONG, Rex Wang-Fung Mak, Jonathan Patrick Ng and Patrick Shu-Hang Yung in The American Journal of Sports Medicine
Footnotes
Submitted October 20, 2025; accepted January 11, 2026.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Joint Chinese University of Hong Kong, New Territories East Cluster Clinical Research Ethics Committee (Ref no. CREC-2021.332).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
