Abstract
Background:
Meniscal preservation has been demonstrated to contribute to long-term knee health and has been a successful intervention in isolation and in patients with anterior cruciate ligament reconstruction (ACLR). The long-term results of meniscal repair in the setting of revision ACLR have yet to be documented.
Purpose:
To report the incidence of meniscal repair failures at the 6-year follow-up in a cohort of patients who underwent concurrent revision ACLR and primary meniscal repair.
Study Design:
Prospective cohort study; Level of evidence, 2.
Methods:
All revision ACLRs with concomitant primary meniscal repair cases from a multicenter group between 2006 and 2011 were selected. Six-year follow-up was obtained to determine whether any subsequent surgery had occurred since their initial revision ACLR. If so, operative reports were obtained, whenever possible, to verify pathological condition and treatment.
Results:
In total, 221 patients from 1234 revision ACLRs underwent concurrent primary meniscal repairs (18% of the cohort). There were 238 repairs performed: 173 medial and 65 lateral. The majority of these repairs (n = 181; 76%) were performed with an all-inside technique. Six-year surgical follow-up was obtained in 77% (171/221) of the cohort, or 189 of 238 (79%) of the repairs (136 medial, 53 lateral). The meniscal repair failure rate, defined as reoperation, was 16% (31/189) at 6 years. Of the 31 failures, 28 were medial (24 all-inside, 4 inside-out; 28/136 = 20.6% failure rate) and 3 were lateral (2 all-inside, 1 inside-out; 3/53 = 5.7% failure rate). Three medial failures were treated in conjunction with a subsequent repeat revision ACLR. Medial tears underwent reoperation for failure at a significantly higher rate than lateral tears (20.6% vs 5.7%;
Conclusion:
Meniscal repair in the revision ACLR setting has a 16% failure rate at 6 years. Failure rates for medial tears (20.6%) were found to be higher than that for lateral tears (5.7%), which aligns with previous studies in both the revision and primary ACLR setting.
Meniscal injury is often seen in conjunction with anterior cruciate ligament (ACL) injuries.36,40,45,50 In both ACL-intact and primary ACL-reconstructed knees, meniscal repair has been shown to be effective in the short term, due to its advantageous properties of secondary knee stabilization and distribution of tibiofemoral contact forces.9,21 The integrity of the meniscus transmits axial loads across the joint surface evenly via a tensile stress referred to as “hoop stress,” which prevents uneven loading and cartilage breakdown.16,18 When the meniscus is repaired appropriately, it is thought to regain the ability to evenly distribute hoop stresses. 22 Previous studies have reported that 4% to 16% of meniscal repairs undergo reoperation at the 2-year follow-up in the setting of primary ACL reconstruction (ACLR).11,19,23,27,48,51 In studies with a minimum of 5 years of follow-up, meniscal repair reoperation rates increased to 11% to 28%.3,32,53 Two systematic reviews looking at a total of 2180 meniscal repairs with follow-up >5 years showed pooled failure rates of 20% and 23%, respectively.33,34
It has been well documented that revision ACLRs have more complications and poorer outcomes compared with primary ACLRs.10,15,20,54,55 This can be attributed to multiple factors, including the potential for additional meniscal and chondral damage, lower extremity malalignment, and differences in graft choice. 28 A previous study by the MARS Group conducted on the same cohort of revision ACLR patients evaluated primary meniscal repair at the 2-year follow-up and showed a failure rate of 8.6% at 2 years. 29 Previously documented risk factors for meniscal repair failure include the presence of bucket-handle tears and lack of integrity of the collateral ligaments.6,38,49 The multicenter approach to this study is advantageous as it demonstrates the reproducibility of successful repair across different sites among multiple surgeons of various training backgrounds while using a variety of surgical techniques and implants. The multicenter approach also increases cohort size, which allows for the formulation of relevant conclusions and increases generalizability.
The purpose of this study was to perform a 6-year follow-up inclusive of a previously reported cohort of patients 29 who underwent concurrent revision ACLR and primary meniscal repair. While primary meniscal repair in the setting of revision ACLR has demonstrated low reoperation rates at 2 years, we hypothesized that several knee-specific patient-reported outcome measures (PROMs) would deteriorate and ipsilateral reoperations for meniscal pathology would increase at the 6-year follow-up. Despite this hypothesis, we postulated that meniscal repair would still demonstrate relatively low failure rates at the 6-year follow-up and prove to be a successful management strategy in the setting of revision ACLR.
Methods
Study Design and Setting
The MARS Group was assembled in cooperation with the American Orthopaedic Society for Sports Medicine, originally consisting of 83 sports medicine fellowship-trained surgeons working at 52 sites. The surgeons were a mix of academic (54%) and private (46%) practitioners. Surgeons performed specific ACL revision techniques based on their own practice preferences. If an allograft was chosen for reconstruction, the surgeon was required to use a Musculoskeletal Transplant Foundation (MTF) graft to standardize and record allograft preparation methods (MTF Biologics; Edison). 2 The objective of this consortium is to assess outcomes after revision ACLR over time and determine how the risk factors and treatment at the time of revision surgery are associated with ACL revision outcomes. This study design involves a longitudinal prospective cohort.
Study Patients
After institutional review board approval from each institution, 1234 patients with documented ACLR failure who underwent revision ACLR surgery between 2006 and 2011 qualified for and provided consent to be in our prospective, longitudinal cohort study. Study inclusion criteria were revision ACLRs performed by a study surgeon on ACL-deficient knees that had failed a previous ACLR as identified by either magnetic resonance imaging (MRI), physical examination (positive pivot shift and Lachman test), KT-1000 testing demonstrating >5-mm side-to-side difference, functional instability, or arthroscopic confirmation. Surgeons were given the freedom to perform the surgeries as they saw best fit. The only surgical requirement was that in an allograft case, an MTF graft had to be used to standardize the graft source across sites. Patients were excluded if they had a multiligament reconstruction, infection, or fracture. For this particular study, all revision ACLRs that had concomitant primary meniscal repair cases were identified and selected.
