Abstract
Background:
Matrix-induced autologous chondrocyte implantation (MACI), the third-generation of the technique, is an established procedure for the treatment of focal cartilage defects in the knee. However, the literature lacks long-term results of MACI with good statistical power.
Purpose:
To determine long-term survival and patient-reported outcomes (PROs) in a representative cohort and to identify patient- and surgery-related parameters that may influence long-term clinical outcomes.
Study Design:
Case series; Level of evidence, 4.
Methods:
A total of 103 patients were clinically evaluated at the current follow-up of 8.1 years (range, 5-11.9 years). PRO measures (PROMs) included the Knee injury and Osteoarthritis Outcome Score (KOOS), EQ-5D, visual analog scale for pain, and Tegner Activity Scale. Magnetic resonance imaging results were evaluated by using the AMADEUS (area measurement and depth and underlying structures) and MOCART (magnetic resonance observation of cartilage repair tissue) 2.0 knee score classification systems. Potential factors influencing PROs were first identified univariately and investigated in a multivariate regression model.
Results:
The defects had a mean size of 4.8 cm2 (range, 1.2-12 cm2) and were predominantly femorotibial (66%). The mean Kaplan-Meier survival rate of revision for any reason was 97.2% ± 1.6% at 10 years. In comparison to preoperative values, all PROMs were significantly improved at the current follow-up (P < .05). The MOCART 2.0 score peaked at 12 months (mean, 80.2 ± 15.3 months) and showed no significant change at 96 months (mean, 76.1 ± 19.5 months; P = .142). The linear multivariate regression model identified an association of body mass index (BMI), MOCART 2.0 score, and number of previous knee surgeries with KOOS (R2 = 0.41; f2 = 0.69). Further analysis of the individual determinants revealed an optimal BMI range of 20 to 29 for favorable PROs at 96 months. Significant correlations of MOCART subscores with the overall KOOS were found for graft surface and structure, bony reaction, and subchondral detectable changes. Only 30% of patients with2 previous surgeries and 20% of patients with 3 previous surgeries achieved a Patient Acceptable Symptom State (χ2 = 10.93; P = .012).
Conclusion:
The present study shows consistently good long-term clinical outcomes after MACI with a low revision rate and high patient satisfaction. BMI and number of previous knee surgeries may influence clinical outcomes and should be considered in patient selection and education. There is a correlation between graft structure, subchondral bone changes on magnetic resonance imaging, and long-term PROMs.
Autologous chondrocyte implantation (ACI) is a procedure for the treatment of focal cartilage defects in the knee that was established in the 1990s and has been continuously improved since then. 5 Stage-appropriate therapy with long-term therapeutic success is of great socioeconomic importance given the high prevalence of focal cartilage defects of up to 60% in the general population and the associated risk of accelerated osteoarthritis development. 24
With >10 years of follow-up, the first generation of ACIs with periosteal coverage showed good medium- and long-term results.27,30 Meanwhile, the third generation of the technique, matrix-induced ACI (MACI), is a widely used treatment method for focal cartilage defects >2 cm2 in the knee. 28 The reliability of third-generation ACI (MACI) has been well studied in the short and medium term and shows a relevant improvement of symptoms, function, and quality of life of patients.6,13
Long-term outcomes in MACI are particularly important given the young age of patients. However, published studies are scarce and lack statistical power and magnetic resonance imaging (MRI) data.1,2,17,29 A recently published study showed promising long-term patient-reported outcomes (PROs) in 87 individuals, with a low rate of graft failure (9.1%) and a remarkable patient satisfaction rate of 88.5%. However, the representativeness of the results was limited by the small mean defect size of 3.2 cm2 (no treated defect >4 cm2) and the greater age of the patients (up to 58 years). 12 No factors that might influence long-term PROs could be identified. 12 Therefore, survival data may not be generalizable, and potential factors influencing long-term clinical and radiologic outcomes should be further investigated.
The purpose of this study was to determine long-term survival and PROs after MACI in a representative cohort and to identify patient- and surgery-related parameters that may influence long-term clinical outcomes.
Methods
Study Cohort
Between January 2012 and December 2018, 150 patients who underwent MACI for focal cartilage damage in the knee were enrolled preoperatively and evaluated at 1, 2, and at least 5 years postoperatively. Concomitant procedures were performed as indicated (see Appendix Table A1, available in the online version of this article). Patients (minimum age, 18 years) with symptomatic, isolated femorotibial or patellofemoral full-thickness cartilage defects of the knee corresponding to International Cartilage Regeneration & Joint Preservation Society grade 3 and 4 who underwent MACI were included. Exclusion criteria at the time of surgery were advanced osteoarthritis (Kellgren-Lawrence grade >2), ligamentous instability, varus/valgus deformity (>5° mechanical tibiofemoral angle), inflammatory arthritis, hemophilia, bipolar cartilage lesions, and subtotal meniscal loss, according to the indication criteria for cartilage therapy of the Tissue Regeneration Working Group of the DGOU (German Society of Orthopaedics and Trauma). 28 All 150 patients were contacted at 1, 2, and at least 5 years postoperatively and invited to our single-center academic institution for clinical and MRI-based radiologic review. Written informed consent for study participation and institutional review board approval were obtained before study implementation. The study was conducted in accordance with the Declaration of Helsinki of 2008. 37
At the current follow-up, complete clinical data were available for 103 patients (103 grafts), of whom 75 patients underwent MRI-based radiologic assessment (Figure 1).
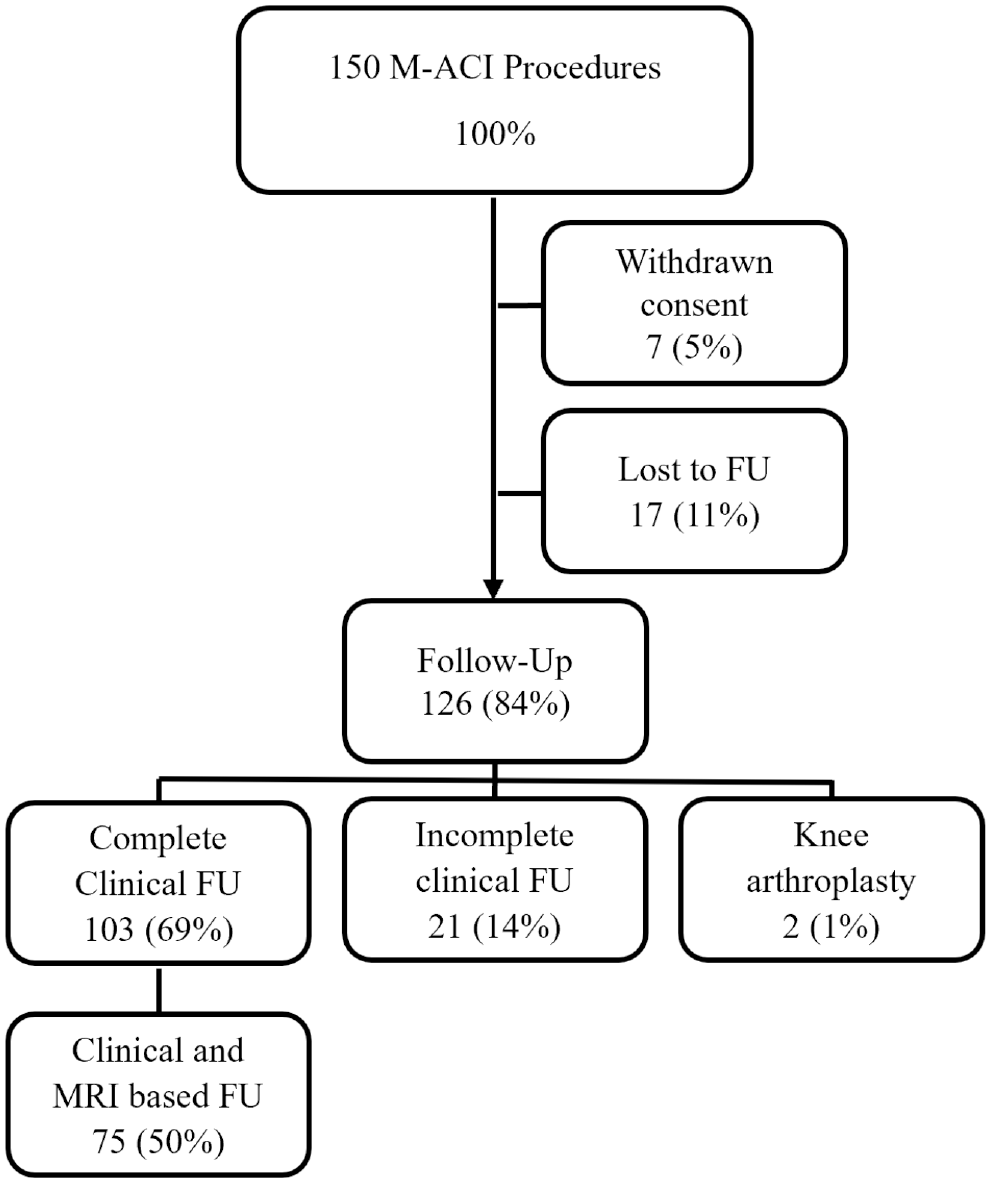
Flowchart visualizing patient selection at present, 96 months of follow-up (FU). M-ACI, matrix-induced autologous chondrocyte implantation; MRI, magnetic resonance imaging.
