Abstract
Background:
The knee is the most commonly afflicted joint in osteoarthritis (OA). Injection of intra-articular of hyaluronic acid (IAHA) is a frequently used therapy for the management of knee OA with varying product characteristics.
Purpose:
To describe and characterize the mechanism of action (MoA) of IAHA products concerning nociception, chondroprotection, and anti-inflammatory properties via a targeted literature review.
Study Design:
Systematic review; Level of evidence, 2.
Methods:
We followed the standard methodologies for conducting and reporting targeted reviews as recommended by the Cochrane Handbook for Systematic Reviews of Interventions, adapted for conducting a targeted literature review. Relevant studies were identified by searching the Embase database using predefined search strategies via the Ovid platform. The results of the review were reported according to the PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-analyses).
Results:
A total of 182 studies were included in this targeted literature review. Of these, 107 reported chondroprotective action, 59 anti-inflammatory activity, 18 analgesic properties, 30 proteoglycan or glycosaminoglycan synthesis, 8 subchondral bone effects, 2 mechanical effects, and 1 other effects of IAHA. These MoAs were studied through diverse types of studies: in vitro biochemistry, animal physiological studies, or human physiological and clinical studies. The chondroprotective effect was the most studied MoA and showed an increase in anabolic biomarkers, such as collagen types II, IX, and XI, and a reduction in catabolic biomarkers, such as matrix metalloproteinases, which play a primary role in the downstream signaling pathways in OA and cartilage degradation in the synovial fluid. IAHA was widely reported by studies to reduce soluble inflammatory mediators, such as interleukins 1β and 6 and tumor necrosis factor α, thereby decreasing the production of degradative enzymes (eg, matrix metalloproteinases, aggrecanases). IAHA was also reported to enhance the synthesis of intrinsic proteoglycan (eg, aggrecan) and glycosaminoglycans, thus delaying the progression of OA. IAHA also reported improvement in the mechanical function of the knee by increasing the viscosity of the synovial fluid, reducing the coefficient of friction, and improving its lubrication. Overall, a significant decrease in knee pain was observed after IAHA treatments.
Conclusion:
Preclinical and clinical studies established evidence for varied MoAs by which IAHA preparations may produce a desired effect in patients with knee OA.
Keywords
Osteoarthritis (OA) is characterized by chronic pain, stiffness, and reduced mobility in weightbearing joints, leading to increased disability in the adult population, and it is especially prevalent in elderly and obese populations. 18 The knee is the most frequently affected joint, with an estimated prevalence of 654 million people affected worldwide in 2020.18,50 Knee OA (KOA) leads to a gradual deterioration of the entire joint structure, which includes the articular cartilage (AC), subchondral bone, synovial capsule, and membrane, as well as the surrounding periarticular tissues comprising connective and muscular elements.55,73 Furthermore, OA extends its influence to the soft tissues within the knee, including ligaments, tendons, and the menisci. This has led to a significant adverse effect on the quality of life of affected individuals and an increased economic burden. 44
Treatment options for KOA are multifaceted, encompassing nonpharmacological and pharmacological approaches. Nonpharmacological strategies include patient education and lifestyle adjustments such as activity modification, exercise programs involving “core to floor” muscle strengthening, weight management, bracing, ambulatory aids, and the use of cooling or heating applications. 42 Encouraging physical activity tailored to each patient's capabilities is essential for maintaining joint function and reducing pain. 15 Nonsteroidal anti-inflammatory drugs (topical or oral) are frequently prescribed pharmacological treatments. However, long-term use of systemic or oral nonsteroidal anti-inflammatory drugs can lead to serious adverse renal, gastrointestinal, and cardiovascular events in addition to proteoglycan catabolism.84,86 Intra-articular injections have been recommended in KOA for achieving maximum local benefits with limited side effects.9,32 Intra-articular steroid injections reduce pain and inflammation, although the relief provided is short-term (up to 4 weeks). 8 Yet, repeat use of intra-articular steroid injections may cause deleterious effects on AC, including a decrease in protein and matrix synthesis, mutation of cartilage cell shape, and prevention of new cartilage growth, as well as cartilage surface deterioration, such as edema, pitting, shredding, ulceration, and erosions, which may enhance cartilage destruction. 31
Intra-articular hyaluronic acid (IAHA) injections have been deemed safe for patients with symptomatic KOA. They offer longer-lasting benefits as compared with steroid injections but may pose a higher risk of nonserious and temporary local reactions as compared with saline injections.8,57,75 Initially regulated as medical devices owing to their assumed shock absorption properties, 87 IAHA products approved by the Food and Drug Administration in 1997 are now being reclassified as pharmaceutical drugs based on evidence of hyaluronic acid’s (HA’s) diverse mechanisms of action (MoAs) beyond shock absorption.5,70,74
IAHA exerts its effect by binding through different receptors, such as CD44, intercellular adhesion molecule (ICAM-1) receptors, or receptors for hyaluronan-mediated motility, which are present on synoviocytes or chondrocytes within the synovial joint5,6,30 and mediate therapeutic actions such as anti-inflammatory and analgesic effects, chondroprotection, proteoglycan synthesis, joint lubrication, shock absorption, and effects on subchondral bone.4,6 IAHA, characterized by its molecular weight (MW), source/origin, and structural properties based on cross-linking, demonstrates varying clinical effectiveness, with recent studies highlighting the differences among low MW (LMW), medium MW (MMW), and high MW (HMW).14,24,71,88 IAHA products are derived from either avian sources or biological fermentation, which has raised concerns regarding their safety. 4
The present literature review aims to describe the MoA of different IAHA products utilized in the treatment of KOA and to explore findings supporting a correlation between IAHA MW and their MoA. We hypothesize that a correlation exists between IAHA MW and its MoA in the treatment of KOA, with HMW IAHA products potentially demonstrating superior therapeutic effects as compared with lower MW counterparts.
Methods
This study was carried out in adherence to the Cochrane Handbook for Systematic Reviews of Interventions and is reported in accordance with the guidelines set by the PRISMA statement (Preferred Reporting Items for Systematic Reviews and Meta-analyses).33,59 Literature was searched using a predefined search strategy in Embase via Ovid from 1974 to March 6, 2023 (Appendix Table A1). The searched articles were reviewed for relevant titles and abstracts, followed by full-text screening. Articles reporting the MoA of IAHA in KOA were included. Editorials, letters, case reports, and non-English publications were excluded. Data from publications were extracted using a standardized template and included the IAHA product utilized, MW of HA, and MoA. Categories of MoAs were assigned during the data extraction stage and included chondroprotection, effects on proteoglycan/glycosaminoglycan (GAG) synthesis, anti-inflammatory effects, analgesic effects, effects on subchondral bone, mechanical effects, and other actions. Publications reporting multiple MoAs were included in multiple categories. Across all studies, the main results and conclusions for MoAs were synthesized, followed by preclinical or clinical reporting.
