Abstract
Background:
The microvascular anatomy of the meniscus of the human knee is regarded as a crucial factor in the injury response. Previous studies have investigated the zone-dependent distribution pattern, but no quantitative data exist on vascular density and its age-related changes.
Hypothesis/Purpose:
The aim of the present study was to histologically analyze the vascular anatomy of the meniscus as a function of age. It was hypothesized that vascular density would decrease with increasing age.
Study Design:
Descriptive laboratory study.
Methods:
Human menisci were retrieved from patients who underwent tumor resection or who received total knee replacement because of osteoarthritis. A total of 51 menisci were collected from 28 patients over 9 years (mean age, 25.6 ± 20.4 years; range 3-79 years). Immunohistological staining (alpha–smooth muscle actin) in combination with serial sections and standardized software-based contrast detection were used for the quantitative analysis. Data were analyzed using multiple t tests and the analysis of variance for trends, with a statistical significance level of P < .05.
Results:
The overall vascular density in the meniscus was lower in the 61- to 80-year age group than in the age groups of 0 to 10, 11 to 20, and 21 to 30 years (P < .01). A negative linear trend was detected with increasing age (slope, -0.007; P = .016). Within the red-white (RW) zone, a low vessel density was detected for the age groups of 0 to 10 and 11 to 20 years. Beyond these age groups, no vasculature was found in the RW zone. For the white-white (WW) zone, no vessel formations were noted in any age group. Almost 95% of the vessels in the meniscus were located in the capsule.
Conclusion:
This study reports quantitative histological data for microvascular anatomy as a function of age in a broad cohort of human knee menisci. The overall vascular density decreased with increasing age. No vessel formations were detected in the RW and WW zones after adolescence. Additionally, the capsule is far more densely vascularized than any other part of the meniscus.
Clinical Relevance:
Vascular density might be an additional factor to consider, along with tear location and patient age, for future treatment options.
The blood supply of the meniscus of the human knee was described in detail by Arnoczky and Warren 1 in 1982. They illustrated a perimeniscal capillary plexus within the synovial and capsular tissues of the knee joint that arises from branches of the lateral and medial geniculate arteries. This plexus is oriented predominantly in a circumferential pattern, with radial branches penetrating into the outer regions of the meniscal tissue. Similar findings were reported in the anatomic cadaveric study of Day et al. 9 This has led to singular classification systems that divide the meniscus into several zones according to its blood supply. The differentiation between the red-red (RR), red-white (RW), and white-white zones (WW) is widely used.6,15
Based on these anatomic considerations and tear type and location, clinical decision making regarding meniscal repair has greatly been influenced. DeHaven and Arnoczky 10 established the theoretical concept of a zone-dependent meniscal healing capacity. Tears in the RR and RW zones have the best prognosis for healing because of their functional blood supply. According to their concept, a tear within the completely avascular WW zone has the worst healing outcome.
In clinical practice, many studies have reported healing rates above 80% for arthroscopically repaired meniscal tears in the RW zone in younger patients.3,20,24 Cinque et al 4 demonstrated significant improvements in subjective outcome measures at a minimum 2-year follow-up, regardless of the meniscal tear zone. The authors recommend repair for meniscal tears in all 3 vascular zones, although improved outcomes can be achieved when the repairs are performed in the RR and RW zones. 4
This contradiction between anatomic concepts and clinical reality can only partially be explained by age-related changes in the microvasculature anatomy of the meniscus. In their immunohistochemical analysis, Petersen and Tillmann 21 found that after the age of 50, only the lateral quarter and the outer margin of the meniscus are vascularized. Other authors reported similar results.5,25 Although these studies illustrate the fundamental anatomic principle of decreasing meniscal vascularity, they lack detailed quantitative data in pivotal age groups between 10 and 50 years. This is because specimens from both intact menisci are not regularly available for analysis in this age group.
Thus, the aim of this study was to collect a broad sample of human knee meniscal tissue and report quantitative data on age-related changes in microvasculature anatomy and density. The hypothesis was that vascular density would decrease with increasing age. Additionally, we hypothesized that the density would decrease from the outer to the inner meniscal zones.
Methods
Patients
The study was reviewed and approved by the local institutional review board (2019-044-f-S), and patients gave their written consent. All menisci were collected in 2 departments (general orthopaedic and trauma) of a single university hospital. The specimens were retrieved from observing 2 groups: (1) patients who underwent wide resection of a malignant bone tumor at the distal femur or the proximal tibia, according to the Enneking system; (2) patients who received total knee replacement because of osteoarthritis (OA).
A total of 51 menisci were collected from 28 patients over 9 years. There were 13 male and 15 female patients. The mean age of patients was 25.6 ± 20.4 years (range, 3-79 years). Because of the resection margins, it was not possible to harvest both menisci from every patient. If the destruction because of the OA was too heavy, only the contralateral meniscus was harvested. For 23 patients, both the medial and the lateral menisci could be collected. In 5 patients, only 1 meniscus could be obtained. The specimens were divided into different age groups (Table 1).