Surgery and Rehabilitation
MACI is an established 2-stage surgical procedure. 38 Initially, diagnostic arthroscopy with cell harvest was performed to confirm the correct indication. In a 4-week interval, in vitro cell proliferation was performed followed by transfer to a type 1 or 3 collagen matrix with subsequent safety and quality control (Novocart 3D; TETEC). In a second procedure, a mini-open arthrotomy was performed to prepare the defect bed down to the subchondral lamina and to create a stable wall of adjacent vital cartilage. Any existing subchondral sclerosis was carefully removed. Additional bone grafting was required in cases of relevant subchondral bone involvement >2 mm. The chondrocyte-seeded membrane was then conformed and fixed to the defect bed with resorbable fibers or fibrin glue.
The postoperative treatment was adapted according to the localization of the defect. Patients were first mobilized with physical therapy 48 hours after surgery. The knee was stabilized with a brace with limited range of motion 24 hours a day. For femorotibial defects, partial weightbearing (sole contact) with range of motion restriction of 0°/0°/60° of extension/flexion was performed for 6 weeks, followed by increasing weightbearing for 2 weeks and full range of motion. For patellofemoral defects, postoperative full weightbearing was allowed after 48 hours in extension only. Range of motion was initially limited to 0°/0°/30° of extension/flexion for 2 weeks. Flexion was increased in 30° increments every 2 weeks. Full range of motion was allowed at 8 weeks. In addition, the affected extremity was treated with a continuous passive motion device for 3 to 8 hours daily. Moderate sports activities such as swimming or cycling could be started 6 to 8 weeks postoperatively and increased 12 weeks after MACI.
Clinical and Radiologic Review
Demographics, complications, revisions, and PROMs were collected preoperatively and at 12, 24, and 96 months postoperatively. Preoperative and postoperative PRO measures (PROMs) included the Knee injury and Osteoarthritis Outcome Score (KOOS) with values for the Symptoms, Pain, Activities of Daily Living (ADL), Sports, and Knee-related Quality of Life (QOL) subscales; the EQ-5D to measure general quality of life; the visual analog scale (VAS) to assess pain; and the Tegner Activity Scale to assess sports activity level.20,31,34,36 KOOS and the change in KOOS from preoperative values (ΔKOOS) were plotted over time. The recently published Patient Acceptable Symptom State (PASS) for KOOS and KOOS subscale values and the minimal clinically important difference (MCID) for ΔKOOS subscale values were used to assess PROMs at each follow-up time point. 7 In addition, the proportion of patients achieving the MCID was determined as the clinical response rate. The MCID for the overall ΔKOOS was set at 10 based on the current literature and published recommendations.15,35 In addition, return to sports (RTS) and satisfaction with surgery were recorded. Kaplan-Meier analysis based on revisions for any cause was used to calculate long-term survival. Potential factors influencing PROs were first identified univariately and investigated in a multivariate regression model as recently described. 21
MRI-based analysis was performed preoperatively and at each postoperative follow-up to determine graft repair tissue using high-resolution 3-T MRI. Because of the structural modifications at the investigating institution, the MRI scans for the current follow-up were performed using a new MRI unit. The preoperative and 1- and 2-year postoperative scans were performed on a 3-T whole-body scanner (Magnetom Verio; Siemens Healthineers) equipped with an 18-channel total imaging matrix in combination with a dedicated 15-channel knee coil, as previously described. 2 The current follow-up was performed using a latest-generation 3-T MR scanner of the same manufacturer (Magnetom Vida; Siemens Healthineers) and an 18-channel knee coil. Imaging sequences were comparable between both scanners and included high-resolution proton density–weighted turbo spin echo (with and without fat saturation) in all imaging planes and T1 spin echo sequences in coronal and sagittal planes.
Preoperative and postoperative MRI results were evaluated by 2 independent investigators (J.We. and J.Wi.) using the AMADEUS (area measurement and depth and underlying structures) and MOCART (magnetic resonance observation of cartilage repair tissue) 2.0 knee score classification systems.
Statistical Analysis
Descriptive data and clinical and radiologic scores were calculated as absolute frequencies and means with standard deviations. The Kolmogorov-Smirnov test was used to determine parametric distributions. Continuous variables were analyzed using the unpaired t test for parametric data and the Mann-Whitney U test for nonparametric data. Chi-square tests were used to compare the distribution of categorical variables.
Survival with the endpoint of reoperation was estimated using Kaplan-Meier analysis with 95% confidence intervals. Patients were censored at the time of reoperation or at the end of follow-up, whichever occurred first. Based on the sample size, survival was calculated to be 9.5 years with at least 21 patients still in follow-up. 11
Intraobserver reliability for radiologic measurements was performed in a blinded fashion on days 1 and 8. Inter- and intraobserver reliability for radiologic measurements were excellent (interobserver OR, 0.84 [95% CI, 0.82-0.86]; intraobserver OR, 0.92 [95% CI, 0.89-0.95]).
Associations between patient- or surgery-related factors were first determined univariately with Pearson correlation and then examined in a multivariate linear regression model. A total of 59 treated patients were included in the regression model. A post hoc power analysis was performed to determine the validity of our findings. With a calculated f2 effect size of .69, 59 available patients, an alpha of .05, and 5 predictors, the calculated statistical power to detect an effect on postoperative PROs as measured using the KOOS was 0.99. The effect size was reported and classified according to Cohen (0.1, small effect; 0.3, medium effect; 0.5, large effect). 8
The significance level was set at P < .05. A Bonferroni correction was performed for the analysis of the association between previous surgery and MCID or PASS achievement, with the significance levels set at P = .016 and P = .025, respectively. Statistical analysis was performed using SPSS Version 27.0 (IBM Corp) and G*Power Version 3.1 (Heinrich Heine University).