Data Sources
After informed consent was obtained, each patient completed a self-reported questionnaire examining demographics, injury characteristics, sports participation history, and health status before their revision ACLR surgery. Within this questionnaire, each patient completed a series of validated general and knee-specific outcome instruments, including the Knee injury and Osteoarthritis Outcome Score (KOOS), 42 the International Knee Documentation Committee (IKDC) 12 subjective form, and the Marx activity rating scale. 30 Surgeons completed a questionnaire that included previous surgical history (if known), current revision surgical technique utilized, intra-articular findings, and surgical management of meniscal and chondral pathology.
Completed forms were mailed from each site to our data coordinating center (Vanderbilt University Medical Center, Nashville, Tennessee) and scanned using TeleForm software (OpenText), and data were verified and exported to a master study database. A series of custom logical error and quality control checks were systematically performed before data analyses.
Patient Follow-up
At 6 years, the same questionnaire was administered as at baseline and the 2-year follow-up to all eligible patients in the study who were able to be contacted (patients who had previously refused or withdrawn, were currently incarcerated, were known to have had a total knee arthroplasty on the involved knee, or had died were excluded). Patients were also contacted by telephone or email to determine if subsequent graft failure and/or any additional knee surgery had occurred. If so, operative reports were obtained (when possible) to verify the patient's pathology and treatment.
Statistical Analysis
Descriptive statistics of variables were calculated. Baseline characteristics and surgical variables were compared between patients who had a documented subsequent meniscal repair failure procedure and patients whose index meniscal repairs did not fail (as defined by not having undergone a subsequent meniscus-related surgical procedure). To examine evidence for unadjusted associations with meniscal failure status, Wilcoxon rank-sum tests were used for continuous variables and Pearson chi-square tests for categorical variables. Cox regression was used to analyze time-to-event data. Statistical analysis was performed using open-source R statistical software (www.cran.r-project.org). A
Results
In total, 221 patients from 1234 revision ACLRs underwent 238 concurrent primary meniscal repairs (18% of the cohort). Of these 238 repairs, 173 were medial and 65 lateral. The majority of these repairs (n = 181; 76%) were performed with an all-inside technique (Table 1). Six-year surgical follow-up was obtained on 77% (171/221) of the cohort, or 189 of 238 (79%) of the repairs (136 medial, 53 lateral).
Patient and Surgical Characteristics Between the Meniscal Failure and Nonfailure Groups
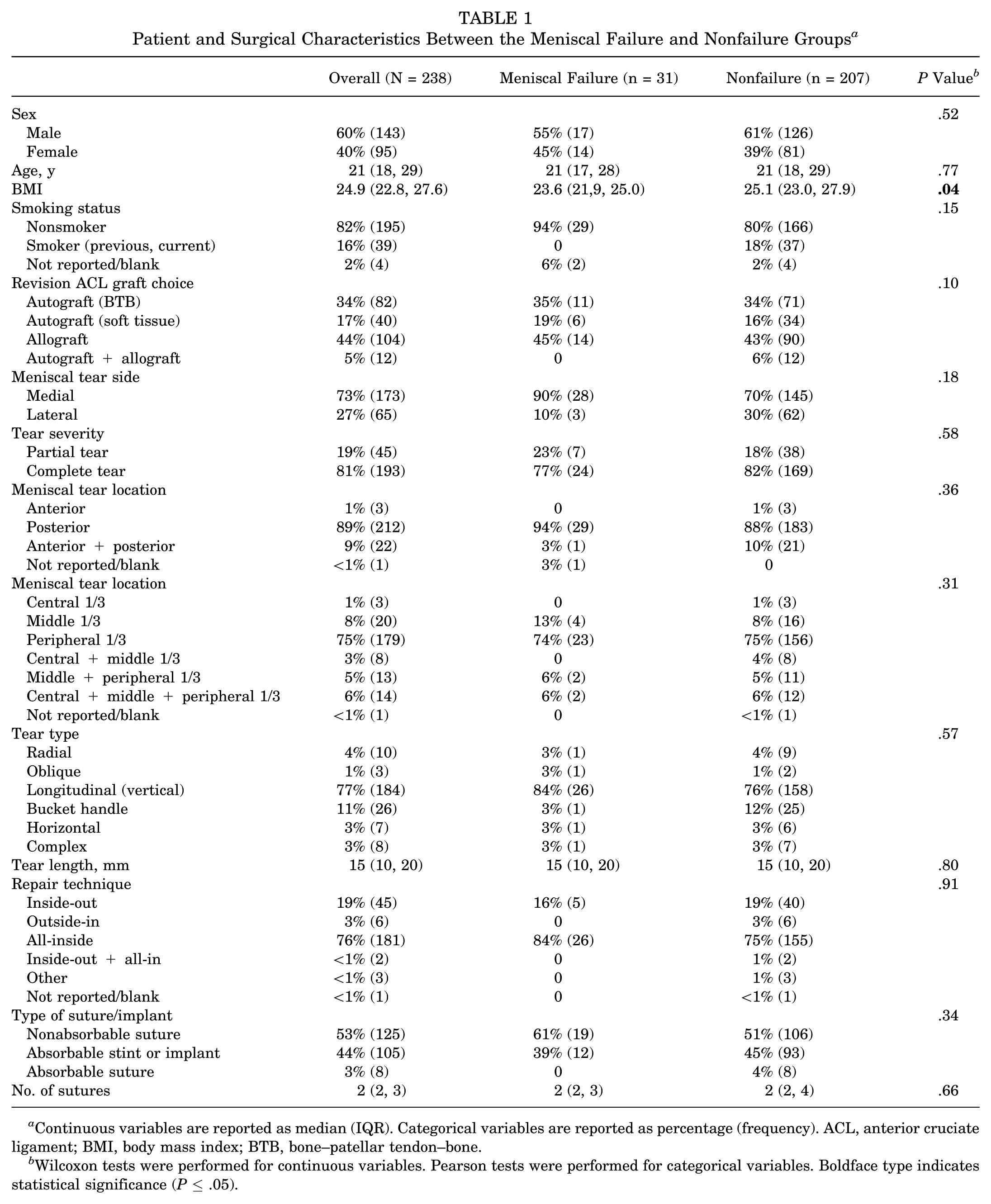
Continuous variables are reported as median (IQR). Categorical variables are reported as percentage (frequency). ACL, anterior cruciate ligament; BMI, body mass index; BTB, bone–patellar tendon–bone.