Number of Specimens in Every Age Group

For the quantitative analysis of the meniscal vascularity in relation to the anatomic zones, immunohistological staining in combination with serial sections and standardized software-based contrast detection were used.
Specimen Preparation
After isolating the medial and lateral menisci from the tibial plateau, the surrounding soft tissue was carefully removed. Care was taken to preserve the capsular attachment. Whole menisci were fixed for 24 hours in 4% paraformaldehyde. Before further processing, the specimens were cut into parts of 1 cm each. Three specimens, 1 each from the anterior and posterior horn and the meniscal body, were used for further analysis. The microvasculature of the anterior and posterior horn of the meniscus is known to be heterogenic; thus, we did not use the outermost parts. 8 Those specimens were decalcified for 3 months in 4% ethylenediaminetetraacetic acid (EDTA). After dehydration in an ascending series of ethanol, the specimens were embedded in paraffin, and coronal sections of 5 µm each were cut with a microtome.
Immunohistological staining—α-SMA
For the histological analysis, alpha–smooth muscle actin (α-SMA) staining was used. Cytoplasmatic actins belong to the microfilament system of cytoskeletal proteins and consist of 6 different isoforms. The α-isoforms are specific for smooth muscle tissue and are widely utilized as antigen targets to immunohistochemically label blood vessels.26,27
The paraffin coronal sections were pretreated with a descending series of ethanol and rehydrated with aqua dest. For antigen retrieval, Tris-buffered saline (TBS) (pH, 7.4-7.6; room temperature) and trypsin (37°C; 59418C) (Sigma-Aldrich Chemie GmbH) were used. For staining, monoclonal mouse antihuman SMA (Clone 1A4; code, M0851; Dako) was used as a primary antibody at a dilution of 1:200. For visualization, biotinylated horse antimouse IgG (BA-2001; Vector Laboratories) was used as a secondary antibody at a dilution of 1:50.
Five serial sections with a minimum distance of 20 µm for each of the 3 parts were obtained. In total, 15 serial coronal sections for a single (medial or lateral) meniscus and 30 serial coronal sections per patient were analyzed.
Immunohistological staining—CD34
The CD34 is a type I membrane protein that is expressed on human hemopoietic progenitor and vascular endothelial cells. It is widely used as a vascular marker and is capable of labeling arterioles and capillaries. 14 It was applied as a control for α-SMA staining to check for vessels smaller in size than arterioles (without smooth muscle cells).
Paraffin coronal sections were pretreated with a descending series of ethanol and rehydrated with aqua dest. For antigen retrieval, TBS (pH, 7.4-7.6; room temperature) and Tris-Borat-EDTA (pH 8.4; steam cooker) were used. For staining, the mouse antihuman CD34 (790-2927; Ventana Medical Systems) was applied at a dilution of 1:1. The secondary antibody was the same as mentioned above (BA-2001).
Of the 2 serial coronal sections with a distance of 5 µm, 1 was labeled with the antibody against α-SMA, and 1 was with CD34. This was done once for every part of the meniscus and allowed for qualitive comparison of the 2 staining methods. For demonstration purposes, some additional sections were stained with light green (Lichtgrün SF, 7706; Carl Roth GmbH). These were not used for the analysis.
Quantitative Analysis
For the quantitative analysis, 15 sections of every meniscus were scanned with an Olympus BX51 microscope (Olympus Europa SE) at 10×magnification. Further digital processing was performed with the cellSense Dimension program (Olympus). A single investigator (A.H.) defined several regions of interest (ROIs) by hand for all sections, according to the established zone concept (Figure 1):
- Total meniscus
- Capsule
- RR
- RW
- WW
For every ROI, the total area in square millimeters was measured. After the ROI definition, a color-channel measurement was performed. A threshold for the color red was defined and separately adjusted for every section. Care was taken that all areas with a red stain (vasculature) were detected, but no artifacts (eg, cell nuclei) were included in the measurement. The detected area (yellow in Figure 2) was then measured and could be used to calculate the vasculature density for every zone. A density below 0.01% was considered as artifact detection.