Results
Study Selection
The Embase search yielded 1442 articles, and an additional 84 were identified through manual searching. After removal of duplicates, 1488 articles underwent title and abstract screening. Among these, 242 underwent full-text screening, and ultimately 182 studies were included in this literature review. The PRISMA flow diagram is presented in Figure 1.
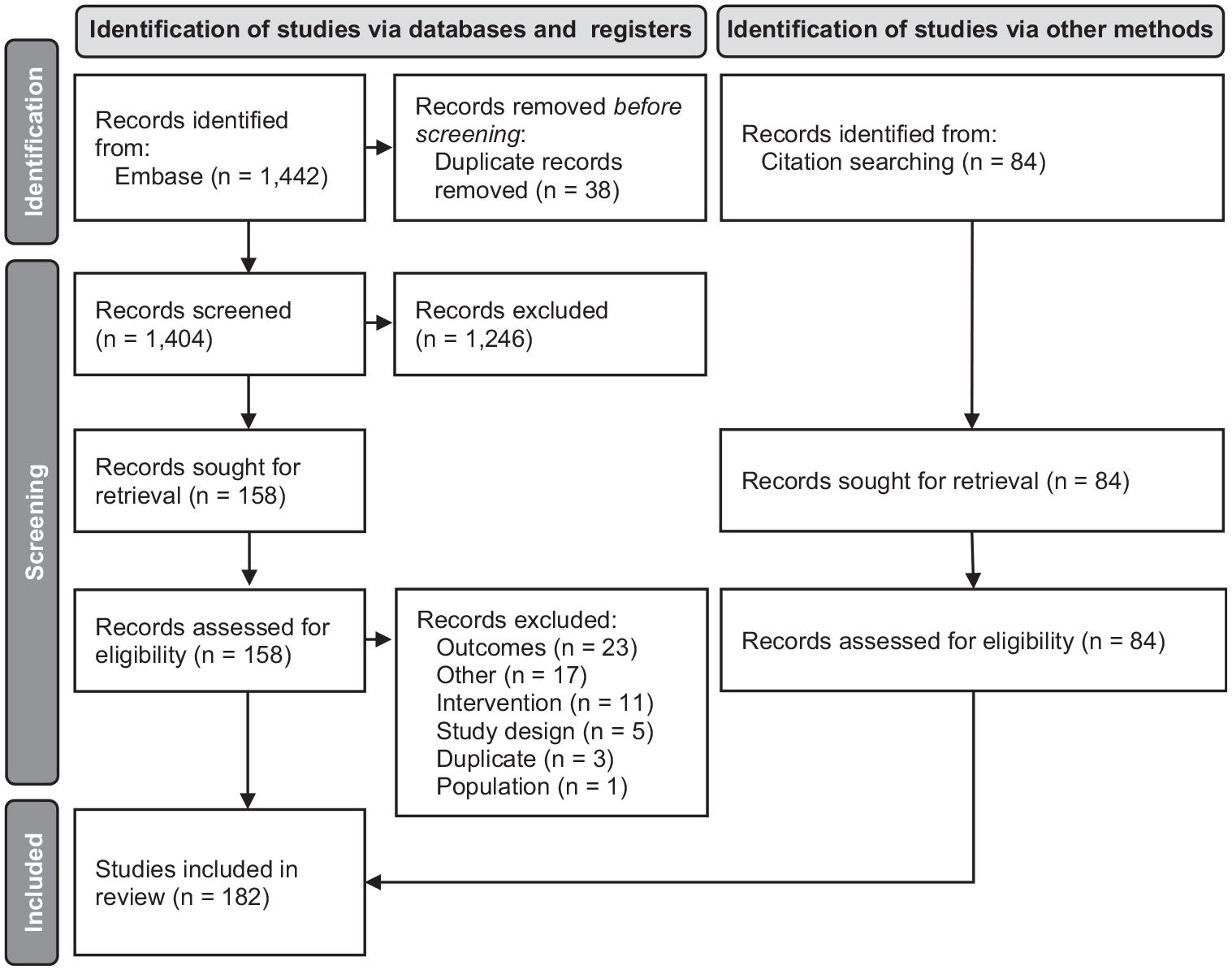
PRISMA flow diagram.
Study Characteristics
Among 182 studies, 55 utilized in vitro cell-based models for investigating the MoA of IAHA products; 51, animal-based cell models; 41, human models; 24, animal models; and 12, cells derived from human sources (Figure 2).
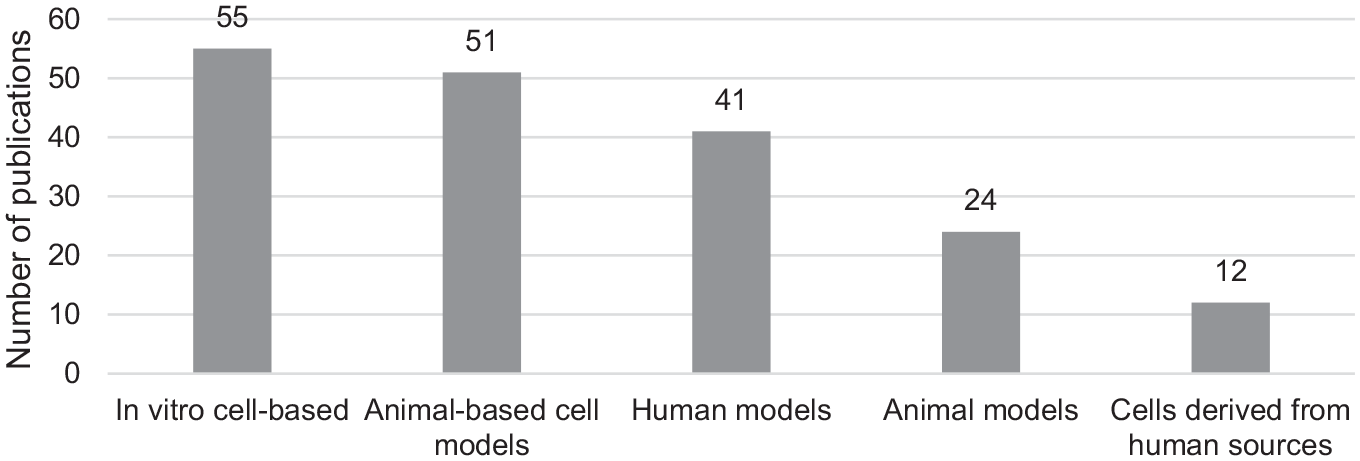
Models reported for investigating the mechanism of action of intra-articular hyaluronic acid products.