Results
At the current follow-up after a mean of 8.1 years (range, 5-11.9 years), complete clinical data were available for 103 patients. Demographic and baseline characteristics are shown in Table 1.
Baseline Characteristics of Patients a
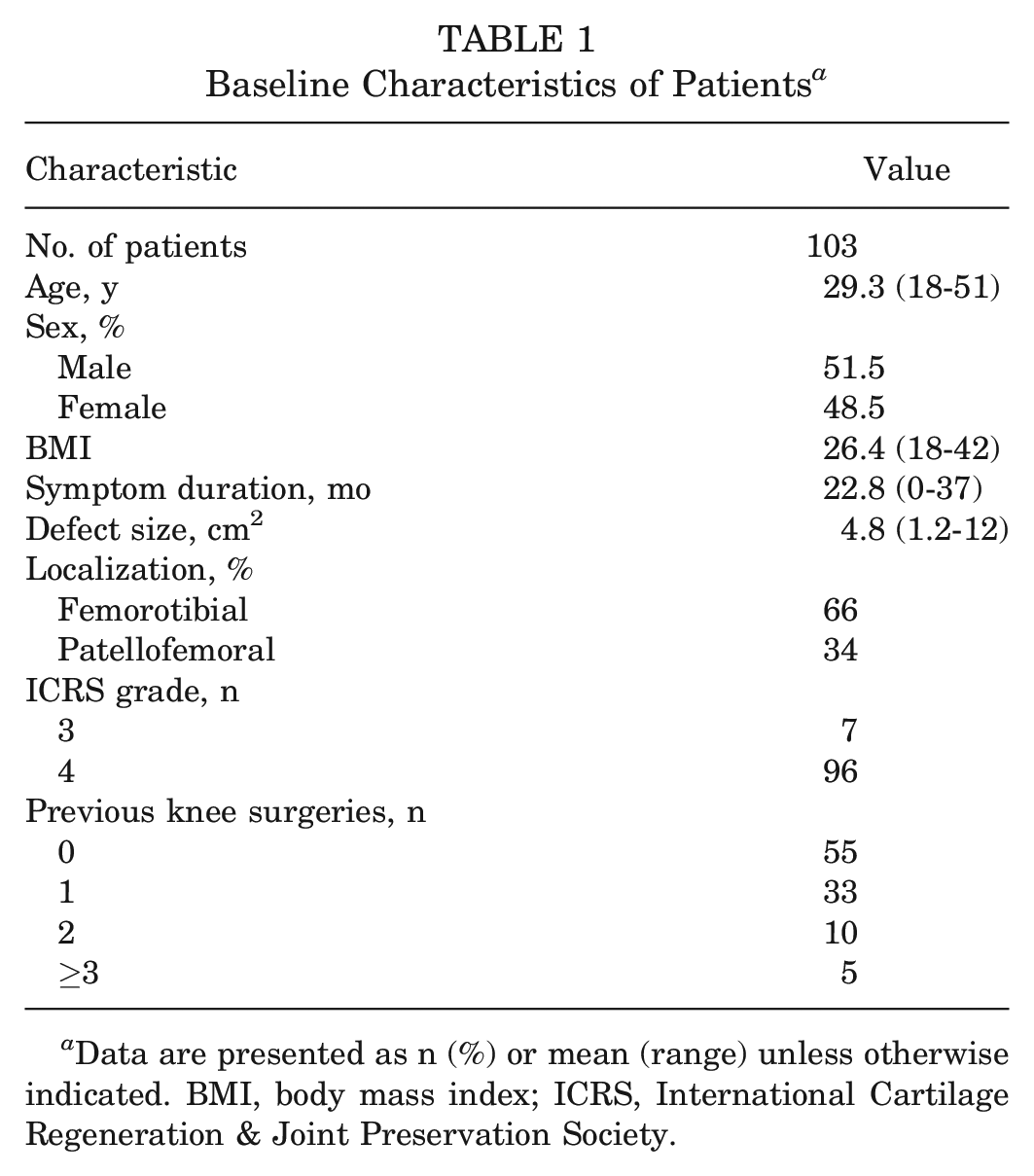
Data are presented as n (%) or mean (range) unless otherwise indicated. BMI, body mass index; ICRS, International Cartilage Regeneration & Joint Preservation Society.
Clinical Review
Compared with preoperative and 12-month postoperative parameters, all PROMs (Table 2) improved significantly (P < .05) at 96 months. The proportion of patients achieving PASS increased steadily throughout the follow-up period (χ2 = 15.775; P < .001).
PROMs at the Various Follow-up Time Points a
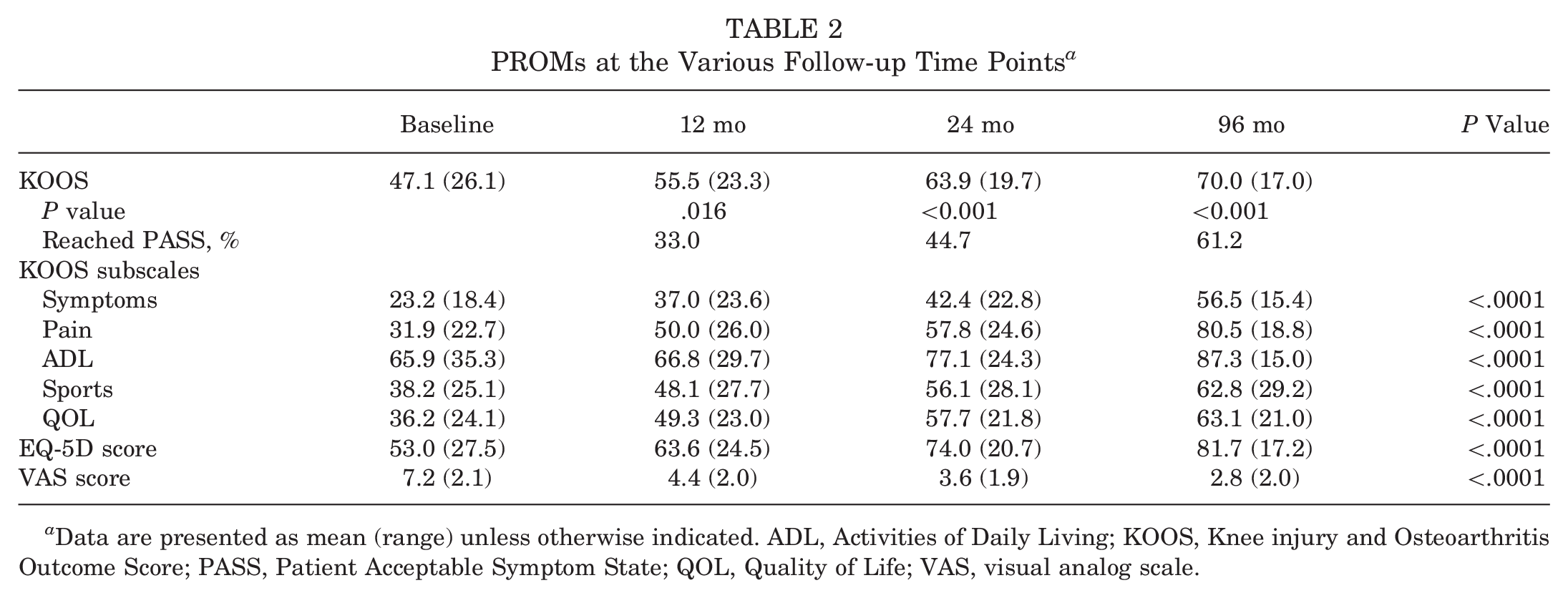
Data are presented as mean (range) unless otherwise indicated. ADL, Activities of Daily Living; KOOS, Knee injury and Osteoarthritis Outcome Score; PASS, Patient Acceptable Symptom State; QOL, Quality of Life; VAS, visual analog scale.