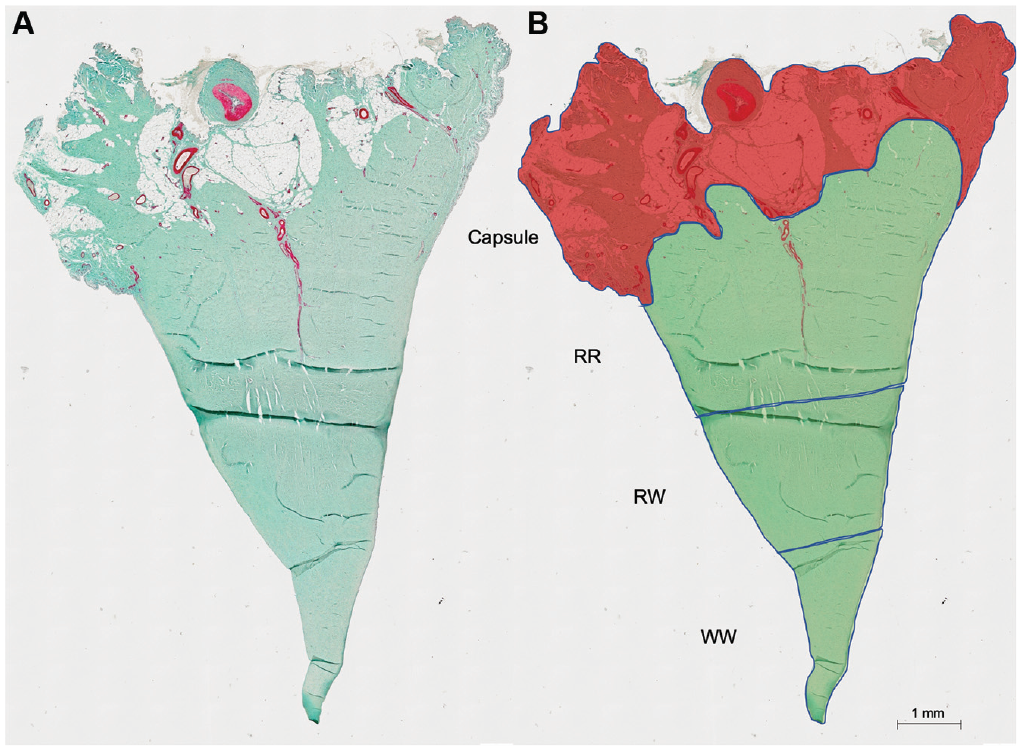
Coronal meniscal sections with α-SMA and light green staining. (A) Native section (total meniscus) before analysis; the red color (α-SMA) shows the stained vasculature. (B) The meniscal section is divided into separate ROIs: capsule, RR, RW, and WW zones. Area of the capsule is marked in light red. α-SMA, alpha–smooth muscle actin; ROI, region of interest; RR, red-red; RW, red-white; WW, white-white.
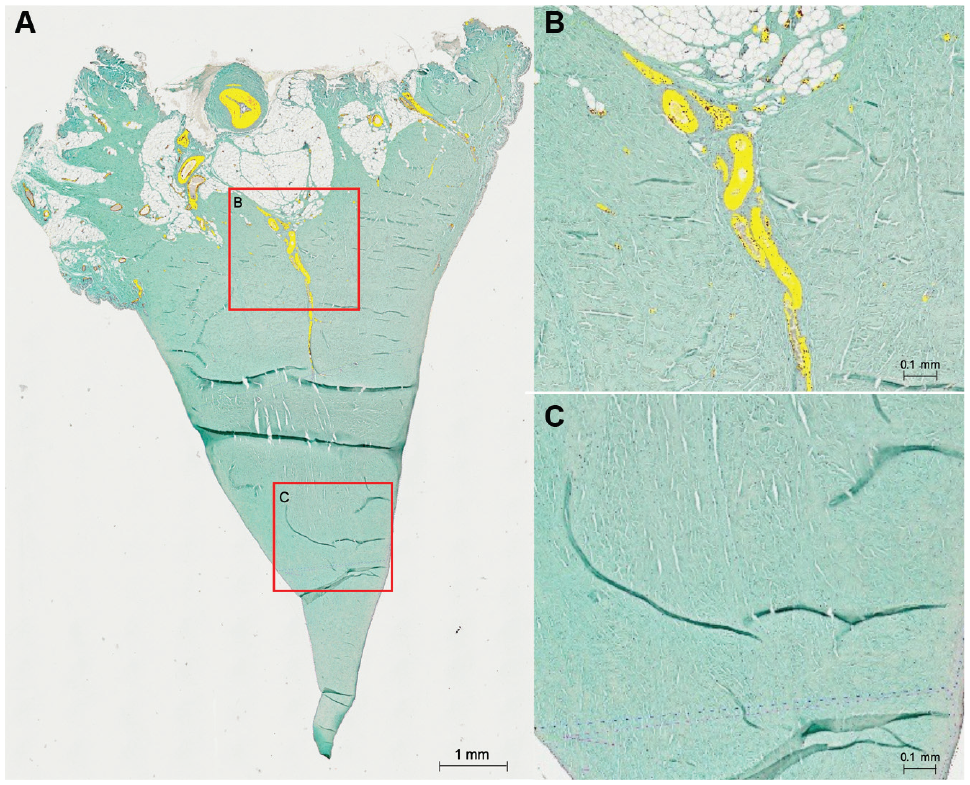
(A) Overview of a meniscal section after color channel detection. The yellow areas represent the red-stained and detected vasculature. (B) Magnification of detected vessels in the RR zone. (C) Magnification in the RW zone without any vasculature. RR, red-red; RW, red-white.
