Abstract
Background:
Previous studies have shown that dexamethasone has a positive effect on postoperative pain control, opioid consumption, nausea, and vomiting and length of hospital stay after arthroplasty surgery.
Purpose/Hypothesis:
The purpose of this study was to assess whether adding perioperative dexamethasone to our current pain regimen after hip arthroscopy is more effective than a placebo. It was hypothesized that dexamethasone would reduce postoperative pain, reduce opioid consumption, improve subjective pain and nausea scores, and reduce the number of vomiting events.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
A total of 50 patients requiring unilateral elective hip arthroscopy were randomized to receive intravenous dexamethasone immediately before induction of anesthesia and at 8 am on the first postoperative day (2 ×12 mg) or a placebo (sodium chloride 0.9%). The patient, the surgeons, the treating anesthesiologist, and the involved nursing and physical therapy personnel were blinded to group assignment. The primary outcome was postoperative pain, and secondary outcomes were opioid consumption and nausea scores—assessed using a translated revised version of the American Pain Society Patient Outcome Questionnaire 6 hours postoperatively and on days 1 and 2—and vomiting events. A clinical follow-up was performed 12 weeks postoperatively to assess adverse events.
Results:
The mean age at inclusion was 29 years in both groups. Postoperative pain levels did not differ significantly in most instances. Opioid requirements during the hospitalization in the dexamethasone group were significantly lower than those in the placebo group (31.96 ± 20.56 mg vs 51.43 ± 38 mg;
Conclusion:
Perioperative intravenous dexamethasone significantly reduced postoperative opioid consumption by 40% without compromising pain level and safety, as no corticosteroid-related side effects were observed. Dexamethasone may be a valuable adjuvant to a multimodal systemic pain regimen after hip arthroscopy.
Registration:
NCT04610398 (ClinicalTrials.gov identifier).
Keywords
Hip arthroscopy is an increasingly common procedure and has widely replaced surgical hip dislocation because of its benefits in terms of lower complication rates and faster rehabilitation.16,27,39 The number of hip arthroscopy procedures performed has been steadily rising. 11
Although postoperative rehabilitation is significantly shorter for hip arthroscopy than for the more invasive procedure of surgical hip dislocation, postoperative pain is common and clinically relevant after this “minor” intervention. 31 Hip arthroscopy is performed as an inpatient and/or outpatient procedure depending on the country, and adequate postoperative pain control is of great importance. The hip joint is innervated by several different nerves, including the femoral nerve, obturator nerve, sciatic nerve, and superior gluteal nerve. 6 Thus, achieving adequate postoperative pain management using nerve blocks as a basis for rapid rehabilitation is challenging. 37
Many multimodal regimens exist to minimize postoperative pain, nausea, and vomiting after hip arthroscopy.31,37 Postoperative pain, nausea, and vomiting are often cited as the most common adverse events after anesthesia, with a high incidence of >80% after orthopaedic surgery, and are known to lead to lower patient satisfaction.12,15,24
Opioids are an important component in the treatment of postoperative pain despite the well-known relevant side effects of severe nausea and the risk of persistent postoperative opioid use. Surgery is associated with an elevated risk of persistent postoperative opioid use, particularly when opioid naïve patients are provided with opioid prescriptions for the posthospitalization period.1,9,10,17 Given that hip arthroscopies are primarily performed in young patients in an outpatient setting or with brief hospital stays, minimizing opioid consumption while ensuring adequate pain control is of great significance, especially given the persisting opioid crisis.22,26 With respect to prescribing opioids, orthopaedic surgeons rank fourth among subspecialists.33,35
Dexamethasone is a greatly potent long-acting glucocorticoid that has been extensively studied in perioperative settings. This synthetic glucocorticoid has minimal mineralocorticoid side effects. 30 It can be administered intravenously or orally and is rapidly absorbed. Also, its strong analgesic, anti-inflammatory, euphorigenic, and prophylactic effects for nausea and vomiting have been clearly demonstrated.2,13,14,19,30 Despite these advantages, its widespread use in the perioperative setting has not yet been established, mostly because of concerns regarding possible adverse effects, especially infections. However, a large study of joint replacements did not show a higher risk of infections. 36 The literature has demonstrated the advantages of administering perioperative dexamethasone with dosages ranging from 10 to 40 mg in joint arthroplasty, foot osteotomy, and spine surgery as a single dose or in 2 separate doses.2,3,13,28,29 The risks of short-term glucocorticoid therapy are low if the contraindications for the drugs are respected. Undesirable side effects are not expected, especially the typical side effects of long-term steroid therapy. The literature has demonstrated the safety and reversibility of both short-term, high-dose steroid therapy on the hypothalamic-pituitary-adrenal axis and the use of steroids for postoperative pain management.19,30
This compelling evidence has made us challenge our current multimodal systemic pain regimen. Our goal was to be able to reduce postoperative pain, reduce the need for opioids, and reduce postoperative nausea and vomiting in the young patient population after this variably painful procedure. We hypothesized that the addition of 2 × 12 mg intravenous (IV) dexamethasone to our current perioperative pain regimen would reduce postoperative pain and opioid consumption, improve nausea scores, reduce antiemetics consumption, and reduce the number of vomiting events.