The clinical response rate was 56.3% at 12 months postoperatively, increasing to 63.1% at 24 months and 71.8% at the current follow-up (χ2 = 5.406; P = .067). When analyzing the KOOS subscale values, an improvement in KOOS Symptoms and Pain was initially observed in the early postoperative interval, exceeding the MCID at 12 months (Figure 2). At 24 months, the mean scores for ADL and QOL also exceeded the MCID. The mean score for KOOS Sports did not reach the MCID throughout the postoperative interval.
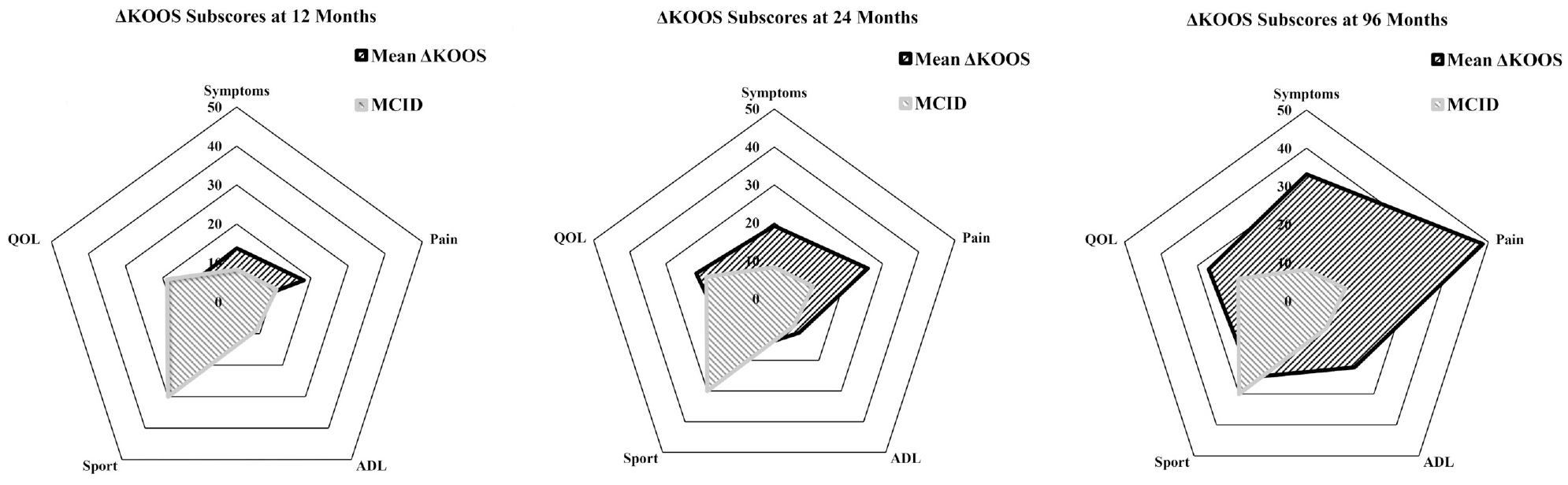
Change in Knee injury and Osteoarthritis Outcome Score (KOOS) subscale values at various follow-up time points, reported relative to the minimal clinically important difference (MCID). ADL, Activities of Daily Living; QOL, Quality of Life.
The mean Tegner Activity Scale score increased from 3.1 (1.9) preoperatively to 4.8 (2.0) at the last follow-up (P < .0001). Of 103 patients, 49.5% (n = 51) were able to return to their desired level of sports at 96 months and 83.5% (n = 86) stated they would have the surgery again.
Radiologic Review
At the current 96-month follow-up, 75 patients were available for MRI-based evaluation (Figure 3). The preoperative AMADEUS score was 34.7 (SD, 14.8; n = 103). The mean MOCART 2.0 score at 12 months after surgery was 80.2 (SD, 15.3; n = 83) and did not show significant decline at 96 months (mean, 76.1; SD, 19.5; P = .142; n = 75). Only 2 graft failures (2.7%), defined as complete delamination or unfilled graft bed on MRI, were observed among 75 patients (75 grafts). Complete defect filling was observed in 51 cases (68%).
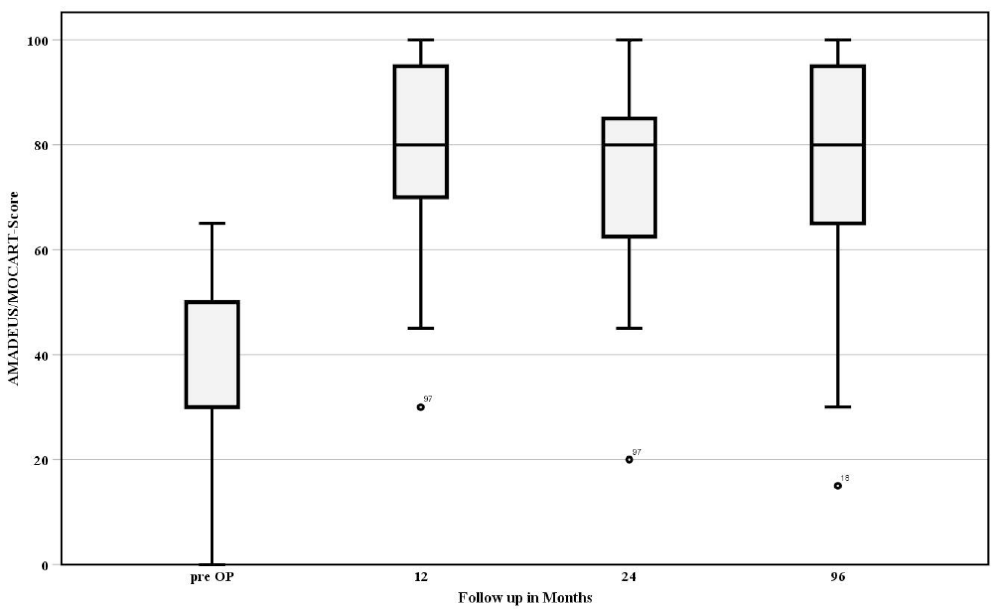
Radiologic outcome, preoperatively measured by the AMADEUS (area measurement and depth and underlying structures) score and postoperatively measured by the MOCART (magnetic resonance observation of cartilage repair tissue) 2.0 score, at various follow-up time points.
When stratified by defect location (femorotibial vs patellofemoral), the AMADEUS and MOCART 2.0 scores did not differ significantly at the different time points (Table 3). In the MOCART 2.0 subscores, patients with femorotibial cartilage defects showed slightly increased bony reactions at 12 months (P = .040) and increased subchondral changes at 96 months (P = .041) (Table 3).
