Abstract
Background:
Bone stress injuries (BSIs) are common sports injuries that occur because of an imbalance between microdamage accumulation and removal through bone remodeling. The underlying bone phenotype has been assumed to be a contributing factor. However, the bone microarchitecture of athletes with BSI is not well characterized, and no study has investigated whether impaired bone microarchitecture is associated with bone composition or anatomic site of injury.
Purpose/Hypothesis:
This cross-sectional study characterizes the bone microarchitecture at distal radial and tibial reference locations in athletes with BSI. Based on previous dual-energy X-ray absorptiometry (DXA) findings, the aim was to compare anatomic injury sites, hypothesizing that athletes with BSIs in bones with greater trabecular composition show impaired bone microarchitecture parameters compared with those with BSIs in bones with greater cortical composition.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
Athletes who had presented to our outpatient clinic because of a high-grade BSI (ie, stress fracture) were retrospectively included. Blood and urine samples were collected. Areal bone mineral density (aBMD) was assessed by DXA at the lumbar spine and both hips. Bone microarchitecture was analyzed by high-resolution peripheral quantitative computed tomography (HR-pQCT) at the distal radius and tibia. HR-pQCT parameters were expressed in relation to available sex-, age-, and device-adjusted reference values and compared with a cohort of 53 age- and sex-matched controls.
Results:
In total, 53 athletes had a BSI of the foot (n = 20), tibia/fibula (n = 18), pelvis (n = 9), femur (n = 5), or sternum (n = 1). Based on DXA measurements, a Z-score of −1.0 or lower was found in 32 of 53 (60.4%) of the athletes, of whom 16 of 53 (30.2%) had a Z score −2.0 or lower. While an impairment of cortical area (P = .034 and P = .001) and thickness (P = .029 and P < .001) was detected at the distal radius and tibia in the BSI cohort compared with controls, no differences in BMD or bone microarchitecture were observed between anatomic injury sites. Furthermore, no difference was revealed when BSIs were grouped into cortical- and trabecular-rich sites.
Conclusion:
Reduced aBMD and impaired cortical bone microarchitecture were present in a considerable number of athletes with BSI. Neither aBMD nor bone microarchitecture was related to the injury site, highlighting the multifactorial etiology of BSI.
Regular physical activity generally has a positive effect on bone health.8,37 However, repetitive and excessive loading may cause a form of skeletal overuse injury termed bone stress injury (BSI). 16 Clinically, these injuries are characterized by pain and tenderness at the injury site. Although return to sports is commonly seen, BSI results in a significant injury burden in athletes, as it causes considerable absence from sports participation, 17 and failure to diagnose or treat may lead to complete fracture, reinjury, delayed union, or avascular necrosis. 4
BSI may represent a pathologic continuum from mild edema or periosteal reaction to radiologic evidence of a fracture line demarcation (referred to as stress fracture). 16 For early establishment of the diagnosis and especially for grading of the lesion, magnetic resonance imaging (MRI) is the gold standard within routine clinical practice. 17 BSIs are more prevalent in the lower limbs and pelvis than in the upper limbs, and common anatomic sites of BSIs are the metatarsals, tibia, pelvis, and femoral neck. 41 Paying attention to the anatomic injury site is of value from both the pathophysiologic and routine clinical points of view, especially since the expected time to return to sports and risk for complication are higher for injuries occurring in certain regions, such as the femoral neck, navicular, or fifth metatarsal base (ie, high-risk regions).7,16,17,27
The etiology of BSI is multifactorial, and several risk factors have been identified, including low energy availability, 28 female sex, 11 history of BSI, 19 and an increased acute:chronic workload ratio.12,13,43 Moreover, it has been shown that impaired bone health may contribute to the occurrence of BSI in athletes.1,25,41 Specifically, low areal bone mineral density (aBMD) was observed in 55% of female athletes and 43% of male athletes with BSI.24,41 Furthermore, a lower bone material strength assessed via microindentation was prevalent in women with previous BSI. 14 Low aBMD was shown to relate to BSI at trabecular-rich sites,24,41 suggesting an influence of poor bone quality on the site-specific development of BSI.
While previous data were based on measurements of aBMD using dual-energy X-ray absorptiometry (DXA), high-resolution peripheral quantitative computed tomography (HR-pQCT) allows a more in-depth and 3-dimensional measurement of volumetric BMD (vBMD) and bone microarchitecture. However, only a few studies have performed HR-pQCT examinations in patients with BSI.1,33,35,36,46 Importantly, there have been no HR-pQCT studies investigating the association between alterations in bone microarchitecture and anatomic injury site. Therefore, the aim of this study was to assess bone microarchitecture by HR-pQCT in athletes with BSI and to compare findings between injury sites. The hypothesis was that patients with BSI show an impaired bone microarchitecture, with specific sites (eg, trabecular-rich bones) demonstrating a more pronounced impairment of bone microarchitecture.
Methods
Study Design and Setting
Athletes (aged >17 and <55 years) with a BSI presenting to our outpatient clinic specializing in skeletal disorders between 2019 and 2021 were retrospectively analyzed. BSI was confirmed by MRI and classified as Arendt grade 4 5 or modified Fredericson grade 4b (ie, stress fracture).15,20 As a further inclusion criterion, individuals had to have an onset of symptoms <6 weeks before and ≥4 hours of self-reported weightbearing exercise per week for ≥6 months before injury. Individuals were excluded from the analysis if they had a hereditary bone disorder or chronic diseases known to cause secondary osteoporosis, such as cancer and renal, liver, rheumatic, metabolic, or endocrine disease; history of prolonged immobilization; or treatment-induced bone loss (eg, prolonged glucocorticoid treatment). Height and weight were measured in all individuals at presentation, and body mass index (BMI) was calculated. Medical histories of previous fractures and BSI were obtained through self-reported measures and from medical records.
The BSIs were grouped by the 4 most prevalent sites (sacrum, femoral neck, tibia, metatarsal), as well as cortical- versus trabecular-rich sites adapted from Tenforde et al 41 (cortical-rich sites: tibia, fibula, metatarsal; trabecular-rich sites: sacrum, femoral neck, talus, cuboid, calcaneus, sternum). The present study was carried out in accordance with the local ethics committee (2021-300011-WF) and the ethical standards in the 1964 Declaration of Helsinki. Informed consent was obtained from all individuals.
