Abstract
Objective. Quantification of meniscus vascularity has been limited with previous techniques, and minimal data exist describing differential vascular zones in the skeletally immature meniscus. The objective of this study is to use quantitative contrast-enhanced magnetic resonance imaging (MRI) to compare meniscal vascularity in neonatal specimens with adults. We hypothesized that the developing meniscus has greater and more uniform vascularity throughout all zones. Design. Ten fresh-frozen human cadaveric knees (5 neonatal, age 0-6 months; 5 adult, 34-67 years) underwent gadolinium-enhanced MRI using an established vascularity quantification protocol. Regions of interest corresponding to peripheral and central zones of the meniscus were identified on pre-contrast coronal images, and signal enhancement within the same regions (normalized against background tissue) was compared between pre- and post-contrast images. Results. The medial and lateral menisci had similar distribution of perfusion (45.8% ± 8.1% medial vs. 54.2% ± 8.1% lateral in neonatal knees; 50.6% ± 11.3% medial vs. 49.4% ± 11.3% lateral in adult knees, P = 0.47). Increased perfusion was demonstrated in the periphery compared with the central zone (2.3:1 in neonatal knees and 3.25:1 in adult knees, P = 0.31). Neonatal specimens demonstrated 6.0-fold greater overall post-contrast meniscal signal enhancement compared with adults (P < 0.0001), with the 0-month specimen demonstrating the greatest proportional signal enhancement. Conclusions. While blood flow to the periphery is greater than to central zones in all menisci, younger menisci receive proportionally greater overall blood flow compared to adults, including to the central zone, suggesting that the immature meniscus is a more biologically active tissue than its adult counterpart.
Keywords
Introduction
Meniscal tears are a common orthopedic condition causing significant pain and dysfunction within the general population, in both adults and children.1,2 Historically, meniscal tears were treated with meniscectomy; however, with greater understanding of the biomechanical role of the meniscus for the long-term health of the knee, there has been a shift toward meniscal preservation.3-8 Despite advancements in surgical techniques for meniscal repair as well as novel adjunctive therapies such as platelet-rich plasma, bone marrow aspirate concentrate, and adipose-derived mesenchymal stromal cells, the fundamental limitation of meniscus healing is its relative hypovascularity, resulting in poor intrinsic healing capacity.4,5,9,10 Numerous studies, such as the landmark study by Arnoczky and Warren, 11 have shown that meniscal vascularity is greatest along the periphery, with 3 generally accepted zones based on degree of vascularity from peripheral to central: red-red, red-white, and white-white, with most blood flow occurring in the red-red and essentially no vascularity in the white-white zone. Prior studies demonstrate that vascularity of the meniscus recedes to the outer 10% to 30% during development.9,12-14 While integral for establishing our current paradigm for meniscal vascularity, techniques used previously in the literature allow for only qualitative analysis. With improved surgical techniques, implants, adjunctive therapies, and diagnostic precision, it is reasonable to further study the microscopic anatomy of the meniscus to further our understanding of the intrinsic healing capacity of this tissue.
As children and adolescents have become increasingly active in sports, there has been a growing incidence of meniscal tears in younger patients, and some authors have reported that these patients have different healing potential than adults. 15 Previous studies of meniscectomy in children have shown poor long-term outcomes, largely related to the premature development of osteoarthritis.1,16 However, studies of meniscal repairs in pediatric patients have shown promising results, with some authors reporting better outcomes than in adult meniscal tears.17,18 Recent studies have demonstrated relatively good healing potential after repairs in young patients with tears in what was traditionally considered the avascular zone of the meniscus.19,20 This suggests that in the pediatric population, there may be a greater intrinsic healing capacity, which may be due at least in part to vascularity that extends beyond the generally accepted vascular boundaries that were established in adult studies.
