Abstract
Background:
Revision anterior cruciate ligament (ACL) reconstruction has been documented to have inferior outcomes compared with primary ACL reconstruction. The reasons why remain unknown.
Purpose:
To determine whether surgical factors performed at the time of revision ACL reconstruction can influence a patient’s outcome at 6-year follow-up.
Study Design:
Cohort study; Level of evidence, 2.
Methods:
Patients who underwent revision ACL reconstruction were identified and prospectively enrolled between 2006 and 2011. Data collected included baseline patient characteristics, surgical technique and pathology, and a series of validated patient-reported outcome instruments: Knee injury and Osteoarthritis Outcome Score (KOOS), International Knee Documentation Committee (IKDC) subjective form, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and Marx activity rating score. Patients were followed up for 6 years and asked to complete the identical set of outcome instruments. Regression analysis was used to control for baseline patient characteristics and surgical variables to assess the surgical risk factors for clinical outcomes 6 years after surgery.
Results:
A total of 1234 patients were enrolled (716 men, 58%; median age, 26 years), and 6-year follow-up was obtained on 79% of patients (980/1234). Using an interference screw for femoral fixation compared with a cross-pin resulted in significantly better outcomes in 6-year IKDC scores (odds ratio [OR], 2.2; 95% CI, 1.2-3.9; P = .008) and KOOS sports/recreation and quality of life subscale scores (OR range, 2.2-2.7; 95% CI, 1.2-4.8; P < .01). Use of an interference screw compared with a cross-pin resulted in a 2.6 times less likely chance of having a subsequent surgery within 6 years. Use of an interference screw for tibial fixation compared with any combination of tibial fixation techniques resulted in significantly improved scores for IKDC (OR, 1.96; 95% CI, 1.3-2.9; P = .001); KOOS pain, activities of daily living, and sports/recreation subscales (OR range, 1.5-1.6; 95% CI, 1.0-2.4; P < .05); and WOMAC pain and activities of daily living subscales (OR range, 1.5-1.8; 95% CI, 1.0-2.7; P < .05). Use of a transtibial surgical approach compared with an anteromedial portal approach resulted in significantly improved KOOS pain and quality of life subscale scores at 6 years (OR, 1.5; 95% CI, 1.02-2.2; P≤ .04).
Conclusion:
There are surgical variables at the time of ACL revision that can modify clinical outcomes at 6 years. Opting for a transtibial surgical approach and choosing an interference screw for femoral and tibial fixation improved patients’ odds of having a significantly better 6-year clinical outcome in this cohort.
Revision anterior cruciate ligament (ACL) reconstruction continues to offer challenges to the sports medicine practitioner. The goal of establishing knee stability and returning patients to their previous level of activities remains difficult. To elucidate the predictors of outcome, the Multicenter ACL Revision Study (MARS) Group has collected a prospective cohort of patients who underwent revision ACL reconstruction. 9 Obviously, modifiable factors are the most enticing, as they can be adopted by surgeons to improve outcome. Previously, the group has demonstrated that autograft choice can decrease rerupture rate and potentially increase activity level. 7 Identifying additional modifiable factors involving surgical and technical decisions could assist in improving outcomes of revision ACL reconstructions.
Little evidence exists regarding patient outcomes >5 years after revision ACL reconstruction. Only 259 patients across 7 studies have been followed for a minimum of 5 years.1,3,6,10,12,13,15 Thus, our cohort of 1234 patients is critical in assessing midterm and longer outcomes after revision ACL reconstruction. Previous analysis of this group at 6 years included graft choice effect and meniscal and chondral predictors of outcome.18,19 This showed a graft failure rate of 5.8% for the cohort. 18
Previously, our group evaluated the effect of surgical factors at minimum 2-year follow-up in our study cohort. 8 Factors that demonstrated positive effects on outcomes at 2 years included opting for an anteromedial portal or transtibial surgical exposure, choosing a metal interference screw for femoral fixation, and not performing notchplasty. 8 We hypothesized that at longer follow-up, a decreasing number of surgical factors would have a significant effect. The purpose of this study was to determine whether surgical factors performed at the time of revision ACL reconstruction can influence a patient’s outcome at 6-year follow-up.
Methods
Study Design and Setting
The MARS Group was assembled in cooperation with the American Orthopaedic Society for Sports Medicine as a collection of 83 sports medicine fellowship–trained surgeons working at 52 sites. The surgeons are a mix of practitioners in academic settings (54%) and private practice (46%). Surgeons could perform the ACL revision surgery according to their own practice preferences. If an allograft was chosen for reconstruction, the surgeon was required to use a Musculoskeletal Transplant Foundation graft (MTF Biologics) to standardize and record allograft preparation methods.
The objective of this consortium has been to assess both the short- and long-term outcomes after revision ACL reconstruction and to determine how the initial factors at the time of revision surgery may influence and predict disease progression. This study design involves a longitudinal prospective cohort for whom we currently have baseline, 2-year, and 6-year follow-up data.