Statistical Analysis
Statistical calculations were performed using GraphPad Prism 8 software. Data were analyzed using multiple t tests, with a post hoc Bonferroni correction for multiple comparisons. To check for linear trends, the analysis of variance test for trends was used. Statistical significance was set at P < .05.
Results
The vascular density in the total meniscus was lower in the 61- to 80-year age group than in the age groups of 0 to 10, 11 to 20, and 21 to 30 years (P < .01). A negative linear trend was detected with increasing age (slope, -0.007; P = .016; R2 = 0.12) (Figure 3A). For the capsular area, the vascular density was decreased in the 61- to 80-year age group compared with the 0- to 10-year age group (P < .01). Additionally, a negative linear trend was noted with increasing age (slope, -0.012; P = .01; R2 = 0.14) (Figure 3B). The vascular density in the RR zone was lower in the 51- to 60-year age group than in the age groups of 0 to 10, 11 to 20, and 21 to 30 years (P < .01). A negative linear trend was present (slope, -0.003; P < .0001; R2 = 0.31) (Figure 3C).
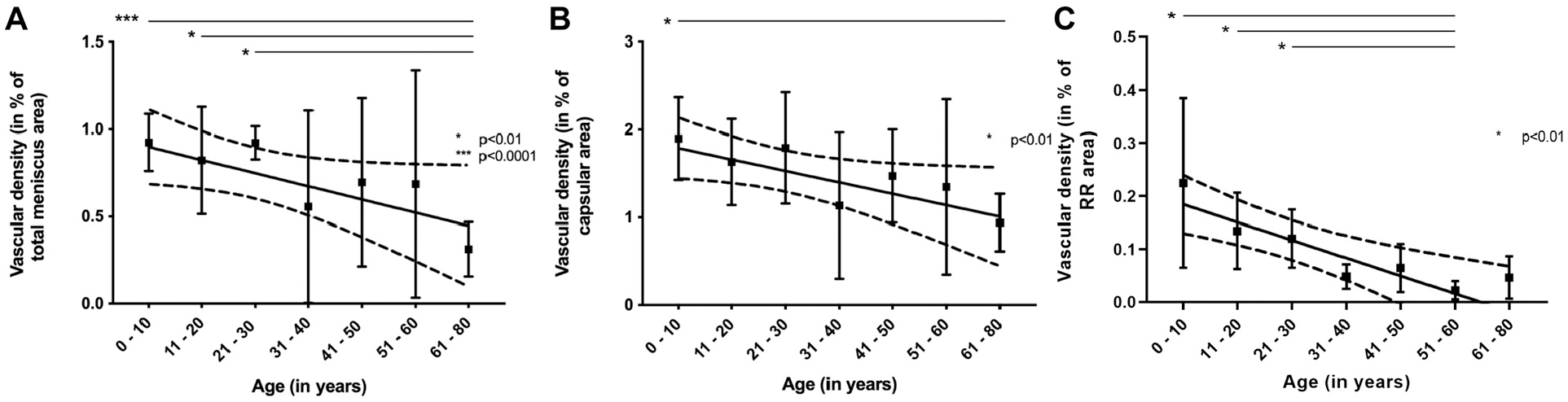
Vascular density in relation to a specific meniscal area. (A) Total meniscus. (B) Capsule. (C) The RR zone in the different age groups. Please note the disparate scales between figures. RR, red-red. *P < .01, ***P < .0001.
Within the RW zone, a low vessel density was detected for the age groups of 0 to 10 and 11 to 20 years. Beyond these age groups, no vasculature was found. For the WW zone, no vessel formation was found in any age group (Figures 4 and 7).
