Abstract
Background:
Total meniscectomy for treating massive meniscal tears may lead to joint instability, cartilage degeneration, and even progressive osteoarthritis. The meniscal substitution strategies for advancing reconstruction of the meniscus deserve further investigation.
Hypothesis:
A decellularized meniscal scaffold (DMS) modified with collagen affinity stromal cell–derived factor (C-SDF1α) may facilitate meniscal regeneration and protect cartilage from abrasion.
Study Design:
Controlled laboratory study.
Methods:
The authors first modified DMS with C-SDF1α to fabricate a new meniscal graft (DMS-CBD [collagen-binding domain]). Second, they performed in vitro studies to evaluate the release dynamics, biocompatibility, and differentiation inducibility (osteogenic, chondrogenic, and tenogenic differentiation) on human bone marrow mesenchymal stem cells. Using in vivo studies, they subjected rabbits that received medial meniscectomy to a transplantation procedure to implement their meniscal graft. At postoperative weeks 6 and 12, the meniscal regeneration outcomes and chondroprotective efficacy of the new meniscal graft were evaluated by macroscopic observation, histology, micromechanics, and immunohistochemistry tests.
Results:
In in vitro studies, the optimized DMS-CBD graft showed notable biocompatibility, releasing efficiency, and chondrogenic inducibility. In in vivo studies, the implanted DMS-CBD graft after total meniscectomy promoted the migration of cells and extracellular matrix deposition in transplantation and further facilitated meniscal regeneration and protected articular cartilage from degeneration.
Conclusion:
The new meniscal graft (DMS-CBD) accelerated extracellular matrix deposition and meniscal regeneration and protected articular cartilage from degeneration.
Clinical Relevance:
The results demonstrate that the DMS-CBD graft can serve as a potential meniscal substitution after meniscectomy.
Keywords
Meniscal tears are the most prevalent injury of the knee joint, causing reduced joint instability and leading to cartilage degeneration or even progressive osteoarthritis after meniscal surgery.8,14 Unfortunately, meniscal injuries are difficult to heal without surgical intervention because of the inadequate blood supply. 14 Clinically, the routine treatment strategies for meniscal tears are mainly surgical methods, including arthroscopic suture, meniscectomy, and meniscal allograft transplantation. 14 Although surgical techniques have progressed during the past decades, there still remain challenges in complex meniscal injuries, for which the outcomes of suture or meniscectomy are dissatisfactory and uncertain. To circumvent these problems, allograft transplantation has been introduced into clinical practice for patients with massive meniscal injuries and experiencing early stages of arthritis.24,36 However, the wide utilization of commercialized meniscal allografts is limited by several obstacles, such as a shortage of donors, implant shrinkage, and immune rejection. To date, considering the poor self-healing potential of the meniscus, how to promote the reconstruction of massive meniscal tears represents a formidable challenge for sports medicine surgeons.
Given the rapid onset of meniscal injuries and growing demand, coupled with the limited availability and narrow inclusion criteria of meniscal allograft, bioengineering technologies are being developed to provide us with ideal scaffolds to address the problem. 10 In general, meniscus-engineered scaffolds are fabricated by either synthetic or natural materials, such as novel artificial materials to bear tensile or compression loads and to absorb shock in joint movement. 53 The real challenges are to mimic both the natural structure and biochemistry of the meniscus and its geometric properties. 38 Excitingly, printing technology advancements have provided us with possibilities to fabricate biomimetic meniscal scaffolds with a native-like anisotropic architecture, biomechanical microenvironment, and bioactive components.15,29
On the other hand, decellularized menisci derived from animal sources have been explored, and fortunately, the appearance of the meniscal acellular matrix has provided us a shortcut, thanks to its low immunogenicity, high biocompatibility, and preservation of native extracellular matrix (ECM).18,40 To date, menisci from a variety of species have been decellularized, and these xenogenic meniscal tissues have similar anatomy and matrices to the human meniscus and are easy to access, and thus have become promising alternatives for transplantation.6,52 According to previous studies, the entire decellularized meniscus has advantages in architecture compatiblity, which provides a natural microenvironment for meniscal cells, like promoting chondrogenic differentiation of the mesenchymal stem cells (MSCs).39,43,57 This type of acellular matrix displays promising results in the functional reconstruction of the injured menisci in rabbit, ovine, and porcine models.22,57 However, concerns over the longevity and revitalization of the acellular meniscal matrix have risen, because only 10% to 25% of mature meniscus contains blood vessels 7 ; how to promote the cell migration onto exogenous transplantation needs further investigation.
Up to now, the most accepted method to facilitate the revitalization of acellular matrix is to load grafts with seeding cells. 23 However, the majority of loaded cells are wasted under an injured microenvironment after implantation, and only a few will survive to regenerate the tissue.11,50 Our team has developed effective cell-free transplantation strategies by modifying the ECM with recombinant collagen affinity stromal cell–derived factor-1α (C-SDF1α), to give grafts the ability to recruit stem cells into the healing site.3,27 Stromal cell–derived factor 1α (SDF1α) is known to be involved in numerous stem cell biological and therapeutic processes, including cellular trafficking and particularly chondrogenic stem cell homing,32,56 and extensive research using SDF1α to recruit bone marrow MSCs (BMSCs) for cartilage repair has been launched. 26
In this study, we hypothesized that the decellularized meniscal scaffold (DMS) fabricated along with C-SDF1α (DMS-CBD [collagen-binding domain]) could promote revitalization of graft and benefit recovery after meniscal transplantation. To test this hypothesis, a rabbit medial meniscal transplantation model was used, and the SDF1α release dynamics, chemotactic functions, chondrogenic effects, and in vivo evaluation of knee joints were evaluated. Abbreviations used in this paper are provided in Table 1.
Abbreviations

Methods
All experimental protocols were approved by the Animal Ethics Committee of Central South University (No. 201912524).
Preparation of DMS
The meniscal samples for preparation of DMS were obtained from New Zealand White rabbits (3.0-3.5 kg). The medial menisci of rabbits were harvested and subject to decellularization on the germ-free operation table according to previously reported protocols with some adjustments.1,47 Briefly, menisci were washed with phosphate-buffered saline (PBS) 3 times and received 5 freeze-thaw cycles (freezing: liquid nitrogen for 5 minutes; thawing: 37°C water bath for 10 minutes) to break down the cell membrane. The samples were then submerged in 0.1% Triton X-100 containing 1% penicillin-streptomycin (replaced every 24 hours) and placed in a shaker with unremitting agitation for 3 days, 7 days, and 14 days at 4°C, respectively. Then, the samples were immersed in PBS containing 1% penicillin-streptomycin with agitation 3 times (2 hours each time) at 4°C, followed by incubation in a saline solution containing 300 U/mL DNase (Sigma-Aldrich) and 500 µg/mL RNase A (Sigma-Aldrich) at 37°C for 24 hours. After the mixed nuclease solution was discarded, the samples were placed in a rotator shaker with vigorous agitation 3 times (6 hours each time) to completely remove the debris. Subsequently, the native menisci and decellularized menisci were lyophilized in the vacuum freeze drier (FD8-5T; SIM) for 24 hours. Ethylene oxide disinfection was applied to sterilize the dehydrated menisci. Then, the dry weight of the dehydrated menisci was measured.
Morphological and Content Characterization of Decellularized Meniscus
The native menisci and 3 groups of menisci under different decellularization times (3 days, 7 days, and 14 days) were fixed in 4% (vol/vol) paraformaldehyde for 24 hours and embedded in paraffin followed by sectioning into 5-µm slices. To assess the residual nucleus debris and the preservation of ECM components, the meniscal slices were separately subjected to hematoxylin and eosin (H&E), toluidine blue, Sirius red, and 4′,6-diamidino-2-phenylindole (DAPI) staining, according to the manufacturers’ guidelines.
To quantify the contents of meniscal proteoglycan and collagen fiber after different days of decellularization, a synchrotron radiation Fourier transform infrared spectroscopy (SR-FTIR) 59 test was performed at BL01B beamline of the National Facility for Protein Science Shanghai (Shanghai Synchrotron Radiation Facility, China). The peak areas of amide I (1720-1590 cm−1) and carbohydrate (1140-985 cm−1) in the infrared spectrum were calculated to characterize the distribution and content of collagen and proteoglycans according to previous literature.47,60
Scanning electron microscopy (SEM) was performed to define the microstructure change after decellularization. The native menisci and optimal decellularized menisci were fixed in 2.5% glutaraldehyde and dehydrated for SEM to observe the ultrastructural characteristics.
Synthesis of C-SDF1α
C-SDF1α was synthesized according to the published literature. 27 Briefly, the C-terminus of SDF1α was designed to incorporate the CBD connected with the linker sequence (GSAGSAAGSGG). The pET vector carrying the C-SDF1α gene was inserted into the Escherichia coli (Novagen) BL21 (DE3) strain to express C-SDF1α. Subsequently, C-SDF1α was purified with a nickel affinity chromatography column (GE Healthcare).