Participants
After institutional review board approval was obtained from each institution, 1234 patients with documented ACL reconstruction failure who underwent revision ACL reconstruction surgery qualified for and were consented to be in this study (Figure 1). This multicenter consortium began patient enrollment in 2006 and ended in 2011. Study inclusion criteria were revision ACL reconstructions performed by a MARS surgeon on patients with ACL deficiency for whom a previous ACL reconstruction had failed, as identified using either magnetic resonance imaging, physical examination (positive pivot-shift and Lachman test), KT-1000 arthrometer testing demonstrating >5-mm side-to-side difference, functional instability, or arthroscopic confirmation.
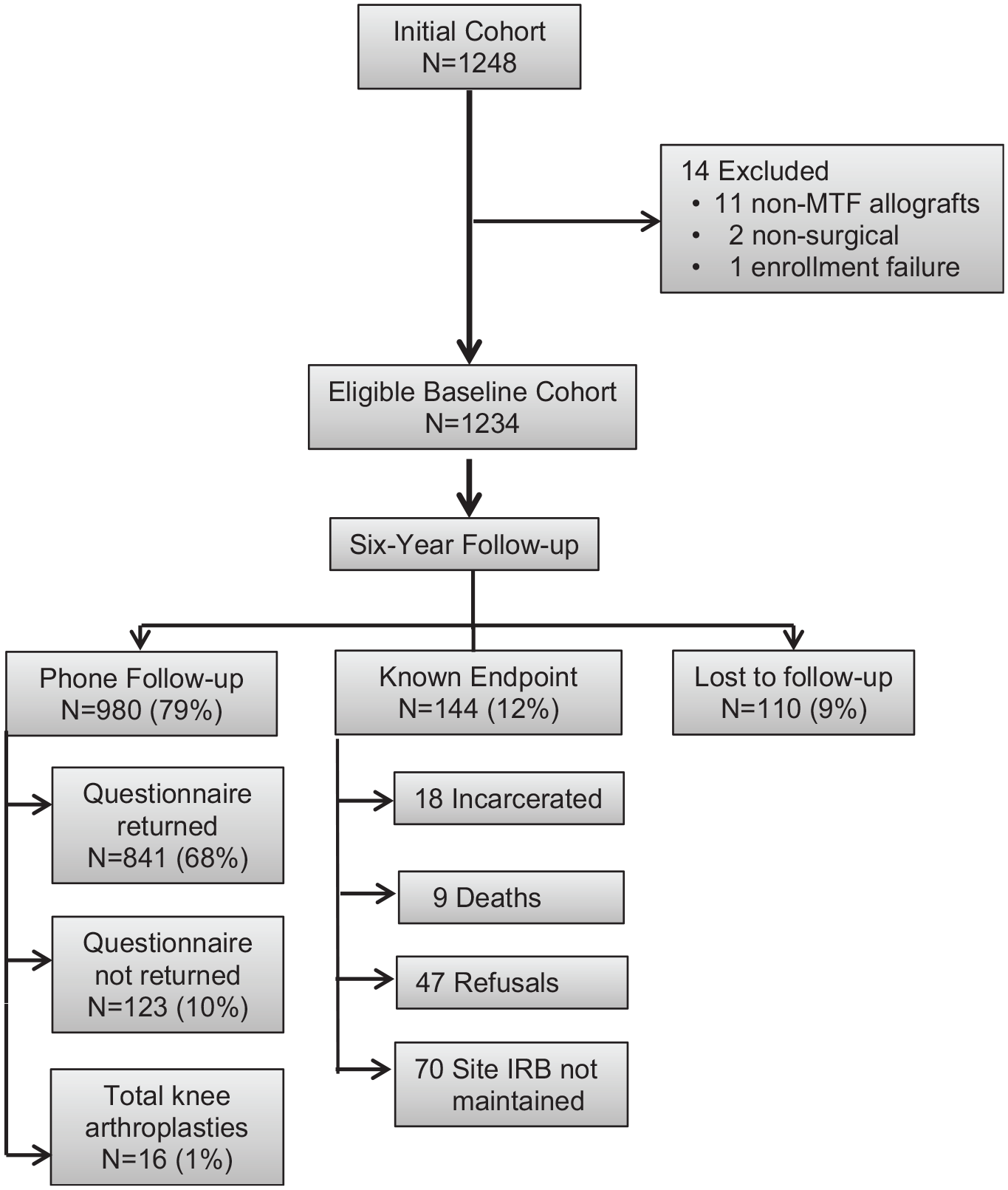
Patient enrollment flow diagram. IRB, institutional review board; MTF, Musculoskeletal Transplant Foundation.
Data Sources
After providing informed consent, the patients completed a self-reported questionnaire examining patient characteristics, injury characteristics, sports participation history, and health status before their revision ACL reconstruction surgery. Within this questionnaire, each participant completed a series of validated general and knee-specific outcome instruments, including the Knee injury and Osteoarthritis Outcome Score (KOOS), the International Knee Documentation Committee (IKDC) subjective form, the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and the Marx activity rating scale. Surgeons filled out a questionnaire that included physical examination findings, previous surgical history (if known), current revision surgical technique used, and the intra-articular findings and surgical management of meniscal and chondral pathology.