Previous studies that have attempted to characterize the vascularity of the human meniscus have used cadaveric tissue from older donors. Furthermore, it is difficult to accurately visualize or quantify blood flow in the meniscus using available diagnostic imaging modalities, including magnetic resonance imaging (MRI). 21 In this study, meniscal vascularity in the developing knee was assessed by applying a previously established MRI protocol for quantifying vascularity within musculoskeletal tissues.22-25 This technique differs from others in that it uses normalized changes in signal intensity to quantify and compare relative regional blood flow independent of visualizing individual vessels. Thus, the objective of this study was to compare meniscus vascularity in neonatal versus adult cadaveric specimens. Given that the immature knee has increased vascularity due to high metabolic demand during development, we hypothesized that neonatal meniscus specimens would demonstrate greater overall vascularity in a more even distribution (proportionally greater blood supply to the central portion) compared with mature meniscus specimens.
Methods
Cadaveric Specimens
Approval was granted by the institutional review board of the study institution for obtaining and investigating 10 fresh-frozen human cadaveric knees for this research investigation. Cadaveric specimens (from mid-femur to mid-tibia) utilized for this study included the following: 5 human immature knees, age 0-6 months, including 1 female and 4 males; and 5 adult knees, age 34 to 67 years, including 2 females and 3 males. No specimen had an associated history of vascular disease, prior lower extremity surgery, or a history of lower extremity trauma.
Dissection and Preparation for Imaging
Specimen preparation was performed at the Laboratory for Joint Tissue Repair and Regeneration and the Bioskills Education Laboratory, an American College of Surgeons accredited dissection laboratory at the study institution. Minimal dissection was performed at the level of the knee joint, with care taken to isolate the superficial femoral artery proximal to the joint at the femoral stump. No arthrotomy or intra-articular dissection was performed, and no deep structures were exposed. Using a previously described vessel cannulation protocol,22,23 each cadaveric knee underwent cannulation of the superficial femoral artery, at the level of the adductor canal, to include the genicular arterial system supplying the menisci. DLP model 30000 vessel cannulas (Medtronic, Minneapolis, MN) were utilized. Normal saline was infused through the implanted cannula, and any large or visible extravasating vessels were tied off to allow adequate infusion and retention of contrast material.
Imaging Acquisition
Imaging was performed using a 3.0 Tesla Excite HD GE MRI scanner (General Electric Healthcare, Milwaukee, Wisconsin); an 8-channel wrist coil was used for all neonatal knee specimen and an 8-channel knee coil was used for the adult knee specimens. Images were obtained using a previously described protocol.22,23 High-resolution fat-suppressed 3-dimensional gradient echo sequences were acquired both before and after MRI contrast agent administration. MRI images were acquired at 2-mm slice thickness pre- and post-contrast solution infusion with Gd-DTPA (gadolinium-diethylenetriamine pentaacetic acid) diluted with saline at a 3:1 saline to contrast agent ratio. Fat-suppressed MRI images were utilized for quantitative MRI assessment to remove signal generated by bone marrow while enhancing Gd-DTPA imaging detail. Contrast solution infusion consisted of 61 mL for adult specimen and 5 mL for all neonatal knee specimens. Images were reconstructed to a resolution of 0.4 × 0.4 × 1.0 mm. Parameters for acquisition consisted of repetition and echo times of 18.6 and 5.3 ms, respectively, with a 35° flip angle.