Evaluating the Binding and Releasing Ability of C-SDF1α on DMS Scaffold
The collagen-binding ability of C-SDF1α was tested according to previous studies.27,46 Briefly, DMS samples with similar weights were placed in a 48-well plate. Then, C-SDF1α or N-SDF1α (nature SDF1α) with concentrations ranging from 0 to 40 μM dissolved in the same volume of PBS was added into the wells. After incubation overnight at 4°C, the samples were gently washed 3 times to remove the unbound factors. A primary antibody against His6 tag (1:1000; Sigma) and an alkaline phosphatase–conjugated goat anti-mouse IgG (1:10,000; Sigma) was applied to detect the residual factors bound to collagen. The optical density (OD) values of the reaction system were measured at 405 nm with a microplate reader (Thermo Fisher).
The collagen-binding ability of C-SDF1α was tested according to previous studies.27,46 The dehydrated DMS specimens were placed in a 48-well plate, and then immersed in 400 μL PBS solution containing 32 μg C-SDF1α or N-SDF1α. After the unbound factors were removed, the specimens were placed in a 48-well plate and rinsed with 500 μL PBS. The mixed system was placed on a rocker platform (37°C, 80g) to imitate body fluid erosion, and the solution containing released factors was collected and replaced every 24 hours. At the same time point for each day (days 0 to 11), the released factors were detected and analyzed with a human SDF1α ELISA kit (EK1119; Multi Sciences).
Bioevaluation of DMS-CBD on hBMSCs
Cell viability of DMS-CBD was observed by live/dead test and proliferation assay. Human bone marrow MSCs (hBMSCs) (Appendix Figure A1, available in the online version of this article) were seeded at a density of 2.5 × 105/cm2 onto DMS-CBD or tissue culture plates (TCPs) (control group) for 3 days. After the culture plate was washed with PBS, a calcein-AM/PI double stain kit (40747ES76; Yeasen) was applied to detect the survival status of cultured cells in the plate. The cell viability (green: live; red: dead) was observed using a fluorescence microscope (Leica) with an excitation wavelength of 488/594 nm. Moreover, hBMSCs were seeded at a density of 2.5 × 105/cm2 onto DMS, DMS-CBD, or TCPs (control group), followed by testing with a Cell Counting Kit-8 (CCK-8) (CA1210; Solarbio) at days 1, 5, 7, 9, and 11. A microplate reader (Thermo Scientific) was used to measure the absorbance at 450 nm.
The chemotactic ability of DMS-CBD was tested by transwell assay. DMS or DMS-CBD was placed in the lower chamber containing 600 μL complete medium containing 500 ng natural SDF1α (N-SDF1α) or C-SDF1α, and the transwell plate (3422; Corning Costar) wells without scaffolds were set as the control. In total, 1 × 104 hBMSCs were suspended in 100 μL serum-free culture medium and added into the upper chamber. After incubation for 24 hours, the chambers were washed with PBS and the cells were fixed and stained with 0.5% crystal violet solution. Subsequently, the migrated cells were observed under the microscope.
Chondrogenic, osteogenic, and tenogenic inducibility of DMS or DMS-CBD on hBMSC differentiation was tested in co-culture with hBMSCs. Briefly, hBMSCs with a density of 2.5 × 105/cm2 were seeded onto DMS, DMS-CBD, or culture plate (control group). After co-culture for 14 days, the culture plate was washed with PBS and fixed with 4% paraformaldehyde. After the cell membranes were ruptured with 0.1% Triton X-100 (Sigma-Aldrich), 3% bovine serum albumin was applied to block the nonspecific binding. Then, the primary antibodies against SOX9 (ab185966; Abcam), RUNX2 (ab192256; Abcam), or SCX (ab58655; Abcam) were added. After the secondary related corresponding antibodies were combined with the primary antibodies, a confocal microscope (TCS-SP8; Leica) was used to capture the image, and the positive cells were calculated. Moreover, the RNA was extracted and a quantitative real-time polymerase chain reaction (qRT-PCR) assay was performed to analyze the expression of ANCA, SOX9, COL10, RUNX2, BMP2, BMP4, OPG, COL1, SCX, and TNMD in the co-cultured cells. The primer sequences are listed in Appendix Table A1 (available online).
Grouping and Surgical Technique
New Zealand White rabbits were randomly allocated into 4 groups: total meniscectomy with DMS-CBD (DMS-CBD group; n = 20), total meniscectomy with DMS (DMS group; n = 20), total meniscectomy with no graft implant (positive control group; n = 20), and sham operation group. At a time point of 6 weeks (n = 10 for each group) or 12 weeks (n = 10 for each group) postoperatively, the rabbits were euthanized and the specimens were harvested for evaluation of gross view and histological, biomechanical, and immunohistochemical tests.
The meniscal transplantation was performed as previous reported.28,58 Briefly, after intramuscular injection of penicillin (100,000 U/kg) for preoperative prophylaxis, the rabbits were anesthetized using intramuscular injections of 160 mg ketamine and 12 mg xylazine. The right medial meniscus was exposed through a medial parapatellar incision, and the medial collateral ligament was partially cut down to preserve postoperative stability. A total meniscectomy was performed to dissect the medial meniscus except for the external rim. The anterior and posterior horns of the DMS or CBD-DMS were sutured to the corresponding reserved rim. Then, the capsular ligament and incision were closed. After the operation, the rabbits were returned to the cages and allowed to move and feed freely.
Macroscopic Observations
The degree of the reconstructed meniscus was evaluated via gross examination, and the fully exposed femoral condyle, tibial plateau, and neo-menisci were observed and photographed. The presence of osteophytes was measured from 0 for no osteophytes to 3 for marked osteophyte formation according to a previous report. 49
Evaluation of Articular Cartilage Degradation
The osteochondral tissue of the femoral condyle and tibial plateau surface were isolated and fixed in 4% (vol/vol) paraformaldehyde, followed by 0.5 M EDTA decalcifying fluid. The decalcified specimens were embedded in paraffin and sectioned into 5-μm slides in the coronal plane at the midpoint of the tibial plateau and in the sagittal plane at the midpoint of the medial femoral condyle. 21 The slides were then subject to H&E, toluidine blue/fast green, and safranin O/fast green staining. To evaluate the degradation of articular cartilages, the International Cartilage Regeneration & Joint Preservation Society (ICRS) and Mankin grading systems were blindly performed by 3 colleagues.2,31
Histological and Immunohistochemical Analyses of the Implants
The regenerated menisci from the DMS and DMS-CBD groups were sectioned into 3 parts (outer, median, and inner) and then fixed in 4% paraformaldehyde and embedded in paraffin. The embedded specimens were sliced into 5 μm sections, which were then subject to H&E, toluidine blue, and Sirius red for histological evaluation. Moreover, immunohistochemistry was performed to evaluate the content of collagen types 1 (COL1) and 2 (COL2). Integral optical density (IDO) was measured to calculate the immunohistochemical positive rate of COL1 and COL2.
Biomechanical Assay
The implants at postoperative 6 and 12 weeks and the native meniscus were analyzed for biomechanical properties as previously described.13,44 The Young modulus of the samples was measured using the nano-indenter (UNHT3; Anton Paar) (see Figure 8A). The sample was cut into 7 μm in thickness and tested at a load of 200 μN. The maximum indentation depth was 500 nm. In this study, the Poisson ratio of the meniscus was assumed as 0.3. The microscopic geomorphology of the indentation zones was captured using a microscanning apparatus. The strain-stress curves were used to calculate the Young modulus. The relative Young modulus was calculated as follows: Et/E0 × 100%, where E0 indicates the Young modulus of the native meniscus and Et is the Young modulus of implants.
Statistical Analysis
One-way ANOVA and the Student t test were performed to assess whether there were statistically significant differences in the results between time groups. Histological scoring of tendon insertions was evaluated by the nonparametric Mann-Whitney test. Values of P < .05 were considered to be statistically significant. Data were analyzed using Prism 7 software (GraphPad).
Results
Morphological and Content Characterization of Decellularized Meniscus
We first sought to find an optimized protocol to decellularize the menisci and preserve the ECM as much as possible. As shown in Figure 1A, H&E staining demonstrated that the residual cells were significantly removed with decellularize time increasing, while most of the meniscal structure was preserved after decellularization. DAPI (which could effectively dye the nucleus) and H&E staining showed that cellular components were effectively removed at 7 days and 14 days, while cellular debris was still observable after 3 days of decellularization. There was no obvious difference between 7 days and 14 days regarding aspects of residual cellular debris, suggesting 7 days of our meniscal decellularization was enough to remove cellular components inside the meniscus (Figure 1A).