Completed forms were mailed from each site to our data coordinating center (Vanderbilt University). Data from both the patient and surgeon questionnaires were scanned using Teleform software (OpenText) using optical character recognition, and the scanned data were verified and exported to a master study database. A series of custom logical error and quality control checks were systematically performed before data analyses.
Patient Follow-up
At 6 years, the same questionnaire was administered as at baseline and at 2-year follow-up.Patients were also contacted via telephone or email to determine whether subsequent graft failure and/or any additional knee surgery had occurred.
Statistical Analysis
Descriptive statistics for the baseline patient and surgical characteristics were collected and reported. The effects of the independent (risk factor) variables on the outcome measures of IKDC, KOOS, WOMAC, and Marx activity scale were modeled with proportional odds logistic regression, and the effects on binary outcome of subsequent surgery (yes/no) were modeled using logistic multivariable regression. This statistical approach allows control of multiple factors with assessment of predictors independent of all other factors. Odds ratios (ORs) and 95% CIs were obtained via exponentiation in the parameter estimates. Patient-related covariates and previous and current surgical-related covariates were included and controlled for in the models. Patient-related covariates included sex (male/female), age at the time of revision ACL reconstruction, body mass index, smoking status (nonsmoker, quit, current), baseline Marx activity level, and baseline outcome measures (IKDC, KOOS, WOMAC, Marx). Covariates related to previous surgical information included time (in years) since the patient’s last ACL reconstruction, previous ACL reconstruction on the contralateral knee (yes/no), and surgeon’s revision of his or her own failed procedure (yes/no). Covariates related to current surgical information included surgeon years of experience, surgical technique (1 incision transtibial, 1 incision anteromedial portal, 2 incisions), femoral and tibial aperture position, femoral and tibial fixation, graft type (bone–patellar tendon–bone [BPTB] autograft, soft tissue autograft, BPTB allograft, soft tissue allograft), and biological enhancement used (yes/no). Three-knot restricted cubic splines were used for all continuous covariates to allow for nonlinear relationships with the outcomes.
The changes in outcome scores between baseline and 6 years were assessed via a comparison of median and interquartile range at each time point and tested using Kruskal-Wallis test. Additionally, the minimal clinically important difference was examined between time points. Minimal clinically important difference was 11 points for IKDC, 8 to 10 points for each of the 5 KOOS subscales, 8 to 10 points for the WOMAC, and 2 points for Marx activity scale. Alpha was set at .05 for all statistical tests. Multiple imputation using predictive mean matching was used to address missing data. The Hmisc and rms packages of the open source R statistical software (https://www.r-project.org) were used for statistical analysis.
Results