Differences in MOCART 2.0 Scores and Subscores at Various Follow-up Time Points a
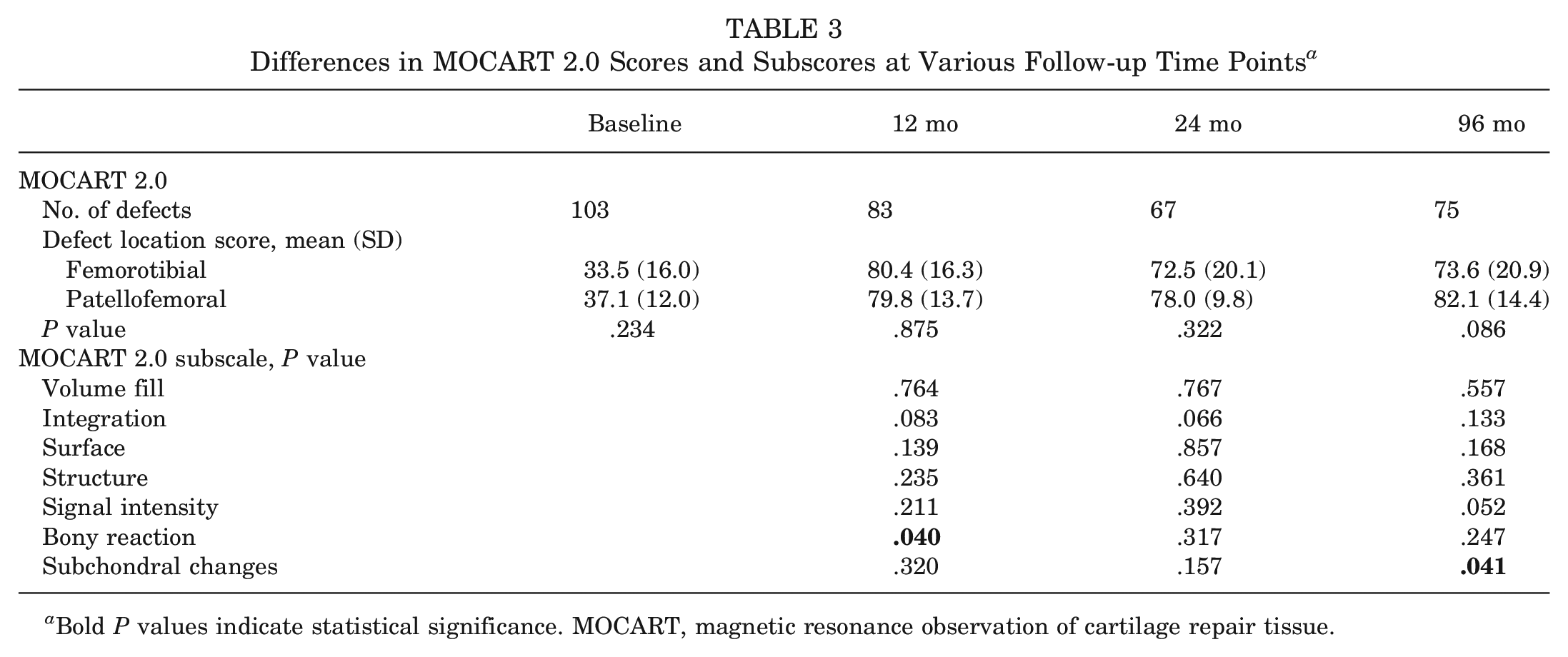
Bold P values indicate statistical significance. MOCART, magnetic resonance observation of cartilage repair tissue.
PRO Influencing Factors
In the univariate analysis, body mass index (BMI) (Pearson r = −0.3), age (r = −0.2), MOCART 2.0 score at 96 months (r = −0.4), previous knee surgery (r = −0.4), and cell count (r = 0.1) were identified as potential influencing factors on long-term PROs as measured by KOOS overall at 96 months (P < .05). In contrast, AMADEUS score, MOCART score at 12 and 24 months, symptom duration, defect size and localization, cell viability, smoking status, and sex did not influence postoperative outcome (P > .05).
The linear multivariate regression model identified BMI, MOCART 2.0 score, and number of previous knee surgeries as factors influencing long-term KOOS (Table 4). The model has a goodness of fit of correlation: an R2 of 0.41 with a high effect size of f2 of 0.69.
Multivariate Linear Regression Model Illustrating the Influence of BMI, MOCART Score, and Previous Knee Surgery on Clinical Outcome as Measured by KOOS (R2 = 0.41, f2 = 0.69) a
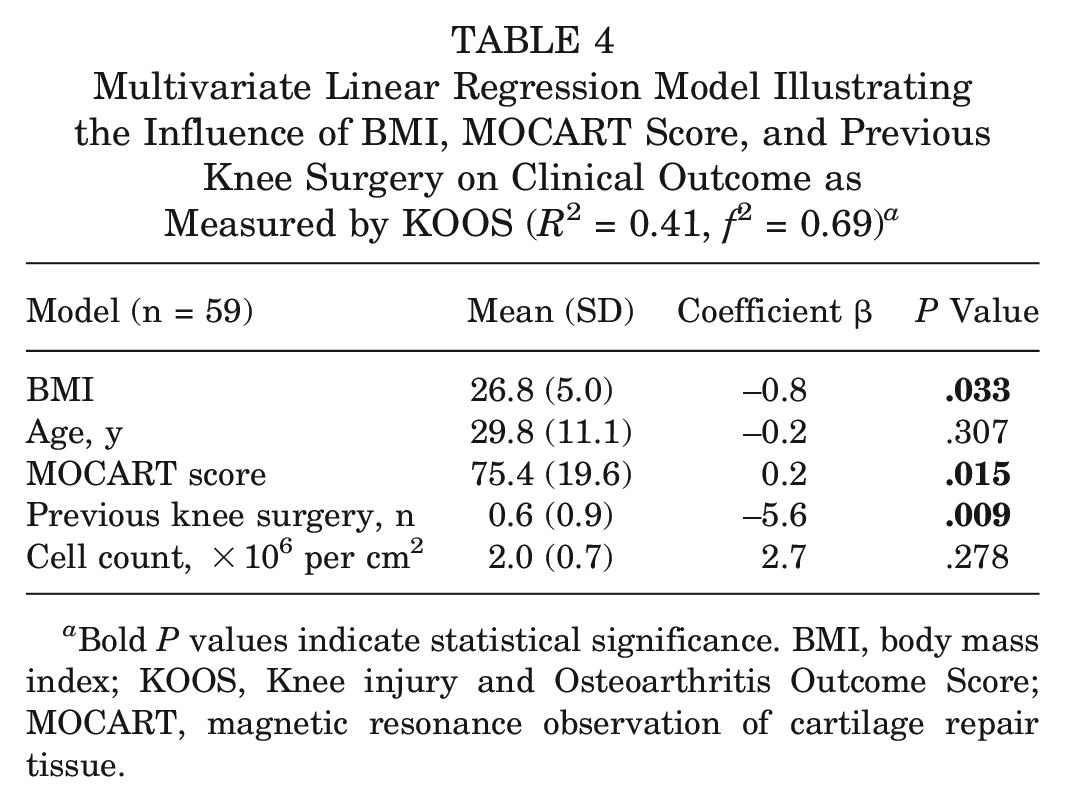
Bold P values indicate statistical significance. BMI, body mass index; KOOS, Knee injury and Osteoarthritis Outcome Score; MOCART, magnetic resonance observation of cartilage repair tissue.
Further analysis of individual determinants revealed an optimal BMI range of 20 to 29 for favorable PROs at 96 months (Figure 4).