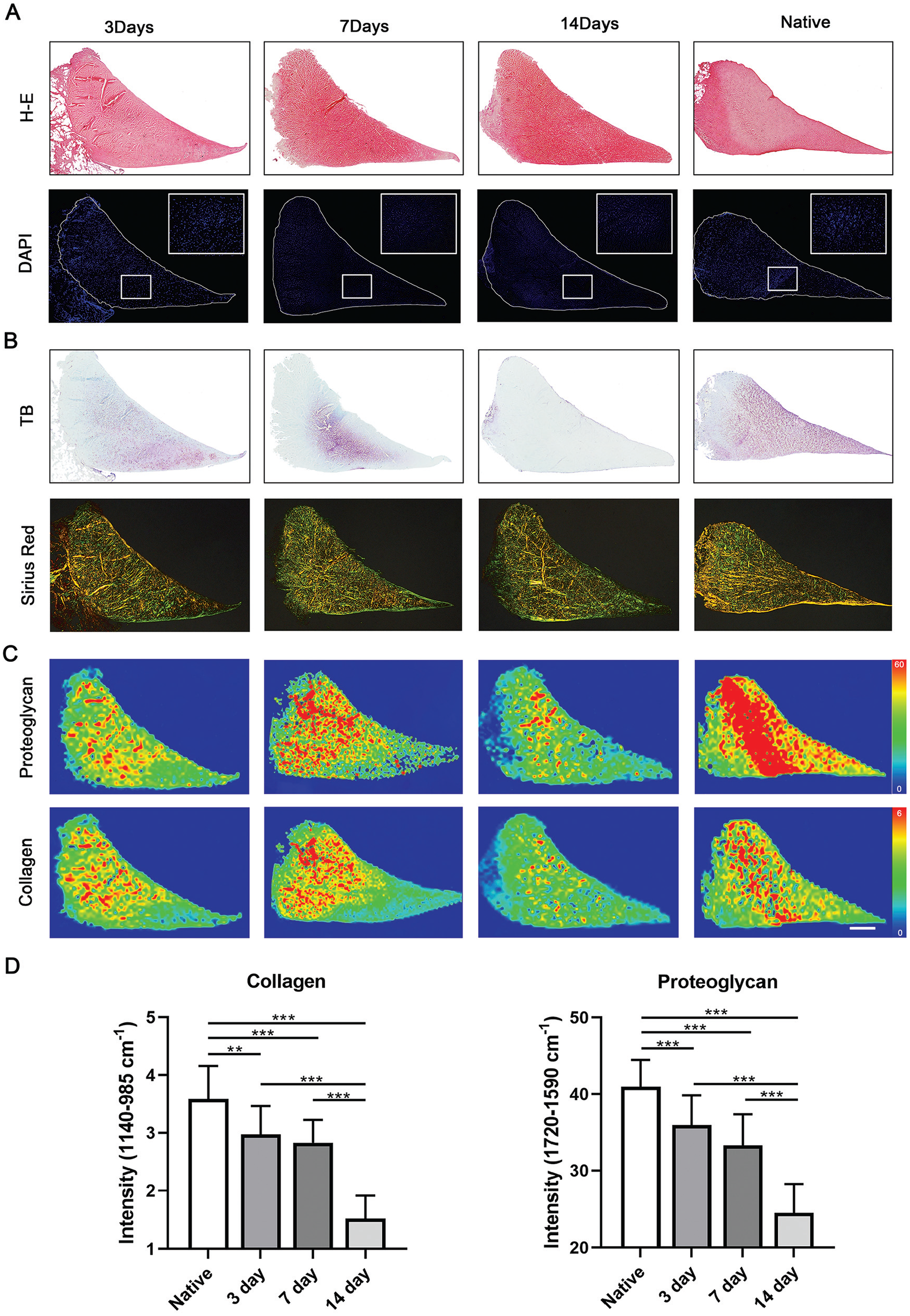
Seven days of decellularization effectively removed meniscal cells and satisfactorily retained most ECM content. (A) H&E and DAPI staining of the decellularized meniscus reveal the overall structure change and residual cells with different decellularizing time points. (B) Toluidine blue (TB) and Sirius red staining show the preservation of meniscal proteoglycan and collagen left after different time points. (C) SR-FTIR mappings including proteoglycan and collagen distribution of native or decellularized scaffolds in different time groups. (D) Semiquantitative analysis of proteoglycan and collagen content of native or acellular scaffolds. Scale bar: 100 μm. **P < .01; ***P < .001.
We next stained the extracellular proteoglycan and collagen with toluidine blue and Sirius red, respectively. These showed that most of the proteoglycan and collagen was retained in the 7-day group, while most of the proteoglycan was lost in the 14-day group (Figure 1B). Innovatively, we performed SR-FTIR analysis to quantify the contents of proteoglycan and collagen fiber after different days of decellularization. The results of SR-FTIR showed a striking decrease in collagen and proteoglycan content after 14 days, while collagen and proteoglycan was mostly preserved in the 7-day group in comparison with the 3-day group (Figure 1, C and D). Taken together, these results show that 7 days of meniscal decellularization achieved a balance between cell removal and ECM preservation.
Macroscopic and Microscopic Observation of DMS
As shown in Figure 2A, the length of the native meniscus or DMS from the anterior facet to the posterior facet was approximately 13 mm. Moreover, DMS kept a similar appearance as a native meniscus. As for the microstructure, SEM images showed a more porous structure in DMS than in the native meniscus (Figure 2B). Additionally, there was a noticeable loss of dry weight in DMS compared with the native meniscus (Figure 2C).
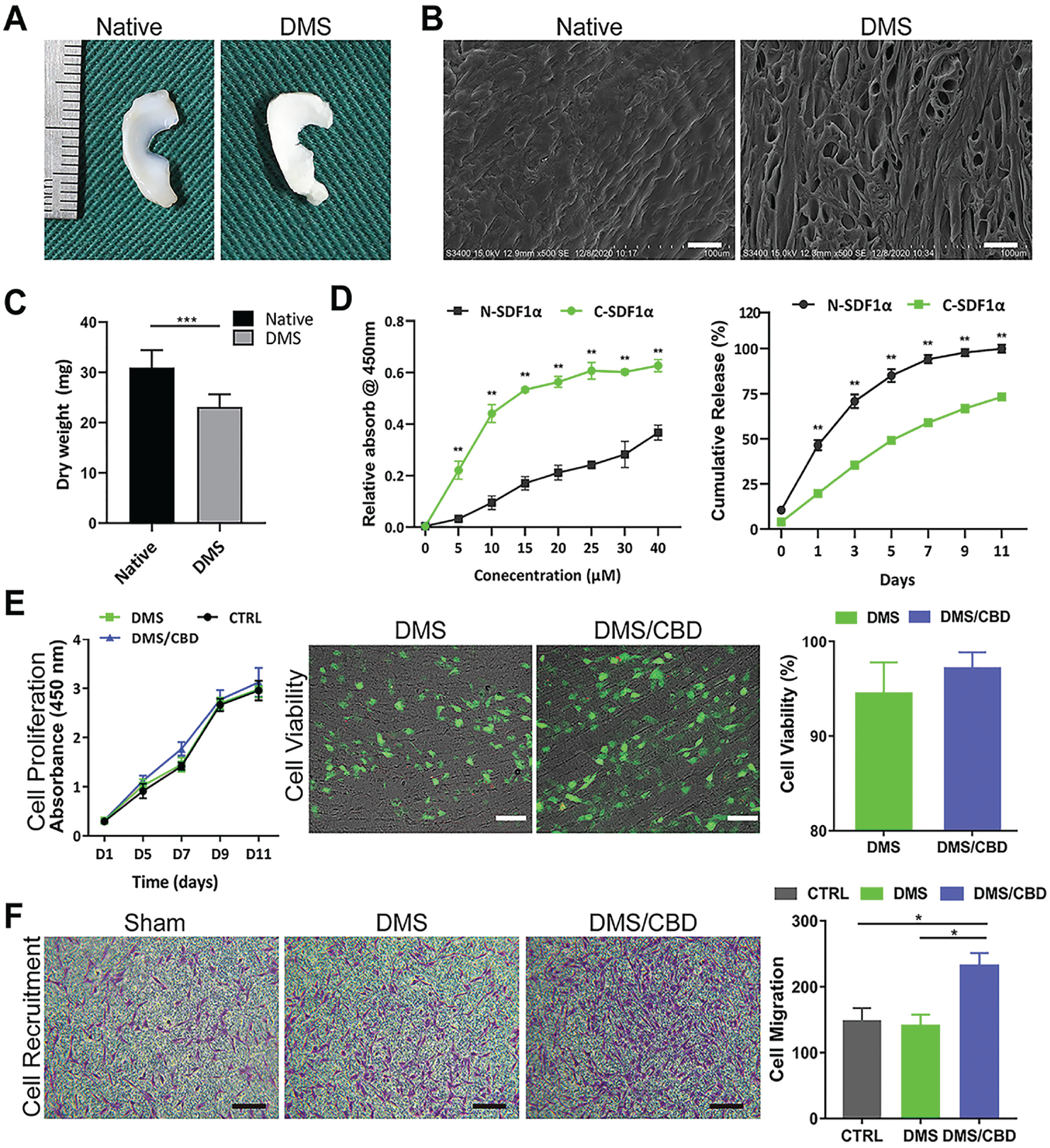
Biophysical features, cytocompatibility, and chemotactic effect of C-SDF1α–conjugated DMS. (A) Macroscopic images of native medial meniscus and DMS. (B) Scanning electron microscopy revealed the microstructure change inside the native medial meniscus and DMS. Scale bar: 150 μm. (C) Dry weight of the native meniscus and DMS (n = 10). (D) Comparative curve graphs showing the binding and releasing of N-SDF1α and C-SDF1α on decellularized meniscus (n = 3). (E) Left: CCK-8 assay showing the viability of hBMSCs loaded on DMS or C-SDF1α–conjugated DMS (DMS-CBD) (n = 4). Center: Live/dead assay showing that hBMSCs were alive (stained green) and dead (stained red) after loading onto DMS or C-SDF1α–conjugated DMS (DMS-CBD) (n = 3). Right: Survival rate of cells loaded on DMS-CBD was slightly higher than that in the DMS. (F) Transwell results show the cytocompatibility and chemotaxis differences between DMS and DMS-CBD scaffold (n = 4). Scale bar: 50 μm. The graph shows differences in cell migration between the DMS-CBD and DMS. *P < .05; **P < .01; ***P < .001. Ctrl, control.