Biochemical Analysis
For biochemical analyses, nonfasted blood and urine samples were collected at the time of presentation. Serum calcium, phosphate, alkaline phosphatase (ALP), creatinine, ferritin, parathyroid hormone, serum markers of bone formation (ie, osteocalcin and bone-specific alkaline phosphatase [b-ALP]), and the urinary bone resorption marker deoxypyridinoline/creatinine were measured. Automated immune assays were performed to measure 25-hydroxycholecalciferol levels. All parameters were compared with the references derived from the local laboratory.
DXA and HR-pQCT
At presentation, aBMD was measured using DXA (Lunar iDXA; GE Healthcare) in all individuals at the lumbar spine (L1-L4) and both proximal femora. aBMD T- and Z-scores (ie, standard deviation to sex-matched 20- to 29-year-old and sex- and age-matched individuals from the National Health and Nutrition Examination Survey III database, respectively) were calculated using the software provided by the manufacturer. While the lower value of both sides was used for the evaluation of the aBMD hip data, the lowest value of all 3 measurement sites was used for the allocation to the aBMD categories.
To assess the 3-dimensional bone microarchitecture, HR-pQCT scans were carried out at the distal radius and tibia. Patients were scanned using either first- or second-generation HR-pQCT using the default in vivo settings (HR-pQCT, XtremeCT with 60 kVp, 900 μA, 100-ms integration time, and 82-μm voxel size or XtremeCT II with 68 kVp, 1470 μA, 43-ms integration time, 60.7-μm voxel size; Scanco Medical). In each case, the distal tibia contralateral to the side of injury and the nondominant distal radius were scanned. A reference line was placed at the inflection point of the endplate, 44 and the bone microarchitecture was measured using a fixed offset. If motion artifacts of grade 4 or 5 were observed, 29 the respective individuals were excluded from further analysis. The volume of interest was contoured semiautomatically and evaluated using the manufacturer’s standard software. The nomenclature of the obtained parameters follows the recommendations defined by the International Osteoporosis Foundation–American Society for Bone and Mineral Research– European Calcified Tissue Society working group. 44
For HR-pQCT data interpretation, individual results were compared with those of a control group of 53 patients without BSI. This cohort was retrospectively generated from our database and consisted of individuals without a BSI who presented to our department for skeletal health evaluation. The mean DXA Z-score was −0.4 ± 1.0 and 0.2 ± 1.1 in the lumbar spine and hip, respectively. The same exclusion criteria as defined for the BSI cohort were applied. This group was comparable in regard to age (34.2 ± 11.0 years; P = .935) and sex distribution (31 women and 22 men; P≥ .999) but was not matched according to the level of exercise. Furthermore, the distribution of patients measured with first- or second-generation HR-pQCT devices was equal between the BSI and control groups (27:26). Since 2 generations of HR-pQCT scanners with different resolutions were used, normalization of the data was performed with reference data specific to each scanner model, analogous to the specification of aBMD Z-scores. The values are presented as the percentage of the median of published age-, sex-, and device-specific references.10,45
Statistical Analysis
Statistical analysis was carried out using GraphPad Prism (Version 8.4.0; GraphPad Software) and SPSS 27 (Version 27.0; SPSS, Inc). The normality of the distribution of the data was tested using the Shapiro-Wilk test, and the Student t test or the Mann-Whitney U test was used for parametric or nonparametric data analysis, respectively. For categorical data, the Fisher exact test was used. If ≥3 groups were compared, 1-way analysis of variance and repeated measures with Tukey correction for parametric data and the Kruskal-Wallis test with Dunn multiple comparison test were performed. Based on the study of Tenforde et al, 41 an effect size d = 1.0 was estimated, resulting in a required sample size of at least 22 for each cortical-rich and trabecular-rich location. The results are given as absolute values or the mean ± standard deviation. The level of significance was defined as P < .05. Exact P values are reported unless P < .001.
Results
Patient Cohort
A total of 53 athletes were included in this study, and the sample was mainly composed of middle- and long-distance runners (n = 40/53; 75.5%) but also included triathletes (n = 4/53; 7.5%), dancers (n = 3/53; 5.7%), and soccer players (n = 2/53; 3.8%), among others (Table 1). The median Tegner activity level was 5 (range, 4-9), indicating that the athletes participated in either recreational or competitive sports. While women were more frequently runners (P = .026), only men participated in the triathlon (P = .025). For the other disciplines, no significant differences in the proportion of affected women and men were revealed (all P > .05). Women and men were of equal age (33.8 ± 10.4 years and 34.3 ± 13.0 years; P = .866) (Table 2), whereas men had a higher BMI (21.5 ± 3.6 and 23.3 ± 2.3; P = .046) (Table 2). All descriptive data are presented in Table 2. Overall, 38 of 53 (71.7%) diagnosed injuries were detected in the foot (n = 20; 37.7%) or tibia/fibula (n = 18; 34.0%), whereas 9 (17.0%) were detected in the pelvis; 5 (9.4%), in the femur; and 1 (1.9%), in the sternum (Table 2). Comparing the frequency of BSI regions between women and men, we observed no significant differences. Moreover, the frequency of BSIs according to their specific anatomic site was analyzed. In total, most of the BSIs occurred at the tibia (n = 14/53; 26.4%), followed by the metatarsal bones (n = 13/53; 24.5%) and sacrum (n = 9/53; 17.0%) (Table 2). When subdivided according to cortical-rich versus trabecular-rich bone composition, 31 of 53 (58.5%) BSIs were present in cortical-rich bones, while 22 of 53 (41.5%) BSIs were detected in trabecular-rich bones. No significant differences were observed concerning the anatomic site or bone composition between women and men (Table 2).
