Abstract
Background:
It is unclear whether the results of osteochondral transplant using autografts or allografts for talar osteochondral defect are equivalent.
Purpose:
A systematic review of the literature was conducted to compare allografts and autografts in terms of patient-reported outcome measures (PROMs), MRI findings, and complications.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
This study was conducted according to the PRISMA guidelines. The literature search was conducted in February 2021. All studies investigating the outcomes of allograft and/or autograft osteochondral transplant as management for osteochondral defects of the talus were accessed. The outcomes of interest were visual analog scale (VAS) score for pain, American Orthopaedic Foot and Ankle Society (AOFAS) score, and Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) score. Data concerning the rates of failure and revision surgery were also collected. Continuous data were analyzed using the mean difference (MD), whereas binary data were evaluated with the odds ratio (OR) effect measure.
Results:
Data from 40 studies (1174 procedures) with a mean follow-up of 46.5 ± 25 months were retrieved. There was comparability concerning the length of follow-up, male to female ratio, mean age, body mass index, defect size, VAS score, and AOFAS score (P > .1) between the groups at baseline. At the last follow-up, the MOCART (MD, 10.5; P = .04) and AOFAS (MD, 4.8; P = .04) scores were better in the autograft group. The VAS score was similar between the 2 groups (P = .4). At the last follow-up, autografts demonstrated lower rate of revision surgery (OR, 7.2; P < .0001) and failure (OR, 5.1; P < .0001).
Conclusion:
Based on the main findings of the present systematic review, talar osteochondral transplant using allografts was associated with higher rates of failure and revision compared with autografts at midterm follow-up.
Osteochondral lesions of the talus are common. 50 Given the avascular and hypocellular nature of articular cartilage, the management of talar osteochondral lesions is challenging.15,49 Several surgical procedures have been described.57,66 Smaller defects up to 1.5 cm 2 can be treated arthroscopically using the microfractures technique.4,52,74 For larger defects, osteochondral transplant has been commonly used.16,69 Osteochondral transplant can be performed using single or multiple plugs (mosaicplasty).13,68 Indications for osteochondral transplant are full-thickness symptomatic defects of 1 cm 2 up to approximately 4 cm 2 .23,24 Both osteochondral allografts and autografts can be used.39,47 Common donor sites for autologous osteochondral plugs are the anterosuperior condyle region and the lateral aspect of the intercondylar notch of the ipsilateral femur. 68 The use of allografts avoids donor site morbidity.9,76 Several clinical studies have evaluated the outcomes of autografts and allografts.** Whether osteochondral transplant using autografts performs better than that using allografts has not been fully clarified. The present study updates current evidence concerning the use of allografts and autografts for osteochondral transplant in talar osteochondral defects at midterm follow-up. A systematic review of the literature was conducted to compare allografts versus autografts in terms of patient-reported outcome measures (PROMs), MRI findings, and complications.
Methods
Search Strategy
This study was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 51 The PICO algorithm was as follows:
P (Problem): talus osteochondral defects
I (Intervention): osteochondral transplant
C (Comparison): autograft versus allograft
O (Outcomes): clinical scores, MRI findings, and complications
Data Source
The literature search was conducted in February 2021. Two independent authors (F.M. and H.S.) accessed the main online databases: PubMed, Google Scholar, Embase, and Scopus. The following keywords were used in combination: talus, ankle, chondral, cartilage, articular, osteochondral, damage, defect, injury, chondropathy, pain, autologous, allograft, autograft, transplantation, therapy, management, surgery, outcomes, failure, revision, reoperation, recurrence. The same reviewers selected the articles of interest and accessed the full-text. The bibliographies were also checked. Disagreements were solved by a third author (N.M.).
Eligibility Criteria
All studies investigating the surgical outcomes of allograft and/or autograft osteochondral transplant as management for osteochondral defects of the talus were accessed. Given the authors’ language capabilities, articles in English, German, Italian, French, and Spanish were considered. Studies with level of evidence 1 to 4, according to the Oxford Centre for Evidence-Based Medicine, 37 were eligible. Registries, letters, abstracts, reviews, editorials, and opinions were not considered. Animal, computational, biomechanics, and in vitro studies were excluded. Studies augmenting autologous matrix-induced chondrogenesis (AMIC) or matrix-induced autologous chondrocyte implantation (mACI) with less committed cells (eg, mesenchymal stem cells) were not considered. Studies reporting data on patients with end-stage joint osteoarthritis were not considered. Missing data under the outcomes of interest warranted exclusion from this study.