Analysis
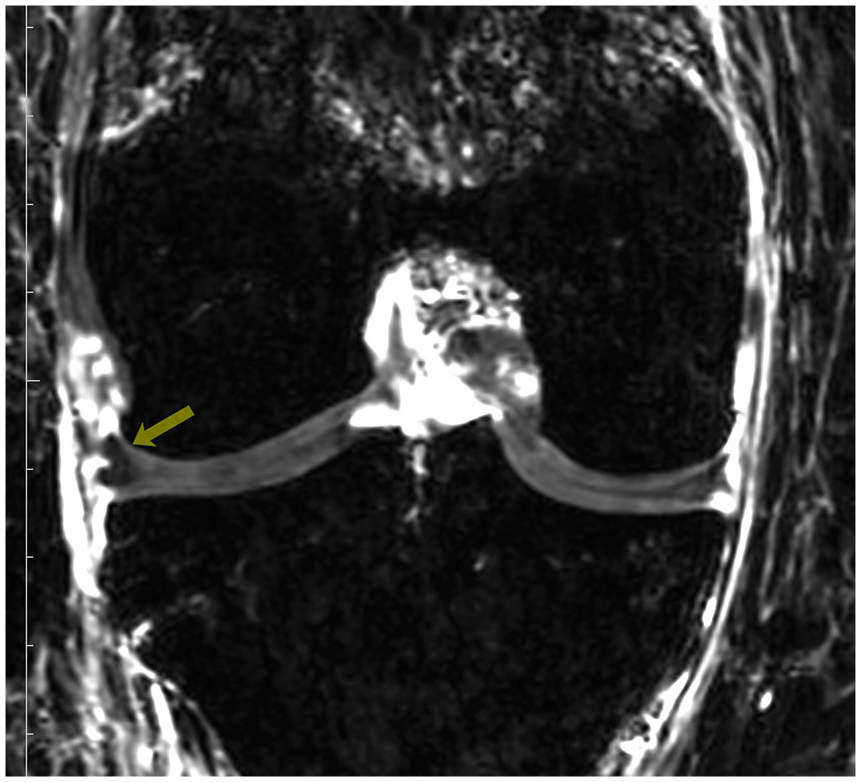
For quantitative analysis, regions of interest (ROIs) corresponding to peripheral and central zones of the meniscus were identified and manually defined on pre-contrast coronal images (
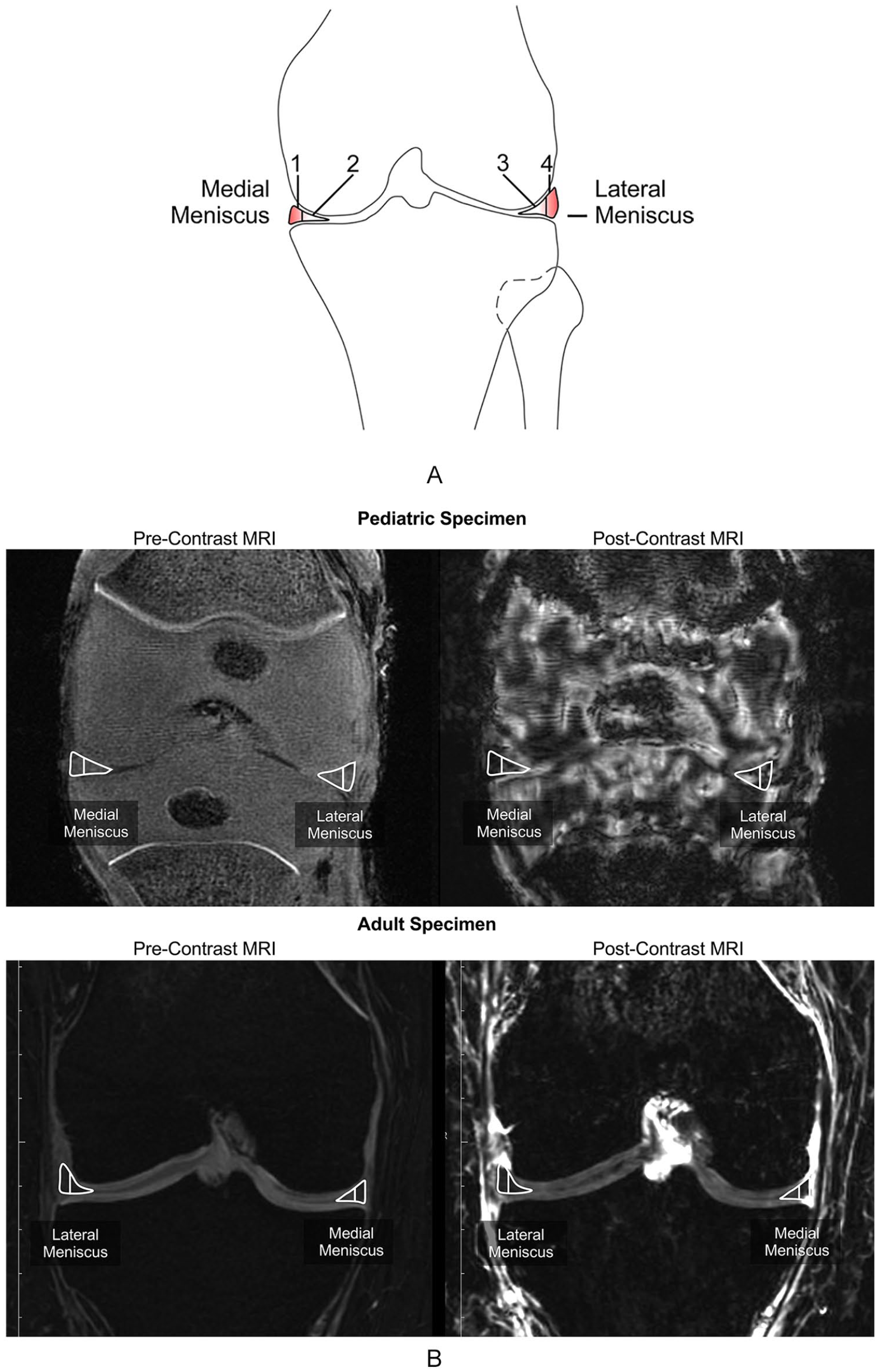
Analysis of meniscal vascularity using contrast-enhanced magnetic resonance imaging. (
Statistical Analysis
Descriptive statistical analysis was performed comparing signal enhancement between peripheral and central zones of the medial and lateral menisci individually, the peripheral versus central zones of the medial and lateral menisci aggregated, and total overall meniscal signal enhancement across different age groups. Comparative statistics were computed using the 2-tailed t test. A post hoc power analysis was calculated for 2 independent study groups with a continuous primary endpoint.
Results