Distribution of Sports Disciplines in the Study Cohort a
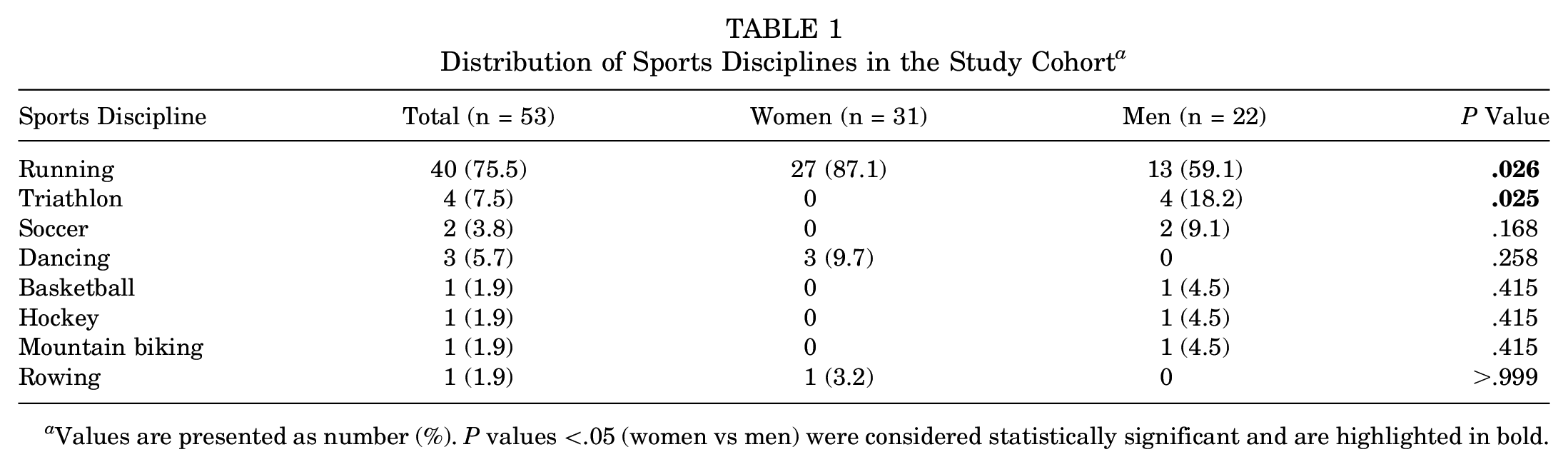
Values are presented as number (%). P values <.05 (women vs men) were considered statistically significant and are highlighted in bold.
Characterization of the Study Cohort a
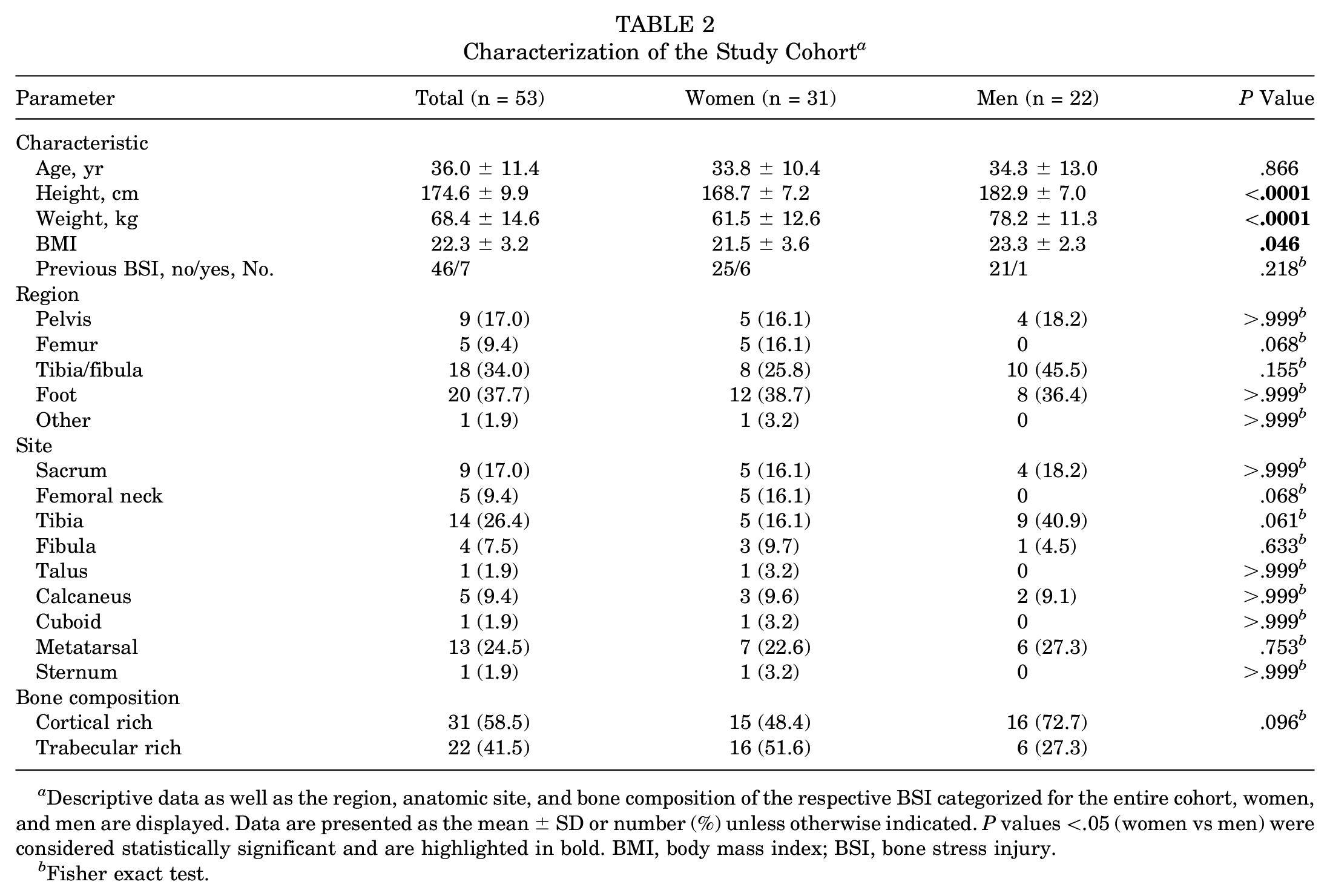
Descriptive data as well as the region, anatomic site, and bone composition of the respective BSI categorized for the entire cohort, women, and men are displayed. Data are presented as the mean ± SD or number (%) unless otherwise indicated. P values <.05 (women vs men) were considered statistically significant and are highlighted in bold. BMI, body mass index; BSI, bone stress injury.