A total of 1234 patients (716 men; 58%) met the inclusion criteria and were successfully enrolled (Table 1). The median age of the patients was 26 years at the time of enrollment, and the median time since their most recent ACL reconstruction before the current revision was 3.3 years. At 6 years, follow-up related to incidence of subsequent surgery was obtained on 79% of the cohort (980/1234) (“Phone Follow-up” in Figure 1), whereas follow-up related to patient-reported outcomes at 6 years was obtained on 68% of the cohort (841/1234) (“Questionnaire returned” in Figure 1).
Baseline Cohort Characteristics at the Time of Revision ACL Reconstruction (N = 1234) a
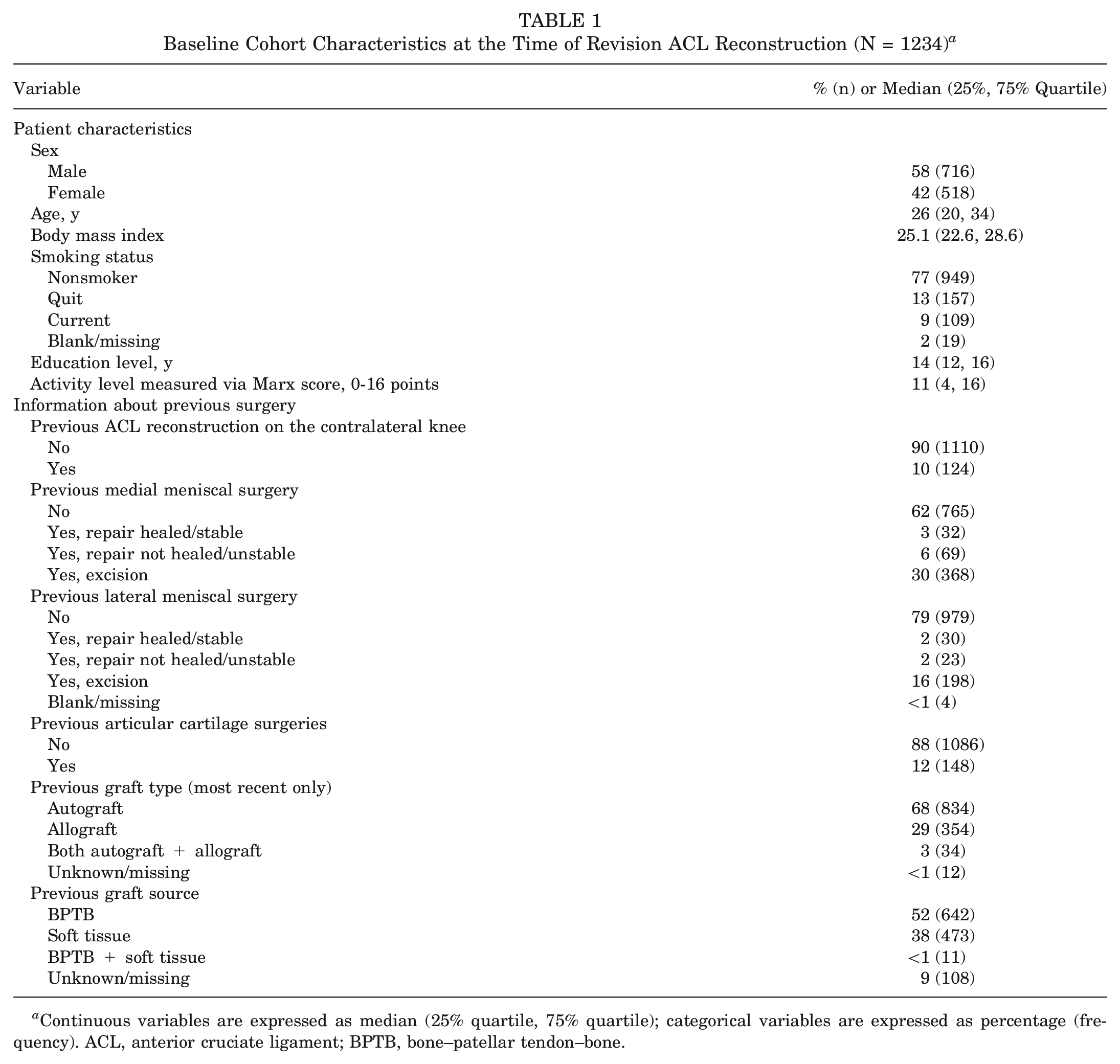
Continuous variables are expressed as median (25% quartile, 75% quartile); categorical variables are expressed as percentage (frequency). ACL, anterior cruciate ligament; BPTB, bone–patellar tendon–bone.
Surgical Approach
Several surgical factors at the time of revision surgery were found to be significant drivers of poorer outcomes at 6 years (Appendix Table, available in the online version of this article). Use of a transtibial surgical approach compared with an anteromedial portal approach resulted in significantly improved KOOS pain and quality of life (QOL) subscale scores (OR, 1.5; 95% CI, 1.02-2.2; P≤ .04) as well as WOMAC stiffness scores (OR, 1.46; 95% CI, 1.00-2.14; P = .05) at 6 years. Using an anteromedial portal approach compared with a 2-incision/rear-entry approach resulted in significantly improved patient activity levels at 6 years (OR, 1.67; 95% CI, 1.07-2.63; P = .024). Surgical approach did not influence graft rerupture rates at 6 years. Outcomes were not predicted according to which individual surgeon performed the revision ACL reconstruction.
Tunnel Position
Regarding tunnel position at the time of the revision surgery, patients whose surgeons noted that the tibial tunnel aperture was in the “optimum position” fared significantly worse in terms of 6-year IKDC scores (OR, 0.6; 95% CI, 0.4-0.8; P = .003), Marx activity levels (OR, 0.20; 95% CI, 0.07-0.6; P = .005), and KOOS symptoms, pain, sports/recreation, and QOL subscale scores (OR range, 0.56-0.68; 95% CI, 0.38-1.00; P≤ .05) compared with patients whose surgeons opted for either a blended new tunnel or noted that the previous tunnel had the same tunnel aperture but a “compromised position.”
Graft Fixation
The most consistent surgical variables driving outcome in patients with revision surgery were related to femoral and tibial fixation. Using an interference screw for femoral fixation compared with a cross-pin resulted in significantly better outcomes in 6-year IKDC scores (OR, 2.2; 95% CI, 1.2-3.9; P = .008) (Table 2) and KOOS sports/recreation and QOL subscale scores (OR range, 2.2-2.7; 95% CI, 1.2-4.8; P < .01). Using an interference screw compared with a cross-pin for femoral graft fixation resulted in a 2.6 times less likelihood of having a subsequent surgery within the 6 years (P = .02) (Table 2).
Baseline Surgical Characteristics at the Time of Revision ACL Reconstruction (N = 1234) a

Continuous variables are expressed as median (25% quartile, 75% quartile); categorical variables are expressed as percentage (frequency). ACL, anterior cruciate ligament; BPTB, bone–patellar tendon–bone; LFC, lateral femoral condyle; LTP, lateral tibial plateau; MFC, medial femoral condyle; MTP, medial tibial plateau.