Comparison of Binding Capability and Release Pattern Between N-SDF1α and C-SDF1α on Decellularized Meniscus
The CBD was fused to the N-terminus of natural SDF1α (N-SDF1α) with a linker sequence to avoid influencing its bioactivity, making it easier to bind onto scaffolds enriched in collagens, such as cartilage. 27 To detect the collagen-binding ability of CBD-fused SDF1α (C-SDF1α) that bound to our decellularized meniscus, an equal amount of N-SDF1α or C-SDF1α was incubated with DMS for the same period and then detected by ELISA, and the OD values were measured. As shown in Figure 2D (left graph), the binding volume of N-SDF1α or C-SDF1α was dose dependent, where C-SDF1α exhibited stronger binding capability ranging from 5 to 40 μM and reached saturation at the incubation concentration of 25 μM.
Meanwhile, the releasing assay was performed. As shown in Figure 2D (right graph), C-SDF1α was sustainedly released in a gentle manner, with 75% of the total until the end of observation (day 11). Nearly the entire amount of non–CBD-fused SDF1α (N-SDF1α) was observed to be released by day 9. Therefore, the results demonstrated that CBD-fused SDF1α (C-SDF1α) exhibited a stronger binding capacity and slower release rate than natural SDF1α.
In Vitro Cell Cytotoxicity Evaluation of C-SDF1α–Conjugated DMS
The CCK-8 assay was performed to measure the viability of hBMSCs loaded on DMS or C-SDF1α–conjugated DMS (DMS-CBD) (Figure 2E, left graph). It showed no significant difference regarding proliferation capability of hBMSC load onto DMS or DMS-CBD. The live/dead assay showed that most BMSCs were alive (stained green) and dispersed in this space, and very few BMSCs were dead (stained red). Although the proportion of the survival rate of cells loaded on DMS-CBD was slightly higher than that in the DMS group, there was no statistical significance between the 2 groups (Figure 2E, right graph). The results of the CCK-8 assay and live/dead assay confirmed the favorable cytocompatibility of DMS-CBD.
In Vitro Chemotactic Effect of C-SDF1α–Conjugated DMS
SDF1α has been identified for its cellular trafficking and particularly the chondrogenic stem cell homing effect,33,56 and extensive research has used SDF1α to recruit BMSCs. 26 We performed a transwell assay to test the chemotactic ability of the C-SDF1α–conjugated DMS. As shown in Figure 2F, the DMS-CBD group attracted substantially more cells compared with the control or DMS group, suggestive of excellent chemotaxis effects of C-SDF1α in attracting hBMSCs.
In Vitro Evaluation of hBMSC Differentiation Induced by DMS-CBD
We first stained the chondrogenic (SOX9), osteogenic (RUNX2), and tenogenic (SCX) proteins to measure the differentiation outcomes of hBMSCs after loading onto DMS or DMS-CBD (Figure 3A). Expression level of chondrogenic protein SOX9 was significantly upregulated in the DMS and DMS-CBD groups compared with that in the control group, but there was no statistical difference between the DMS and DMS-CBD groups. Meanwhile, there was no significant difference among the control, DMS, and DMS-CBD groups regarding the expression of osteogenic protein (RUNX2) or tenogenic protein (SCX) (Figure 3A, right panel). In addition, we performed qRT-PCR analysis to test the chondrogenic-, osteogenic-, and tenogenic-related gene expressions in hBMSCs after being loaded onto DMS or DMS-CBD. The results showed that higher-expressed chondrogenic genes (ACAN, SOX9, and COL10) were observed in the DMS and DMS-CBD groups than in the control group, after either 7 days or 14 days of in vitro culture (Figure 3B). Osteogenic-related genes (RUNX2, BMP2, OCN, ALP, BMP2, and BMP4) showed no significant change between the DMS and DMS-CBD groups, except for OPG, whose expressions in the DMS and DMS-CBD groups was higher than that in the control group (Figure 3B and Appendix Figure A2, available online).
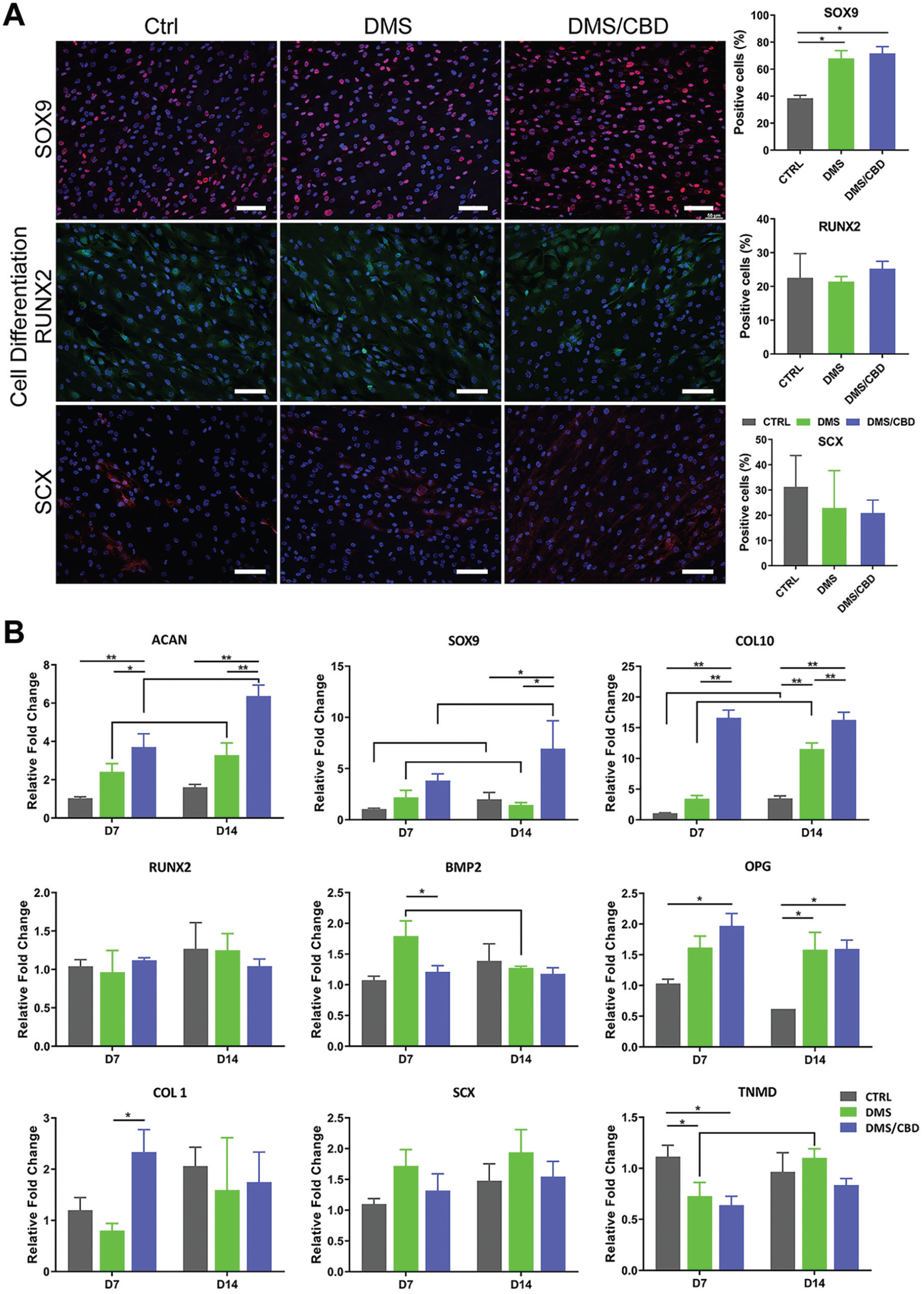
In vitro evaluation of hBMSC differentiation induced by DMS-CBD. (A) Immunofluorescence staining revealing expressions of osteogenic (RUNX2), chondrogenic (SOX9), and tenogenic (SCX) proteins in hBMSCs after loading onto DMS or C-SDF1α–conjugated DMS (DMS-CBD) (n = 4). Scale bar: 100 μm. (B) qRT-PCR results showing the chondrogenic-related (ACAN, SOX9, COL10A), osteogenic-related (RUNX2, BMP2, OPG), and tenogenic-related (COL1A1, SCX, TNMD) gene expressions in hBMSCs after loading onto DMS or DMS-CBD (n = 3). *P < .05; **P < .01. Ctrl, control.