Fisher exact test.
Laboratory and aBMD Data
The mean calcium, parathyroid hormone, phosphate, and ALP levels of the cohort were within the reference range. Creatinine and ALP levels were significantly higher in men than in women (P < .0001 and P = .009) (Table 3). While the bone formation markers osteocalcin and b-ALP were within the lower reference range for both women and men, the bone resorption marker deoxypyridinoline/creatinine was increased in women and men. For b-ALP, significantly lower values were observed in women (P = .002). Vitamin D deficiency, as defined by serum levels of <30 µg/L, was observed in nearly half of athletes (n = 26/53; 49.1%) (Table 3).
Biochemical and DXA Analysis a
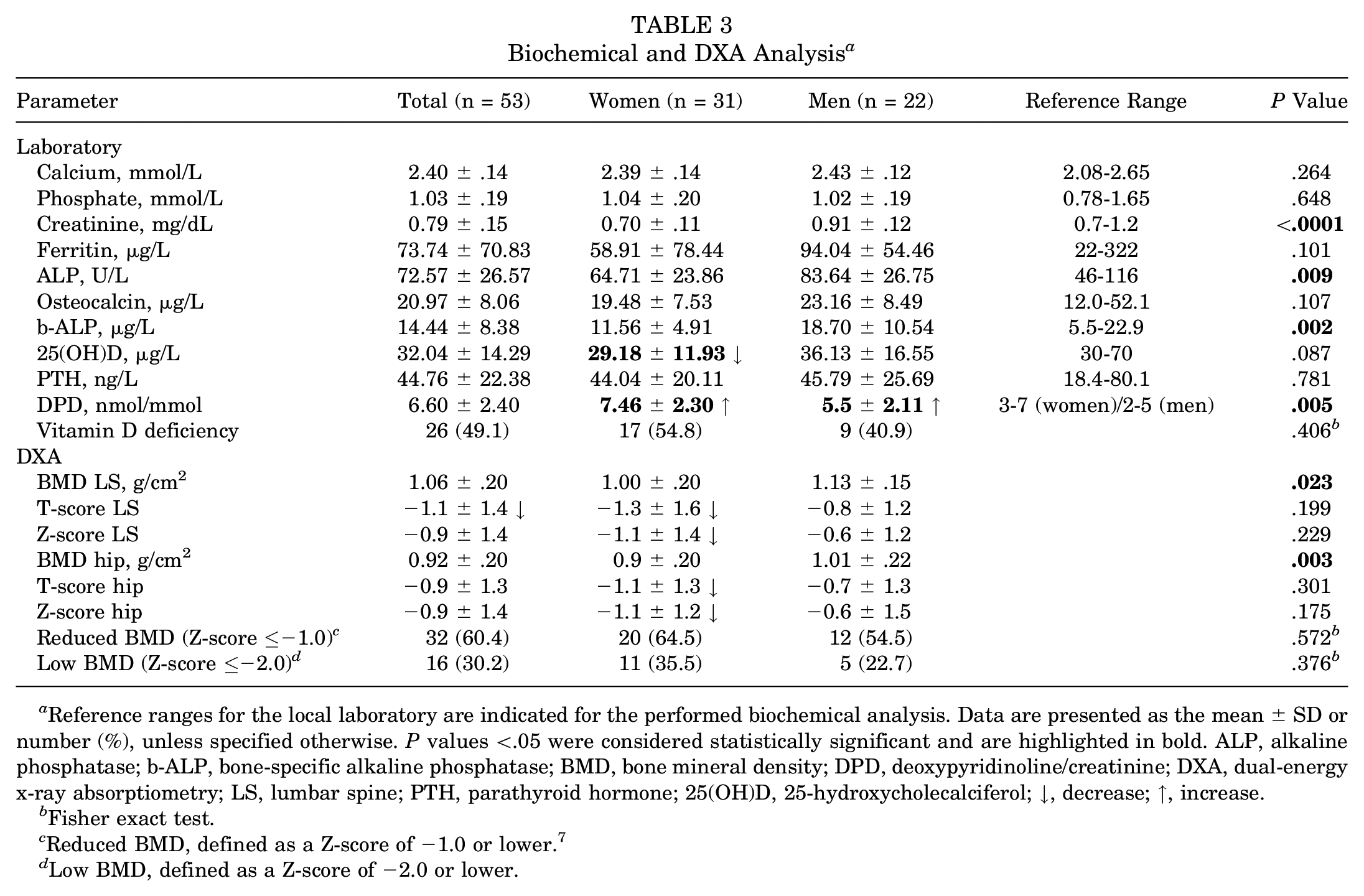
Reference ranges for the local laboratory are indicated for the performed biochemical analysis. Data are presented as the mean ± SD or number (%), unless specified otherwise. P values <.05 were considered statistically significant and are highlighted in bold. ALP, alkaline phosphatase; b-ALP, bone-specific alkaline phosphatase; BMD, bone mineral density; DPD, deoxypyridinoline/creatinine; DXA, dual-energy x-ray absorptiometry; LS, lumbar spine; PTH, parathyroid hormone; 25(OH)D, 25-hydroxycholecalciferol; ↓, decrease;
Fisher exact test.
Reduced BMD, defined as a Z-score of −1.0 or lower. 7
Low BMD, defined as a Z-score of −2.0 or lower.
Evaluation of DXA scans of the lumbar spine and both hips revealed heterogeneous but overall low values (aBMD, T-score, and Z-score) at both sites in women and men (Table 3). Namely, approximately two-thirds (n = 32/53; 60.4%) of the included athletes were classified as having low aBMD (ie, Z-score of −1.0 or lower) according to a previously proposed categorization for the athlete population, 6 and in one-third of the cohort (n = 16/53; 30.2%), a Z-score of −2.0 or lower was detected. No significant difference was observed when comparing women and men. To investigate whether a site- or region-specific pattern of aBMD reduction was prevalent in our cohort, we compared DXA measurements according to the most common anatomic BSI sites, showing no significant differences (Figure 1).