Five neonatal knees, with an average age of 3 months (range 0-6 months), including 1 female and 4 males, and 5 adult knees, with an average age 50.2 years (range 34-60 years), including 2 females and 3 males, were analyzed, for a total of 10 menisci (5 lateral and 5 medial) from each group. Overall, the medial and lateral menisci had similar perfusion to each other in all specimens (45.9% ± 8.3% medial vs. 54.1% ± 8.3% lateral in neonatal knees; 50.5% ± 11.3% medial vs. 49.5% ± 11.3% lateral in adult knees). When comparing the 10 neonatal menisci to the 10 adult menisci, there was similarly increased perfusion in the peripheral zones compared with the central zones, at a ratio of 2.3:1 (70.0% vs. 30.0%) in immature knees, and 3.25:1 (76.5% vs. 23.5%) in mature knees (P = 0.31;
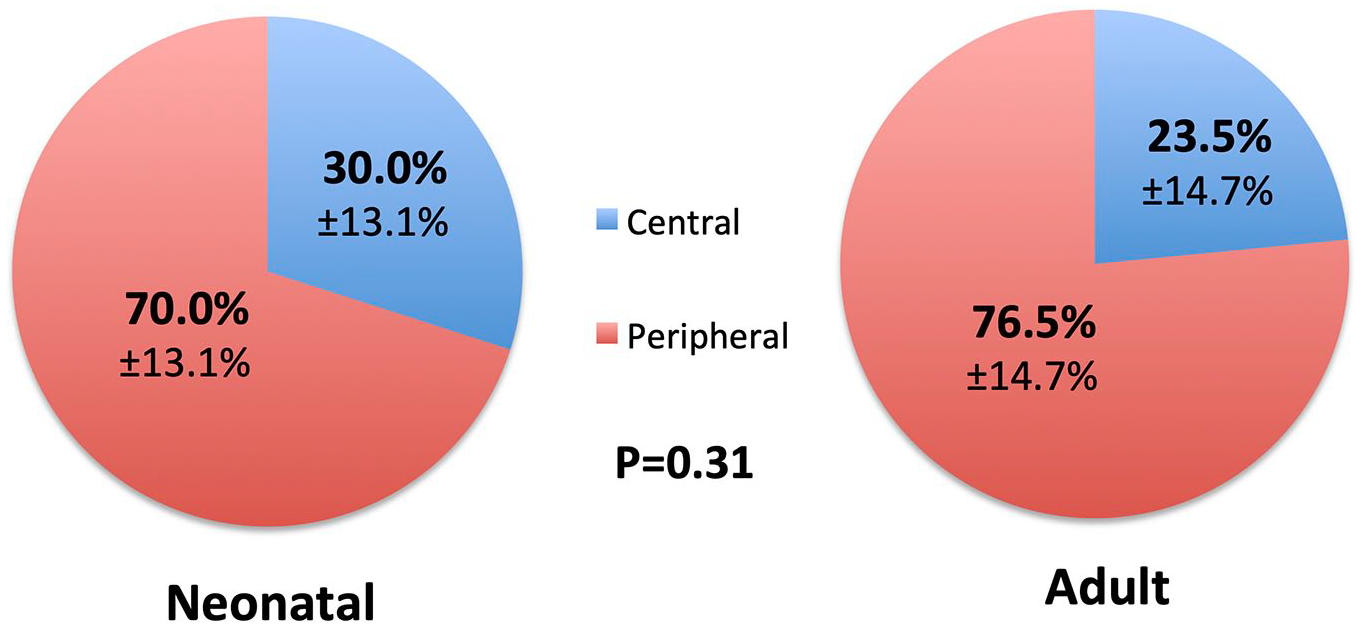