Methods
Study Design
This study was designed as a prospective randomized double-blind placebo-controlled trial with a 12-week intervention period. The study protocol and methodology were approved by the local institutional review board, and written informed consent was obtained from all study patients (KEK ZH: BASEC Nr. 2020-02131; Swiss Medic Nr. 2020-02131). The study was registered on the ClinicalTrials.gov platform (ID No. NCT04610398).
The inclusion criteria were as follows: elective isolated unilateral hip arthroscopy for any reason; age >18 years; general endotracheal anesthesia; no previous hip surgery; and competent German language skills to understand the informed consent form. The exclusion criteria were as follows: chronic lower back pain; steroid or immunosuppressive drugs used within 6 months of surgery; renal or hepatic failure; relevant allergies; pregnancy or active breastfeeding; contraindications for dexamethasone treatment (active infection, allergies); previous enrollment into the present study; known or suspected noncompliance; drug or alcohol abuse; and participation in another study with an investigational drug within the 30 days before entering the present study.
We screened 146 consecutive patients between March 2021 and June 2023. We excluded 46 patients, and 40 patients declined participation. A total of 60 patients were available for allocation using randomized cards and included in the study. After randomization and during hospitalization, 9 patients who had admitted to regularly using cannabis (n = 8) and amphetamines (n = 1) were excluded. One patient was excluded because of use of an antiretroviral medication, which was noted after randomization (Figure 1).
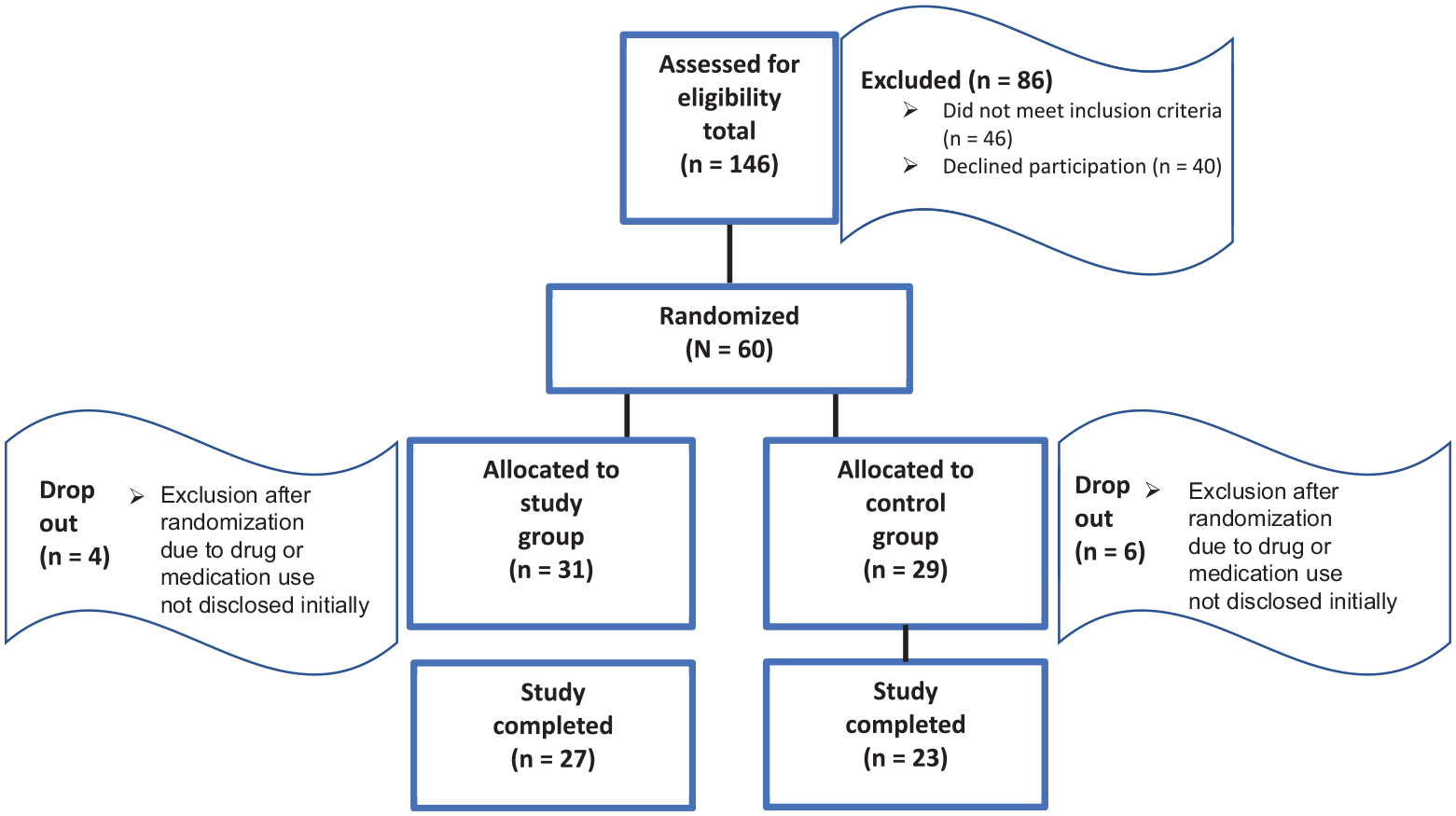
Flowchart of patient assessment, randomization, and completion of study.