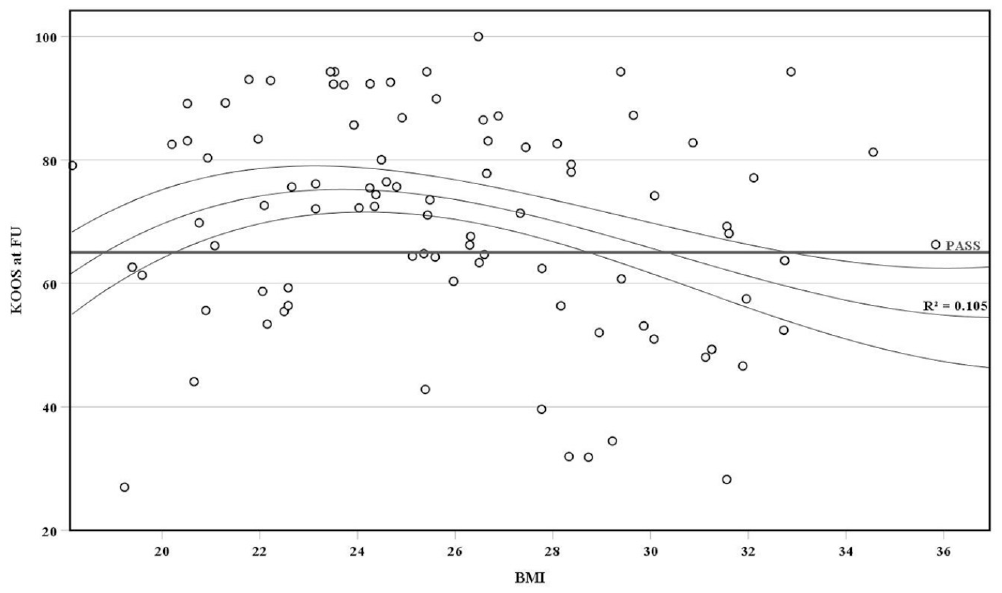
Scatterplot showing the correlation between body mass index (BMI) and Knee injury and Osteoarthritis Outcome Score (KOOS), identifying a range of 20 to 29 for optimal outcome and high probability of exceeding the Patient Acceptable Symptom State (PASS) (R2 = 0.1). FU, follow-up.
A differentiated analysis of the influence of previous knee surgery on PROs as measured by the KOOS at 96 months shows a decrease in mean KOOS with increasing number of previous surgeries (P < .05) (Figure 5).
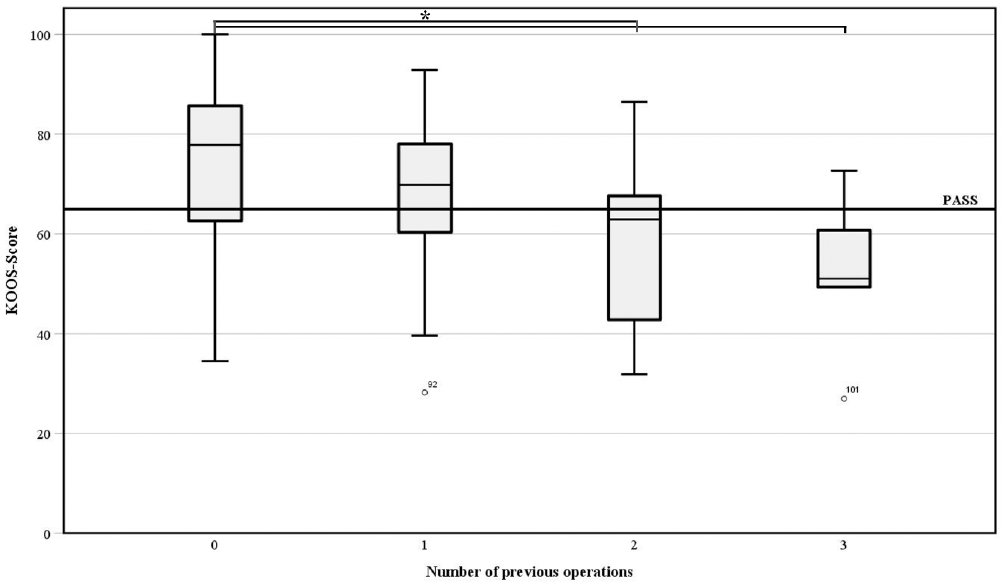
Box plot showing the Knee injury and Osteoarthritis Outcome Score (KOOS) relative to the Patient Acceptable Symptom State (PASS) according to the number of previous surgeries on the affected knee. *Significant.
While 73% of patients with no previous surgery achieved PASS, only 30% of patients with 2 previous surgeries and 20% of patients with 3 previous surgeries achieved PASS (χ2 = 10.93; P = .012). Patients with a BMI in the newly identified range of 20 to 29 had clinically detectable, superior KOOS values and were more likely to achieve the MCID and PASS (Table 5).
Differences in Clinical Outcomes by Number of Previous Knee Surgeries and BMI Range
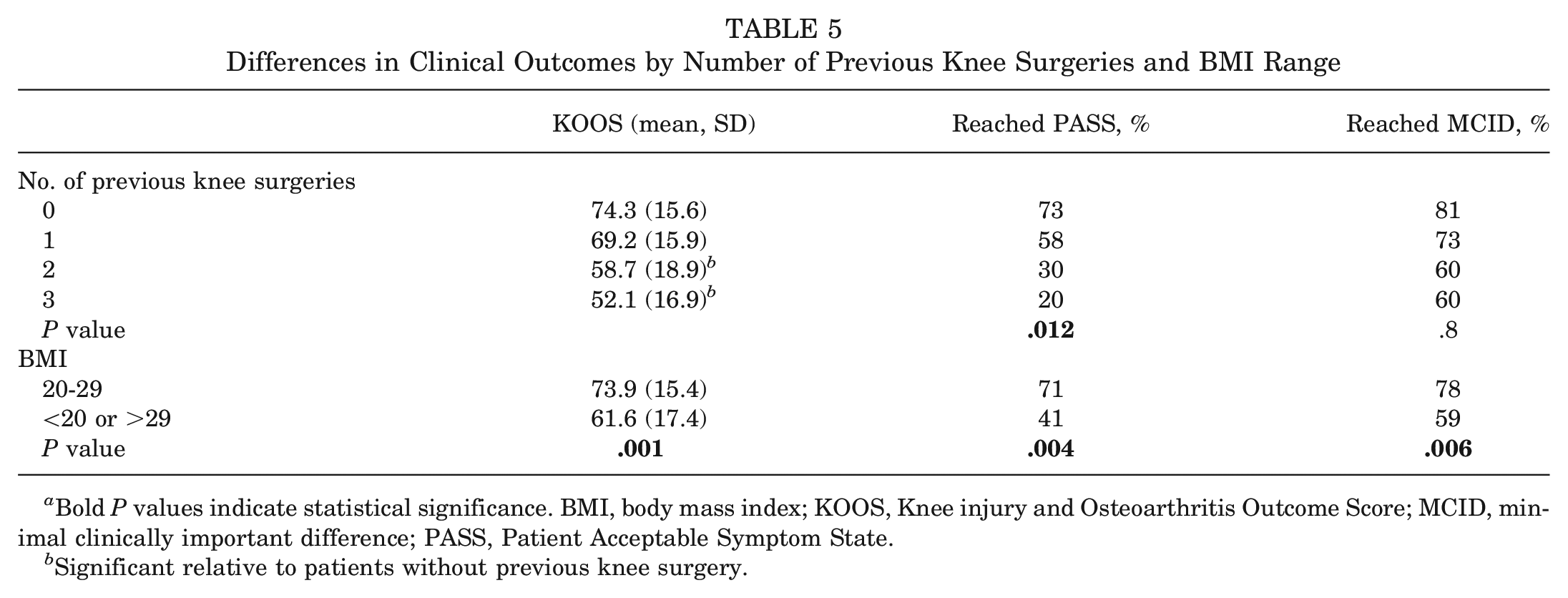
Bold P values indicate statistical significance. BMI, body mass index; KOOS, Knee injury and Osteoarthritis Outcome Score; MCID, minimal clinically important difference; PASS, Patient Acceptable Symptom State.