Distribution of meniscal perfusion by region. Comparison of relative vascularity of peripheral versus central zones in neonatal and adult menisci shows significantly greater blood flow to peripheral zones: 70% peripheral versus 30% central (SD 13.1%) in neonatal menisci and 76.5% peripheral versus 23.5% central (SD 14.7%) in adult menisci. The ratio of peripheral to central perfusion is not significantly different between the neonatal and adult groups (P = 0.31).
When comparing the total aggregate (peripheral plus central) meniscal perfusion, younger specimens demonstrated increased overall normalized meniscal signal enhancement (
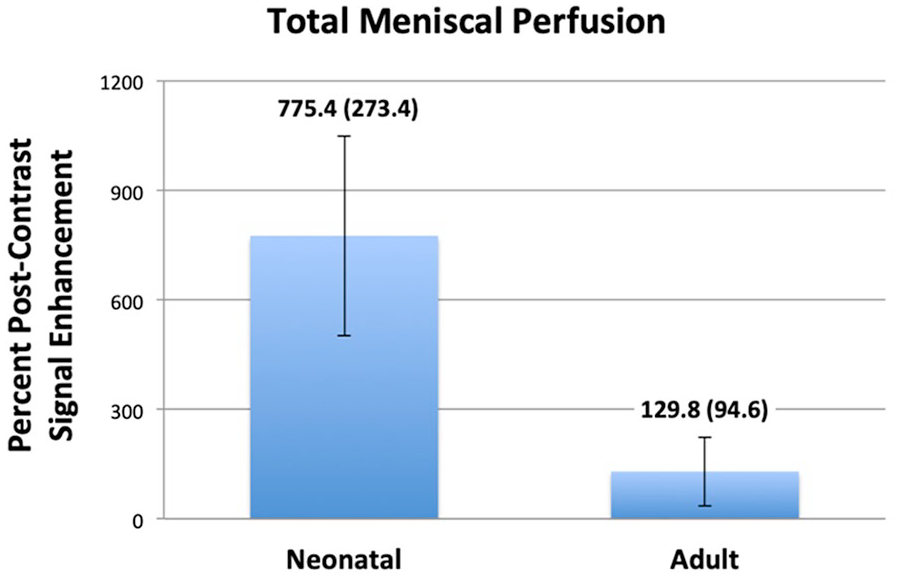
Total relative meniscal perfusion. Quantification of total meniscal post-contrast signal enhancement (peripheral plus central perfusion) shows an approximately 6.0-fold greater overall perfusion to neonatal menisci compared to adults. Neonatal menisci demonstrate 775.4% (SD 273.4%) signal enhancement, compared with 129.8% (SD 94.6%) enhancement in adult menisci (P < 0.0001).