Group 1 (study group) received 12 mg of IV dexamethasone (Fortecortin; Merck) in 0.9% sodium chloride solution immediately before induction of anesthesia and 12 mg of IV dexamethasone in 0.9% sodium chloride solution on the first postoperative day at 8 am.
Group 2 served as the control group and received 0.9% sodium chloride solution without dexamethasone immediately before induction of anesthesia and on the first postoperative day at 8 am. All patients had general anesthesia with endotracheal intubation using fentanyl (analgesia), propofol (sleep and anesthesia maintenance), and rocuronium (muscle relaxation).
Dexamethasone was administered in addition to our multimodal pain regimen, which includes paracetamol (3 to 4 g per day) and diclofenac (50 mg 3 times per day for 24 hours), which was then substituted for indometacin (75 mg once daily for 14 days), morphine patient-controlled analgesia for a minimum of 24 hours or as needed, followed by an oral reserve opioid (usually oxycodone), metamizole (3 to 4 g daily), and a ketamine perfusor for breakthrough pain management, only if necessary. Postoperative nausea and vomiting were managed primarily using ondansetron, as needed. In addition, droperidol and paspertin were administered.
The patient, the surgeons (P.O.Z., A.H.), and the anesthesiologist who performed the anesthesia and was responsible for intraoperative management, the nurses, and physical therapists included in the patient care were all blinded to treatment group. The first and third authors (D.K., D.D.), who were not included in patient care, the study nurse (T.G.), and the anesthesiologists (U.E., H.B., J.A.) who prepared and injected the preoperative study medication, were not blinded.
The nurses administering the dexamethasone and/or placebo on the first postoperative day were blinded as well. The medication was prepared by the first author (D.K.); in his absence, it was prepared by the third author (D.D.), and it was personally handed over to the nurse responsible for administration. As dexamethasone is a clear liquid, it cannot be identified and distinguished from 0.9% sodium chloride solution.
Opioid consumption, as read out on the patient-controlled analgesia, as well as the medical chart, was obtained. All opioids were converted to morphine equivalents. No patients were prescribed opioids on discharge. Subjective pain and nausea/vomiting scores were collected using a questionnaire translated into German based on the Revised American Pain Society Patient Outcome Questionnaire (APS-POQ-R). 7 This questionnaire was completed 6 hours after the surgery and at 8 am every following day until discharge. The completion of the questionnaire was monitored by the research assistant (T.G.) and the first author (D.K.).
In addition, antiemetics consumption, vomiting events, patient age, sex, American Society of Anesthesiology score, surgical time, hip distraction time, and length of hospital stay were obtained from the medical chart. Because of the reimbursement system at our institution, a minimal hospital stay of 2 nights is required. All adverse events of the first 3 months were assessed prospectively at the outpatient follow-up.