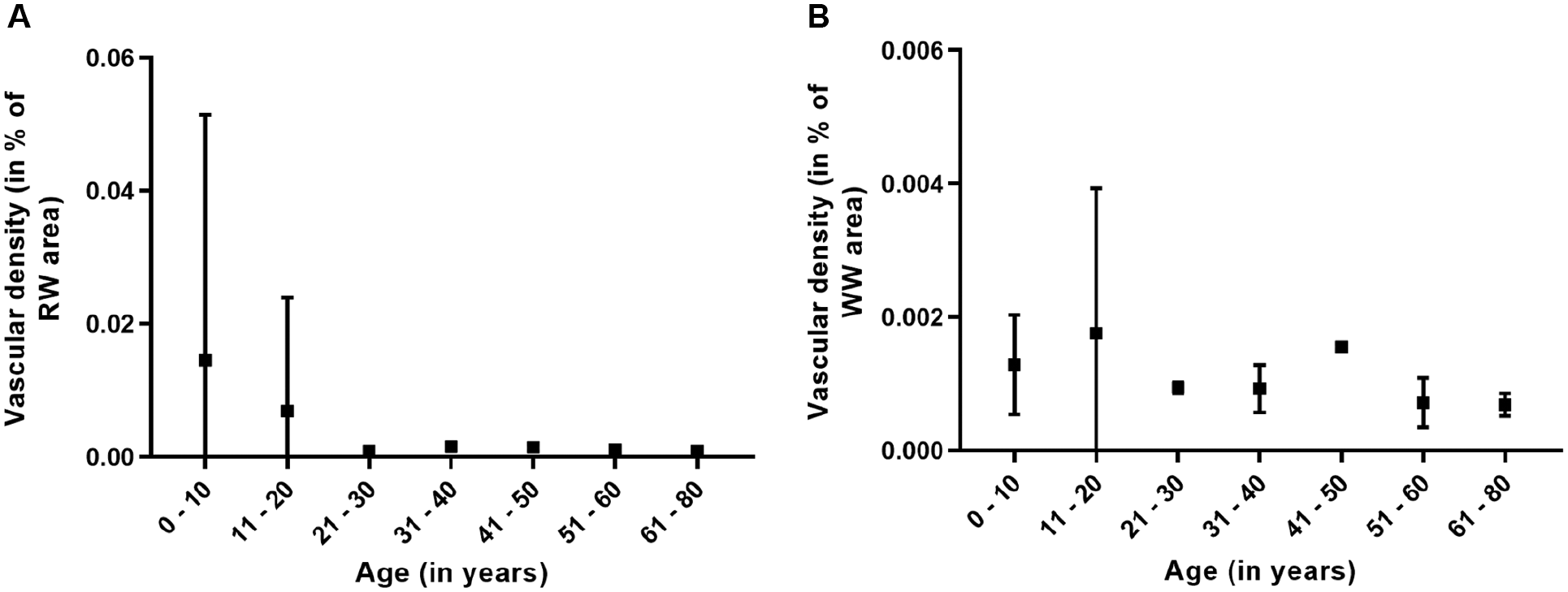
Vascular density in the (A) RW zone and (B) the WW zone. Please note the disparate scales between figures. RW, red-white; WW, white-white.
When pooled over all age groups, the capsule showed the highest vascular density. In relation to the RR zone, the vascular density within the capsule was 12.96-fold increased (P < .0001) (Figure 5A). Without the capsule, the RR zone displayed the highest vascular density (P < .0001) (Figure 5B).
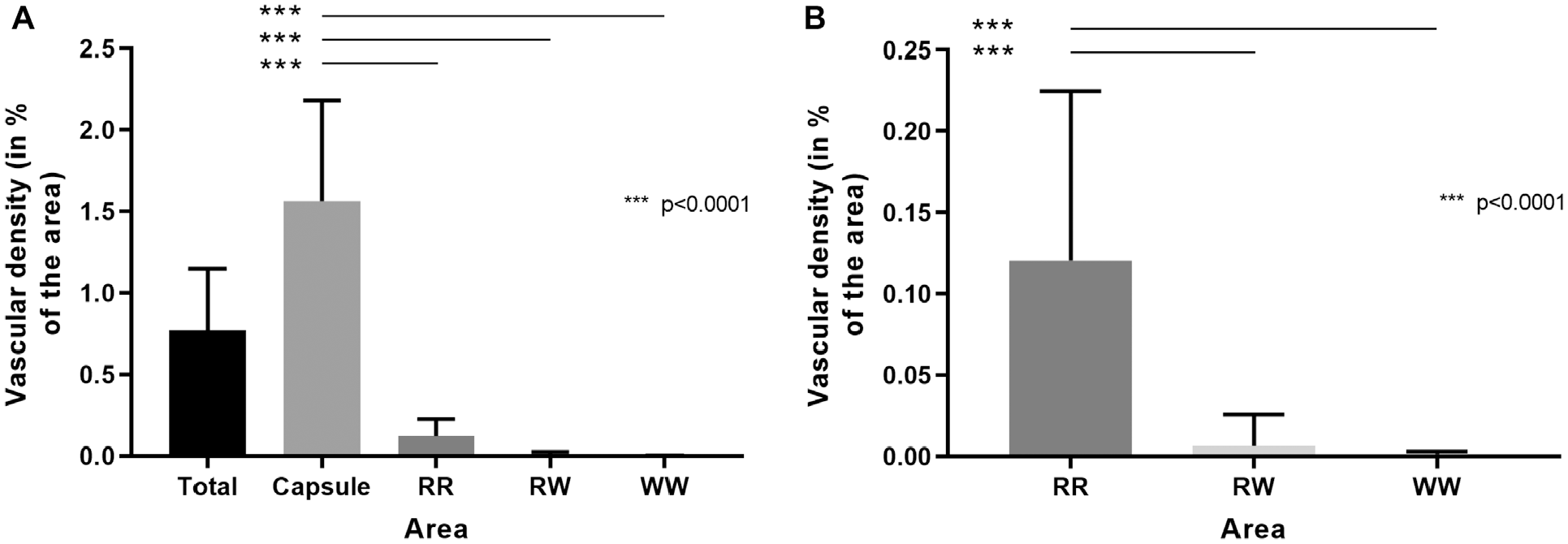
Vascular density pooled over all age groups. (A) Total meniscus with all zones. (B) The meniscal area without the capsule (RR, RW, and WW zones). Please note the disparate scales between figures. RR, red-red; RW, red-white; WW, white-white. ***P < .0001.