Wilcoxon tests were performed for continuous variables. Pearson tests were performed for categorical variables. Boldface type indicates statistical significance (
Overall, the meniscal repair failure rate (defined as reoperation) was 16% (31/189) at 6 years. Of the 31 failures, 28 were medial (24 all-inside, 4 inside-out; 28/136 = 20.6% failure rate) and 3 were lateral (2 all-inside, 1 inside-out; 3/53 = 5.7% failure rate). Three medial failures were treated in conjunction with a subsequent repeat revision ACLR. None of the lateral failures had an associated ACL graft failure needing repeat revision ACLR. Medial tears underwent reoperation for failure at a significantly higher rate than lateral tears (20.6% vs 5.7%;
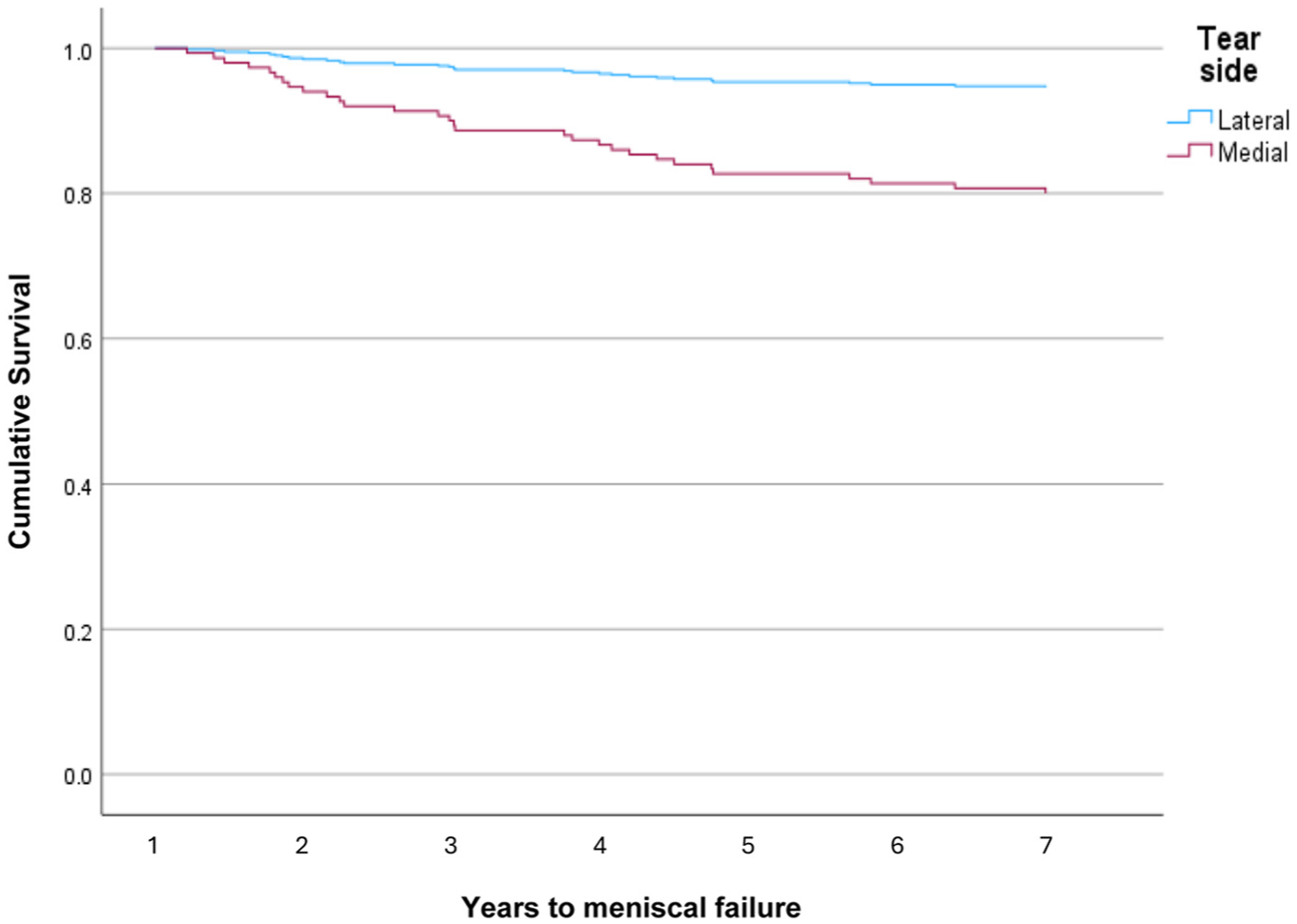
Survival analysis between medial and lateral meniscus repairs.