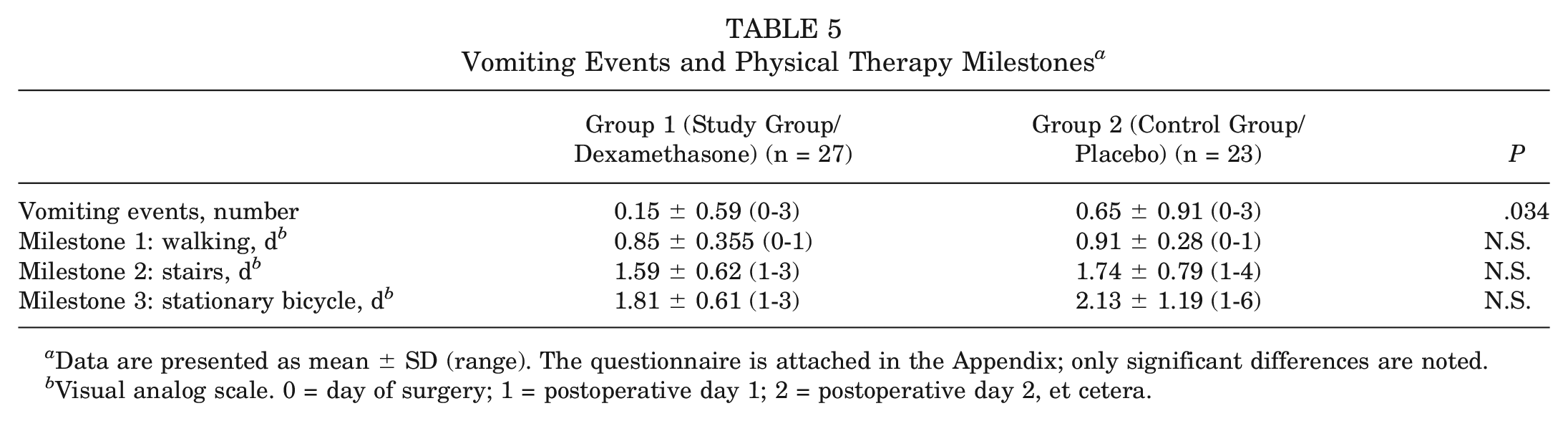
All patients received postoperative physical therapy and were evaluated by their therapist. The achievement of the 3 postoperative milestones of walking, walking stairs, and using the stationary bicycle was obtained from the physical therapy report.
Statistical Analysis
A power analysis before the study resulted in the necessity of enrolling 25 patients per treatment group in the study. This was based on the number needed to demonstrate a significant difference in pain level, 4 with a power of 80% and an alpha level of .05. Accounting for a dropout rate of 15% to 20%, ≥30 patients were necessary per group, adding up to approximately 60 patients.
There were 27 patients in the study group (dexamethasone) and 23 patients in the control group (placebo) who completed the study. The Kolmogorov-Smirnov test was applied to test for normal distribution. The Mann-Whitney
Results
Patient screening, allocation, and completion are presented in Figure 1.
Indications for hip arthroscopy were femoroacetabular impingement in 26 of 27 patients in the study group and 20 of 23 in the control group. A symptomatic labral tear was the indication for the remaining 4 patients—one-quarter had borderline hip dysplasia and three-quarters had anterior rim syndrome. A labral refixation with suture anchors was performed in 21 of 27 patients in the study group and 18 of 23 patients in the control group. Two senior surgeons (P.O.Z.) performed 12 and 9 hip arthroscopies, respectively. One junior surgeon (A.H.) performed 29 hip arthroscopies. The junior surgeon performed 15 of 27 hip arthroscopies in the study group and 14 of 23 hip arthroscopies in the control group.
Patient characteristics, surgical time, and distraction time are summarized in Table 1. No significant differences were noted between the 2 groups.
Patient Characteristics a
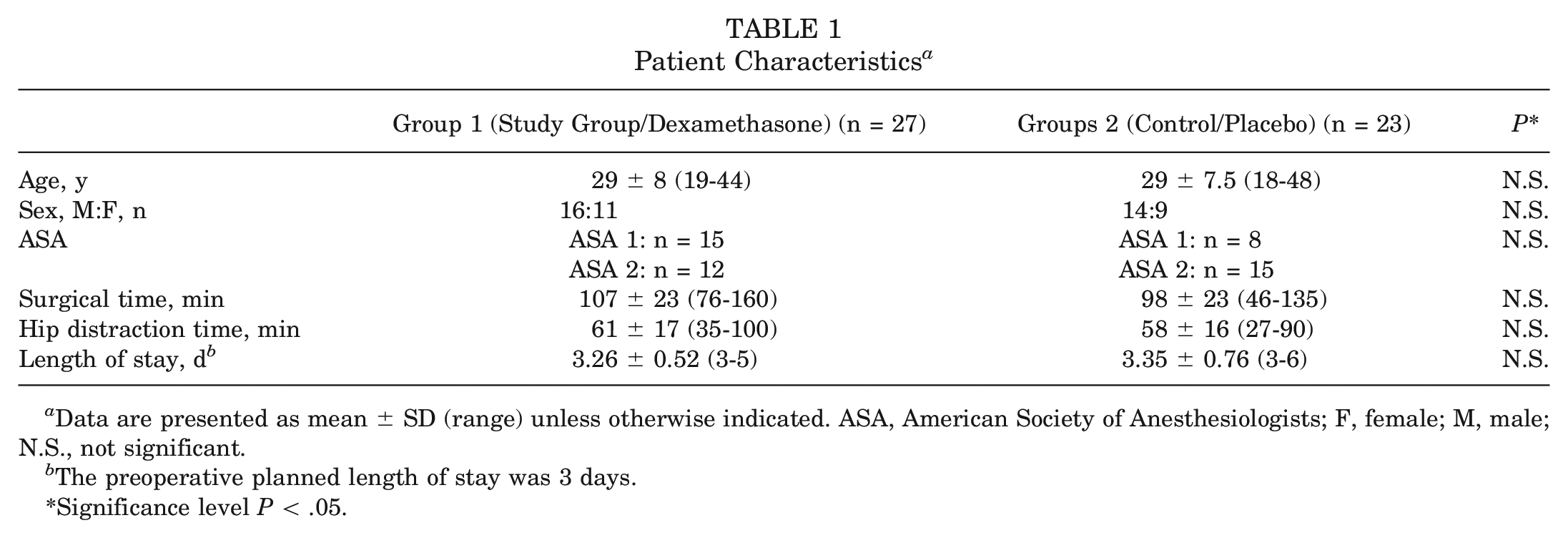
Data are presented as mean ± SD (range) unless otherwise indicated. ASA, American Society of Anesthesiologists; F, female; M, male; N.S., not significant.