The contribution of a specific area to the overall meniscal vasculature showed that 94.4% of the vessels in the meniscus were located in the capsule (Figure 6A). Without the capsule, 92.3% of the vasculature within the meniscus was located in the RR zone (Figure 6B).
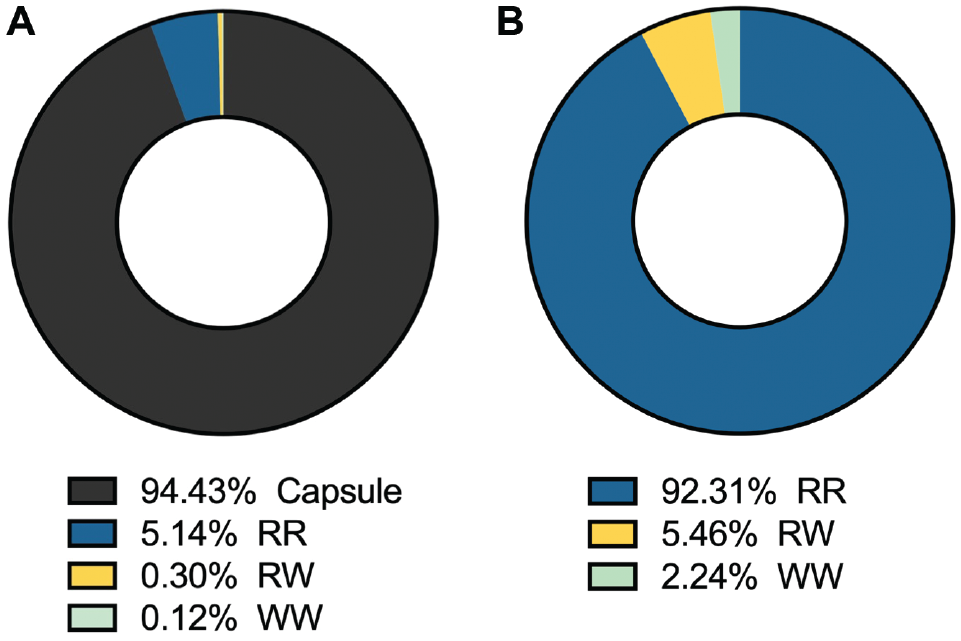
Contribution of the vasculature in a specific area to the overall meniscal vasculature. (A) Total meniscus. (B) Meniscus without the capsule (RR, RW, and WW zones). RR, red-red; RW, red-white; WW, white-white.
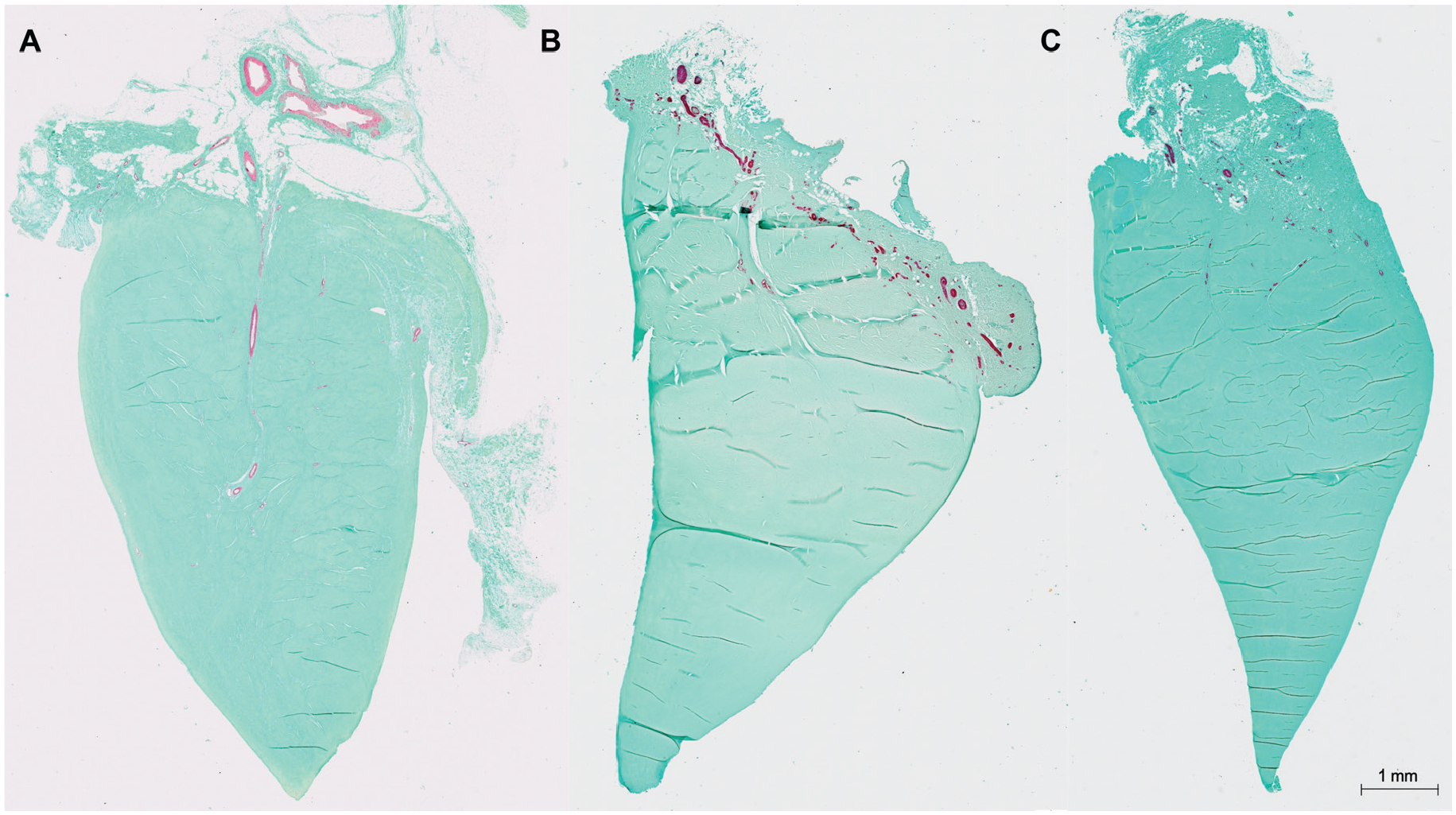
Representative sections with α-SMA and light green staining. (A) Section from a medial meniscus of an 8-year-old boy. Vessels are visible until the RW zone. (B) The lateral meniscus of a 19-year-old patient and (C) 35-year-old female patient. Vasculature is only present in the RR zone and the capsule. α-SMA, alpha–smooth muscle actin; RR, red-red; RW, red-white.
Control staining with CD34 revealed no differences in vessel detection capacity compared with α-SMA staining. No vessels were detected in the RW or WW zones in the analyzed sections after adolescence.
Discussion
The most important finding of the present study was that the overall microvascular density in the meniscus of the human knee decreased with increasing age. Specifically, the vascular density declined in the capsule and the RR zones. No vessel formations were detected in the RW and WW zones after adolescence. The capsule was far more densely vascularized than any other part of the meniscus. These results support the hypothesis of an age- and zone-related vascular distribution pattern.
Anatomic studies have established the concept of zone-dependent vascularization. Arnoczky and Warren 1 used 20 cadaveric knees from donors of various ages from the 6th through 10th decades of life. They reported that a perimeniscal capillary plexus originating in the capsular and synovial tissues of the joint supplies the peripheral 10% to 25% of the meniscus. A recent