Data Extraction
Two authors (F.M. and H.S.) separately performed data extraction. Study generalities (author, year, journal, study design) and length of follow-up were retrieved. Baseline patient data were collected: length of symptoms before intervention, number of procedures, mean body mass index (BMI), mean age, sex, and mean defect size. Further, data concerning the following scores were retrieved: visual analog scale (VAS) for pain, American Orthopaedic Foot and Ankle Society (AOFAS), 75 and Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART). 48 Rate of failures and revision surgeries were also collected.
Methodological Quality Assessment
The methodological quality assessment was performed by 2 independent authors (F.M. and H.S.). The risk of bias graph tool of the Review Manager Software (The Nordic Cochrane Collaboration) was used. Selection, detection, attrition, reporting, and other sources of bias were considered for evaluation.
Statistical Analysis
The statistical analyses were performed by the main author (F.M.) using the IBM SPSS Version 25 program. Continuous data were reported as mean difference (MD), whereas binary variables were reported using the odds ratio (OR) effect measure. The confidence interval (CI) was set at 95%. We performed t tests and χ2 tests for continuous and binary data, respectively, with P < .05 considered statistically significant.
The meta-analyses were performed using Editorial Manager Software Version 5.3 (The Nordic Cochrane Collaboration). Dichotomous data were analyzed through the Mantel-Haenszel method and OR effect measure. The CI was set at 95% in all comparisons. A fixed model effect was set as default. If moderate or high heterogeneity was detected, a random model effect was adopted. Heterogeneity was evaluated through Higgins I2 and χ2 tests. Values of Higgins I2 were interpreted as low (<30%), moderate (30%-60%), and high (>60%). Values of P > .05 were considered statistically significant.
Results
Search Result
The literature search resulted in 357 articles. Of them, 104 were duplicates. A further 213 articles were excluded because of incompatibility with the inclusion criteria: not matching the topic (n = 139), not focusing on the talus (n = 17), study design (n = 21), not reporting quantitative data under the outcomes of interest (n = 11), augmented with other cells (n = 7), reporting data on patients with end-stage joint degeneration (n = 5), language limitations (n = 1), and other (n = 12). This left 40 articles for the present study: 35 retrospective and 4 prospective studies and 1 randomized clinical trial. The literature search results are shown in Figure 1.
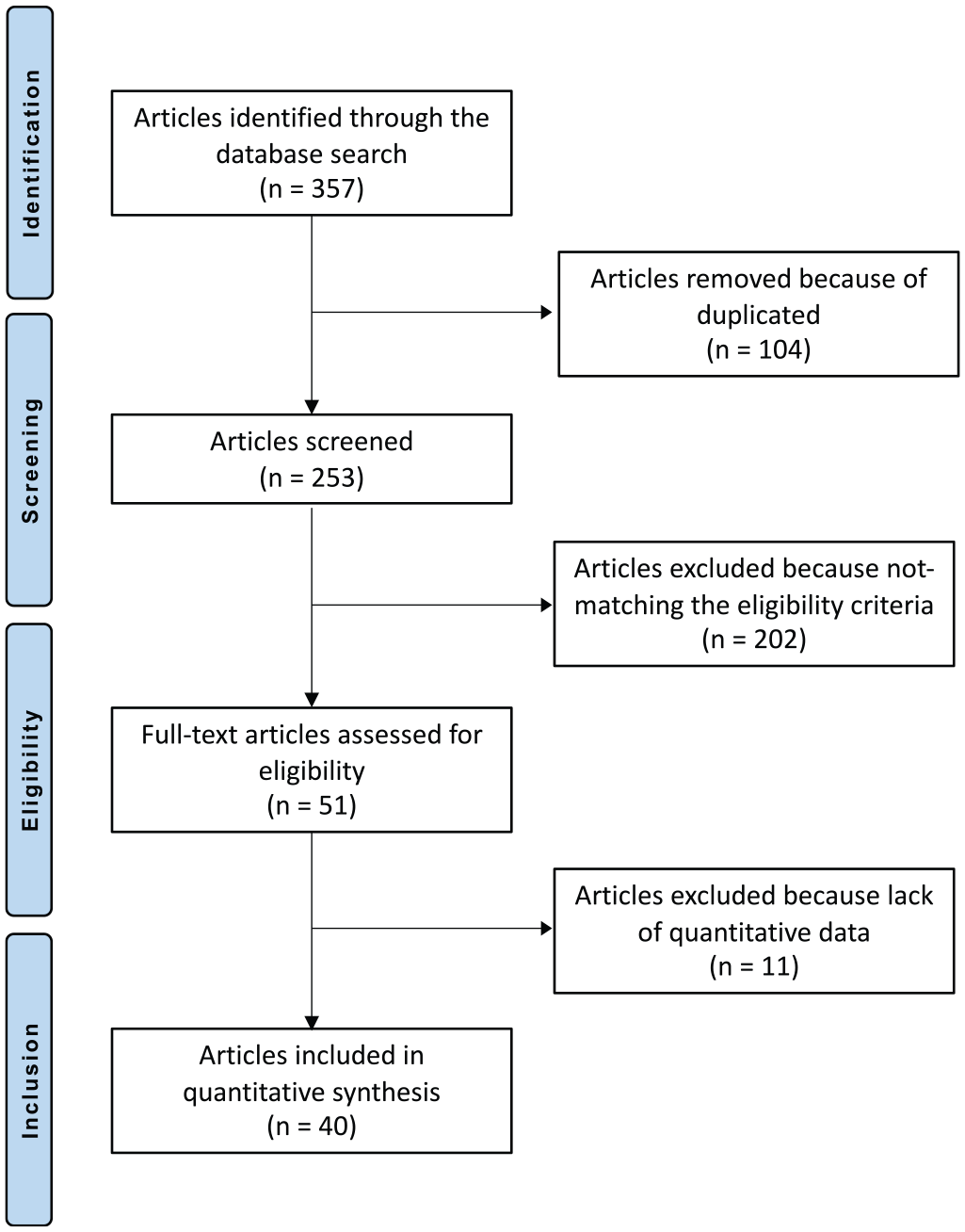
Flow chart of the literature search.
Methodological Quality Assessment
Given the retrospective design of 85% (35/40) of the included studies, the risk of selection bias was moderate-high. Given the overall lack of blinding, detection bias was high. The risk of attrition and reporting bias among all included studies was moderate, as was the risk of other bias. The overall quality of the methodological assessment was fair. The risk of bias graph is shown in Figure 2.
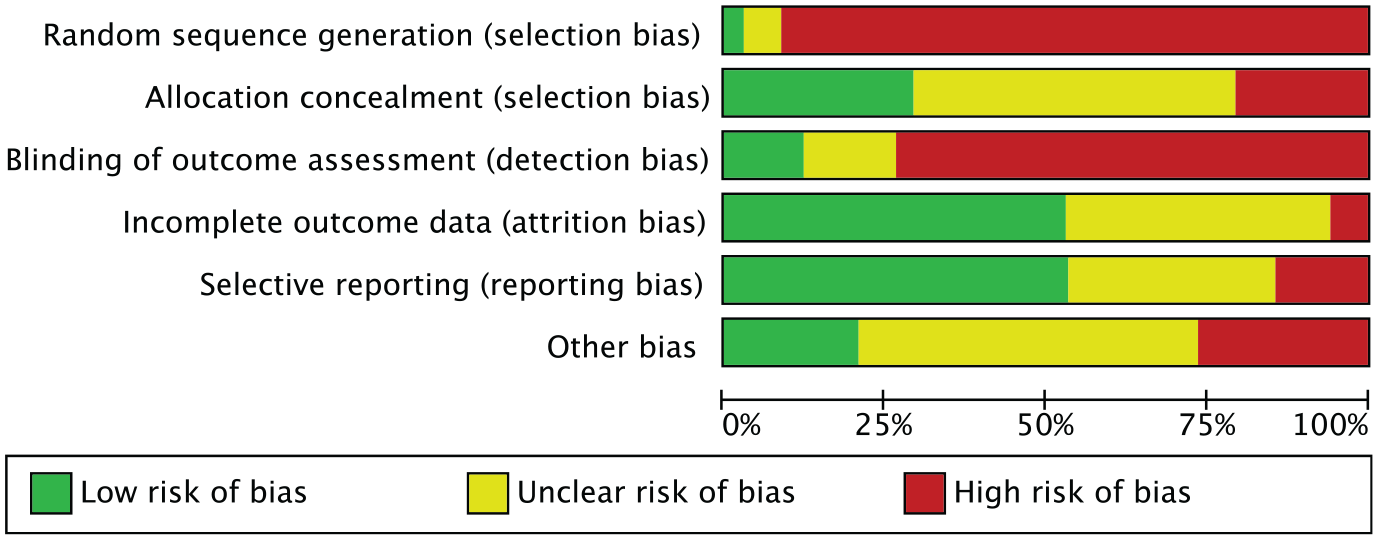
Methodological quality assessment.