Table 1 stratifies the patient and surgical characteristics between the meniscal failure and nonfailure groups. The only significant difference found between the 2 groups was in body mass index (BMI), in that the failure group had a lower BMI than the nonfailure group (
Table 2 stratifies PROMs between the meniscal failure and nonfailure groups at baseline. Interestingly, the meniscal failure group exhibited better KOOS pain scores than the nonfailure group at baseline (median, 81 points vs 75 points;
Baseline Patient-Reported Outcome Characteristics Between the Meniscal Failure and Nonfailure Groups
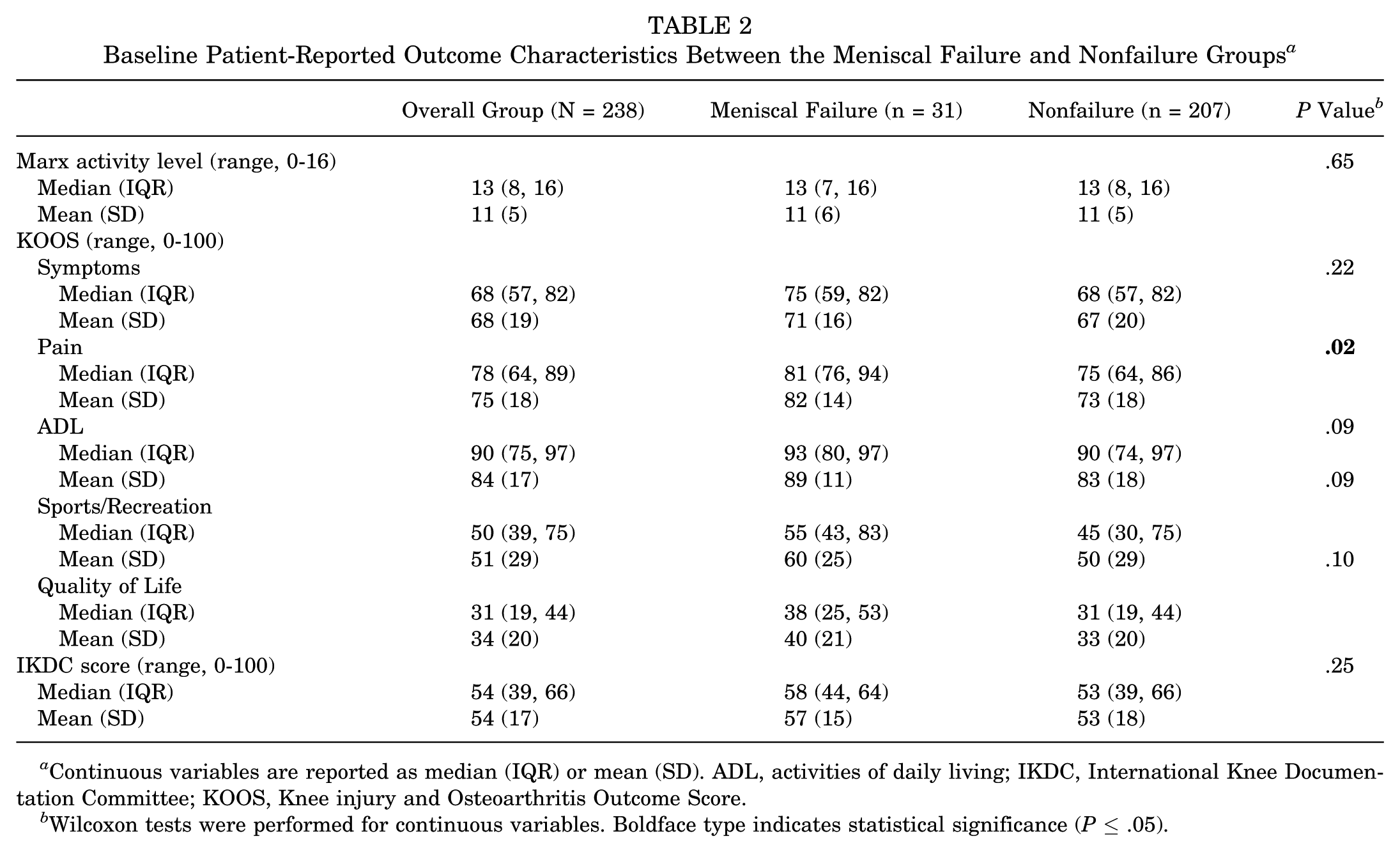
Continuous variables are reported as median (IQR) or mean (SD). ADL, activities of daily living; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score.