Significant relative to patients without previous knee surgery.
When analyzing the MOCART subscores, graft surface and structure, bony reaction, and subchondral detectable changes showed a significant correlation with overall KOOS (Table 6).
Association of Various MOCART 2.0 Subscales With Clinical Outcome at 96 Months, as measured by the KOOS
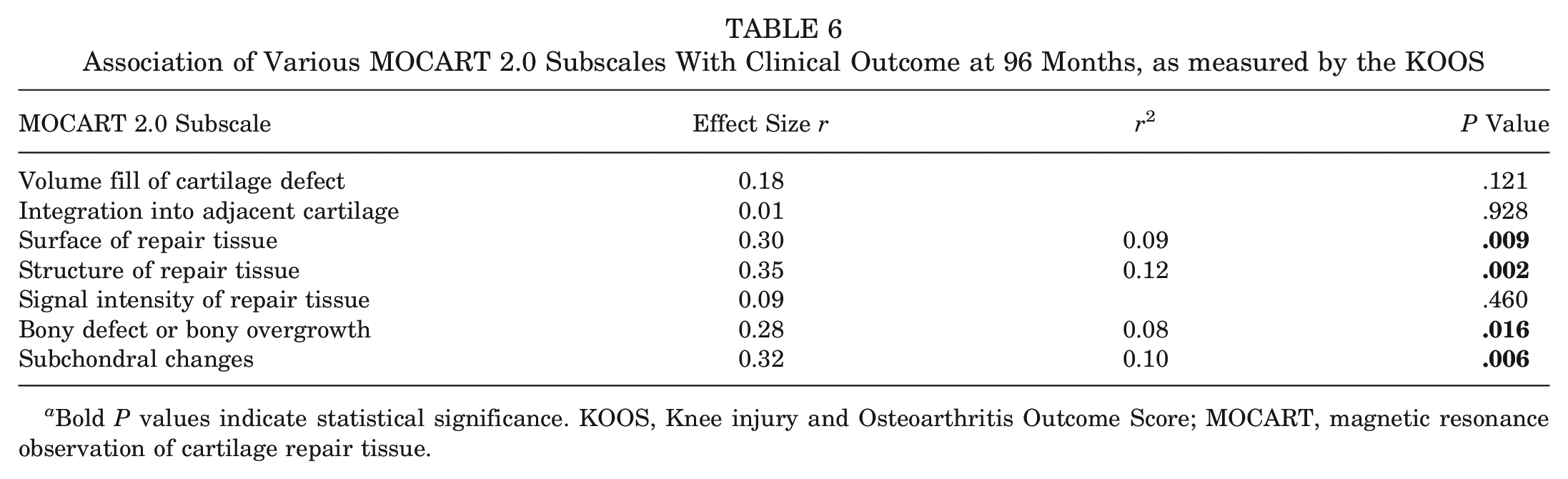
Bold P values indicate statistical significance. KOOS, Knee injury and Osteoarthritis Outcome Score; MOCART, magnetic resonance observation of cartilage repair tissue.
Complications and Reoperations
No surgery-related complications such as postoperative bleeding, arthrofibrosis, thrombosis, or infection were observed in the population studied. Three patients required reoperation. One patient underwent rearthroscopy with microdrilling at 3 years for a symptomatic defect <1 cm2 at the graft site. Two additional patients underwent arthroplasty at 1 and 2 years for refractory pain and rapidly progressing osteoarthritis. The Kaplan-Meier survival rate at 9.5 years for revision for any reason was 97.2% (SD, 1.6%). The Kaplan-Meier survival rate at 9.4 years for reoperation or graft failure was 93.3% (SD, 1.9%). Seventeen patients could not be contacted despite intensive efforts.
Discussion
The main finding of the study is that MACI for symptomatic unifocal femorotibial and patellofemoral cartilage defects showed good PROs with a low graft failure rate of 2.7% and a high survival rate of 97.2% at 9.5 years in a representative cohort of 103 patients. A BMI between 20 and 29 and <2 previous knee surgeries were associated with significantly better clinical outcomes in the long term. MRI findings at 96 months, particularly graft surface and structure, bony reaction, and detectable subchondral changes, correlated with clinical outcomes at long-term follow-up.
The PROMs increased significantly in the immediate postoperative period as well as in the further course up to the present follow-up at 96 months. Sports activity and quality of life seem to plateau after 2 years, although there was an insignificant further increase in both. Although Aldrian et al 1 initially reported a decline in clinical scores from mid- to long-term follow-up, the present study tends to support the findings of other long-term studies, which observed sustained or further improvement in clinical scores over time.12,17,29 A novel finding of the present study is that there was an initial relevant improvement in symptoms and pain at 12 months compared with preoperative values, referenced to the MCID threshold. For ADL and QOL, it took longer for patients to achieve relevant improvements compared with preoperative values, although the MCID threshold was passed at 24 months. This finding highlights the long and challenging rehabilitation process after MACI, and in this context, patient education regarding expectations is critical to achieve compliance and patient satisfaction in the long term.
Somewhat sobering is the fact that the mean postoperative KOOS Sports value did not reach the MCID and only 49.5% of patients achieved their desired level of sport. This finding is not entirely new, as Mithoefer et al 26 early on pointed to a low return-to-performance (RTP) rate of 67% after second-generation ACI. Recently reported RTS and RTP rates between 80% and 100% must be interpreted with caution as they examine short-term data. 9 Although our rate is significantly lower, other long-term data also report a slightly lower rate of long-term satisfaction with sports participation (74.7%). 12 It is therefore possible that the long-term RTS/RTP is overestimated, and it is important to discuss the patient's expectations regarding the cartilage repair procedure in this context. Nevertheless, a high long-term clinical response rate of 71.8% was achieved. To improve long-term clinical outcomes, it is essential to implement appropriate patient selection and education strategies that include the factors identified in this study.
While the clinical benefit of surgery is maintained in the long term, a plateau in the dynamics of MRI scoring using MOCART 2.0 was reached at 12 months postoperatively. Long-term MRI-based results are rare and have involved small populations of 13 or 16 patients.1,29 Nevertheless, Niethammer et al 29 reported good graft survival (Novocart 3D), defined as complete tissue infill, in 76.9% of cases. Aldrian et al 1 reported 73.9% graft survival (Genzyme or Hyalograft-C). In a large-scale study with MRI-based long-term results, Ebert et al 12 reported a graft failure rate of 9.1%, but did not report the number of patients with complete defect filling.
Looking at the MRI results in a differentiated manner, no significant advantage for defect localization could be observed in the studied population. However, a slight advantage for patellofemoral defects was found in the long term (although not significant). In the analysis of the subscores, this finding is likely related to the better condition of the subchondral bone. For reasons of statistical power, the results are limited by the fact that patellar and trochlear defects were grouped as patellofemoral defects in the study population. There is conflicting evidence regarding the difference in MRI findings by location. In their review, Aldrian et al 1 found a trend toward worse MRI results over time after patellofemoral MACI. However, this observation must be interpreted with caution given the small cohort size and the different grafts used in their study. Ebert et al 12 found no difference in long-term MRI results for patellofemoral and femorotibial defects, which is consistent with our findings. Other long-term studies did not address this issue separately.14,17,29 In the present study, a trend toward an advantage of nonweightbearing joint surfaces could be observed in the long-term MRI result.