investigation by Crawford et al 7 confirmed these results in a younger age group. The authors used 13 cadaveric knee specimens with a mean age of 28.5 years (range, 22-34 years). They reported that the degree of vascular penetration (medial meniscus: range, 0%-42%; lateral meniscus: range, 0%-48%) exceeds what was previously reported. However, the median values of vascular depth within different radial locations of the menisci are consistent. Both studies used injection techniques (India ink) and light microscopy for analysis. A major concern with this method is that vascularization is only visualized if the vessels are filled with ink. Petersen and Tillmann 21 pointed out that the absence of vessels inside a dense connective tissue may be caused simply by insufficient filling with the ink. They used immunohistochemical methods (antibody against the basement membrane components laminine and type IV collagen) to analyze a cohort of 20 human menisci that ranged in age from 22 weeks of gestation to 80 years. They found an age-related distribution pattern. At the time of birth, nearly the whole meniscus was vascularized; and in the second decennium, blood vessels occurred only in the lateral third. After 50 years of age, only the lateral quarter of the meniscal base was vascularized. A limitation of this study was that only 1 specimen between the ages of 1.5 and 50 years could be analyzed. In 1983, Clark and Ogden 5 investigated a large longitudinal cohort of human menisci. The authors examined the knee menisci of 109 fetuses and 28 menisci from donors 3 months to 14 years of age. At the fetal stage, blood vessels are numerous and can be found in the whole body of the meniscus. Regarding vascular development, they reported a progressive decrease in vascularity already beginning in the first year of life. By the age of 10 years, blood vessels were primarily located in the peripheral one-third of the meniscus, while smaller vessels could be identified throughout the inner zones.