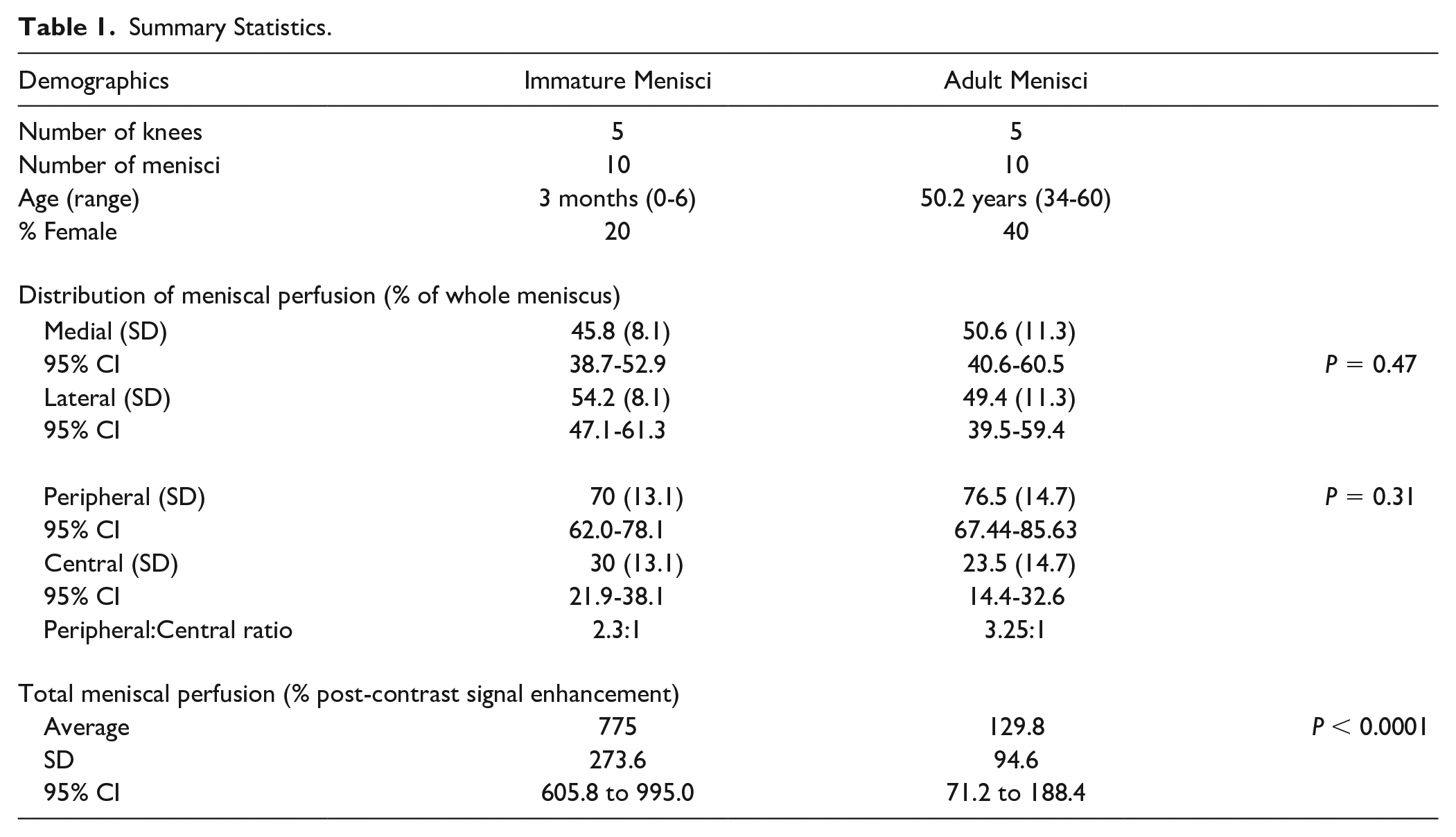
Summary Statistics.
On post-contrast coronal images in both pediatric and adult specimens, the peripheral vascular plexus described in previous literature was visualized (
Representative mid-coronal post-contrast magnetic resonance image showing signal enhancement at the periphery of the meniscus, representing the peripheral vascular plexus described by Arnoczky and Warren. 11
Post hoc power analysis was calculated separately for comparison of peripheral-central perfusion ratio between neonatal and adult menisci, and for comparison of total meniscal perfusion between the 2 groups. Post hoc power was 0.18 for detecting a difference in the peripheral:central ratio, and 1.00 for detecting a difference in total meniscal perfusion between neonatal and adult menisci in this study.