The preoperative planned length of stay was 3 days.
Significance level
Intra- and postoperative medication requirements are summarized in Table 2. A significantly decreased postoperative opioid consumption until hospital discharge was seen in the study group (dexamethasone) (
Intraoperative and Postoperative Medication a
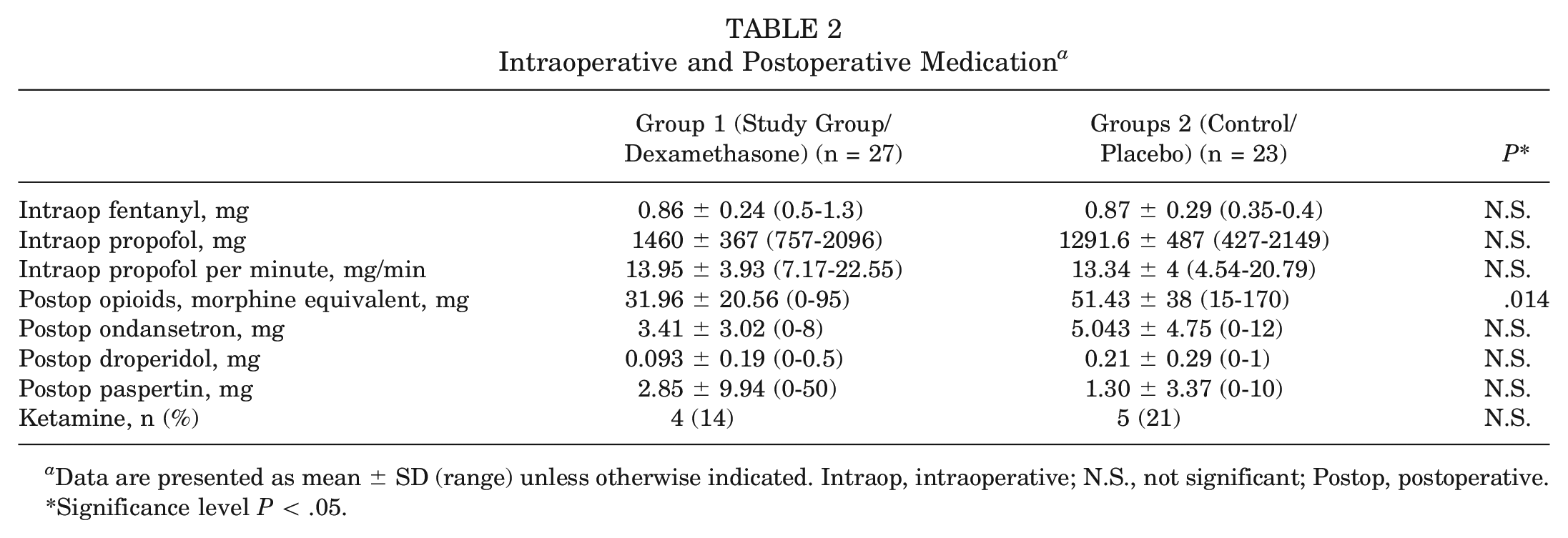
Data are presented as mean ± SD (range) unless otherwise indicated. Intraop, intraoperative; N.S., not significant; Postop, postoperative.
Significance level
Significant differences in the results of the questionnaire are summarized in Table 3. All results are depicted in Table 4. The questionnaire used is attached in the Appendix (available in the online version of this article.).