The IAHA preparations reported in the literature and their characteristics are represented in Table 1. The MW varied widely across the studies, from 500 to >3000 kDa. There were inconsistencies across studies regarding the classification of IAHA products into LMW, MMW, and HMW, with several studies refraining from such classification. Altman et al 4 classified IAHA products LMW (<1500 kDa), MMW (1500-3000 kDa), and HMW (>300 kDa).
Characteristics of IAHA Formulations Reported in the Literature a
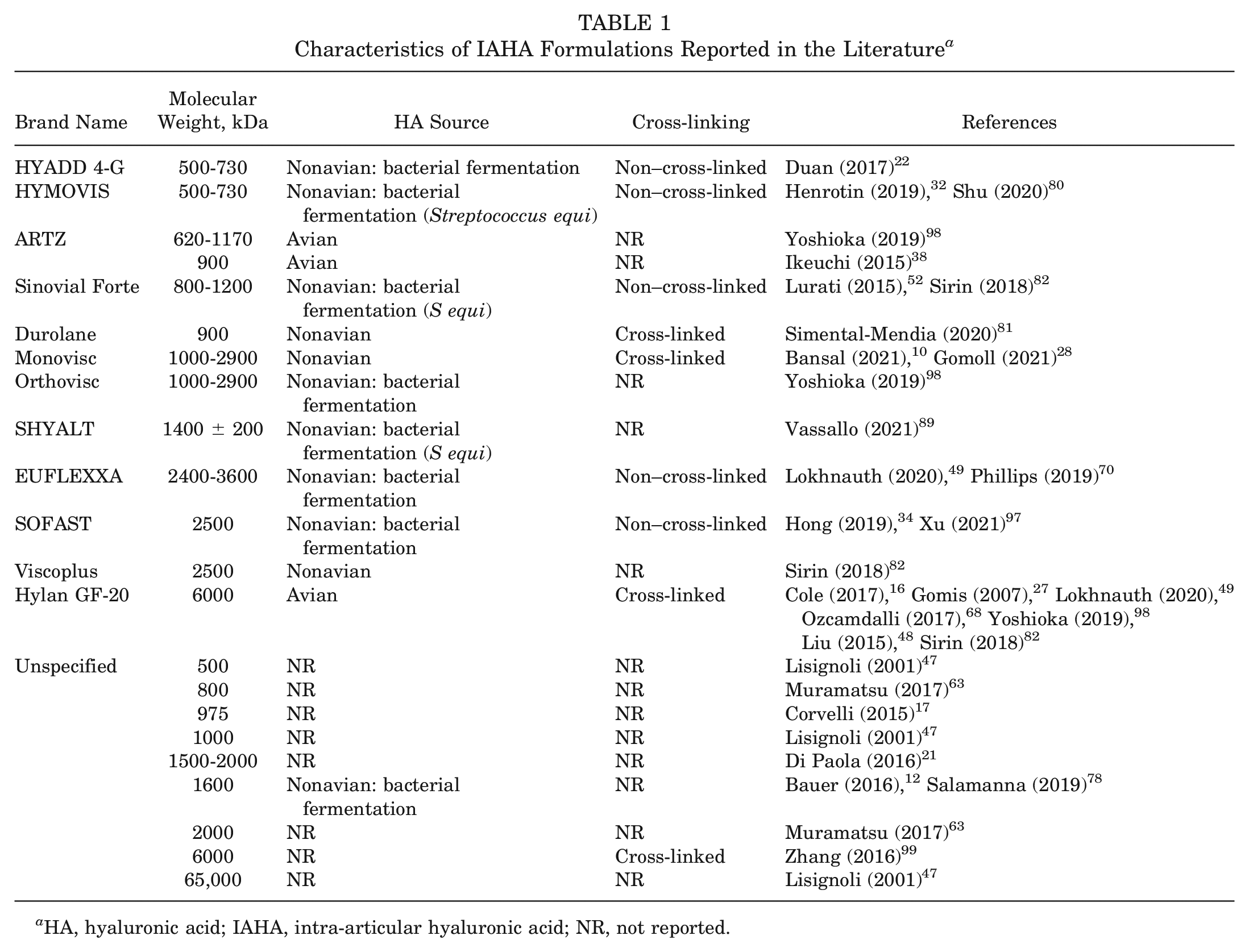
HA, hyaluronic acid; IAHA, intra-articular hyaluronic acid; NR, not reported.
All studies were categorized as high, medium, and low relevance based on the publication impact factors and quality, and finally 47 studies were utilized to summarize the MoA of IAHA. Key findings from the articles are described under various MoA headings: chondroprotective, increased proteoglycan/GAG synthesis, anti-inflammatory, analgesic, effects on subchondral bone, mechanical, and other MoAs (Figure 3).