Discussion
The intrinsic capacity of meniscal tissue to heal after injury is dependent on the biological milieu of the tissue, specifically cell population(s), matrix-synthesizing activity, cell signaling capacity, and blood flow. The biomechanical loading environment likely also plays a critical role in meniscus biology. To our knowledge, this is the first study that quantifies meniscal vascularity through the use of quantitative MRI in the developing human knee. Quantitative contrast-enhanced MRI demonstrates that while arterial contribution to the periphery is greater than that to central zones in all menisci, developing menisci receive proportionally 6-fold greater arterial contribution compared to adults including to the central zone. Greater overall and central vascularity in the developing meniscus suggests improved healing potential, further encouraging meniscal preservation whenever possible.
From a developmental standpoint, the presence of a perfusion gradient from peripheral to central in the developing meniscus, although less pronounced than in the adult meniscus, suggests that development of the local vascularity of the meniscus is likely complete by birth. Early studies using only qualitative methods showed that the prenatal meniscus is a homogenous structure that is fully vascularized, and this abundant vascularization is known to recede throughout development.26,27 Studies in adult specimens have shown that this vascularized region comprises only the outer 10% to 30%.9,12,13,27,28 Our study provides insight into the early postnatal developmental period. Importantly, our findings suggest that at this point in development, although the vascular distribution may be similar to adult specimens, there is increased overall flow to the meniscus, including the presence of flow to the central region (albeit less than to the peripheral region).
Over time, there has been a trend toward meniscal preservation as opposed to meniscectomy when possible, given numerous studies showing an adverse effect on joint contact mechanics and long-term deterioration of the joint following meniscectomy.28-30 Biomechanical studies have shown that removal of 30% of the meniscus can increase peak compartment contact pressures by up to 350%. 31 Clinical and radiographic outcomes have been shown to be superior following meniscus preservation compared with meniscectomy,6-8,32-34 with a reported relative risk ratio of 14 for the development of osteoarthritis at an average 21 years after meniscectomy. 35 However, while meniscal preservation is the favored treatment for meniscal pathology, tissue healing is limited by the relatively poor intrinsic biological capacity of the meniscus for regeneration.27,36 Biological healing potential is imparted by available progenitor cell populations in the meniscus as well as cells derived from synovium, synovial fluid, bone marrow, or the vasculature, inter- and intracellular signaling pathways, and vascularity. While we are unable to draw conclusions regarding true healing potential in this cadaveric study, our results suggest that in neonatal/pediatric specimens, there exists arterial contribution to the central portion of the meniscus. While the distribution of arterial contribution is similar within the neonatal and adult meniscus, there is increased overall vascularity to the meniscus in the neonatal specimens, including to the central region, which was previously thought to be avascular. These findings suggest greater overall plasticity as greater blood flow means greater perfusion with oxygen, growth factors, and possibly progenitor cells. However, while vascularity clearly plays a critical role in meniscus healing, it should be noted that increased vascularity by itself may not be adequate to improve healing, as evidenced by animal studies of local treatment of meniscal tears using vascular endothelial growth factor, which have shown no effect in meniscal angiogenesis nor improved healing. 37
This increased overall vascularity may in part explain the improved outcomes after meniscal repairs in pediatric patients compared to adults.16,17 A recent study of horizontal meniscus tears extending into the “avascular zone” in a young population showed good midterm outcomes following repair, with a slight additional benefit when augmented with platelet-rich plasma. 38 A recent systematic review reported good to excellent results following meniscal repair in children and adolescents regardless of injury pattern, zone, or repair technique. 39 Some authors suggest that in contrast to adult patients, in pediatric patients, repair should be attempted for meniscal injuries in any zone, due to the hypothesized improved vascularity and intrinsic healing capacity.18,40 Nonetheless, further investigation is required to improve our basic understanding of meniscal biology with respect to cellular activity, signaling pathways, and vascularity. Further study is also necessary to better elucidate the clinical differences in outcomes following meniscus repair in immature versus mature knees, as well as the differences in initial injury patterns. Moreover, a fundamental understanding of the biology of the developing meniscus may provide greater insight that can inform development of therapeutic strategies for biological augmentation to improve healing in the adult meniscus.