The results showed that higher-expressed tenogenic-related genes(COL1) were observed in the DMS-CBD groups than in the DMS and control group after 7 days of in vitro culture, while lower-expressed tenogenic-related genes(TNMD) were observed in the DMS and DMS-CBD groups than in control group after 7 days and tenogenic-related genes(COL1) showed no significant change between three groups (Figure 3B).
Macroscopic Evaluation of Implanted Grafts and Joint Cartilage Degeneration
Gross views from macroscopic observation showed that both groups (DMS and the DMS/CBD-scaffold group) formed meniscus-like tissue in situ. The gross appearance of the meniscus in the DMS-CBD group was superior to that in the DMS group at the 6- and 12-week time points, presenting a complete and uniformly regenerated meniscus. Specifically, the implanted meniscal grafts in the DMS-CBD group were slightly degraded and looked similar to the shape of a native meniscus after 6 or 12 weeks of transplantation (Figure 4A). In contrast, meniscal grafts in the DMS group were degraded. At 12 weeks postoperatively, the explants of the DMS group were completely distorted and even infiltrated with hyperplasia tissue (Figure 4B).
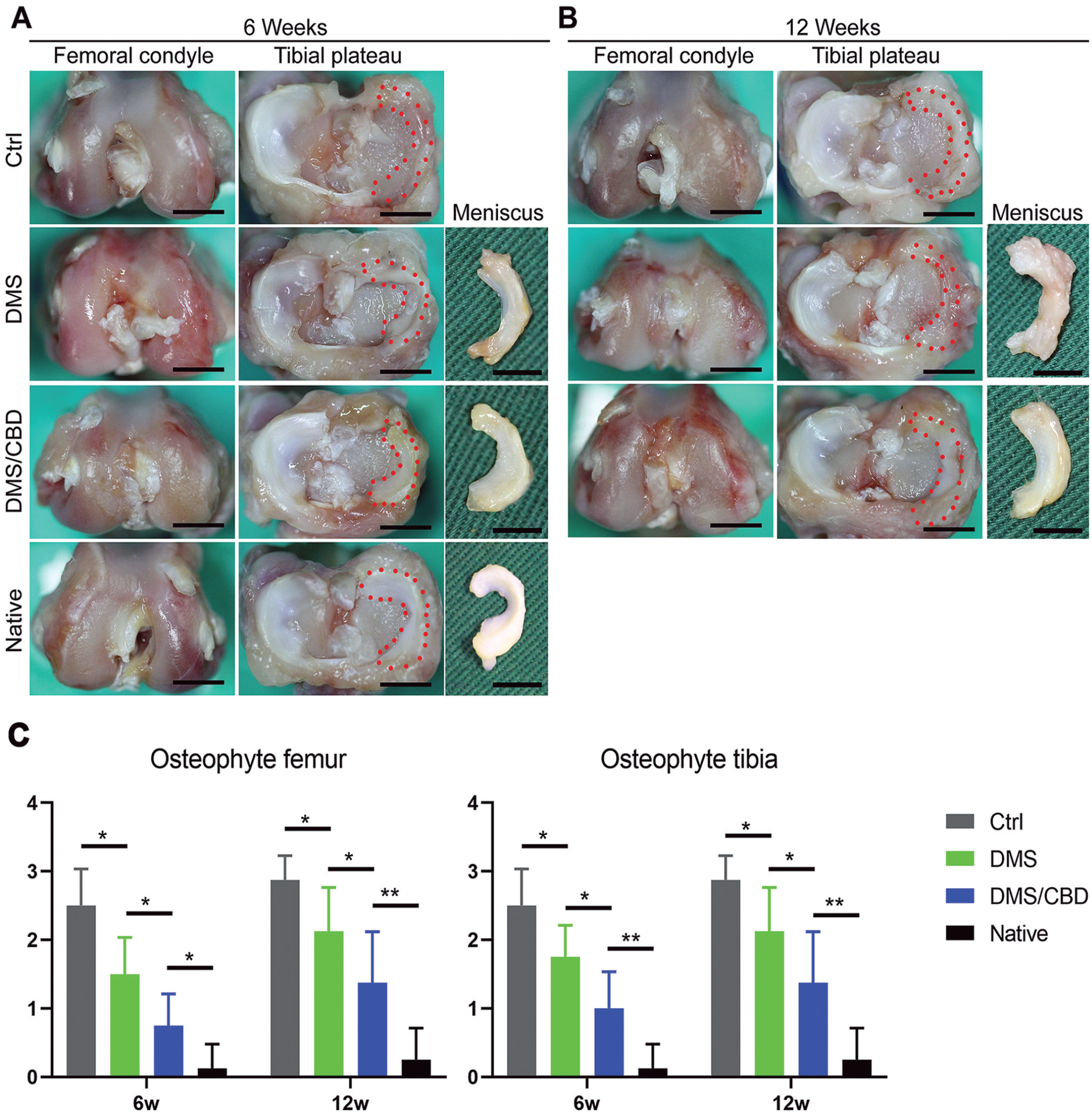
Macroscopic evaluation of implanted grafts and joint cartilage degeneration. Gross observation of tibial plateau, femoral condyles, and meniscus at (A) 6 weeks and (B) 12 weeks after meniscal graft transplantation. (C) Osteophyte grading in the medial femur and tibia. Scale bar: 10 mm. *P < .05; **P < .01. Ctrl, control.
It was important to evaluate the chondroprotective effect of different meniscal transplants. Gross observation of cartilage showed less cartilage degeneration in the DMS-CBD group than in the DMS and control groups at each time point, which was also verified by histological analysis (Figure 5C). Although there was progression in cartilage damage over time in the DMS-CBD group, it was visibly delayed compared with the other 2 groups. After meniscectomy (control group), fibrillation was seen in the articular cartilage of the tibial plateau, with marked osteophytes on the medial side. In the DMS-CBD group, there were no osteophytes found in the lateral compartment, at both postoperative weeks 6 and 12. Furthermore, the presence of osteophytes in each group was graded, and the scores in the DMS-CBD group were graded as significantly better than in the DMS and control groups (Figure 4C).