Manufactured by DePuy Synthes.
The suture button/Endobutton (Smith & Nephew) construct was the second most commonly used femoral fixation method in our revision cohort (21%) (Table 2) compared with the interference screw (56%). We did not find that the interference screw had any significant advantage over the suture button, nor did the suture button have any advantage over the cross-pin (P value range, .10-.88).
Using an interference screw for tibial fixation compared with any combination of tibial fixation techniques resulted in significantly improved IKDC score (OR, 1.96; 95% CI, 1.3-2.9; P = .001); KOOS pain, activities of daily living (ADL), and sports/recreation subscale scores (OR range, 1.5-1.6; 95% CI, 1.0-2.4; P < .05); and WOMAC pain and ADL subscale scores (OR range, 1.5-1.8; 95% CI, 1.0-2.7; P < .05). Biologic enhancement (eg, platelet-rich plasma) at the time of revision surgery did not influence 6-year outcomes in this cohort.
General
Lower baseline outcome scores, lower baseline activity level, being a smoker at the time of the revision, older age, female sex, higher body mass index, shorter time since the patient’s last ACL reconstruction, having a previous ACL reconstruction on the contralateral side, and the surgeon’s revision being his or her own failed procedure all significantly increased the odds of reporting poorer clinical outcomes at 6 years (Table 2). No surgical factors were found to influence the incidence of having a subsequent ACL reconstruction after the index ACL revision surgery (Table 2).
Discussion
Surgical predictors of outcome for revision ACL reconstruction are believed to be a critical issue. The ability to improve what are typically poor outcomes after revision ACL reconstruction depends upon the ability to make decisions at the time of revision that would improve outcomes. Unfortunately, many factors are beyond the control of the surgeon. Our study demonstrates some factors that the surgeon can consider when planning the approach to revision ACL reconstruction. One of the challenges in discussing the results of studies by the MARS Group is the lack of comparison cohorts in the literature. This becomes increasingly relevant as the cohort moves past 5 years of follow-up. Currently, in the literature, only 259 patients have been followed for >5 years and reported on.1,3,6,10,12,13,15 Also, unless a prospective study design was used, many of these surgical factors may not have been measured or identified in other case series. Thus, our report on 980 patients with 6-year follow-up remains unique.
Demographically, the cohort remained consistent with cohorts in previously reported revision ACL reconstruction series. 8 Our follow-up continued to be strong, with 79% of the 1234 patients followed for a minimum of 6 years. Like most ACL series, the majority of our patients were men (58%).
Despite the increasing popularity of the anteromedial portal approach for drilling the femoral tunnel, our results did not demonstrate a clear superiority of the anteromedial approach at 6 years. Transtibial approach was a predictor of improved WOMAC stiffness and KOOS pain and QOL scores. In the previous review of this cohort at 2-year follow-up, a difference in outcomes was not noted between transtibial and anteromedial technique. 8 Most surgeons would not change their transtibial versus anteromedial versus 2-incision approach based on KOOS pain and QOL outcomes at 6 years but rather would be more likely to change their surgical approach if the rerupture rate at 6 years was affected. 18 We have been unable to demonstrate that surgical approach (or, independently, the surgeon performing that approach) affects graft rerupture at 6 years. Arthrotomy, a 2-incision approach, and notchplasty, which were predictors of worse outcome at 2 years, did not affect outcomes with further follow-up at 6 years.
Consistent with the findings at 2-year follow-up, 8 a surgeon’s determination that the tibial tunnel was in the “optimum” position resulted in worse outcomes for 6-year IKDC scores; Marx activity levels; and KOOS symptoms, pain, sports/recreation, and QOL scores. This was in comparison to surgeons who either opted for a blended new tunnel or noted that the previous tunnel had the same tunnel aperture but a “compromised position.” The finding that patients who had a new tibial tunnel drilled when the previous tunnel position was poor were not affected is in contradistinction to the findings at 2 years where this resulted in poor outcomes. Femoral tunnel issues affected only the KOOS pain outcome in this study, in that an “entirely new tunnel” produced a significantly poorer KOOS pain score at 6 years compared with a femoral tunnel in the “optimum position,” as determined by the operating surgeon. At 2 years, a double tunnel on the femur resulted in worse patient-reported outcome scores. These findings raise questions about whether the tibial tunnel has more effect than surgeons typically believe and whether our ability to judge the proper tibial tunnel is inconsistent or impaired in many situations.