There are several limitations to our study. First of all, this study included a small sample size of cadaveric specimens, as immature human cadaveric tissues are exceptionally rare. The post hoc power analysis showed a significantly underpowered comparison of peripheral-central distribution of perfusion between adults and immature specimens; thus, there is a high likelihood of type II error (false negative). Post hoc calculations, however, showed adequate power for determining difference in overall perfusion, due to the large effect size observed. For clinical correlation, analysis of living knees with presence of physiologic circulation would be most relevant. Additionally, analysis of older children or adolescent knees would be more directly applicable, as among the pediatric cohort they are typically the demographic that presents with meniscal injuries. While immature cadaveric tissues are exceedingly rare, a thorough study would require multiple specimens at each age; this is an area of interest for future study. Second, our MRI images are collected using a 3-T MRI. The meniscus, even in the adult specimens, is a small structure. The resolution of the images is enough to fully visualize the meniscus and to divide it into peripheral and central zones on a coronal projection; however, it is very difficult to subdivide further. The traditional breakdown of the meniscal zones includes red-red, red-white, and white-white zones, while our analysis is only able to reliably include peripheral third and central two-thirds. Similarly, due to resolution of the images in relation to the small size of the pediatric specimens, our imaging analysis was limited to the coronal plane. Nonetheless, clear quantitative analysis of signal enhancement changes was obtained using the defined zones in this study, providing valuable information. Another consideration is the contribution of the anterior and posterior horns to the vascularity. Further study using 3-dimensional ROIs in combination with higher resolution imaging capabilities would allow for more precise assessment of blood flow not only in the coronal projection but also within the horns and root attachments. Finally, the amount of contrast solution infused into the adult and pediatric specimens, while constant within each cohort, was different between cohorts: adult specimens received larger volumes of contrast given the greater overall specimen size. In order to control for different overall volumes of contrast administered, all quantifications were performed by normalizing to a standardized peripheral tissue. Furthermore, the overall contrast administered to adult specimens was four times greater than that administered to pediatric specimens; if pure contrast volume could create falsely elevated measurements of contrast enhancement, the adult specimens should theoretically have demonstrated elevated increases in overall signal intensity as compared with pediatric specimens. However, even with the greater contrast volume injected into adult specimens, neonatal menisci still had greater overall relative contrast enhancement, with analysis normalized to surrounding soft tissue. Further study using contrast volumes precisely standardized to specimen size would be beneficial.
Conclusion
This study demonstrates similar perfusion patterns in both immature and mature knees, with the periphery more densely perfused than the central region. Furthermore, younger menisci appear to receive proportionally greater overall arterial contribution even though the distribution of arterial contribution to peripheral and central zones remains similar. This knowledge has direct clinical relevance for meniscal repair in both pediatric and adult patients. Further investigation is warranted in pediatric and adult specimens using a larger sample size and higher resolution imaging to allow for detailed analysis of smaller zones of the meniscus.
Footnotes
Authors’ Note
This investigation was performed at the Hospital for Special Surgery, New York, NY, USA.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was funded by the Hospital for Special Surgery Pediatric Service Research Award, specifically for contrast-enhanced MRI imaging and analysis software. The study investigators gratefully acknowledge AlloSource (Centennial, CO, USA) for provision of immature specimen and Anatomy Gifts Registry (Hanover, MD, USA) for provision of adult specimen used for this vascular research study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Scott A. Rodeo—Flexion Therapeutics (paid consultant), Ortho RTI (Stock or stock options); Daniel W. Green—AO Trauma International (paid presenter or speaker), Arthrex, Inc. (IP royalties, paid consultant, paid presenter or speaker), Pega Medical (IP royalties); David L. Helfet—Fx Devices (stock or stock options), Healthpoint Capital (unpaid consultant), OHK Medical Devices (Stock or stock options, unpaid consultant), TriMedics (unpaid consultant). The remainder of the authors have nothing to disclose.