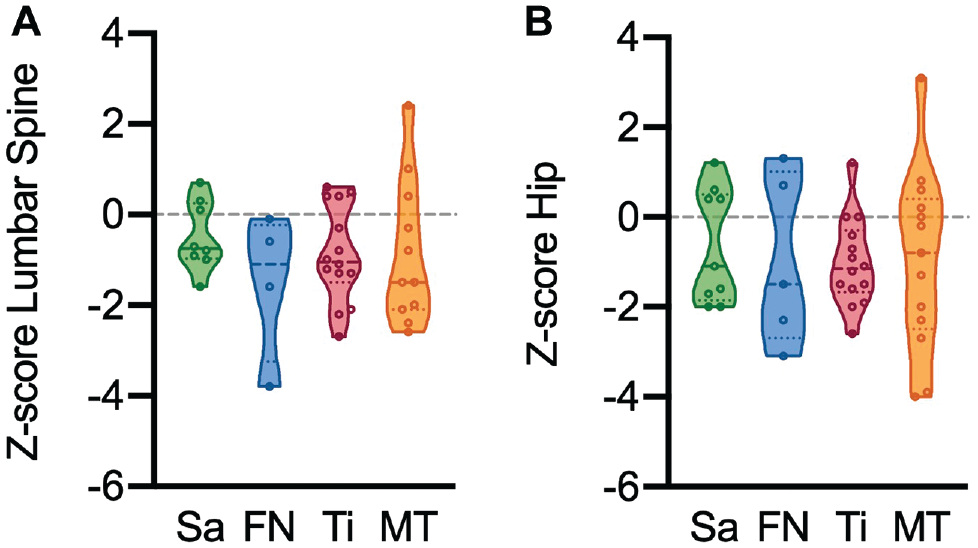
Dual-energy X-ray absorptiometry (DXA) results according to specific anatomic site. Comparison of areal bone mineral density Z-scores among the 4 most common bone stress injury sites assessed by DXA (A) in the lumbar spine and (B) in the hip. FN, femoral neck; MT, metatarsal; Sa, sacrum; Ti, tibia.
Bone Microarchitectural Characteristics
For densitometric and microarchitectural parameters at the distal radius, lower bone volume per tissue volume (BV/TV, 85.5% ± 19.1% vs 101.0% ± 25.7%; P = .019), cortical thickness (Ct.Th, 79.5% ± 21.3% vs 88.4% ± 19.2%; P = .029), and cortical area (Ct.Ar, 80.1% ± 21.5% vs 89.3% ± 21.2%; P = .034) were observed in individuals with BSI compared with the control group (Figure 2A), but no differences were observed in the other HR-pQCT parameters. At the distal tibia (Figure 2B), only cortical parameters showed significant differences with reduced values in BSI (Ct.Th, 79.5% ± 20.2% vs 94.0% ± 21.1%; P = .0007; Ct.Ar, 88.0% ± 18.6% vs 100.8% ± 19.8%; P = .001).
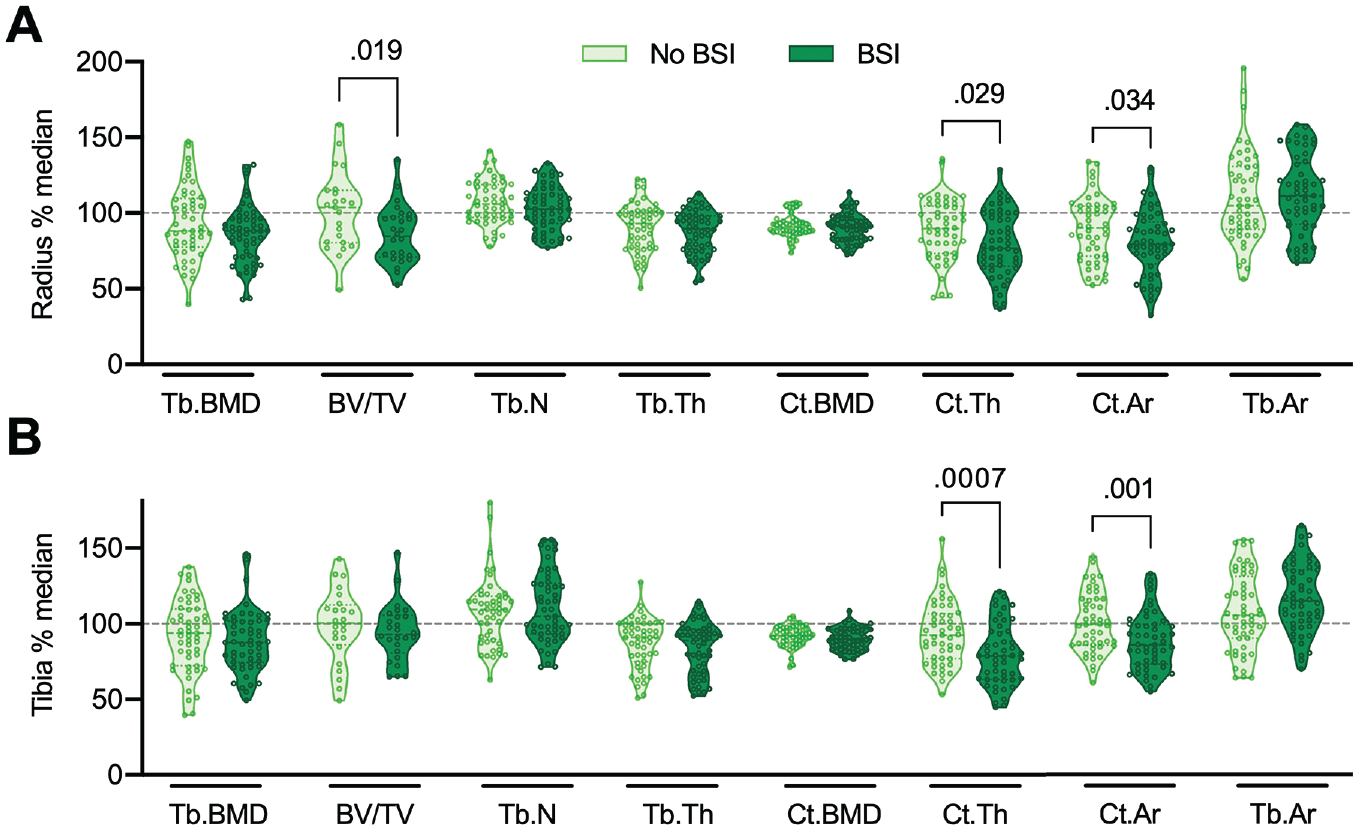
Bone microarchitecture in patients with bone stress injuries (BSIs) as assessed via high-resolution peripheral quantitative computed tomography. Bone microarchitecture parameters assessed (A) at the distal radius and (B) at the distal tibia in patients with BSI versus no BSI (controls). Values are presented as a percentage of the median compared with reference values for the first-generation 10 and second-generation device. 45 The horizontal dashed line indicates the median of the reference. The Shapiro-Wilk test was used to evaluate the normal distribution of the data. The Student t test was used for normally distributed data, and the Mann-Whitney U test was used for nonparametric data. The dashed lines of the truncated violin plots represent the median and quartiles. Exact P values are reported unless P < .001. BV/TV, bone volume per tissue volume; Ct.Ar, cortical area; Ct.BMD, cortical bone mineral density; Ct.Th, cortical thickness; Tb.Ar, trabecular area; Tb.BMD, trabecular bone mineral density; Tb.N, trabecular number; Tb.Th, trabecular thickness.