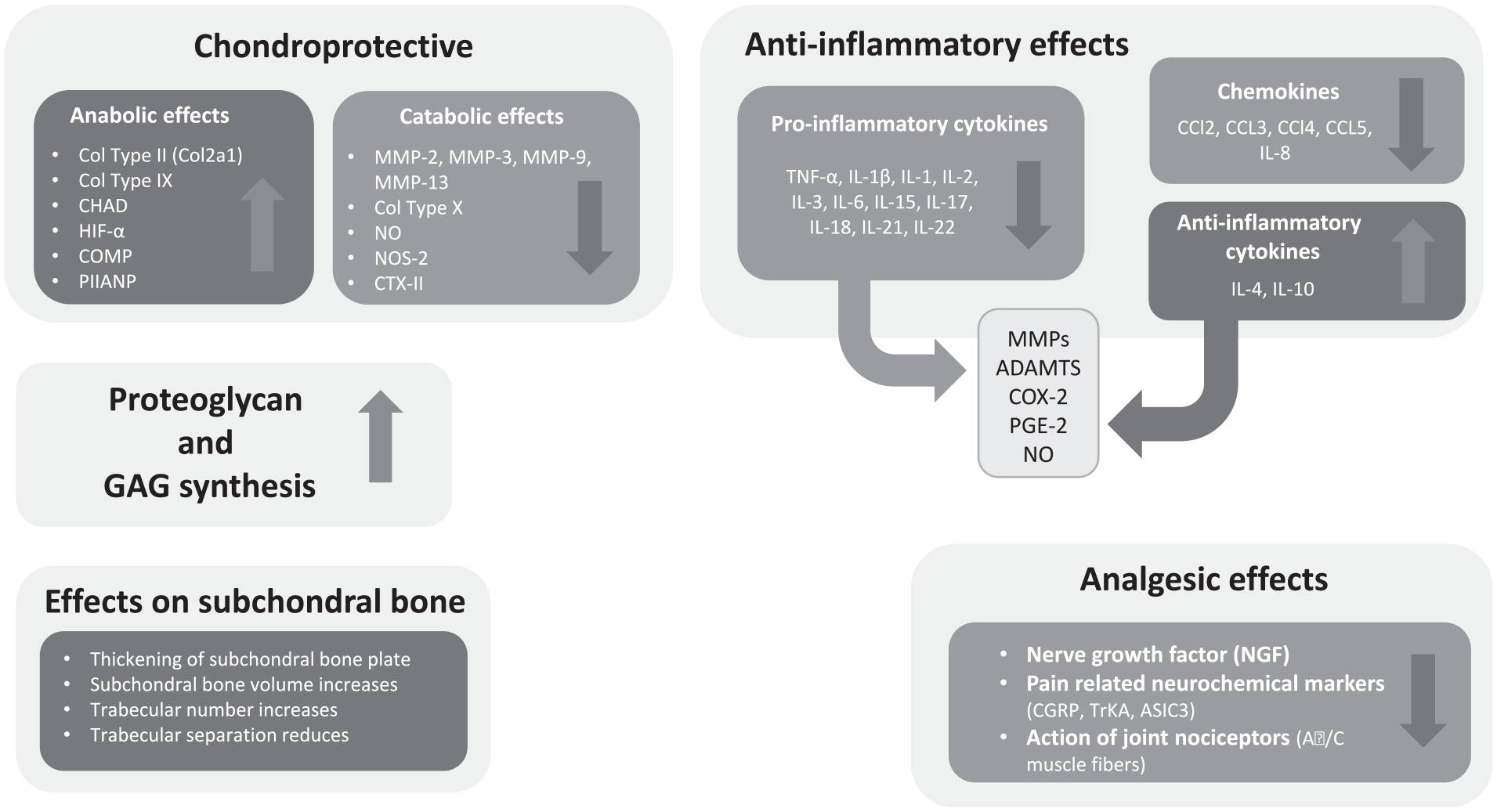
Different mechanisms of action of intra-articular hyaluronic acid treatment in knee osteoarthritis. ADAMTS, a disintegrin and metalloproteinase with thrombospondin motifs; ASIC3, acid-sensing ion channel 3; CCL, chemokine; CGRP, calcitonin gene-related peptide; CHAD, Chondroadherin; COL2A1, collagen type 2a1; COMP, cartilage oligomeric matrix protein; COX-2, cyclooxygenase 2; CTX-II, C-terminal cross-linked telopeptides of type II collagen; GAG, glycosaminoglycan; HIF-α, hypoxia-inducible factor α; IL, interleukin; MMP, matrix metalloproteinases; NO, nitric oxide; NOS-2, nitric oxide synthase 2; PGE-2, prostaglandin E2; PIIANP, procollagen type II N-terminal propeptide; TNF-α, tumor necrosis factor α; TrKA, tropomyosin receptor kinase; ↑, increase; ↓, decrease.
Chondroprotective Effect
Chondroprotective effects of IAHA formed the most frequently reported MoA, appearing in 27 articles from different types of studies: in vitro cells, animal models, or human models (Appendix Table A2). # KOA exerts a significant influence on anabolic and catabolic processes within the AC. Studies of cartilage and synovial fluids assess the anabolic processes by measuring the presence of collagen type II α chain 1 (COL2A1), an important cartilage-specific extracellular matrix (ECM) protein synthesized from proliferative chondrocytes and essential for chondrogenesis, and aggrecans, which are fundamental constituents of hyaline cartilage. With advancing KOA, COL2A1 and aggrecan levels decrease significantly, resulting in a reduction in the anabolic processes. In addition, cartilage oligomeric matrix protein (COMP), synthesized by synoviocytes and chondrocytes, helps in stabilizing their bonds and provides support to type II collagen fibers. Its production increases with degenerative conditions, as it affects the reconstitution of the AC after damage. 25 Chondroadherin (CHAD) mediates signaling between the ECM and chondrocytes and primarily contributes to the development of the collagen fibrillar network in the early stages of skeletal development. Thus, it is theorized that the downregulation of CHAD is pivotal in triggering and advancing KOA. Hypoxia-inducible factor 1α (HIF-1α) regulates the hypoxia-induced ECM synthesis in chondrocytes and thus positively regulates cartilage development and regeneration. Conversely, elevated levels of matrix metalloproteinases (MMPs) such as MMP-3, MMP-9, and MMP-13 serve as prominent indicators of catabolism in OA-affected cartilage, as they play a key role in cartilage matrix degradation. MMP-3 has wide substrate specificity and therefore contributes significantly to the degradation of type II collagen. 46 Tissue inhibitor of metalloproteinase 1 (TIMP-1) is an endogenous inhibitor of different MMPs, including MMP-13, and is thought to play a role in regulating ECM turnover by modulating the degradative capacity of these MMPs. 46 A higher TIMP-1 gene expression might indicate a protective effect against ECM degradation in articular tissues, which otherwise gets downregulated with advancing KOA. 81 The production of reactive oxygen species, such as nitric oxide, also results in degeneration of cartilage through increased chondrocyte apoptosis. 29
Preclinical Studies
The cell cultures obtained from the osteochondral tissue samples of patients with KOA showed a significant increase in the expression and proliferation of COL2A1, CHAD, and HIF-1α and a reduction in the levels of MMP-13 and COMP-2 after treatment with HA.82,89 One in vitro study utilizing HA and adipose-derived mesenchymal stromal/stem cells in an OA coculture model reported a decrease in the level of catabolic MMP-13 and an increase in TIMP-1 levels. 81 Another study noted an increase in the expression levels of anabolic marker COL2a1 and a 3-fold reduction in the expression of COL1a1, thereby increasing the differentiation index (ratio of COL2A1 to COL1A1) after treatment with HA. It also found a significant reduction in the expression of MMP-3, MMP-9, and MMP-13 in chondrocytes derived from human synovial tissue. 12
In animal studies, knees or femurs harvested were either studied histopathologically or scanned using micro–computed tomography for reporting structural changes in the synovium or AC. # Most studies reported the positive chondroprotective effects of IAHA. In a rat anterior cruciate ligament transection OA model, IAHA treatment resulted in a significant reduction in osteophytes observed radiographically. The histological findings also revealed reductions in cartilage lesions and fibrillations of the rat tibial plateau. 99 Several studies recorded the Modified Mankin score, scoring the severity of OA lesions from 0 (normal) to 14 (degraded) before and after treatment of OA with IAHA, and reported a significant reduction in the scores at lateral and medial regions of the knee.1,21,34,38,48,49,70,78 A study utilizing destabilization of a medial meniscus rat model showed an increase in relative bone volume and cartilage thickness after 8 weeks of IAHA administration. 78 A mouse compression joint injury model revealed a reduction in ectopic calcification (number of ectopic nodules/osteophyte formation) after 8 weeks of IAHA treatment, but this result was not statistically significant, likely because of the study’s low power. 22 In an axial mice tibial loading model, IAHA did not produce an observable effect at the cartilage level, which might be due to a shorter period of the study, as bone turnover or remodeling requires a longer study duration to observe such effects. 22