Patient Characteristics
Data from 1174 procedures, with a mean follow-up of 46.5 ± 25 months, were retrieved. Study generalities and patient characteristics are shown in Table 1. There was no difference at baseline between the allograft and autograft groups (Table 2) concerning the duration of symptoms before surgery (P = .05), length of follow-up (P = .6), male to female ratio (P = .5), mean age (P = .4), mean BMI (P = .1), defect size (P = .6), VAS score (P = .8), and AOFAS score (P = .1).
Generalities and Descriptions of the Included Studies a
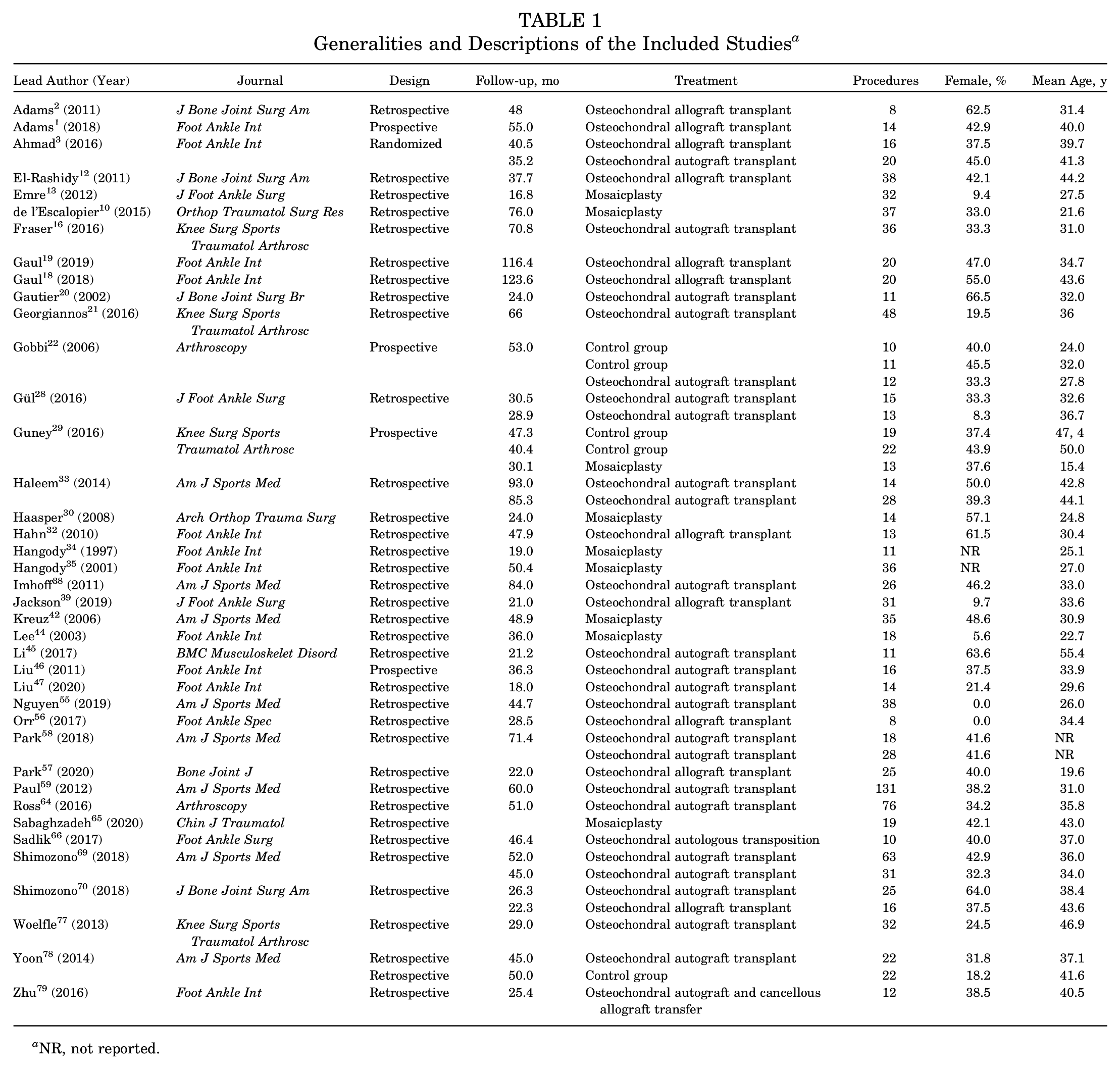
NR, not reported.