Potential determinants of clinical outcome after ACI have been extensively studied for first- and second-generation ACIs, but there is a lack of evidence regarding long-term outcomes after MACI.23,26,32 Modifiable risk factors such as BMI, but also nonmodifiable factors such as defect size and localization, age, duration of symptoms, previous knee surgery, and previous cartilage surgery, are often discussed in the short term.18,23,26 In societies with an increasing prevalence of obesity, BMI is a potentially highly relevant determinant of clinical outcome. In a systematic review, BMI could not be identified as an independent risk factor for short-term outcomes after MACI. 3 This finding is supported by available long-term data but may lack statistical power.12,17 Individual case series have previously reported a correlation between BMI and clinical outcome, showing an association of BMI >27.5 with worse outcome after cartilage therapy.23,26 In the present study, factors such as age, sex, defect size, duration of symptoms, cell count, and smoking status were found to have no influence on long-term clinical outcomes after MACI. In contrast, a BMI between 20 and 29 (at the time of surgery) showed a favorable association with long-term PRO. However, it should be noted that potential weight gain or loss was not captured in the present study because the weight of the treated patients was not measured at the various follow-up time points. In addition, BMI alone has limited explanatory value for the variance of KOOS (R2 = 0.105).
The clinical outcome after MACI also seems to be negatively influenced by the number of previous knee surgeries, especially previous cartilage repair. A registry study showed a negative influence of previous cartilage treatments on the postoperative outcome 3 years after primary ACI, independent of the type of cartilage treatment. 32 However, medium- or long-term results were not available because of the lack of statistical power. After 10 years, Ebert et al 12 found a negative influence of previous knee surgery on the KOOS Sports and Symptoms values as well as defect filling, each with a low statistical effect. The present study demonstrated a negative effect of the number of previous knee surgeries on the KOOS and identified a threshold number of 2 previous knee surgeries at which the PASS is unlikely to be achieved after surgery.
In addition, for the first time, a relevant association between the MRI parameters studied and long-term clinical outcome after MACI was shown. Graft surface and structure, bony reaction, and subchondral detectable changes correlated with the KOOS with moderate effect sizes (r = 0.28-0.35). Radiologic studies showed a correlation between bone marrow edema-like lesions and clinical outcome as measured by the Lysholm score in the medium term, at up to 2 years. 22 In particular, the integrity and quality of the subchondral bone appears to be essential for proper maturation of the cartilage tissue during the first 24 months. 16 This relationship has not been investigated in the long-term MACI studies published to date.1,12,17,29
Several alternative procedures such as microfracture or osteochondral autograft transfer (OAT) have been studied in the long term. PROs after microfracture have shown a decline in the long term after promising clinical results in the short term, up to 2 years. 19 Recently published randomized controlled trials have shown that microfracture is inferior to MACI in the medium term.6,10 Therefore, the indications for microfracture are mainly small defects, although long-term comparative studies are lacking. Solheim et al 33 showed better survival rates for mosaicplasty with OAT cylinders compared with microfracture. However, compared with second-generation ACI, the mosaic technique showed a higher revision rate at 10 years (55% vs 17%). 4 This is likely because of the use of multiple cylinders and the associated interface issues that limit the indication range of OATs to small defects. The long-term revision rate of the MACI has not been well reported. In a retrospective analysis, Ehmann et al 14 found a revision rate of 12% after 9 years. Other studies have not reported MACI revision rates separately, but did report 5% to 10% of the original population converting to total knee arthroplasty.12,17,29 The present study shows that MACI is an effective treatment even in the long term at 96 months, with a high survival rate of 97.2% and low rates of graft failure (2.7%).
Strengths and Limitations
Compared with other studies investigating long-term outcomes after MACI,1,17,29 the present study has a relatively large sample size. However, the mean follow-up was <10 years, and the present study had a large range in the follow-up period (5-11.9 years). Although this may bias possible outcome measures, it allowed us to investigate potential long-term PRO determinants for the first time with sufficient power. In the subgroup analysis of patients with >10 years of follow-up (n = 17), no significant differences in PROMs and MRI-based outcomes, including subscores, could be detected (P > .05). Furthermore, no statement could be made about previous cartilage surgery or the exact type of previous surgery, and only the influence of previous knee surgery on clinical outcome could be investigated. This is because of the small number of previous cartilage surgeries (n = 23). A post hoc power analysis with an effect size of r of −0.165, 103 available patients, and an alpha of .05 showed a calculated statistical power of 0.52, which may not be sufficient to detect an association. However, as described in the Methods section, the regression model had adequate power. Our analysis ignores biomechanical factors that may have a potential effect on postoperative outcome. Given the strict inclusion and exclusion criteria, comorbidities were treated according to current guidelines, reducing potential bias. 28 Nevertheless, their influence on clinical and radiographic outcomes needs to be further investigated. 35 As concomitant procedures address concomitant pathologies and aim to create a more physiological condition for cartilage repair, they tend to objectify the outcome after MACI under a more biomechanically physiological condition. This is also reflected in the comparable KOOS after long-term follow-up in patients with and without concomitant procedures (mean, 70.1 ± 17.3 vs 69.7 ± 16.7; P = .95). The concomitant procedures are listed in Appendix Table A1 (available online). It should be noted that clinical data were available for 83% of patients and radiologic data were available for 50% of patients at the latest follow-up. While clinical compliance is above the recommended threshold for clinical trials, MRI follow-up is not. 25 This may introduce selection bias and limit interpretation of the results, although the present study is the first to show that long-term clinical and radiologic outcomes correlate. Finally, it should be noted that the reference values for MCID and PASS after cartilage repair were taken from the publication by Chahal et al 7 and refer to short-term follow-up. To our knowledge, such data are not yet available for long-term follow-up. Nevertheless, such data provide important reference parameters to better stratify clinical outcomes after MACI.
Conclusion
The present study shows consistently good long-term clinical outcomes after MACI with a low revision rate and high patient satisfaction. BMI and number of previous knee surgeries may influence clinical outcomes and should be considered in patient selection and education. There is a correlation between graft structure and integrity, subchondral bone changes and long-term clinical outcomes. To identify predictive factors associated with progressive graft failure and worse clinical outcomes, it is important to continue to monitor these structural changes and their dynamics.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465241270152 – Supplemental material for Factors Influencing Long-term Outcomes After Matrix-Induced Autologous Chondrocyte Implantation
Supplemental material, sj-pdf-1-ajs-10.1177_03635465241270152 for Factors Influencing Long-term Outcomes After Matrix-Induced Autologous Chondrocyte Implantation by Johannes Weishorn, Johanna Wiegand, Severin Zietzschmann, Kevin-Arno Koch, Christoph Rehnitz, Tobias Renkawitz, Tilman Walker and Yannic Bangert in The American Journal of Sports Medicine
Footnotes
Submitted February 9, 2024; accepted June 17, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: T.R. has received research funding at personal disposal from DePuy, Zimmer, Aesculap, German Federal Ministry of Education and Research, Deutsche Arthrose-Hilfe, OttoBock-Stiftung, German Federal Ministry of Economic and Development, Oskar-Helene-Heim Foundation in Berlin, Vielberth Foundation, and Deutsche Forschungsgemeinschaft (DFG); and reimbursement of costs from DePuy, Zimmer, and Aesculap. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Data Accessibility Statement
The data are available on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