Graft fixation was a significant predictor of outcome at 6 years after revision ACL reconstruction. Use of interference screws for femoral fixation continued to result in improved outcomes compared with other fixation methods. At 6 years, the use of interference screws affected IKDC, KOOS sports/recreation, and KOOS QOL scores. Using an interference screw compared with a cross-pin resulted in a patient being 2.6 times less likely to have a subsequent surgery within the 6 years. This is similar to the 2-year findings where KOOS symptoms, pain, and QOL scores as well as WOMAC stiffness scores were affected. 8 Use of the tibial interference screw also predicted improved outcomes at 6 years. Improved outcomes at 6 years were demonstrated by the IKDC score; the KOOS pain, ADL, and sports/recreation subscale scores; and the WOMAC pain and ADL subscale scores. Similarly, we demonstrated at 2 years that using a metal interference screw for current revision tibial fixation (compared with using a combination of fixation devices) was associated with significantly better IKDC and WOMAC stiffness scores. 8 Graft fixation in the primary ACL reconstruction setting has rarely demonstrated a significant effect on outcome.4,5,11,14,16,17,20 It is uncertain why graft fixation has a more significant effect in the revision ACL reconstruction setting. This effect may be related to previous old, sclerotic, and slightly enlarged tunnels and providing the most significant fixation via an interference screw, but these subtle nuances are difficult to measure. Previously, this group published a study of bone grafting demonstrating worse baseline and 2-year patient-reported outcomes and activity levels in patients who underwent 2-stage bone grafting compared with patients who underwent no bone grafting or single-stage bone grafting. 2 This group has also previously reported that similar to graft fixation, the graft choice and meniscal and chondral pathology significantly affect 6-year outcomes.18,19 Specifically, use of a BPTB autograft for revision resulted in patients being 4.2 times less likely to sustain a subsequent graft rupture compared with use of a BPTB allograft (P = .011; 95% CI, 1.56-11.27). No significant differences were found in graft rerupture rates between BPTB autograft and soft tissue autografts (P = .87) or between BPTB autografts and soft tissue allografts (P = .36). 18 Meniscal and chondral pathology at the time of revision ACL reconstruction had continued significant detrimental effects on patient-reported outcomes at 6 years after revision ACL surgery; the most consistent factors driving poorer outcomes were having a medial meniscal excision (either before or at the time of revision surgery) and the presence of patellofemoral articular cartilage pathology at the time of the revision surgery. 19
Strengths of the study include the prospective data collection of validated patient-reported outcome measures with the largest prospective revision ACL reconstruction cohort collected to date. This allows multivariable analysis to control for a large number of variables. Limitations and weaknesses include no onsite follow-up, surgeon variation in tunnel drilling as to blended versus previous tunnel use, and inability to control indications for tunnel placement and fixation choice by surgeons.
Conclusions
There are surgical variables at the time of an ACL revision that can modify clinical outcomes at 6 years. Opting for a transtibial surgical approach and choosing an inference screw for femoral and tibial fixation improved the patients’ odds of having a significantly better 6-year clinical outcome in this cohort.
Supplemental Material
sj-xlsx-1-ajs-10.1177_03635465241288227 – Supplemental material for Surgical Predictors of Clinical Outcome 6 Years After Revision ACL Reconstruction
Supplemental material, sj-xlsx-1-ajs-10.1177_03635465241288227 for Surgical Predictors of Clinical Outcome 6 Years After Revision ACL Reconstruction by Rick W. Wright, Laura J. Huston, Amanda K. Haas, Jacquelyn S. Pennings, Christina R. Allen, Daniel E. Cooper, Thomas M. DeBerardino, Warren R. Dunn, Brett (Brick) A. Lantz, Kurt P. Spindler, Michael J. Stuart, Annunziato (Ned) Amendola, Christopher C. Annunziata, Robert A. Arciero, Bernard R. Bach Jr, Champ L. Baker, Arthur R. Bartolozzi, Keith M. Baumgarten, Jeffrey H. Berg, Geoffrey A. Bernas, Stephen F. Brockmeier, Robert H. Brophy, Charles A. Bush-Joseph, J. Brad Butler V, James L. Carey, James E. Carpenter, Brian J. Cole, Jonathan M. Cooper, Charles L. Cox, R. Alexander Creighton, Tal S. David, David C. Flanigan, Robert W. Frederick, Theodore J. Ganley, Charles J. Gatt Jr, Steven R. Gecha, James Robert Giffin, Sharon L. Hame, Jo A. Hannafin, Christopher D. Harner, Norman Lindsay Harris Jr, Keith S. Hechtman, Elliott B. Hershman, Rudolf G. Hoellrich, David C. Johnson, Timothy S. Johnson, Morgan H. Jones, Christopher C. Kaeding, Ganesh V. Kamath, Thomas E. Klootwyk, Bruce A. Levy, C. Benjamin Ma, G. Peter Maiers, Robert G. Marx, Matthew J. Matava, Gregory M. Mathien, David R. McAllister, Eric C. McCarty, Robert G. McCormack, Bruce S. Miller, Carl W. Nissen, Daniel F. O’Neill, Brett D. Owens, Richard D. Parker, Mark L. Purnell, Arun J. Ramappa, Michael A. Rauh, Arthur C. Rettig, Jon K. Sekiya, Kevin G. Shea, Orrin H. Sherman, James R. Slauterbeck, Matthew V. Smith, Jeffrey T. Spang, Colonel (retired) Steven J. Svoboda, Timothy N. Taft, Joachim J. Tenuta, Edwin M. Tingstad, Armando F. Vidal, Darius G. Viskontas, Richard A. White, James S. Williams Jr, Michelle L. Wolcott, Brian R. Wolf and James J. York in The American Journal of Sports Medicine
Footnotes
Acknowledgements
The authors express their appreciation to the late Barton Mann, PhD (AOSSM, Rosemont, IL, USA), Timothy M. Hosea, MD (University Orthopaedic Associates LLC, Princeton, NJ, USA), Allen F. Anderson, MD (Tennessee Orthopaedic Alliance, Nashville, TN, USA), and Jeffery R. Bechler, MD (University Orthopaedic Associates LLC, Princeton, NJ, USA), whose contribution to this work was of great significance.