Characteristics of the 2 Cohorts at Baseline a
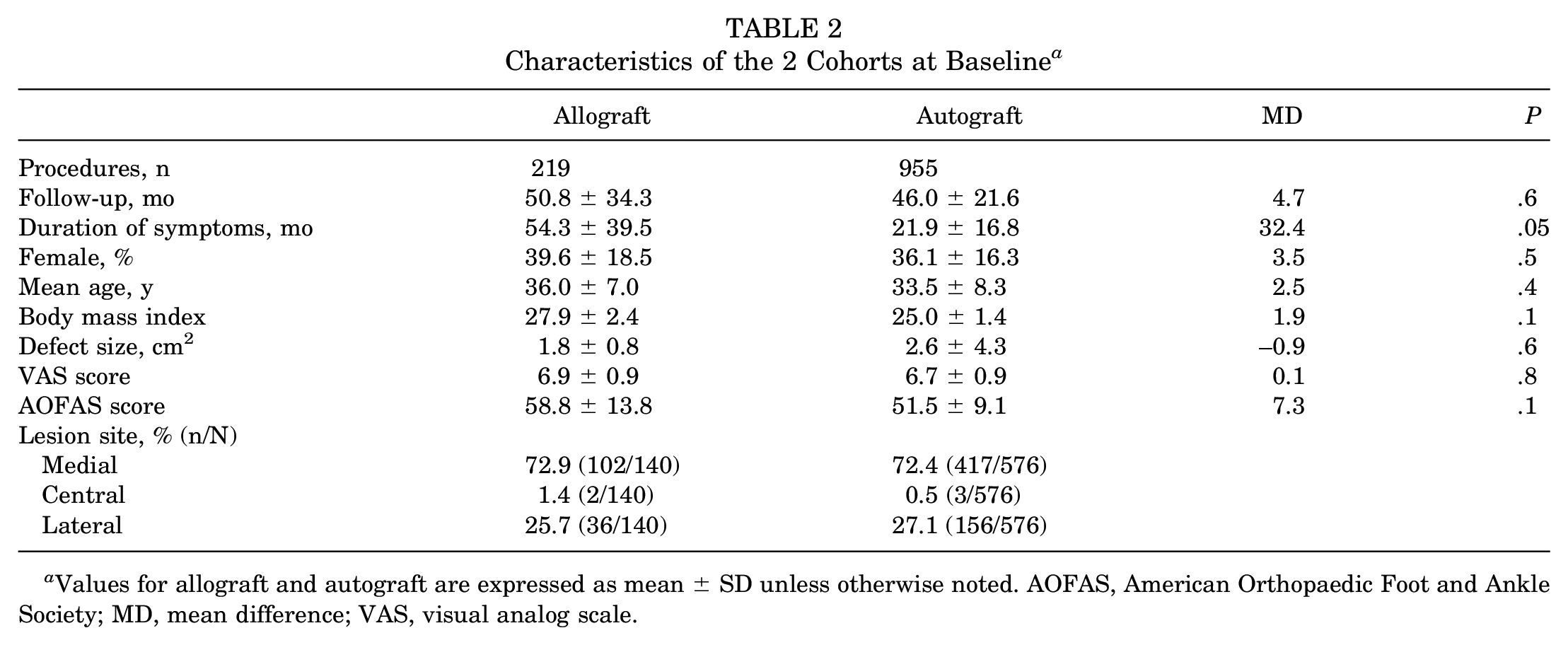
Values for allograft and autograft are expressed as mean ± SD unless otherwise noted. AOFAS, American Orthopaedic Foot and Ankle Society; MD, mean difference; VAS, visual analog scale.
Outcomes of Interest
At last follow-up, the autograft group had higher MOCART scores (MD, 10.5; P = .04). Similarly, the AOFAS score was higher in the autograft group (MD, 4.8; P = .04). The VAS score was similar between the 2 groups (P = .4) (Table 3).