Immunohistochemical or gene expression analyses of dissected femur or tibial specimens after IAHA administration showed increased expression of cartilage synthesis biomarkers such as COL2a1 and collagen type IX.1,23,63,78,99 In contrast, galectin I and III, which reflect cartilage degeneration, were downregulated after IAHA treatment in OA-induced models. 78 Another study identified induction of the SRY-related protein 9 (SOX-9) gene, which is responsible for ECM construction, after IAHA treatment, but no effect was observed on the extracellular cartilage matrix protein COMP and HA binding site CD44. 22 Coherently, a reduction in cartilage catabolic biomarkers (eg, MMPs, nitric oxide, and collagen type X) was noted. Levels of various MMPs, such as MMP-2, MMP-3, MMP-13, nitric oxide, and collagen type X, were significantly reduced in all the studies after IAHA treatment, marking a reversal of cartilage degeneration processes.1,21,23,29,63,78,80,99 Hylan G-F 20 also showed a significant increase in the expression of the neuronal PAS domain protein 2 (NPAS2) gene, a transcription factor belonging to the basic helix-loop-helix-PAS family, which forms a heterodimer with brain muscle Arnt-like 1 (BMAL1) genes to regulate the circadian rhythm and cartilage matrix homeostasis. Another circadian rhythm gene, period 2, was increased, albeit nonsignificantly, after injection with Hylan G-F 20. 48 Circadian clock genes (NPAS2 and period 2) located in the cartilage help in regulating the cartilage matrix homeostasis. In cartilage degeneration and inflammatory joint diseases, dysregulation of clock-controlled genes has been observed under mechanical stress conditions.
Clinical Studies
Several clinical studies reported improvements in the structure of synovium and cartilage after treatment with IAHA products. Outcomes were assessed using various imaging techniques, such as radiography, high-frequency Doppler imaging, magnetic resonance imaging, whole-organ magnetic resonance imaging score (WORMS), and magnetic resonance observation of cartilage repair tissue score.10,32,34,68,94,97
A prospective, multicenter, pilot study was conducted in 46 patients with KOA treated with 2 cycles of Hymovis (LMW product) by rheumatologists and rehabilitation medicine physicians in hospitals in Belgium and France. 32 This study reported a significant increase in the WORMS total cartilage score in lateral femoral and trochlear regions of the knee and significant reductions in the WORMS effusion score, representative of synovitis. The structural findings also correlated with the biomarker levels after IAHA administration from patients with KOA, with a significant increase in the anabolic markers over time (eg, COL2a1, COLL2-NO2, N-propeptide of collagen IIA [PIIANP]). Furthermore, Coll2-1/PIIANP and CTX-II/PIIANP ratios decreased, showing a reduction in cartilage catabolism processes. 32 Significant improvement in the synovial thickness of the medial and lateral femoral condyles was reported after 6 months of IAHA administration. 97 Other studies noted improvement of AC defects and overall improvement in the knee structures.10,34,94 Besides structural improvements, biomarker analysis of serum, urine, or synovial fluid revealed a significant increase in COL2a1 concentration, a cartilage synthesis biomarker.32,94 Similar results were observed with a decrease in MMP-3, cross-linked C-terminal telopeptide of type 2 collagen (CTX-II), and other catabolic biomarkers, accompanied by an increase in TIMP-1 levels, indicative of metalloproteinase degradation.32,68
Increased Synthesis of Proteoglycan/GAG
Thirty studies suggested proteoglycan/GAG synthesis as an MoA for reducing cartilage degeneration (Appendix Table A3). ** The ECM of AC within the synovial joints is made up of water and various organic components, such as collagen, aggrecan, proteoglycans, GAGs, and glycoproteins (eg, chondroitin sulfate, keratan sulfate). 73 The negatively charged GAGs are bound to the aggrecan core protein and form the proteoglycan aggregates linked to the HA backbone, which further remain entrapped within the collagen type II fibril meshwork. 73 With KOA progression, intrinsic GAGs and proteoglycans are degraded by MMPs and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS), specifically ADAMTS4 and ADAMTS5. Aggrecan is the primary proteoglycan present within the AC, which degenerates with the progression of OA and is thus released in the form of fragments from ECM into the synovial fluid.
Preclinical Studies
Animal model studies demonstrated that IAHA treatment stimulated proteoglycan, aggrecan, and GAG synthesis, which may delay the progression of OA. ** Few other studies reported a reduction in proteoglycan loss over time after IAHA treatment. 80 Additionally, a few in vitro cell-based studies showed increased concentrations of aggrecan, GAG, and proteoglycans after treatment with IAHA.12,17,81,89
Clinical Studies
Two clinical studies reported data related to proteoglycan.32,68 Chondroitin sulfate, a sulfate proteoglycan, occurs within the collagen framework of the knee joint. As OA advances, various aggrecanases or chondroitinases degrade the AC components, thereby releasing the fragments of chondroitin sulfate. The prospective, multicenter, trial study of symptomatic patients with KOA revealed no significant change over time in the levels of chondroitin sulfate. 11 Another clinical trial including patients with mild to moderate KOA found decreases in the concentration of collagen sulfate in the synovial fluid.32,68
In our present review, several studies demonstrated that IAHA treatment stimulated proteoglycan and GAG synthesis, which may delay the progression of KOA.22,23,63,69,96,99 Aggrecan is the primary proteoglycan within AC, and IAHA treatment has been shown to suppress aggrecan degradation as well as promote intrinsic aggrecan development. 6 The enzyme MMP-13 plays a critical role in the degeneration of AC in OA. It is known for its potent ability to degrade AC’s proteoglycans. 25 Treatment with HA resulted in a significant decrease in MMP-13 levels, suggesting a potential inhibitory effect on its secretion. This, in turn, may contribute to the rescue of collagen type II and proteoglycan expression.