The authors sincerely appreciate Elizabeth Garofoli’s years of dedicated work and effort on this study. The authors also extend their gratitude to Jack T. Andrish, MD (Cleveland Clinic, Cleveland, OH, USA), John D. Campbell, MD (Bridger Orthopedic and Sports Medicine, Bozeman, MT, USA), John P. Albright, MD (University of Iowa Hospitals and Clinics, Iowa City, IA, USA), and Diane L. Dahm, MD (Mayo Clinic, Rochester, MN, USA) for their effort and leadership on this project. All are enjoying a well-deserved and happy retirement after many years of dedication to the advancement of orthopaedics.
Authors
The MARS Group; Rick W. Wright, MD (Vanderbilt University, Nashville, TN, USA); Laura J. Huston, MS (Vanderbilt University, Nashville, TN, USA); Amanda K. Haas, MA (Washington University in St Louis, St Louis, MO, USA); Jacquelyn S. Pennings, PhD (Vanderbilt University, Nashville, TN, USA); Christina R. Allen, MD (Yale University, New Haven, CT, USA); Daniel E. Cooper, MD (W.B. Carrell Memorial Clinic, Dallas, TX, USA); Thomas M. DeBerardino, MD (UT Health, San Antonio, TX, USA); Warren R. Dunn, MD, MPH (Fondren Orthopedic Group, Houston, TX, USA); Brett (Brick) A. Lantz, MD (Slocum Research and Education Foundation, Eugene, OR, USA); Kurt P. Spindler, MD (Cleveland Clinic, Cleveland, OH, USA); Michael J. Stuart, MD (Mayo Clinic, Rochester, MN, USA); Annunziato (Ned) Amendola, MD (Duke University, Durham, NC, USA); Christopher C. Annunziata, MD (Commonwealth Orthopaedics & Rehabilitation, Arlington, VA, USA); Robert A. Arciero, MD (University of Connecticut Health Center, Farmington, CT, USA); Bernard R. Bach Jr, MD (Rush University Medical Center, Chicago, IL, USA); Champ L. Baker III, MD (The Hughston Clinic, Columbus, GA, USA); Arthur R. Bartolozzi, MD (3B Orthopaedics, University of Pennsylvania Health System, Philadelphia, PA, USA); Keith M. Baumgarten, MD (Orthopedic Institute, Sioux Falls, SD, USA); Jeffrey H. Berg, MD (Town Center Orthopaedic Associates, Reston, VA, USA); Geoffrey A. Bernas, MD (State University of New York at Buffalo, Buffalo, NY, USA); Stephen F. Brockmeier, MD (University of Virginia, Charlottesville, VA, USA); Robert H. Brophy, MD (Washington University in St Louis, St Louis, MO, USA); Charles A. Bush-Joseph, MD (Rush University Medical Center, Chicago, IL, USA); J. Brad Butler V, MD (Orthopedic and Fracture Clinic, Portland, OR, USA); James L. Carey, MD, MPH (University of Pennsylvania, Philadelphia, PA, USA); James E. Carpenter, MD (University of Michigan, Ann Arbor, MI, USA); Brian J. Cole, MD (Rush University Medical Center, Chicago, IL, USA); Jonathan M. Cooper, DO (HealthPartners Specialty Center, St Paul, MN, USA); Charles L. Cox, MD, MPH (Vanderbilt University, Nashville, TN, USA); R. Alexander Creighton, MD (University of North Carolina Medical Center, Chapel Hill, NC, USA); Tal S. David, MD (Synergy Specialists Medical Group, San Diego, CA, USA); David C. Flanigan, MD (The Ohio State University, Columbus, OH, USA); Robert W. Frederick, MD (The Rothman Institute/Thomas Jefferson University, Philadelphia, PA, USA); Theodore J. Ganley, MD (Children’s Hospital of Philadelphia, Philadelphia, PA, USA); Charles J. Gatt Jr, MD (University Orthopaedic Associates LLC, Princeton, NJ, USA); Steven R. Gecha, MD (Princeton Orthopaedic Associates, Princeton, NJ, USA); James Robert Giffin, MD (Fowler Kennedy Sport Medicine Clinic, University of Western Ontario, London, ON, Canada); Sharon L. Hame, MD (University of California Los Angeles, Los Angeles, CA, USA); Jo A. Hannafin, MD, PhD (Hospital for Special Surgery, New York, NY, USA); Christopher D. Harner, MD (University of Texas Health Center, Houston, TX, USA); Norman Lindsay Harris Jr, MD (Grand River Health, Rifle, CO, USA); Keith S. Hechtman, MD (Miami Orthopedics and