The results of the present study support these findings and, for the first time, report quantitative histological data on the microvascular density of human knee menisci. In general, the wound repair process occurs in almost all tissues after exposure to a destructive stimulus. 16 It is widely accepted that wound healing requires a dynamic angiogenic response. New capillaries grow into the wound, forming a network of blood vessels that is 2, 3, or even up to 10 times more dense than in normal tissue. 11 Reduced microvascular density (eg, in patients with diabetes) can lead to delayed or complicated wound healing. 12 It is therefore of great importance to both understand the distribution pattern of vessels and the exact microvascular density within the specific tissue. Although vessel penetration was detected within the RW zone in the present study, the overall vascular density in this zone was extremely low, even in the younger age groups. Almost 95% of the overall meniscal vascularity was found to be located within the capsule.
Vascular anatomy is especially important because it is thought to be one of the key factors in the healing of meniscal tears. Arnoczky and Warren 2 demonstrated in a dog study that avascular meniscal tears show healing potential when connected to the peripheral synovial tissue by a vascular access channel. Angiogenic approaches to meniscal healing have shown only limited success so far, although a variety of growth factors, such as the vascular endothelial growth factor, have been evaluated. Williams and Adesida 28 described a significant knowledge gap in our understanding of meniscal healing. While it is commonly understood that vascularization and healing of avascular meniscal injuries occur simultaneously, there is little evidence as to why this occurs and, hence, how it can be manipulated to improve healing. A recent concept proposes that not the meniscal stroma, with its intrinsic vascularity, but the extrinsic surrounding synovium is the driver of meniscal healing. A study by Kim et al 17 in rabbits showed improved healing of meniscal tears with an intact synovium. Based on the study results, it was concluded that synovial cells were the origin of the regenerated cells in the meniscal defects and accounted for the majority of cells contributing to meniscal regeneration. In light of these studies, our data underline the importance of the synovial tissue for the meniscal healing potential, as almost 95% of the meniscal vasculature is located there. Further basic science research and clinical studies are needed to fully elucidate this subject.
Clinically, better surgical outcomes are reported for tears in the outer meniscal zones. Nevertheless, some studies demonstrated healing rates above 80% for arthroscopically repaired meniscal tears in the RW zone in younger patients.3,20,24 Some authors even showed successful repairs in all 3 vascular zones, although improved outcomes were achieved when performed in the RR and RW zones. 4 Poland et al 22 reported that an age of 40 years or older is not associated with an increased risk of meniscal repair failure after 5 years. Unfortunately, a significant number of meniscal repairs fail, and younger patients may be at higher risk of failure of revision meniscal repair. 18 Rai et al 23 concluded that a number of intrinsic and extrinsic factors seem to influence the meniscal injury response and that our lack of understanding of the intricacies of meniscal biology is a major reason why current treatments are only partially successful. Future clinical studies investigating different repair techniques that respect the synovial microvasculature might be an interesting approach.
The present data indicate that the zone-dependent distribution of vessels might play a role in both the injury response and the overall microvascular density and its age-related decline. Clinically, this is highly relevant, as vascular density might be another factor to consider—in addition to meniscal tear morphology, localization, and patient age—in optimizing individual treatment decisions. A study conducted by Lin et al 19 indicated that meniscal perfusion might even be measurable preoperatively by contrast magnetic resonance imaging.
A limitation of this study was the unequal distribution of specimens between the different age groups. In some groups, we had only very limited access to meniscal specimens. This was mainly because of the age pattern of malignant bone tumors. These groups might be only partially representative. Nevertheless, this study provides one of the largest and broadest cohorts in the literature, given the difficulties of obtaining intact specimens of human knee menisci in age groups <35 years. Another limitation concerns the differentiation between the medial and the lateral meniscus. This was because the specimens were mainly obtained from patients after a wide tumor resection. After pathological examination and preparation of the specimens, key anatomic landmarks were missing. A correct identification was therefore not possible for every case. Because of the rarity of the specimens, we decided to pool the data. Fedje-Johnston et al 13 recently reported a higher cell density for the medial compared with the lateral meniscus in a cohort aged 1 month to 11 years. Crawford et al 7 could not detect a difference in vessel penetration between the medial and lateral meniscus. In our study, no differentiation between the medial and lateral meniscus was made. Another limitation concerns the use of meniscal specimens from patients with OA. The vasculature in these specimens may be different compared with those from non-OA knees. Additionally, this was an ex vivo histological study, which limits the possibility of drawing conclusions for the in vivo meniscal injury response.
Conclusion
This study reported quantitative histological data on age-related changes in microvascular anatomy in a broad cohort of human knee menisci. Vascular density decreased with increasing age, and no vessel formations were detected in the RW and WW zones after adolescence. Additionally, the capsule was far more densely vascularized than any other part of the meniscus. Clinically, vascular density might be an additional factor to consider, along with tear location and patient age, for future treatment options.
Footnotes
Acknowledgements
The authors thank Simone Niehues for technical assistance. The authors thank Wolfgang Hartmann for support with the histological analysis.
Submitted January 24, 2021; accepted May 17, 2021.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