Anti-inflammatory Effect
The anti-inflammatory MoA was reported by 24 studies (Appendix Table A4). †† The activation of inflammatory and catabolic events in AC attributed to the activation of Toll-like receptors in chondrocytes resulting in synovial inflammation is well documented.11,58 Furthermore, Toll-like receptors 2 and 4 enhance the expression of catabolic biomarkers (eg, MMP-13) and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling through the cytosolic adaptor myeloid differentiation factor 88 (MyD88). 11 IL-1β is a key mediator in the inflammatory effects of HA and is regulated through binding of HA with CD44. IL-1β suppression results in downregulation of MMPs, which aids in the anti-inflammatory effect of HA.4-6 In the context of KOA, synoviocytes also produce cytokines and several other inflammatory mediators, such as NF-κB, chemokines (interleukin 8 [IL-8], monocyte chemotactic and activating factor, CCL2, CCL3, CCL4, CCL5), and matrix metalloproteases (eg, MMP-1, MMP-2, MMP-3, MMP-9, MMP-13) and their specific tissue inhibitor (TIMP-1). 95
Preclinical Studies
IL-1β is a key mediator involved in the inflammatory processes within the synovial fluid as well as the chondrocytes.5,95 The majority of preclinical research investigations have reported downregulation of IL-1β after IAHA treatments.12,81,89 TNF-α also plays a significant role in the inflammatory cascade, as it has been shown to induce the production of MMPs and thus to increase cartilage degeneration mechanisms.60,95 TNF-α has been widely studied in the immunohistochemical staining of femur or tibial sections or by gene expression analysis, wherein it was downregulated after treatment with IAHA in all such studies.12,21,23,29,34,48,49,89 In addition, a few other proinflammatory cytokines, such as IL-1 and IL-6, were indicated with marked reduction after IAHA treatment.12,34,62,80,89 A significant reduction in synovial ADAMTS4 was noted over time from weeks 4 to 12 after IAHA treatment. 80 Studies utilizing in vitro cultures of synovial tissue from patients with KOA have also shown a decrease in the expression levels of key inflammatory biomarkers upon treatment with IAHA, such as NF-κB, MyD88, and MMP-13. 81 Similar findings were observed in the chondrocytes of in vitro cultures as well. 89 Finally, several other proinflammatory markers (IL-5, IL-6, IL-7, interferon γ [INF-γ], and chemokines) were downregulated after IAHA treatment.12,81,89 In addition, increased levels of anti-inflammatory cytokines (IL-4 and IL-10) were reported. 89
Clinical Studies
Several clinical studies reported a reduction in the levels of proinflammatory mediators (IL-2, IL-5, IL-6, IL-8, IL-12p70, IL-17A, IL-17F, IL-22, INF-γ), inflammatory mediators (IL-1β, TNF-α, TNF-β), cytokines, macrophages (M1 and M2), and C-reactive proteins in the blood samples or synovial fluid samples withdrawn from patients with KOA after IAHA treatment. ‡‡ In addition, increased levels of anti-inflammatory cytokines (IL-4 and IL-10) were noted in the literature.40,56
Analgesic Effect
From the included studies, 18 reported an analgesic MoA for different IAHA treatments (Appendix Table A5). §§ Pain serves as the primary symptom driving individuals with OA to seek medical assistance. This pain is frequently linked to joint damage in patients with OA, which mostly originates from the synovium and subchondral bone. All the components of the knee appendage possess abundant nociceptors responsible for initiating the sensation of pain. Upon mechanical, thermal, or chemical stimuli, these nociceptors get stimulated and transduce the signals along the dorsal root ganglion and the dorsal horn of the spinal cord, thereby finally transmitting the signals to higher brain centers. The persistent inflammatory microenvironment in the OA joint induces peripheral and central nerve sensitization, resulting in mechanical allodynia and hyperalgesia around the joint. 85
It has been demonstrated that IL-1β induces an increase in levels of nerve growth factor (NGF), a key factor in hyperalgesia associated with inflammation, which is found in the synovial fluid of patients with KOA. Several signaling pathways are associated with OA pain: NGF activates its receptor tyrosine receptor kinase A (TrkA), mediating pain transmission. Calcitonin gene-related peptide (CGRP) is released from C-fiber terminals and binds to calcitonin receptor-like receptor and receptor activity-modifying protein 1 (RAMP1) on adjacent Aδ nerve fibers. The dorsal root ganglion and dorsal horn of the spinal cord exhibit high expression of chemokines, such as CCL2 and CX3CL1, with immune mediators such as colony-stimulating factor 1 (CSF-1) and CGRP. 85 Treatment with HA significantly reduced proinflammatory cytokine levels and NGF expression, thus alleviating algesia.
Preclinical Studies
IAHA injection significantly reduced mechanical hyperalgesia of the paw in animal models, thereby increasing the paw withdrawal threshold and weightbearing capacity. 62 IAHA helped in alleviating pain in the joints by reducing the action of joint nociceptors. It downregulated the pain-related neurochemical markers CGRP, TrkA, and acid-sensing ion channel 3 (ASIC3) in joint afferents. 38 IAHA also caused a reduction of movement-evoked nerve impulse activity in fine afferent fibers (Aδ/C muscle fibers). 27 Another study reported a reduction in the levels of NGF after treatment with IAHA. 21 Some other preclinical studies noted reductions in the severity of KOA pain.21,80
Clinical Studies
Several clinical studies assessing the pain severity in patients with KOA evaluated the effects of IAHA treatments over time utilizing various assessment tools: Knee injury and Osteoarthritis Outcome Score, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Lequesne Pain functional index, numeric rating scale, and visual analog scale. All the studies reported statistically significant reductions in these scores, establishing the analgesic effect of IAHA treatments. ∥∥ In addition, a significant correlation was observed between HMW and pain score reduction. Various Systemic Literature Review/Meta-analysis/Network Meta-analysis have demonstrated a higher and significant reduction in pain scores with HMW IAHA preparations in comparison with LMW preparations.6,14,36,71,77,93
Effect on Subchondral Bone
Eight studies reported the effect of IAHA treatment on the subchondral bone regions (Appendix Table A6).21-23,29,34,69,80,96 The subchondral bone structures were studied utilizing different techniques, including histopathology or micro–computed tomography in the studies. With the progressive loss of AC, OA is characterized by increased subchondral bone sclerosis with thickening of the cortical plate, extensive remodeling of the trabeculae, formation of new bone at the joint margins (osteophytes), and development of subchondral bone cysts. Changes to the subchondral bone occur because of abnormal bone remodeling with a subsequent decrease in bone density.