A significantly lower rate of pruritus (
Significant Results Between the Study and Control Groups Obtained From the Questionnaire a
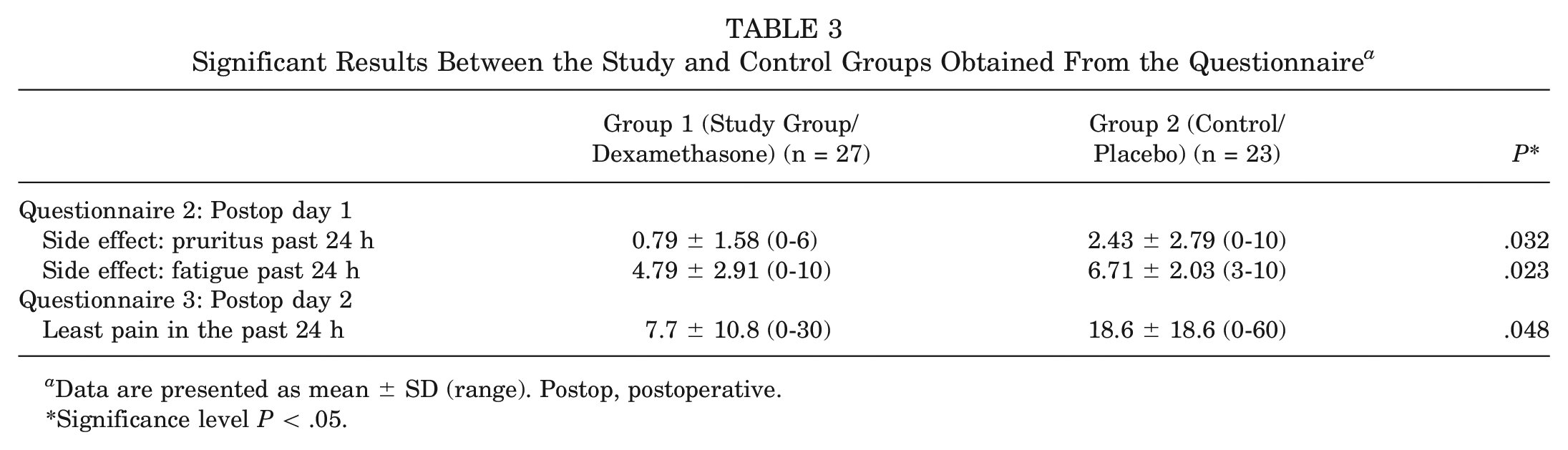
Data are presented as mean ± SD (range). Postop, postoperative.
Significance level
Results Between the Study and Control Groups Obtained From the Questionnaires a
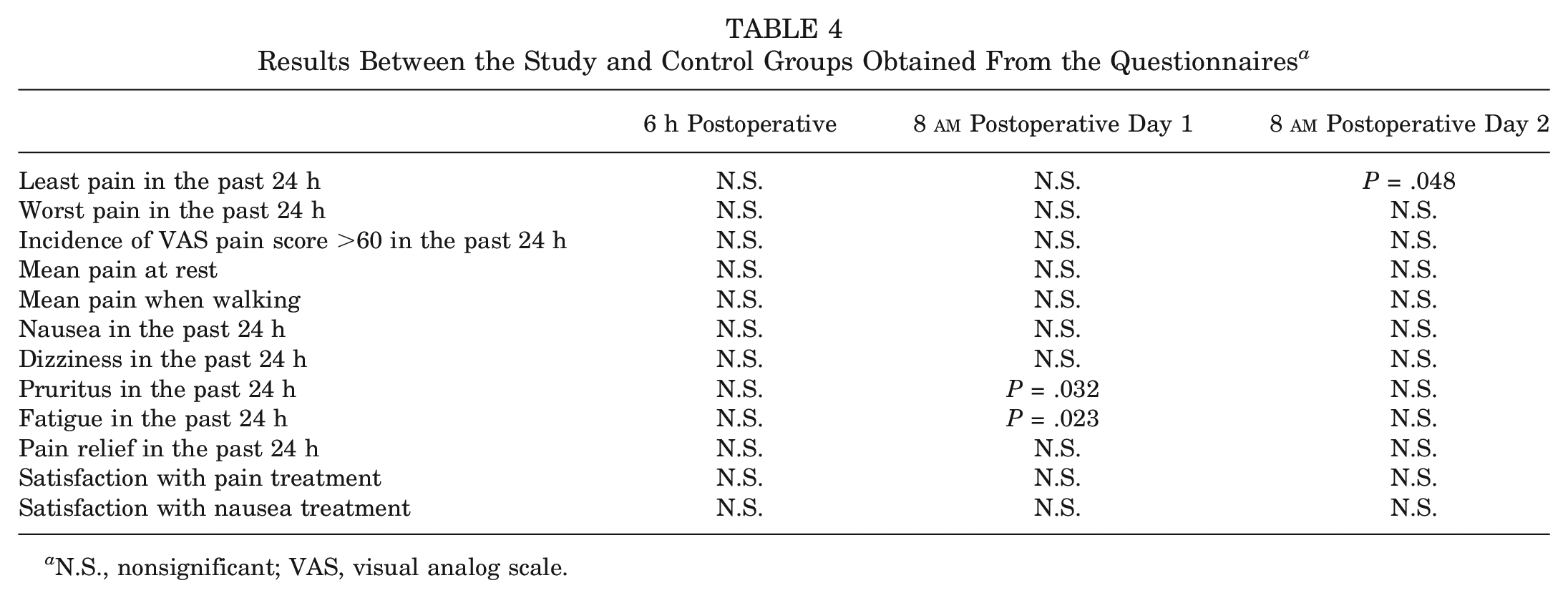
N.S., nonsignificant; VAS, visual analog scale.