Comparison of HR-pQCT parameters according to the most prevalent specific anatomic BSI sites (Figure 3) showed no significant differences among anatomic sites except for significantly lower distal tibial trabecular thickness (Tb.Th) in patients with a BSI in the femoral neck compared with the sacrum (P = .042).
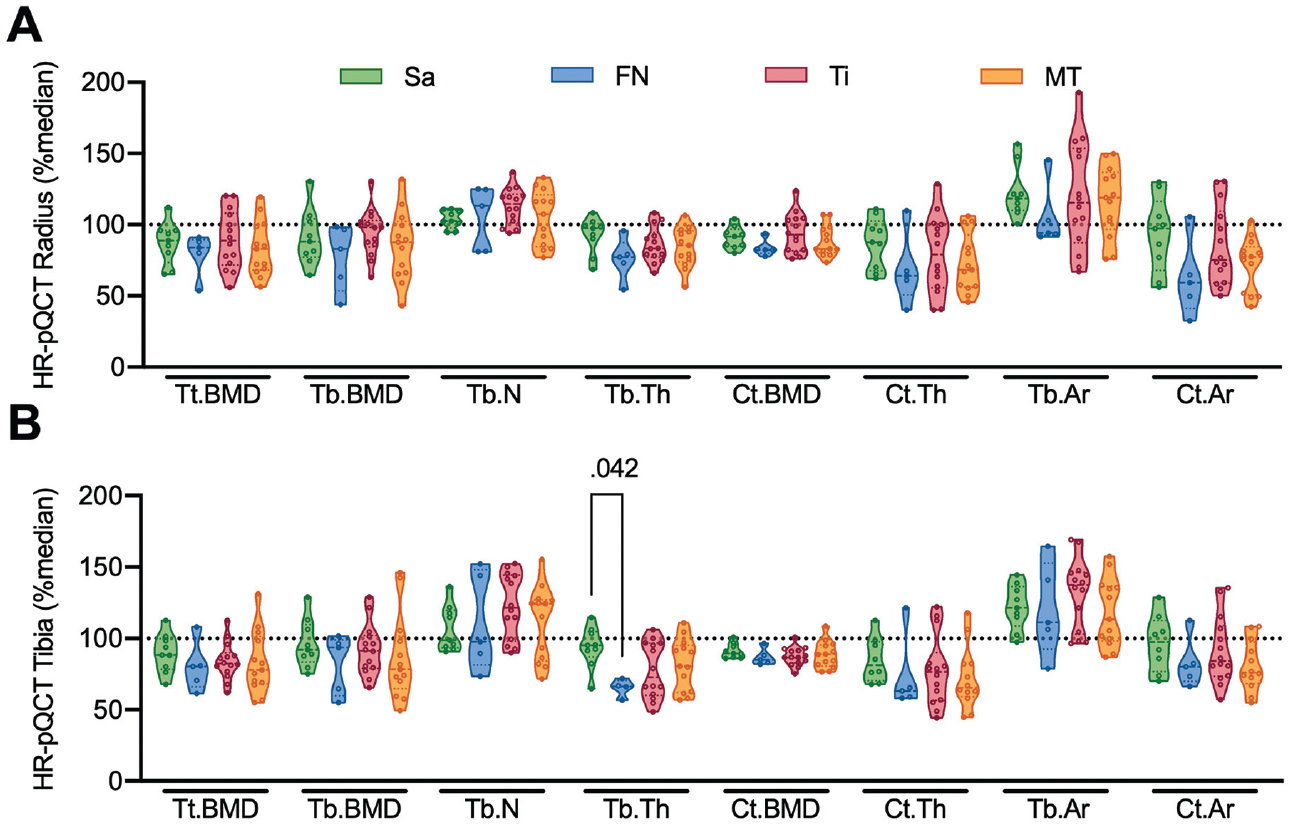
High-resolution peripheral quantitative computed tomography (HR-pQCT) results according to specific anatomic site. Comparison of (A) distal radius HR-pQCT parameters and (B) distal tibial HR-pQCT parameters among the 4 most common bone stress injury sites. The horizontal dashed line indicates the median of the reference. Ct.Ar, cortical area; Ct.BMD, cortical bone mineral density; Ct.Th, cortical thickness; FN, femoral neck; MT, metatarsal; Sa, sacrum; Tb.Ar, trabecular area; Tb.BMD, trabecular bone mineral density; Tb.N, trabecular number; Tb.Th, trabecular thickness; Ti, tibia; Tt.BMD, total bone mineral density.
Investigation of potential differences in aBMD and bone microarchitecture parameters between individuals who had a BSI in a cortical-rich or trabecular-rich site showed no differences (Figure 4). Last, when comparing athletes with the first diagnosis of a BSI versus those with a positive medical history of BSI (Appendix Figure A1, available in the online version of this article), no significant differences in DXA or HR-pQCT parameters were detected.