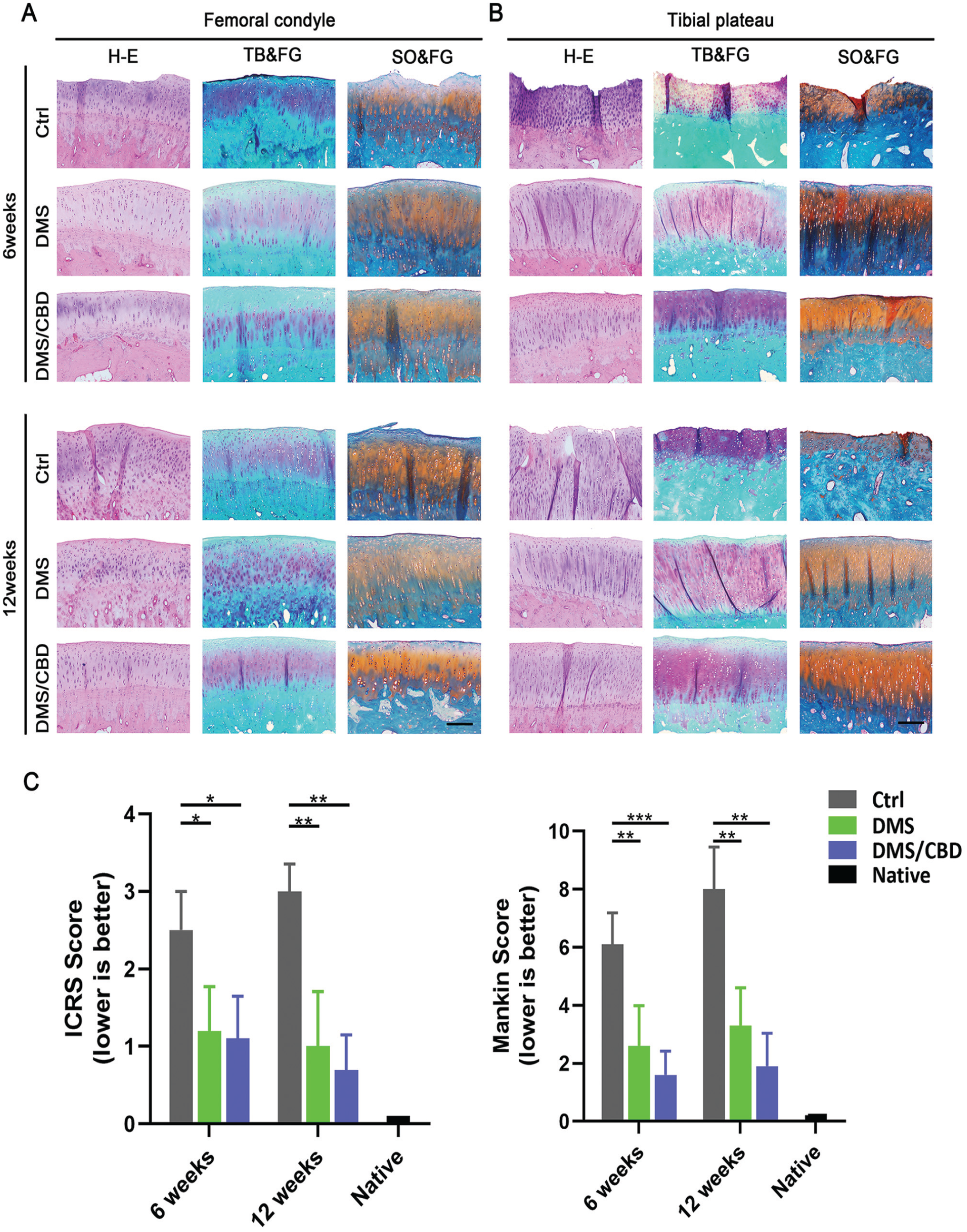
Histological evaluation of joint cartilage degeneration. H&E, toluidine blue/fast green (TB&FG), and safranin O/fast green (SO&FG) staining of the sections of (A) femoral condyle and (B) tibial plateau at 6 and 12 weeks postoperatively. (C) International Cartilage Regeneration & Joint Preservation Society (ICRS) and Mankin scores of articular cartilage surfaces in the femoral condyle and tibial plateau (n = 5). Scale bar: 200 μm. *P < .05; **P < .01; ***P < .001. Ctrl, control.
Histological Evaluation of Joint Cartilage Degeneration
To evaluate the degree of corresponding articular cartilage injury after the operation, histological staining and cartilage damage scoring, including ICRS and Mankin scoring, were performed. As shown in Figure 5 (A and B), the articular cartilage of the tibial plateau and femoral condyles presented progressive osteoarthritis-like changes over time, manifested as disorganized arrangement and hypertrophy of chondrocytes, and even extensive exposure of subchondral bone. In comparison with the articular damage in the control group, the joint surfaces in the DMS and DMS-CBD groups were relatively less damaged, as less cartilage degeneration and fewer cartilage clefts were observed. However, the cartilage of the DMS and DMS-CBD groups was gradually damaged to a certain degree over time compared with the native joints. Semiquantitively, the ICRS and Mankin scores showed the best cartilage performance progressing with time in the DMS-CBD group followed by the DMS group, significantly better than that in the control group. Overall, the DMS-CBD group showed the best cartilage outcomes as flatter surfaces with scarcely any cracks.
Histological Evaluation of Meniscal Graft Regeneration
The histological features of implants were assessed to analyze the meniscal regeneration. Toluidine blue/fast green staining was performed to show the content of proteoglycan inside the meniscal graft. As shown in Figure 6A, in the DMS and DMS-CBD groups, proteoglycan is stained purple and is spread all over the outer and median regions at week 6 postoperatively. Although not as much as the native meniscus, the cartilaginous matrix deposition in the DMS-CBD group was greater than in the DMS or control group. With healing time increasing, the amount of proteoglycan in the DMS-CBD group appeared similar to that of the native meniscus after 12 weeks of regeneration.
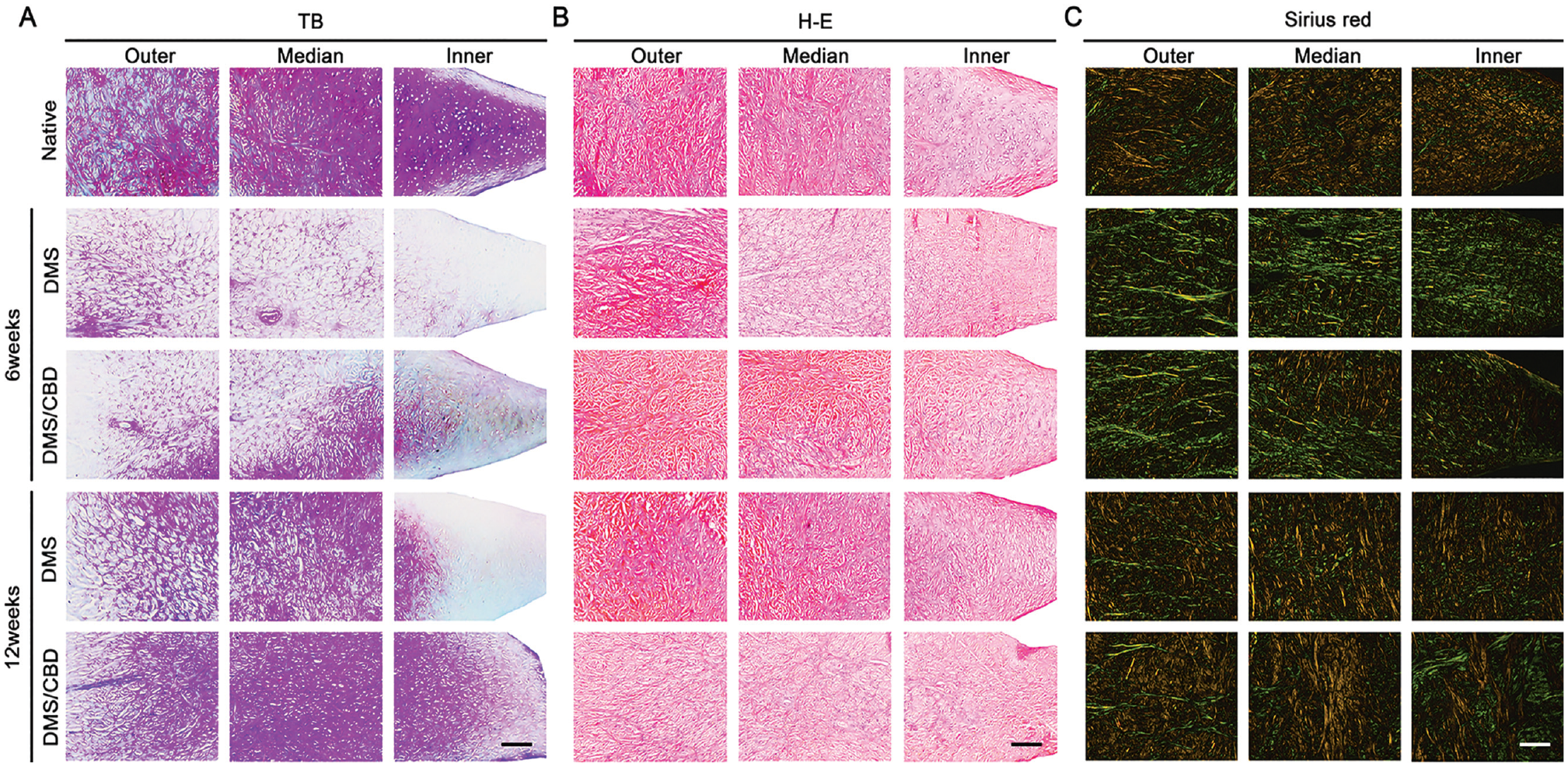
Histological evaluation of meniscal graft regeneration. Histological analysis of native meniscus and meniscal graft at postoperative weeks 6 and 12, including (A) toluidine blue/fast green (TB), (B) H&E, and (C) Sirius red staining. Scale bar: 200 μm.