Wilcoxon tests were performed for continuous variables. Boldface type indicates statistical significance (
At 6 years, KOOS values and IKDC scores were both significantly improved compared with baseline, while Marx activity scores declined at the 6-year follow-up (Table 3). There were 2 statistically significant differences between the meniscal failure group and the nonfailure group at 6 years. First was the KOOS knee-related quality of life score, for which the meniscal failure group had a significantly lower score compared with the nonfailure group (median, 50 points vs 69 points;
Six-Year Patient-Reported Outcome Characteristics Between the Meniscal Failure and Nonfailure Groups
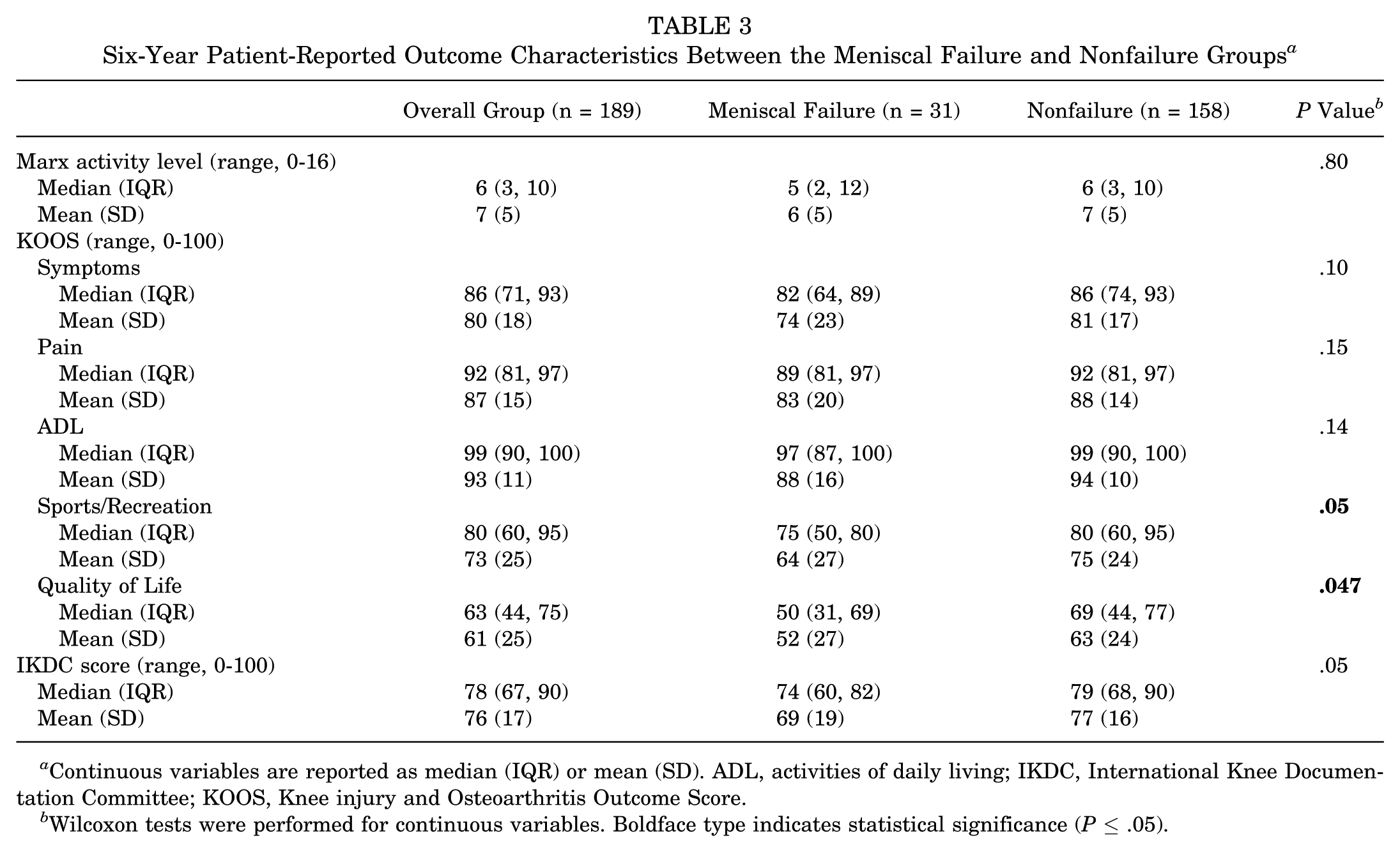
Continuous variables are reported as median (IQR) or mean (SD). ADL, activities of daily living; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score.
Wilcoxon tests were performed for continuous variables. Boldface type indicates statistical significance (
Discussion
In the setting of primary ACLR, meniscal repair has been demonstrated to have good results with relatively low failure rates in both short and midterm follow-up.3,19,23,27,32,48,51,53 When it comes to revision ACLR, there is only 1 prospective cohort study reported in the literature that evaluated the efficacy of primary meniscal repair, and it is the 2-year data for the cohort being described in this study. 29 However, in general, it is known that revision ACLRs have worse outcomes than in the primary setting.54,56 It is important for orthopaedic surgeons to understand the unique and challenging differences presented when considering meniscal repair in the setting of a revision ACLR. One example is the prevalence of articular cartilage injury, which is reported to increase 17% compared with primary ACLR. 57 The current study provides important longer-term follow-up on meniscal repairs in the revision ACLR setting.
The meniscal failure rate, defined as reoperation, reported in the current study with a minimum 6-year follow-up was 16%. This aligns with our hypothesis that as follow-up increases, we expect the number of reoperations to increase. This failure rate increased compared with our previously reported failure rate for meniscal repair in the setting of revision ACLR of 8.6% at the 2-year minimum follow-up. 29 One plausible explanation for this increase is the increased exposure to injury with time and return to activity. In the setting of primary ACLR, Lee and Diduch 23 showed that the failure rate of their meniscal repairs deteriorated from 10.4% at 2 years to 28.6% at 6 years. In their study, they attributed this partially to the fact that meniscal repair in conjunction with primary ACLR has been associated with incomplete meniscal healing, which 2 other studies cite at a rate of 15% 1 and 19%, 47 respectively, using second-look arthroscopy. Additionally, a study by Siebold et al 44 demonstrated an 8% increase in the meniscal repair failure rate in the primary ACLR setting from the 2- to 6-year follow-up, with a 23% medial retear rate and a 5% lateral retear rate.
Our data demonstrated a 6-year failure rate of 20.6% after medial meniscus repair and 5.7% after lateral meniscus repair. Our 2-year study also demonstrated a significantly higher rate of failure in medial repairs (8.8%) compared with lateral repairs (3.0%).