No adverse events associated with dexamethasone (infections, delayed wound healing, and superficial wound healing) were seen 3 months postoperatively, as summarized in Table 6.
Adverse Events in the First 3 Months Postoperatively a
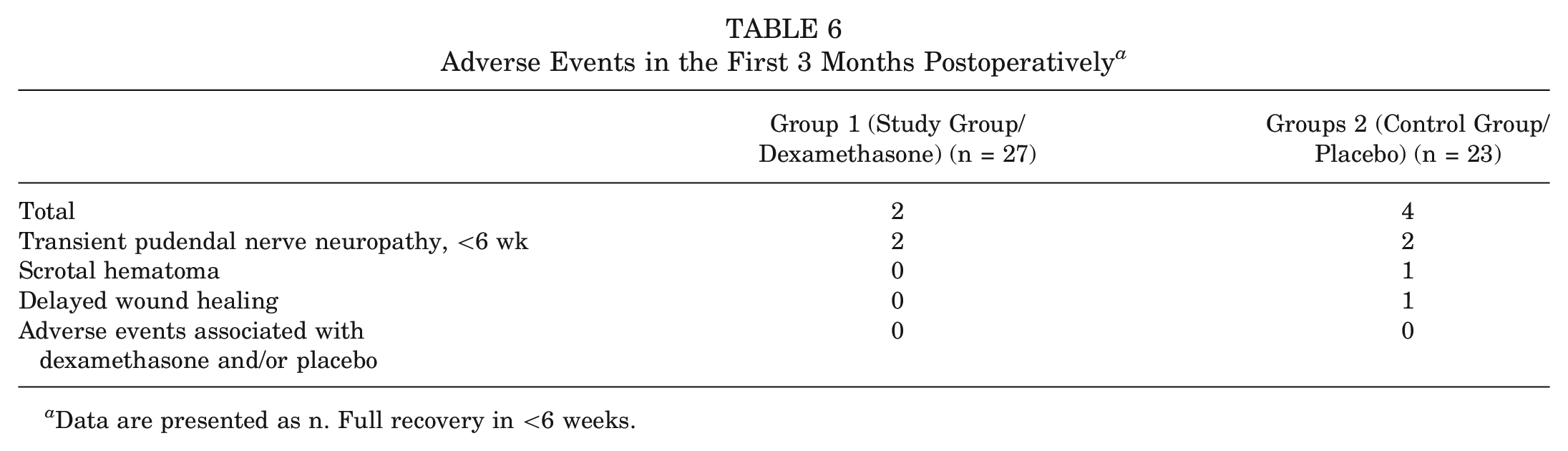
Data are presented as n. Full recovery in <6 weeks.
Discussion
Perioperative administration of dexamethasone significantly reduced opiate consumption after unilateral elective hip arthroscopy by almost 40%. Moreover, a significant reduction of pruritus and fatigue was seen on postoperative day 1 as assessed using the questionnaire, and a significantly lower pain score was recorded on postoperative day 2. A significant reduction in postoperative vomiting events was also noted (
Our results are comparable with those of other studies showing positive effects of perioperative dexamethasone after orthopaedic surgery with significant reduction of postoperative opioid requirements.2,3,21,28,34,38
We did not see a significant difference in self-assessed postoperative pain levels or self-assessed nausea scores. The antiemetics consumption did not significantly differ, albeit a tendency to less ondansetron, droperidol, and metoclopramide need was noted in the study group.
The mean opioid consumption during the hospitalization in the study group was 31.96 (SD, 20.56) mg of morphine equivalents compared with 51.43 (SD, 38) mg of morphine equivalents in the control group. Despite our multimodal pain regimen, the mean maximal pain level perceived in the previous 24 hours by all patients was a visual analog scale (VAS) score of 66 (SD, 22.6) 6 hours postoperatively, a VAS of 57.5 (SD, 25.8) at 8 am on postoperative day 1, and a VAS of 37.9 (SD, 25.4) at 8 am on postoperative day 2. The large amount of >50 mg of morphine equivalents in the control group confirms the painful nature of this procedure. Ketamine was necessary more often in the control group (21%) than in the study group (14%), again potentially speaking for the analgesic potential of dexamethasone.