From the H&E staining pictures, no obvious inflammatory cell infiltration was observed in any group (Figure 6B). After 6 weeks of implantation, elongated fibroblast-like cells accounted for a significant proportion and were mostly distributed in the outer and median parts of the regenerated meniscus. At postoperative week 12, larger amounts of circular chondrocyte-like cells were embedded in grafts of the DMS-CBD group, which represented better cartilage metaplasia and regeneration of meniscal tissue. Sirius red staining (Figure 6C) showed that COL1 and collagen type 3 (COL3) fibers exhibited thick and tightly arranged yellow or green fibers, respectively, while COL2 fibers were arranged as a loose network with weak birefringence of various colors. COL1 and COL3 in the DMS and DMS-CBD groups were similar at postoperative week 6. Then the collagens in the DMS-CBD group increased more noticeably than the DMS group in the outer and median region of the graft at week 12 postoperatively, demonstrating a better-organized arrangement of collagen.
Expression Levels of COL1 and COL2 in Regenerated Implants
The expression levels of COL1 and COL2 were assayed by immunohistochemical analysis (Figure 7). At 6 and 12 weeks after surgery, the DMS-CBD group showed higher expression of COL1 than the DMS or control group. Quantitatively, the expression level of COL1 was confirmed by statistically analyzing the results of the IOD value (Figure 7C). As for the expression of COL2 in meniscal graft, the DMS-CBD group showed significantly higher COL2 content than the DMS group at postoperative week 6, while no significant difference was observed at postoperative week 12 (Figure 7C).
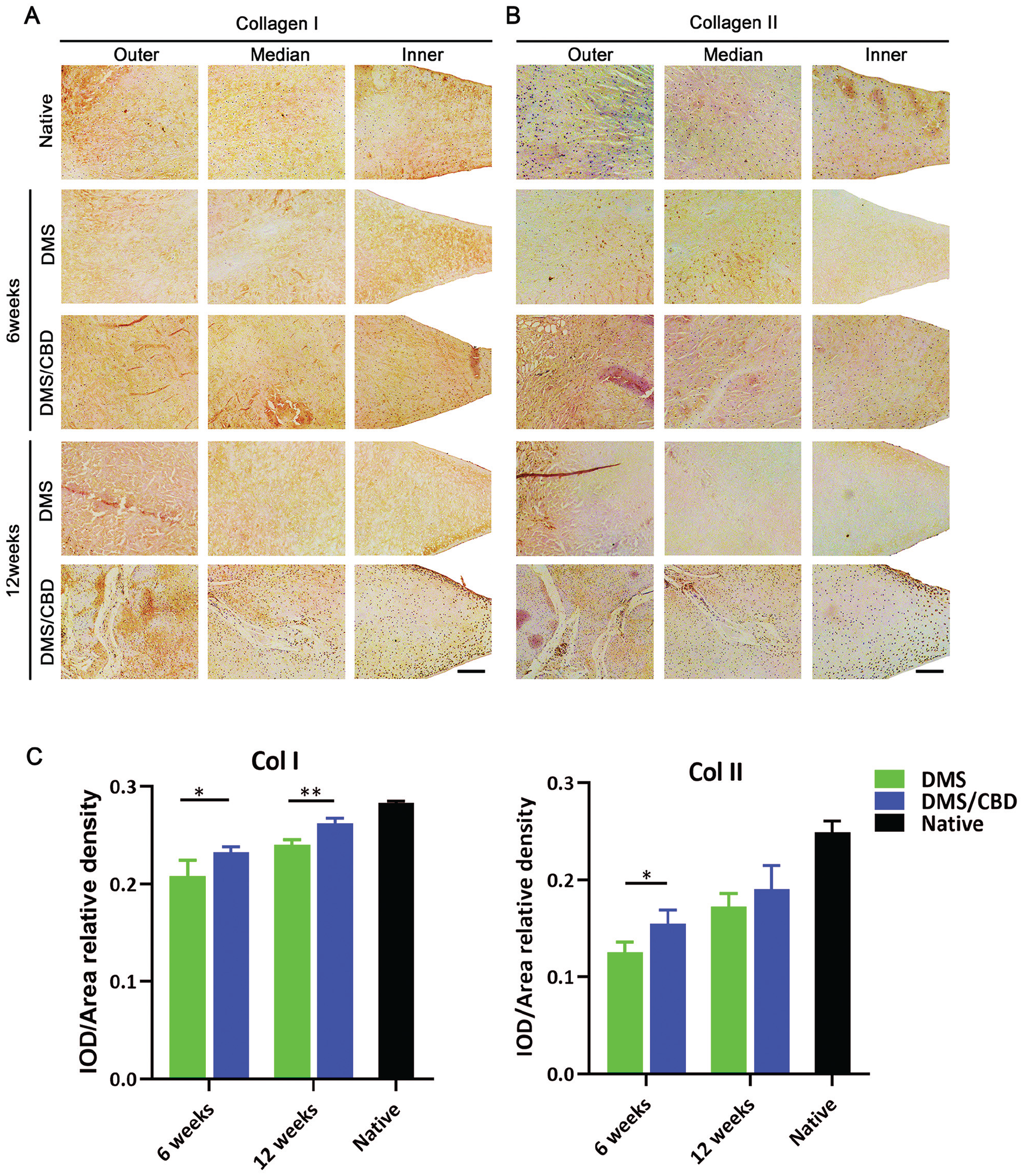
Immunohistochemical staining revealing the expression levels of collagen types (A) 1 (Col I) and (B) 2 (Col II) in meniscal grafts at postoperative weeks 6 and 12 (n = 5). (C) Semiquantitive comparison of integrated optical density (IOD) value. Scale bar: 200 μm. *P < .05; **P < .01.
Biomechanical Properties of Implants
The biomechanical properties of regenerated meniscus were analyzed at 6 and 12 weeks postoperatively, and the results are presented as a percentage of values tested from the native meniscus (Figure 8B). At 6 weeks, the biomechanical properties of implants of both the DMS and DMS-CBD groups were inferior to those in the native meniscus, while the biomechanical properties in the DMS-CBD group were stronger than those of the DMS group. At 12 weeks, the biomechanical properties in the DMS-CBD group were stronger than those of the DMS group, showing higher similarity to the native meniscus (Figure 8B).
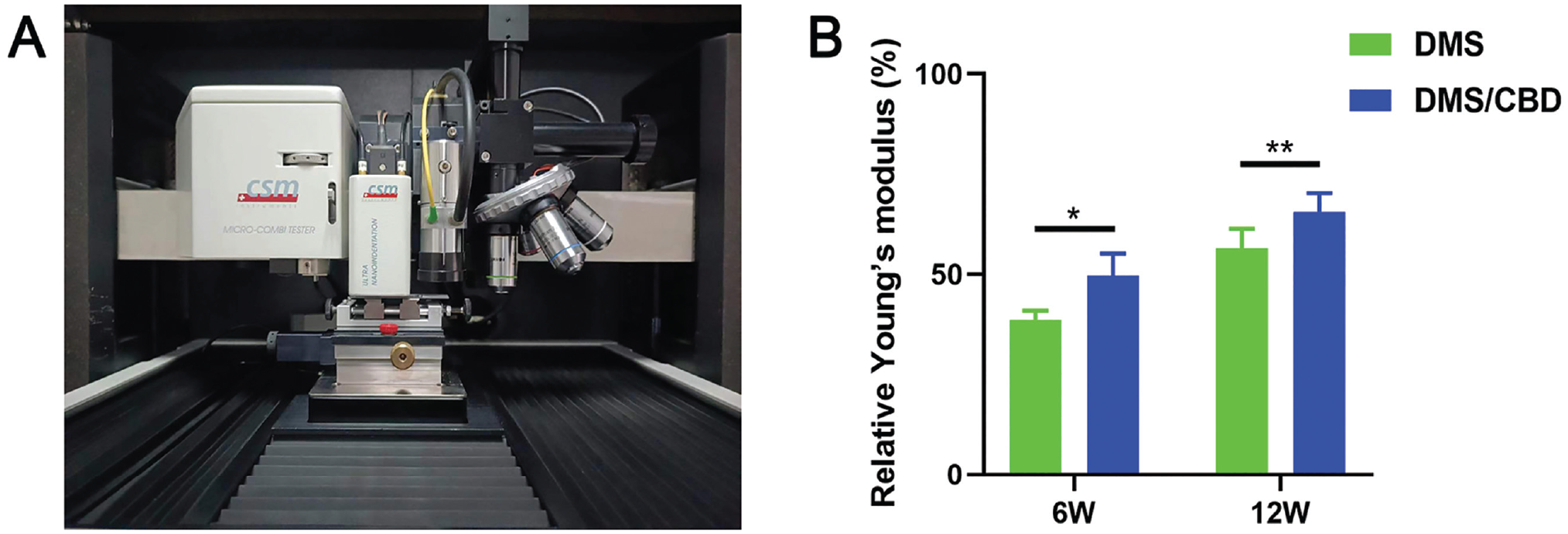
Biomechanical properties of implants from DMS and DMS-CBD groups at 6 and 12 weeks postoperatively. (A) The Young modulus of the samples was measured using the nano-indenter (UNHT3; Anton Paar). (B) The results are presented as a percentage of values tested from the native meniscus (n = 4). *P < .05; **P < .01.