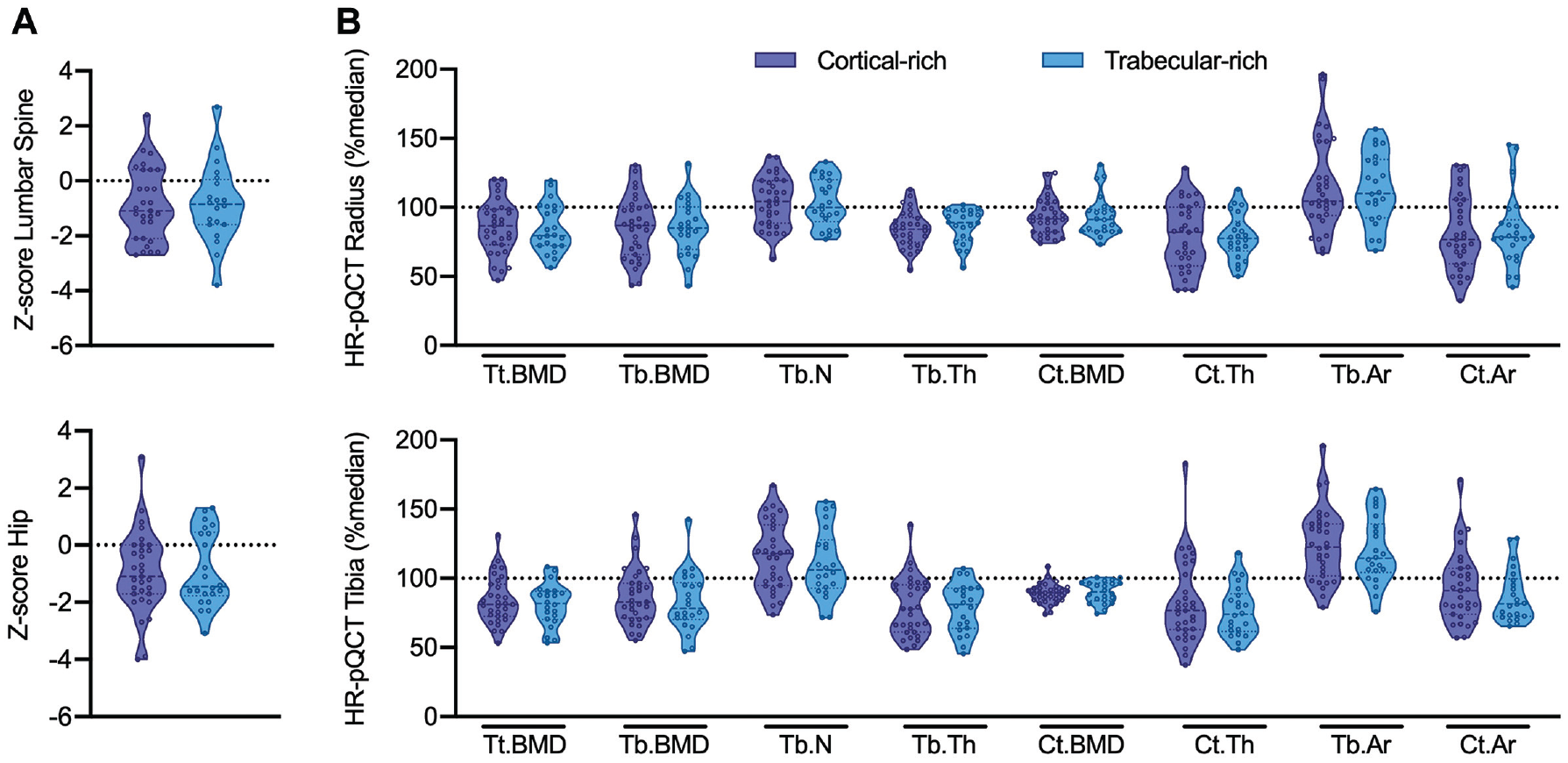
Bone mineral density and microarchitecture at reference locations according to bone stress injuries (BSIs) at cortical-rich versus trabecular-rich sites. (A) Comparison of areal bone mineral density Z-scores at the lumbar spine and hip and (B) bone microarchitecture parameters at the distal radius and tibia between individuals with BSI at cortical-rich and trabecular-rich sites. Values were compared with reference values for the first-generation 10 and second-generation devices 45 and are presented as the percentage of the median. The horizontal dashed line indicates the median of the reference. Ct.Ar, cortical area; Ct.BMD, cortical bone mineral density; Ct.Th, cortical thickness; HR-pQCT, high-resolution peripheral quantitative computed tomography; Tb.Ar, trabecular area; Tb.BMD, trabecular bone mineral density; Tb.N, trabecular number; Tb.Th, trabecular thickness; Tt.BMD, total bone mineral density.
Discussion
As specific risk factors for BSI development remain poorly understood, knowledge of potential bone microarchitectural alterations could provide additional insights in this context. In this study, mainly cortical microarchitecture parameters were lower in athletes with BSI at distal radial and tibial reference locations than in controls. To our knowledge, this is the largest BSI cohort in which biochemical, DXA, and HR-pQCT data have been assessed, shedding light on the involvement of impaired bone microarchitecture impairment as a possible contributing factor for BSI development. Nonetheless, the fact that no association between bone microarchitecture and anatomic fracture site or its bone composition could be demonstrated highlights the difficulties and unknowns associated with BSI in athletes.
Low aBMD measured via DXA was observed in nearly two-thirds of the athletes with BSI, supporting previous reports claiming low aBMD as a risk factor for BSI.22,40,41 In addition to the observed potential harmful effect of vitamin D deficiency and other factors, such as amenorrhea, 2 relative energy deficiency in sports is assumed to play a decisive role in BMD reduction in weightbearing exercise endurance athletes.18,26 However, considering that aBMD Z-scores were heterogeneous and some were even within the upper reference range, aBMD alone may provide only a limited explanation for the occurrence of BSI. 33 This finding was expected, as it is known that BSI can occur in the presence of high stress and strain without a reduced bone-loading capacity (ie, normal bone and remodeling). 16 Contrary to previous reports, no differences in the aBMD Z-score were revealed among anatomic sites, and no difference was observed when comparing bones with greater cortical versus trabecular composition.24,27,41 Differences in age and a selection bias among studies might serve as potential explanations; however, these findings point to the necessity for future studies to evaluate aBMD in an unbiased, prospective fashion, not only when low aBMD is assumed. 41 As only athletes with a stress fracture and not low-grade BSI were included in this study, the demonstrated findings may only apply to high-grade or late-stage manifestations of the BSI continuum.