Sports Medicine Institute, Coral Gables, FL, USA); Elliott B. Hershman, MD (Lenox Hill Hospital, New York, NY, USA); Rudolf G. Hoellrich, MD (Slocum Research and Education Foundation, Eugene, OR, USA); David C. Johnson, MD, (National Sports Medicine Institute, Leesburg, VA, USA); Timothy S. Johnson, MD (National Sports Medicine Institute, Leesburg, VA, USA); Morgan H. Jones, MD (Brigham and Women’s Hospital, Boston, MA, USA); Christopher C. Kaeding, MD (The Ohio State University, Columbus, OH, USA); Ganesh V. Kamath, MD (University of North Carolina Medical Center, Chapel Hill, NC, USA); Thomas E. Klootwyk, MD (Forte Sports Medicine and Orthopedics, Indianapolis, IN, USA); Bruce A. Levy, MD (Orlando Health, Orlando, FL, USA); C. Benjamin Ma, MD (University of California, San Francisco, CA, USA); G. Peter Maiers II, MD (Forte Sports Medicine and Orthopedics, Indianapolis, IN, USA); Robert G. Marx, MD (Hospital for Special Surgery, New York, NY, USA); Matthew J. Matava, MD (Washington University in St Louis, St Louis, MO, USA); Gregory M. Mathien, MD (Knoxville Orthopedic Clinic, Knoxville, TN, USA); David R. McAllister, MD (University of California Los Angeles, Los Angeles, CA, USA); Eric C. McCarty, MD (University of Colorado Denver School of Medicine, Denver, CO, USA); Robert G. McCormack, MD (University of British Columbia/Fraser Health Authority, Vancouver, BC, Canada); Bruce S. Miller, MD, MS (University of Michigan, Ann Arbor, MI, USA); Carl W. Nissen, MD (Connecticut Children’s Medical Center, Hartford, CT, USA); Daniel F. O’Neill, MD, EdD (The Alpine Clinic, Plymouth, NH, USA); Brett D. Owens, MD (Warren Alpert Medical School, Brown University, Providence, RI, USA); Richard D. Parker, MD (Cleveland Clinic, Cleveland, OH, USA); Mark L. Purnell, MD (Valley Ortho, Aspen, CO, USA); Arun J. Ramappa, MD (Beth Israel Deaconess Medical Center, Boston, MA, USA); Michael A. Rauh, MD (State University of New York at Buffalo, Buffalo, NY, USA); Arthur C. Rettig, MD (Forte Sports Medicine and Orthopedics, Indianapolis, IN, USA); Jon K. Sekiya, MD (University of Michigan, Ann Arbor, MI, USA); Kevin G. Shea, MD (Stanford University, Palo Alto, CA, USA); Orrin H. Sherman, MD (NYU Hospital for Joint Diseases, New York, NY, USA); James R. Slauterbeck, MD (UNC Health Southeastern, Lumberton, NC, USA); Matthew V. Smith, MD (Washington University in St Louis, St Louis, MO, USA); Jeffrey T. Spang, MD (University of North Carolina Medical Center, Chapel Hill, NC, USA); Colonel (retired) Steven J. Svoboda, MD (MedStar Orthopaedic and Sports Center, Washington, DC, USA); Timothy N. Taft, MD (University of North Carolina Medical Center, Chapel Hill, NC, USA); Joachim J. Tenuta, MD (Albany Medical Center, Albany, NY, USA); Edwin M. Tingstad, MD (Inland Orthopaedic Surgery and Sports Medicine Clinic, Pullman, WA, USA); Armando F. Vidal, MD (The Steadman Clinic, Vail, CO, USA); Darius G. Viskontas, MD (Fraser Orthopaedic Institute, New Westminster, Vancouver, BC, Canada); Richard A. White, MD (Fitzgibbon’s Hospital, Marshall, MO, USA); James S. Williams Jr, MD (Cleveland Clinic, Euclid, OH, USA); Michelle L. Wolcott, MD (University of Colorado Anschutz Medical Campus, Aurora, CO, USA); Brian R. Wolf, MD (University of Iowa Hospitals and Clinics, Iowa City, IA, USA); James J. York, MD (Luminis Health Orthopedics, Pasadena, MD, USA).
Submitted June 6, 2023; accepted July 8, 2024.
Presented at the annual meeting of the AOSSM, Nashville, Tennessee, July 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This project was funded by grant No. 5R01-AR060846 from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. All author disclosures are listed in the Appendix (available in the online version of this article). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