29
These results align with data reported in a systematic review conducted by Paxton et al,
37
which showed that medial meniscus repairs have higher failure rates compared with lateral repairs at the 10-year follow-up. In a systematic review with >5 years of follow-up, Nepple et al
33
reaffirmed the notion that medial repairs fail more frequently; they showed a failure rate of 23.9% in medial repairs and 12.6% in lateral repairs (
Few studies have documented validated PROMs when evaluating long-term follow-up of concurrent revision ACLR and meniscal repair. Our study reports the IKDC score, KOOS, and Marx activity rating level. The IKDC is a validated clinical assessment tool for evaluating outcomes in patients with ligamentous and meniscal knee injuries.12,13 Our previous study on combined revision ACLR and primary meniscal repair demonstrated mean IKDC scores of 73 (repair failure) and 75 (repair success) at the 2-year follow-up, with no significant difference between the failure and nonfailure groups. 29 This indicated that patients experienced successful outcomes at 2 years after combined revision ACLR and meniscal repair irrespective of meniscal healing status. Our 6-year follow-up data had IKDC mean scores of 76 (overall cohort), 69 (repair failure), and 77 (repair success), demonstrating sustained clinical success compared to baseline with longer-term follow-up. Melton et al 32 reported an IKDC score of 84.2 at a mean 7.7-year follow-up in 44 patients who underwent combined meniscal repair and primary ACLR. Westermann et al52,53 reported similar data in patients undergoing combined primary ACLR and meniscal repair with median IKDC scores of 87.4 in those who underwent successful repair at the 6-year follow-up compared with 47.1 at baseline. Additionally, Logan et al 25 examined 42 athletes who underwent meniscal repair either in isolation (17%) or combined with primary ACLR (83%), and they reported a mean IKDC score of 89.6 at a mean follow-up of 8.5 years. Our IKDC scores in the revision ACLR population are similar to those in previously reported primary ACLR patients, which is surprising given that revision patient populations are fraught with multiple confounding factors, possibly leading to worse functional outcomes. Additionally, at the 6-year follow-up, our cohort demonstrated improvement greater than the minimal clinically important difference (MCID), indicating clinical success of the combined surgery.
The MCID for each subscale in the KOOS is approximately 8 to 10 points. 41 The data from our 2-year follow-up of meniscal repairs in revision ACLR showed that each subscale of the KOOS improved by at least 8 points, 29 while the current 6-year follow-up of this cohort showed mean KOOS subscale scores rising by at least 9 points. The KOOS data from the 2- and 6-year follow-ups indicate that patients experienced successful outcomes as a result of combined revision ACLR and meniscal repair at 6 years.
The mean Marx activity level score at the 6-year follow-up in our current study deteriorated to 7 points, down from 11 points (of 16) at baseline. There is no validated study determining clinically significant differences in Marx scores, but a reduction of 50% is generally considered a true change. 53 Westermann et al 53 looked at meniscal repair combined with primary ACLR and found that Marx activity scores decreased from 12 to 9 to 7 points at baseline, 2-year follow-up, and 6-year follow-up, respectively. Their data in the primary ACLR setting shows a similar decrease to what we found in revision cases, indicating that meniscal repair in primary and revision ACLR potentially leads to similar functional status in short- and midterm follow-up.
It is important not only to address the ACL status when discussing success of meniscal repair, but also to investigate the method of fixation when evaluating the success of meniscal repair, as there are multiple techniques and fixation methods available. In our study, 76% (181/238) of the patients had all-inside meniscal repairs performed. There were 5 failures in the inside-out cohort (5/45; 11%), 0 failures in the outside-in cohort (0/6; 0%), and 26 failures in the all-inside group (26/181; 14%). When looking at pooled data, it has been shown that open, outside-in, inside-out, and all-inside repair have similar failure rates ranging from 22.3% to 24.3% at >5 years of follow-up. 28 The inside-out and all-inside techniques are the most common methods preferred by orthopaedic surgeons today, with inside-out preferred for larger tears (>3 cm) and bucket-handle tears requiring multiple sutures. 24 The popularity of the all-inside repair technique continues to increase, with improved implants and new data demonstrating equivalent efficacy to inside-out repair in treating complex pathologies such as bucket-handle tears.4,43
This study has multiple strengths due to the multicenter study design. The large prospective cohort allows us to perform a variety of analyses, which would not be possible in a single site study. The diverse group of surgeons across the country in both private practice and the academic setting allow for generalizability to other sports medicine physicians. An additional strength includes the use of a variety of validated PROMs.
This study is not without weaknesses. One major intuitive perceived weakness is that the large number of contributors from different locations could lead to a varied assessment and identification of meniscal pathology. To combat this weakness, we had meetings before launching the consortium to educate the involved surgeons and try to create consistency in diagnosis and approach. Another weakness is that it has been reported in the literature that fellowship-trained sports medicine surgeons can have good agreement and consistency in evaluating and treating patients with ACL and meniscal pathology.8,31 An additional weakness of the study is that our definition of failure as reoperation could underestimate the true number of failures. However, it is not practical, safe, or cost-efficient for all the patients in the study to undergo second-look arthroscopy or MRI after repair. One method of finding more failures would have been clinically assessing for symptoms of pain in the compartment where meniscal repair was performed, as pain could be an indicator of failure. An additional weakness is the lack of on-site follow-up for patients. While meaningful, we believe on-site evaluation of these patients is less critical to determine meniscal repair outcome compared with other revision ACL outcome measures that assess graft laxity and knee stability. However, we believe that radiographic analysis of these patients to assess subsequent joint space degradation is important in long-term follow-up and is a weakness in this current study. That said, a future plan is to obtain radiographs to assess sagittal and coronal alignment at the 10-year follow-up in a subset of this cohort.
Conclusion
At the 6-year follow-up, the overall primary meniscal repair failure rate as defined by reoperation was 16% in this ACL revision cohort, compared with 8.6% at 2 years. Failure rates for medial repairs were higher than those for lateral repairs, with a significantly shorter survival time in the medial compartment compared with the lateral compartment. No differences were found between meniscal repair failures and nonfailures when it came to tear type, tear length, repair technique utilized, suture/implant type, or number of sutures used between the 2 groups. Ultimately, this study demonstrates that meniscal repair, even in the revision ACLR setting, is worthwhile with a high rate of success.