Within the cohort of this study, the most commonly affected anatomic sites were the tibia and metatarsals. Interestingly, all BSIs of the femoral neck in our cohort occurred in female athletes; this site is also a common site of fragility fractures. 38 Risk stratification systems for the development of BSI have previously been introduced,22,25 showing discrimination of anatomic distribution of BSI by risk category, including the foot as the most prevalent region for the low-risk category. 39 Overall, the clinical relevance to differentiate among sites was highlighted by previous reports showing that bones of higher trabecular composition require a comparably longer time to heal 27 and that the risk for nonunion differs among the sites. 7
Few studies have elaborated on the osseous microstructural alterations in athletes with BSI,1,33,35,36,46 albeit no study comprising biochemical, DXA, and HR-pQCT data in a comparable cohort size has been reported thus far. In our study cohort, cortical thickness and area at the distal radius and tibia as well as trabecular bone volume fraction at the distal radius were reduced compared with those in controls. In line with our results, women with lower limb stress fractures (n = 19) have been reported to have a lower cortical area at the distal tibia. 36 In another previous study, women and men with BSI (n = 26) showed a lower vBMD and cortical cross-sectional area and a higher porosity of the outer and inner transitional zones. 46 Our results are only partly in line with previous observations in male soldiers with BSI (n = 26) who had reduced cortical vBMD and trabecular thickness but not cortical thickness. 35 Together, these results suggest that impaired bone microarchitecture may be a relevant risk factor for BSI, but the results regarding the affected bone compartment or specific parameters are inconsistent. Our data suggest alterations occured in the cortical compartment, which became evident by comparison with a control group and adaptation of the parameters to age-, sex-, and device-specific reference data. Notably, no differences in aBMD or bone microarchitecture were observed between athletes with no history or those with a history of BSI in our study, which is in line with a previous study in which having a history of multiple (≥3) versus a single BSI was not related to alterations in bone microarchitecture but related to menstrual history. 33 However, another study showed that women with amenorrhea with a history of 2 stress fractures had lower total area and trabecular vBMD than those with <2 stress fractures. 1 In a cohort of 30 female athletes with tibial BSI, the occurrence of an additional BSI was not associated with alterations in vBMD, although a decrease in total, trabecular, and cortical vBMD measured via HR-pQCT was observed 12 weeks after the diagnosis of BSI. 30
Previous studies have indicated the value of assessing bone sites with a potentially higher dependency on trabecular-mediated bone stability.7,17,41 Our study revealed no differences among the different anatomic regions or sites or between bone compositions (ie, cortical-rich or trabecular-rich), highlighting the need for future studies to elaborate on this phenomenon. This appears to be of even greater importance regarding treatment strategies, as previous reports have outlined a longer time to return to sports for BSI at trabecular-rich sites. 27 However, based on our findings, BSI at trabecular-rich sites does not pose an increased risk (eg, for delayed healing or recurrence of BSI) due to systemically affected BMD or bone microarchitecture, as these parameters appear equally altered compared with cortical-rich sites.
The results from the biochemical analysis revealed a state of comparably low bone formation and increased bone resorption. Vitamin D deficiency was detected in nearly half of the athletes. The role of vitamin D in bone remodeling and the relevance of balanced calcium homeostasis have been the subject of previous research and have already been highlighted for various skeletal diseases.31,32 Vitamin D deficiency has been shown to be highly prevalent in athletes in general 3 and to represent a distinct risk factor for BSI,34,40 as for other common musculoskeletal disorders, such as osteochondritis dissecans. 23 In line with these previous observations, vitamin D deficiency represented the major biochemical finding in our cohort. To foster sufficient bone mineralization, adequate vitamin D supplementation is generally recommended in athletes,21,25 as is an individualized supplementation regimen with regular biochemical monitoring. 42
Despite its novelty and potential clinical relevance, this study had some limitations that need to be mentioned. Regardless of the cross-sectional study design, an unbiased approach for the assessment of all individuals was used, and all individuals were assessed in the same way, reducing the risk for selection bias. No matching of the exercise level of the control cohort was feasible, and no detailed data concerning the menstrual history of female athletes could be provided, although this was identified as an independent risk factor for multiple BSIs. 33 Furthermore, the mean age of our cohort was slightly older than that of other BSI cohorts, which could limit the interstudy comparability. Whereas the introduction of reference locations for HR-pQCT measurements (ie, distal radius and distal tibia) allows for the comparison with reference values, no assessment of BSI sites or cortical-rich references was performed; this appears to be worthy of further investigation. Further limitations include the fact that collection of HR-pQCT data was performed on 2 different generations of HR-pQCT scanners and the scans were performed with a fixed offset, which could introduce a systematic bias, especially when comparing groups with different bone lengths. 9 To equalize the differences between the 2 scanner generations, we compared all HR-pQCT parameters using device-specific reference values with equal methodology. Moreover, training load was not included in the analysis, posing an interesting question that needs to be answered by future studies.
Conclusion
BSI development is likely influenced by reduced aBMD and impaired compartment-specific microarchitectural properties as assessed via HR-pQCT at distal radial and tibial reference locations. However, aBMD and bone microarchitecture cannot solely explain the occurrence of BSI at specific anatomic sites.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465221120385 – Supplemental material for Impaired Bone Microarchitecture at Distal Radial and Tibial Reference Locations Is Not Related to Injury Site in Athletes With Bone Stress Injury
Supplemental material, sj-pdf-1-ajs-10.1177_03635465221120385 for Impaired Bone Microarchitecture at Distal Radial and Tibial Reference Locations Is Not Related to Injury Site in Athletes With Bone Stress Injury by Julian Stürznickel, Nico Hinz, Maximilian M. Delsmann, Tim Hoenig and Tim Rolvien in The American Journal of Sports Medicine
Footnotes
Submitted February 28, 2022; accepted July 12, 2022.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