Preclinical Studies
The effect of various IAHA treatments on the subchondral bone was reported utilizing various OA-induced animal models. Mixed results were observed for the effects of HA on subchondral bone, as few studies reported an increase in subchondral bone volume/thickness or scores, while others revealed no differences.21-23,29,34,69,80,96
Clinical Studies
In a double-blind randomized clinical trial, 16 patients with grade II to III KOA were subjected to a single-injection treatment of IAHA. After 6 months, knees were studied radiologically using magnetic resonance imaging, and authors found subchondral bone and lamina to be intact in 30% of patients after treatment. 34
Mechanical Effect
Twelve studies reported the mechanical MoA of IAHA (Appendix Table A7). ¶¶ IAHA protects the joint capsule through lubricant action and beneficial shock absorption effects, as it provides cushioning to absorb pressure and vibration within the joint that otherwise may lead to chondrocyte degradation. In addition, IAHA functions as a joint structure stabilizer and flow resistance regulator. 30 Given the dilution of synovial fluid composition by inflammatory effusion, a decrease in HA concentration, and a reduction of synovial fluid viscosity, osteoarthritic knees have higher friction within the joint space than healthy knees.
Preclinical Studies
In most preclinical studies, the effect of IAHA treatments on the biomechanical properties was analyzed by estimating the coefficient of friction (COF) of the synovial fluid within the knee joints. After IAHA treatment, a significant reduction in joint COF values was reported.13,17,41,61,64,92 However, another study found no alterations in COF values in comparison with the disease control group.17,70 IAHA also ameliorated the progression of OA by preventing damage to the AC layer and restoring its elastoviscosity. 51 Additionally, HA products exhibit an analgesic effect through modulation of joint nociceptor discharges, which appears to be primarily dependent on their rheological properties and the associated elastoviscosity. 26 Moreover, IAHA prevented cartilage erosion from its normalization of joint biomechanics and its inhibitory effects on periarticular cells, which are involved in tissue hyperplasia and fibrosis. 72
Clinical Studies
IAHA treatment has been shown to increase the viscosity and elasticity of synovial fluid, lubricate the joint capsule, and prevent degeneration of cartilage through decreased friction.17,70 The reduction of friction within the joint can provide therapeutic effects, as the cartilage is protected from mechanical degradation; it also facilitates the dissipation of loads transmitted across articular surfaces, limits migration of inflammatory cells to AC, and protects articular pain receptors and synoviocytes. Human synovial fluid samples of patients with KOA showed significant reductions in COF values. 39 A phase 1/2 randomized clinical trial including patients with KOA reported significant improvements in the WOMAC function scores after 6 and 12 months of IAHA treatment. 43
Other Actions of IAHA
Vascular endothelial growth factor (VEGF) is a cytokine that promotes angiogenesis and regulates the process of bone development and bone remodeling. 73 The expression of VEGF was notably elevated in the cartilage of OA-afflicted knees as compared with that in individuals with good health, likely attributed to the presence of hypoxia. Additionally, VEGF expression was observed in the synovium of patients with KOA, coinciding with the formation of capillaries and the onset of inflammation. VEGF levels tend to rise during the progression of OA. After IAHA treatment, VEGF was significantly reduced in the synovial fluid in comparison with controls. 45
Effect of MW and Source of HA
New research suggests that the effectiveness of different IAHA MoAs depends on the MW of the IAHA used. Previous studies signify a greater effect on pain alleviation obtained with HMW IAHA preparations in comparison with LMW and MMW preparations (Appendix Table A8).6,14,36,71,77,93 Similar reporting was observed for the chondroprotective MoA, for which an increase in COL2A1, CHAD, and HIF-1α expression and proliferation was observed, particularly in cultures treated with MMW and HMW HA than LMW HA. 82
This was further evident with effects on viscosity and joint lubrication effects. 54 However, among all these profound cartilage restorative effects observed with HMW IAHA preparation, there were reports of an increased incidence of various adverse effects, such as injection site flare-ups or pseudosepsis, a rare complication associated with HA injections. 79 A separate study analyzed subgroups based on IAHA’s MW and production method to investigate the pooled incidence of injection site reactions and effusion. The results did not demonstrate a significant difference in the occurrence of effusion across MW subgroups (HMW, MMW, and LMW). Yet, the incidence of injection site flare-ups was significantly higher for HMW HA as compared with LMW and MMW HA.
Additionally, biological fermentation-derived HA had a significantly smaller incidence of effusion and injection (site flare-ups) than did avian-derived HA. 4 However, results from these studies did not establish a direct correlation between avian-derived or cross-linking and incidence of inflammatory reactions, given the confounding by differences in the MW of products or the mode of administration (ie, local injection) and technique of administration (superolateral vs others). Ong et al 66 described case reports of severe acute localized reactions (SALRs) after IAHA injections for the knee. They evaluated surrogate SALR measures between patients using Hylan G-F 20 and non–Hylan G-F 20 HA products based on whether the products were single- or multiple-injection therapies, avian or nonavian derived, and cross-linked or non–cross-linked. This study showed no clear correlation between avian-derived or cross-linked products and SALR, and it provided evidence against avian-derived products or cross-linking as a source for these reactions. In fact, allergic reactions have also been reported for non–cross-linked, nonanimal, and/or naturally derived HA.7,37,53,76,83
Discussion
The present literature review has delineated the research regarding the MoA of IAHA with regard to chondroprotection, proteoglycan or GAG synthesis, anti-inflammatory and analgesic effects, effects on subchondral bone, and mechanical effects. This literature review helps to understand the potential disease-modifying effects of IAHA treatment on the progression of KOA as it acts at various molecular levels, decreasing the expression of cartilage degeneration molecules and increasing the expression of signaling factors responsible for cartilage and other structural unit synthesis within the synovial joint.
IAHA products demonstrated interaction with CD44 receptors, the major receptor responsible for mediating the effects of HA. 30 Treatment of KOA with IAHA demonstrated an increase in the expression of anabolic factors, COL2a1 majorly, responsible for collagen synthesis and a decrease in the expression of catabolic markers such as MMPs and collagen type X. MMP production is regulated through IL-1β, which is regulated by the NF-κB and MyD88 pathways. Furthermore, IL-1β plays a significant role in the inflammatory processes, wherein it activates NF-κB and mitogen-activated protein kinase. IAHA treatment inhibits the production of IL-1β, thereby hindering the MMP production pathway. This action mediates further effects, including reduced production of aggrecanases (eg, ADAMTS), COX-2 molecules responsible for pain, and nitric oxide. IAHA treatment also helps in alleviating the pain function. In addition to these effects, owing to the decreased production of ADAMTS, proteoglycan degradation reduces within the AC. IAHA treatment restores the proteoglycan loss that occurs during OA progression. In addition, IAHA helps in restoring synovial fluid viscosity, thereby reducing friction and improving joint mobility. It is evident from these reports that IAHA produces its effect through various MoAs, as summarized in Figure 3.