Authors
MARS Group; Jake A. Fox, MD (Vanderbilt University, Nashville, Tennessee, USA); Laura J. Huston, MS (Vanderbilt University, Nashville, Tennessee, USA); Amanda K. Haas, MA (Washington University in St. Louis, St. Louis, Missouri, USA); Jacquelyn S. Pennings, PhD (Vanderbilt University, Nashville, Tennessee, USA); Christina R. Allen, MD (Yale University, New Haven, Connecticut, USA); Daniel E. Cooper, MD (W.B. Carrell Memorial Clinic, Dallas, Texas, USA); Thomas M. DeBerardino, MD (UT Health, San Antonio, Texas, USA); Warren R. Dunn, MD, MPH (Fondren Orthopedic Group, Houston, Texas, USA); Brett (Brick) A. Lantz, MD (Slocum Research and Education Foundation, Eugene, Oregon, USA); Kurt P. Spindler, MD (Cleveland Clinic, Cleveland, Ohio, USA); Michael J. Stuart, MD (Mayo Clinic, Rochester, Minnesota, USA); Annunziato (Ned) Amendola, MD (Duke University, Durham, North Carolina, USA); Christopher C. Annunziata, MD (Commonwealth Orthopaedics & Rehabilitation, Arlington, Virginia, USA); Robert A. Arciero, MD (University of Connecticut Health Center, Farmington, Connecticut, USA); Bernard R. Bach Jr., MD (Rush University Medical Center, Chicago, Illinois, USA); Champ L. Baker III, MD (The Hughston Clinic, Columbus, Georgia, USA); Arthur R. Bartolozzi, MD (3B Orthopaedics, University of Pennsylvania Health System, Philadelphia, Pennsylvania, USA); Keith M. Baumgarten, MD (Orthopedic Institute, Sioux Falls, South Dakota, USA); Jeffrey H. Berg, MD (Town Center Orthopaedic Associates, Reston, Virginia, USA); Geoffrey A. Bernas, MD (State University of New York at Buffalo, Buffalo, New York, USA); Stephen F. Brockmeier, MD (University of Virginia, Charlottesville, Virginia, USA); Robert H. Brophy, MD (Washington University in St. Louis, St. Louis, Missouri, USA); Charles A. Bush-Joseph, MD (Rush University Medical Center, Chicago, Illinois, USA); J. Brad Butler V, MD (Orthopedic and Fracture Clinic, Portland, Oregon, USA); James L. Carey, MD, MPH (University of Pennsylvania, Philadelphia, Pennsylvania, USA); James E. Carpenter, MD (University of Michigan, Ann Arbor, Michigan, USA); Brian J. Cole, MD (Rush University Medical Center, Chicago, Illinois, USA); Jonathan M. Cooper, DO (HealthPartners Specialty Center, St. Paul, Minnesota, USA); Charles L. Cox, MD, MPH (Vanderbilt University, Nashville, Tennessee, USA); R. Alexander Creighton, MD (University of North Carolina Medical Center, Chapel Hill, North Carolina, USA); Tal S. David, MD (Synergy Specialists Medical Group, San Diego, California, USA); David C. Flanigan, MD (The Ohio State University, Columbus, Ohio, USA); Robert W. Frederick, MD (The Rothman Institute/Thomas Jefferson University, Philadelphia, Pennsylvania, USA); Theodore J. Ganley, MD (Children’s Hospital of Philadelphia, Philadelphia, USA); Charles J. Gatt Jr., MD (University Orthopaedic Associates, Princeton, New Jersey, USA); Steven R. Gecha, MD (Princeton Orthopaedic Associates, Princeton, New Jersey, USA); James Robert Giffin, MD (Fowler Kennedy Sport Medicine Clinic, University of Western Ontario, London, Ontario, Canada); Sharon L. Hame, MD (University of California, Los Angeles, California, USA); Jo A. Hannafin, MD, PhD (Hospital for Special Surgery, New York, New York, USA); Christopher D. Harner, MD (University of Texas Health Center, Houston, Texas, USA); Norman Lindsay Harris Jr., MD (Grand River Health, Rifle, Colorado, USA); Keith S. Hechtman, MD (Miami Orthopedics and Sports Medicine Institute, Coral Gables, Florida, USA); Elliott B. Hershman, MD (Lenox Hill Hospital, New York, New York, USA); Rudolf G. Hoellrich, MD (Slocum Research and Education Foundation, Eugene, Oregon, USA); David C. Johnson, MD (National Sports Medicine Institute, Leesburg, Virginia, USA); Timothy S. Johnson, MD (National Sports Medicine Institute, Leesburg, Virginia, USA); Morgan H. Jones, MD (Brigham and Women's Hospital, Boston, Massachusetts, USA); Christopher C. Kaeding, MD (The Ohio State University, Columbus, Ohio, USA); Ganesh V. Kamath, MD (University of North Carolina Medical Center, Chapel Hill, North Carolina, USA); Thomas E. Klootwyk, MD (Forte Sports Medicine and Orthopedics, Indianapolis, Indiana, USA); Bruce A. Levy, MD (Orlando Health, Orlando, Florida, USA); C. Benjamin Ma, MD (University of California, San Francisco, California, USA); G. Peter Maiers II, MD (Forte Sports Medicine and Orthopedics, Indianapolis, Indiana, USA); Robert G. Marx, MD (Hospital for Special Surgery, New York, New York, USA); Matthew J. Matava, MD (Washington University in St. Louis, St. Louis, Missouri, USA); Gregory M. Mathien, MD (Knoxville Orthopedic Clinic, Knoxville, Tennessee, USA); David R. McAllister, MD (University of California, Los Angeles, California, USA); Eric C. McCarty, MD (University of Colorado Denver School of Medicine, Denver, Colorado, USA); Robert G. McCormack, MD (University of British Columbia/Fraser Health Authority, New Westminster, British Columbia, Canada); Bruce S. Miller, MD, MS (University of Michigan, Ann Arbor, Michigan, USA); Carl W. Nissen, MD (Connecticut Children's Medical Center, Hartford, Connecticut, USA); Daniel F. O’Neill, MD, EdD (The Alpine Clinic, Plymouth, New Hampshire, USA); Brett D. Owens, MD (Warren Alpert Medical School, Brown University, Providence, Rhode Island, USA); Richard D. Parker, MD (Cleveland Clinic, Cleveland, Ohio, USA); Mark L. Purnell, MD (Valley Ortho, Aspen, Colorado, USA); Arun J. Ramappa, MD (Beth Israel Deaconess Medical Center, Boston, Massachusetts, USA); Michael A. Rauh, MD (State University of New York at Buffalo, Buffalo, New York, USA); Arthur C. Rettig, MD (Forte Sports Medicine and Orthopedics, Indianapolis, Indiana, USA); Jon K. Sekiya, MD (University of Michigan, Ann Arbor, Michigan, USA); Kevin G. Shea, MD (Stanford University, Palo Alto, California, USA); Orrin H. Sherman, MD (NYU Hospital for Joint Diseases, New York, New York, USA); James R. Slauterbeck, MD (UNC Health Southeastern, Lumberton, North Carolina, USA); Matthew V. Smith, MD (Washington University in St. Louis, St. Louis, Missouri, USA); Jeffrey T. Spang, MD (University of North Carolina Medical Center, Chapel Hill, North Carolina, USA); Col. (Ret.) Steven J. Svoboda, MD (MedStar Orthopaedic and Sports Center, Washington, DC, USA); Timothy N. Taft, MD (University of North Carolina Medical Center, Chapel Hill, North Carolina, USA); Joachim J. Tenuta, MD (Albany Medical Center, Albany, New York, USA); Edwin M. Tingstad, MD (Inland Orthopaedic Surgery and Sports Medicine Clinic, Pullman, Washington, USA); Armando F. Vidal, MD (The Steadman Clinic, Vail, Colorado, USA); Darius G. Viskontas, MD (Fraser Orthopaedic Institute, New Westminster, British Columbia, Canada); Richard A. White, MD (Fitzgibbon’s Hospital, Marshall, Missouri, USA); James S. Williams Jr., MD (Cleveland Clinic, Euclid, Ohio, USA); Michelle L. Wolcott, MD (University of Colorado Anschutz Medical Campus, Aurora, Colorado, USA); Brian R. Wolf, MD (University of Iowa Hospitals and Clinics, Iowa City, Iowa, USA); James J. York, MD (Luminis Health Orthopedics, Pasadena, Maryland, USA); and Rick W. Wright, MD (Vanderbilt University, Nashville, Tennessee, USA).
Footnotes
Acknowledgements
The authors express their appreciation to the late Barton Mann, PhD (AOSSM, Rosemont, Illinois, USA), Allen F. Anderson, MD (Tennessee Orthopaedic Alliance, Nashville, Tennessee, USA), Jeffery R. Bechler, MD (University Orthopaedic Associates, Princeton, New Jersey, USA), and Timothy M. Hosea, MD (University Orthopaedic Associates, Princeton, New Jersey, USA), whose contribution to this work was of great significance. They sincerely appreciate Elizabeth Garofoli's years of dedicated work and effort on this study. They also extend their gratitude to John P. Albright, MD (University of Iowa Hospitals and Clinics, Iowa City, Iowa, USA), Jack T. Andrish, MD (Cleveland Clinic, Cleveland, Ohio, USA), John D. Campbell, MD (Bridger Orthopedic and Sports Medicine, Bozeman, Montana, USA), Diane L. Dahm, MD (Mayo Clinic, Rochester, Minnesota, USA), and Brett (Brick) A. Lantz, MD (Slocum Research and Education Foundation, Eugene, Oregon, USA) for their effort and leadership on this project. All are enjoying a well-deserved and happy retirement after many years of dedication to the advancement of orthopaedics.
Submitted September 20, 2024; accepted September 5, 2025.
Presented at the annual meeting of the AOSSM, Denver, Colorado, USA, July 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This project was funded by grant No. 5R01 AR060846 from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. D.E.C. has received royalties or license from DJO and Stryker; and consulting fees from DJO. T.M.D. has received royalties, consulting fees, and speaking fees from Arthrex; and consulting fees from Kowa Pharmaceuticals American. K.P.S. has received support for education from Summit Surgical and consulting fees from NovoPedics. M.J.S. has received royalties, consulting fees, and speaking fees from Arthrex; and grant and support for education from Smith & Nephew. A.A. has received royalties and consulting fees from Arthrex; consulting fees from LimaCorporate and DJO; travel support from Lima USA; support for education from Southtech Orthopedics and Tricoast Surgical Solutions; and acquisitions and investment interest in Anika Therapeutics. C.C.A. has received support for education from Arthrex and Supreme Orthopedics Systems. R.A.A. has received acquisitions and consulting fees from Linvatec Coporation; and consulting fees from Biorez and Smith & Nephew. B.R.B. has received consulting fees from OsteoCentric Technologies. C.L.B. has received travel support from Arthrex and CGG Medical. K.M.B. has received consulting fees from Miach Orthopaedics, Stryker, and Wright Medical Technology. J.H.B. has received support for education from Supreme Orthopedic Systems. G.A.B. has received support for education from Prodigy Surgical Distribution. S.F.B. has received consulting fees and royalties from Arthrex and Exactech; and royalties from Zimmer Biomet Holdings. C.A.B.J. has received a grant from DJO. J.B.B. has received consulting fees from DePuy and Medical Device Business Services; and support for education from Steelhead Surgical. J.L.C. has received consulting fees and honoraria from Vericel; speaking fees from Arthrex; and consulting fees from Bioventus and Joint Restoration Foundation; he is an associate editor for