Uncontrolled opioids prescribed during and after surgery, as well as at hospital discharge, can increase the risk of persistent postoperative opioid use in patients; thus, it seems of great importance to minimize perioperative opioid consumption.1,8,10,20 It has also been shown that targeted postoperative regimens reducing inpatient opioid use can substantially reduce the rate of postoperative opioid refills between 180 days and 365 days in nonelective hip fracture surgeries. 25
This significant reduction of opioids can be achieved without compromising subjective pain and nausea scores as assessed by the APS-POQ-R questionnaire. There was a substantial reduction in pruritus and fatigue on postoperative day 1, as well as a significantly lower least pain score on postoperative day 2. In addition to the antalgic effect of dexamethasone, this may be attributable to the antipruritic and euphoric effect of this drug. Both effects are desired in the immediate postoperative phase. The significant reduction of vomiting events is noteworthy, as dexamethasone has known antiemetic effects. However, the number of vomiting events was very small in both groups, with a mean of 0.15 (SD, 0.59) in the study group and a mean of 0.65 (0.91) in the control group. We did note a tendency to consume fewer antiemetics, albeit the differences were not significant.
Backes et al 3 showed a significant reduction in length of hospital stay, which was not observed in our study. This is mainly because of the medical reimbursement system in our country, which dictates a minimal length of stay of 2 nights. We have thus adjusted our postoperative regimen to distribute the focus of physical therapy to achieve 1 milestone a day. These milestones include walking on crutches, walking on stairs, and being able to mobilize the joint on a stationary bicycle. Hip arthroscopy is performed as an outpatient surgery in many countries. We are convinced that these patients would benefit from the positive effects of perioperative dexamethasone. In outpatient hip arthroscopy, dexamethasone can be prescribed and administered orally, and thus the dose of 12 mg IV dexamethasone can be readily substituted on postoperative day 1. 32
The short-term use of dexamethasone is deemed safe, and a large systematic review found no evidence for adverse events. 19 We noted 6 adverse events in our study (Table 6). None required an additional intervention, and none was associated with the administration of dexamethasone. Concerns for surgical-site infections regarding perioperative dexamethasone for hip arthroscopy have not yet been investigated. However, a large study did not show increased infection rates in total hip and total knee arthroplasty. 36 As infection rates in hip arthroscopy are markedly lower than those in arthroplasty surgery with rates of <0.14%, 18 we do not believe that the administration of dexamethasone has a relevant adverse clinical effect on surgical-site infections or wound complications after hip arthroscopy. Avascular necrosis of the femoral head is a rare complication associated with chronic glucocorticoid use. Bergeron et al 5 demonstrated that preoperative administration of 40 mg of dexamethasone did not result in an increased incidence of avascular necrosis of the contralateral femoral head after a minimum follow-up of 1 year.
Several limitations need to be considered when interpreting the data of this study. A surprisingly large number of patients refused to participate in the study, although hardly any additional effort was necessary by the patient apart from filling in the questionnaire. We attribute this mostly to the bad reputation of dexamethasone in the general population, as many fear the known complications associated with long-term cortisone therapy. On the other hand, it may also be due to the general study fatigue of young patients who prefer not to deal with "unnecessary" things. Of the 60 patients who were allocated, 10 were excluded after allocation because of drug or medication use, which was not shared in the first screening. This information was shared in the preoperative anesthesiologist consultation. Patients with cannabis use were excluded, as these patients have an altered CYP3A4 metabolism. Postoperative pain and nausea management is influenced by cannabis use as well as cannabis withdrawal. 23 The ratio of patients in whom the junior surgeon performed the hip arthroscopy was slightly greater in the control group (14/23 [61%]) compared with the study group (15/27 [56%]); however, surgical time and hip distraction time as more objective measures were very comparable. Power analysis was performed based on the primary endpoint of expected differences in postoperative pain levels and not based on differences in opioid consumption. However, we did not see any differences in pain levels between the 2 groups, which can reflect the good postoperative pain management at our institution.
Conclusion
Perioperative IV dexamethasone significantly reduced postoperative opioid consumption by 40% without compromising pain level and safety, as no corticosteroid-related side effects were observed. Dexamethasone may be a valuable adjuvant to a multimodal systemic pain regimen after hip arthroscopy.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465241232157 – Supplemental material for Perioperative Intravenous Dexamethasone Significantly Reduces Postoperative Opioid Requirement and Nausea After Unilateral Elective Hip Arthroscopy: A Randomized Double-blinded Placebo-controlled Trial
Supplemental material, sj-pdf-1-ajs-10.1177_03635465241232157 for Perioperative Intravenous Dexamethasone Significantly Reduces Postoperative Opioid Requirement and Nausea After Unilateral Elective Hip Arthroscopy: A Randomized Double-blinded Placebo-controlled Trial by Dominik Kaiser, Armando Hoch, Dimitris Dimitriou, Tanja Groeber, Hagen Bomberg, José A. Aguirre, Urs Eichenberger and Patrick O. Zingg in The American Journal of Sports Medicine
Footnotes
Submitted September 20, 2023; accepted December 20, 2023.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.