These mechanisms were reported by Altman et al 6 in 2015 as well, delineating the MoAs for IAHA. The data gathered in this review confirms these previous findings with the addition of a few biomarkers that were not previously reported. The findings from this targeted literature review support the involvement of COL2a1, known for its anabolic properties, in augmenting collagen synthesis. This extends the knowledge base by incorporating additional biomarkers such as collagen type IX, CHAD, HIF-α, COMP, and PIIANP, providing a broader understanding than previously reported. In addition, evidence was demonstrated for the role of chemokines in the inflammatory pathways and the counteracting effects provided by IL-4 and IL-10, thus providing anti-inflammatory effects. Furthermore, nociceptive sensory mechanisms were delineated for a better understanding of the analgesic effects produced by IAHA. Finally, the MoAs in this targeted literature review include evidence from basic science reports and clinical studies, establishing the background for the various effects of IAHA treatment in KOA.
This review also highlights the role of MW in determining the effectiveness of IAHA for KOA. Limited evidence suggests that HMW IAHA provides superior pain relief and chondroprotective effects as compared with LMW and MMW preparations, attributed to its enhanced viscoelastic properties and increased expression of cartilage-specific proteins, such as COL2A1, CHAD, and HIF-1α. Despite these benefits, HMW IAHA is associated with a higher rate of adverse effects, including injection site flare-ups and pseudosepsis, perhaps owing to its prolonged presence in the joint space. No significant difference in effusion occurrence was found across MW subgroups, but HMW preparations had significantly more injection site reactions. These findings suggest a need for personalized treatment strategies, balancing the therapeutic advantages of HMW IAHA with the potential for increased adverse effects and exploring optimized formulations or biomarkers to predict patient response and minimize risks.
IAHA treatment has demonstrated sufficient efficacy in pain relief in KOA during clinical trials. However, more long-term studies should be conducted to determine its long-term efficacy and safety profile for single and repeated treatment courses, as well as in combination with preparations such as platelet-rich plasma, corticosteroid, amniotic fluid, exosome, and cell-based injections. This can be a suitable alternative to knee replacement by either delaying surgery or completely barring it.2,3,19,20,65,67,90,91
Conclusion
The literature provides evidence to support numerous mechanisms through which IAHA preparations may provide clinical benefit in KOA, including the significant role of IAHA MW in the effectiveness of different MoAs. Chondroprotection was the most frequently reported mechanism, with a reduction in cartilage degeneration and chondrocyte apoptosis and hypertrophy, an increase in anabolic biomarkers, and a reduction in catabolic biomarkers. This was further supported by an increase in proteoglycan and GAG synthesis within the cartilage and a delay in the inflammatory cascade in the synovial joint by inhibiting various inflammatory mediators. Finally, an analgesic effect was provided by IAHA treatment through the action of nociceptors.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465241302820 – Supplemental material for Mechanisms of Action of Intra-articular Hyaluronic Acid Injections for Knee Osteoarthritis: A Targeted Review of the Literature
Supplemental material, sj-pdf-1-ajs-10.1177_03635465241302820 for Mechanisms of Action of Intra-articular Hyaluronic Acid Injections for Knee Osteoarthritis: A Targeted Review of the Literature by Seth L. Sherman, Andrew S. Gudeman, John D. Kelly, Robert J. Dimeff and Jack Farr in The American Journal of Sports Medicine
Footnotes
Acknowledgements
The authors thank Charlotte Singh, MD, Kevin Steele, PharmD, and Wilson Ngai, PharmD, Sanofi, for coordinating the development of this manuscript, facilitating author discussions, and reviewing the manuscript critically and Ramandeep Kaur, Evidinno Outcomes Research Inc, for medical writing support funded by Sanofi.
Submitted April 11, 2024; accepted September 30, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.L.S. has received royalties or licenses from ConMed, DJO, and Ostesys; consulting fees from AO, Arcuro, Arthrex, ConMed, Depuy J&J, DJO, Flexion Orthopedics, Icarus Bracing, JRF, Kinamed, LifeNet, Linksys, NewClip, Olympus America, Osteosys, Pacira, Reparel, Sarcio, Smith & Nephew, Sparta Biomedical, Vericel, and Vivorte; speaking fees from Synthes GmbH; and support for education from Elite Orthopedics and Evolution Surgical. He participates on a data safety monitoring board or advisory board for Allosource, Bioventus, Reparel, Sarcio, Sparta MedixL, Vericel, and Vivorte and holds stock or stock options with Sarcio, Vivorte, Osteosys, and Reparel. J.D.K. has received hospitality payments, support for education, and speaking fees from Arthrex; support for education from Paladin Technology Solutions; hospitality payments from Baxter Healthcare; and consulting fees from Heron Therapeutics. R.J.D. received support for the present manuscript from Sanofi; equipment, materials, drugs, medical writing, gifts, or other service with Ferring Pharmaceuticals; honoraria from Pacira Pharmaceuticals and Ferring Pharmaceuticals; consulting fees from Sanofi-Aventis; and compensation for services other than consulting from Orthogenrx. J.F. has received royalties or licenses with Biopoly LLC, MedShape Inc, and Organogenesis; consulting fees from Arthrex, Collagen Matrix, Vericel Corporation, Joint Restoration Foundation, Cook Biotech Inc, Bioventus, and Organogenesis; support for education from Crossroads Orthopedics; and stock or stock options from ZKR Orthopedics Inc. He participates on a data safety monitoring board or advisory board with Alira Health, Medipost, and Elute. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
An online CME course associated with this article is available for 1 AMA PRA Category 1 Credit™ at https://education.sportsmed.org/Public/Catalog/Home.aspx?CourseSearch=1&Criteria=9&Option=25. In accordance with the standards of the Accreditation Council for Continuing Medical Education (ACCME), it is the policy of The American Orthopaedic Society for Sports Medicine that authors, editors, and planners disclose to the learners all financial relationships during the past 12 months with any commercial interest (A ‘commercial interest’ is any entity producing, marketing, re-selling, or distributing health care goods or services consumed by, or used on, patients). Any and all disclosures are provided in the online journal CME area which is provided to all participants before they actually take the CME activity. In accordance with AOSSM policy, authors, editors, and planners’ participation in this educational activity will be predicated upon timely submission and review of AOSSM disclosure. Noncompliance will result in an author/editor or planner to be stricken from participating in this CME activity.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