Discussion
In the current study, referring to merits in previously published literature,1,27,47,54 we designed a new meniscal substitute for total meniscectomy, composed of DMS and C-SDF1α, named DMS-CBD. Our in vitro results revealed that the DMS-CBD meniscal graft could release C-SDF1α in a sustained manner and exhibit a markedly chemotactic effect on hBMSCs, which subsequently differentiated into chondrocytes because of the excellent biomimetic microstructure of DMS. In vivo, when compared with decellularized meniscus, the implanted DMS-CBD graft showed better healing outcomes as evidenced by abundant cartilage metaplasia and the deposition of proteoglycan, COL1, and COL2, as well as a better protective effect to prevent joint cartilage from abrasion.
Bioengineering has provided us with possibilities to fabricate biomimetic meniscal transplants from either ECM-based materials or artificial sources.10,22 The widely recognized challenge in producing meniscal reconstruction is that the materials used at present can barely meet the requirement in terms of anisotropic microstructure, mechanical functions, and biochemical components of the native meniscus.1,12,30,55 However, progress has been made to overcome these difficulties. Hao et al 15 reported a sufficient biofabrication design of cell-free dual drug-releasing biomimetic scaffolds, and their 3D-printed meniscal scaffold with an anisotropic architecture was able to enhance cell migration and synergistically promote MSC chondrogenic differentiation. Recently, the development of in situ tissue-engineered meniscal scaffolds loaded with growth factors (CTGF and TGF-β3), to provide biochemical signaling to direct endogenous stem/progenitor cell behavior, has been proven to have potential to revolutionize meniscal tissue engineering. 25 On the other hand, decellularized menisci derived from animal sources provided us a shortcut, thanks to its biocompatibility and highly efficient preservation of native ECM.18,40 To date, promising strategies include using synthetic polycaprolactone/hydrogels as supporting scaffold, in combination with natural meniscal ECM to serve as an additive element. 25 These results provide not only a potential substitute that can guide meniscal tissue regeneration but also a promising ready-made cell-instructive meniscal product for clinical use.
Despite the superiority of native ECM illustrated by previous literature, how to better retain the native ECM contents and remove the immunogenic cell components has become the pivotal argument. Immunological rejection might unfortunately lead to the failure of meniscal regeneration, and the main reason is the inadequate decellularization of cell components.1,54 However, excessive decellularization inevitably leads to excessive loss of extracellular components, which constitute key components of the microenvironment that promote cellular growth.19,61 Thus, in this study, an optimized protocol was first investigated to sufficiently decellularize the menisci and to preserve the ECM as much as possible. Our data showed that after 7 days of decellularization (Figure 1), cellular components were completely removed, while collagen and polysaccharide components were retained as much as possible to the hilt. Furthermore, hBMSCs were seeded on DMS-CBD, and immunofluorescence and qRT-PCR were performed to evaluate the chondrogenic, osteogenic, and tenogenic differentiation induced by the retained microstructure of DMS-CBD from the level of both gene transcription and protein expression. It was found that it could effectively promote chondrogenic differentiation of hBMSCs, but no significant difference was found in tendon formation. Moreover, osteogenic-related genes (RUNX2, BMP2, OCN, ALP, BMP2, and BMP4) showed no significant change between the DMS and DMS-CBD groups, except for OPG, whose expressions in the DMS and DMS-CBD groups was higher than that in the control group (Figure 3B and Appendix Figure A2, available online). Given that overexpressed OPG might enhance osteodifferentiation of hBMSCs, we further graded the osteophyte formation in each group. The scores in the DMS-CBD group showed smaller osteophytes than the DMS and control groups, suggesting no osteogenesis was upregulated in the DMS-CBD group.
Among the basis of tissue engineering, seed cells are crucial for the integration, assimilation, and regeneration of scaffolds in vivo.4,41,47 However, applications of exogenous cells are restricted by the host immune rejection and indeterminate survival rate. 16 Therefore, recruiting endogenous cells into explants has been proven to be a better choice to avoid the aforesaid problems. In this study, a CBD with high collagen-binding capacity was fused to SDF1α to promote binding efficiency onto collagen-enriched meniscal cartilage. In previous studies, CBD-SDF1α was tethered to different grafts rich in collagen, and it was found that the modified collagen-abundant scaffolds could sustainedly release SDF1α and recruit endogenous stem cells, thus promoting tissue repair.27,45,46 In our study design, we used SDF1α to recruit endogenous cells into the DMS, which we believed to take place within an early postsurgery period. Therefore, we defined the evaluation time points from 6 weeks and 12 weeks postoperatively, according to previous literature.42,55 In terms of the sources of the recruited cells, previous studies reported that SDF1α could recruit bone marrow–derived stem cells and ligament-derived stem/progenitor cells into articular cartilage and anterior ligament.9,20,51 Newberry et al 34 reported that SDF1α-preconditioned hydroxypropyl cellulose scaffolds mobilized cartilage-derived progenitors and stimulated meniscal fibrocartilage repair in human explant tissue culture. In another study, Nishida et al 35 found that intra-articular injection of stromal SDF1α promoted meniscal healing via macrophage and MSC accumulation in a rat meniscal defect model. Here, our in vitro results showed that DMS modified with CBD-SDF1α could sustainedly release SDF1α to recruit MSCs (Figure 2F), and the in vivo results illustrated that the DMS-CBD group exhibited better meniscal regeneration than the DMS group, suggesting a positive role of our meniscal graft in recruiting endogenous cells to participate in healing.
In this study, immune-related tests about implants were not performed in vitro or in vivo, but relevant studies revealed acellular scaffolds with cellular components completely removed showed low immunogenicity that caused no significant response, appearing as low amounts of neutrophil, eosinophil, and lymphocyte infiltration and no foreign body reaction around or inside the grafts.5,33,37 SDF1α was shown to attract a myriad of cell populations, such as monocytes, lymphocytes, and macrophages and BMSCs, to the injury site. 17 In our previous study, we found DMS-CBD recruited both CXCR4-positive MSCs and inflammatory cells.3,46 Nishida et al 35 found that intra-articular injection of stromal SDF1α enhanced migration of macrophages to the meniscal injury site, as well as MSCs identified as both CD90- and CD105-positive cells. It has been previously reported that macrophages recruited from bone marrow are also involved at an early stage of meniscal healing, and αSMA-positive cells were migrated into the injured meniscal surface and are essential for integration into adjacent meniscal tissue. 48 Taken together, SDF1α could recruit more inflammatory cells into the meniscal site and further benefit regeneration. Future studies to employ single-cell methods will help decipher the immune resource that is recruited to the meniscal site.
There are certain limitations in this study. First, the chemotactic efficacy of DMS-CBD was only assessed in an in vitro experiment, while the source of cells recruited into the explants in vivo needs further study. Second, the interactional mechanisms between migrated cells and DMS-CBD graft remained unclear. Future work will need to investigate the specific cell signaling pathway mechanism induced by C-SDF1α. Except for the listed limitations, our DMS-CBD graft showed great potential in serving as a meniscal substitution after clinical meniscectomy.
Conclusion
We present a modified DMS with C-SDF1α as a new meniscal graft. We found that our optimization strategy was able to achieve superior meniscal regeneration and prevent articular cartilage from degeneration, which may serve as a potential meniscal substitution for clinical meniscectomy.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465231210950 – Supplemental material for Optimized Allogenic Decellularized Meniscal Scaffold Modified by Collagen Affinity Stromal Cell–Derived Factor SDF1α for Meniscal Regeneration: A 6- and 12-Week Animal Study in a Rabbit Model
Supplemental material, sj-pdf-1-ajs-10.1177_03635465231210950 for Optimized Allogenic Decellularized Meniscal Scaffold Modified by Collagen Affinity Stromal Cell–Derived Factor SDF1α for Meniscal Regeneration: A 6- and 12-Week Animal Study in a Rabbit Model by Tao Zhang, Xin Shi, Muzhi Li, Jianzhong Hu and Hongbin Lu in The American Journal of Sports Medicine
Footnotes
Acknowledgements
The authors thank the staff from BL01B beamline of the National Center for Protein Science Shanghai (NCPSS) for their kind assistance during data collection.
Submitted April 7, 2023; accepted September 15, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by the Science and Technology Major Project of Changsha (No. kh2102015) and the National Natural Science Foundation of China (Nos. 82230085 and 82272572). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