Results of VAS, MOCART, and AOFAS Scores a
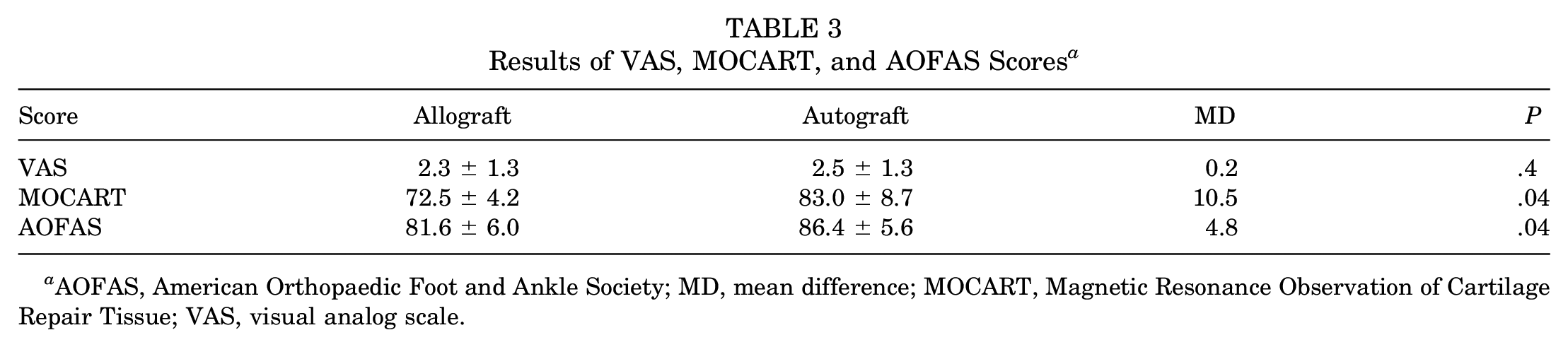
AOFAS, American Orthopaedic Foot and Ankle Society; MD, mean difference; MOCART, Magnetic Resonance Observation of Cartilage Repair Tissue; VAS, visual analog scale.
Complications
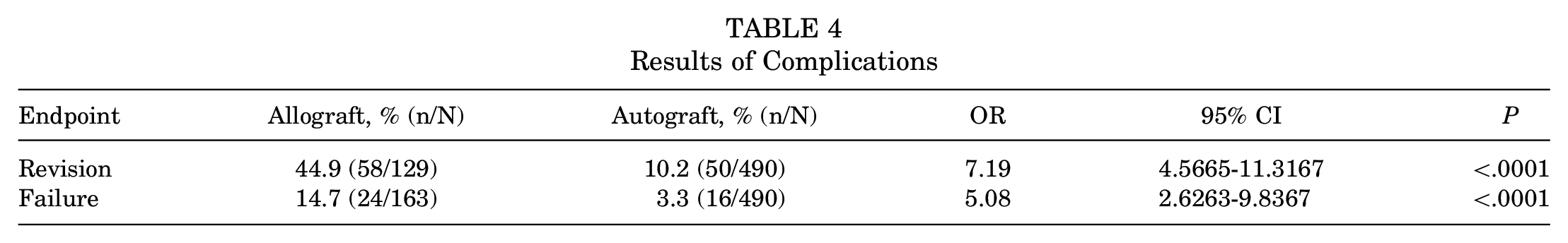
The autograft group demonstrated lower rates of revision surgery (OR, 7.2; P < .0001) and failure (OR, 5.1; P < .0001) (Table 4).
Results of Complications
Meta-analysis
Two comparative studies including 77 patients were included in the meta-analysis.3,70 We noted statistically significant lower rates of revision surgery (OR, 12.0; P = .02) and failure (OR, 13.8; P = .004) in the autograft group. The forest plots are shown in Figure 3.

Forest plots of the comparison: (A) failures and (B) revision surgeries.
Discussion
According to the main findings of the present study, osteochondral transplant for osteochondral defects of the talus using autografts or allograft evidenced similar VAS and AOFAS scores at midterm follow-up. However, given the lower rates of failure and revision, autografts should be preferred during primary surgeries for osteochondral defects of the talus. The primary use of allograft should probably be reserved for patients who do not have a suitable donor site.
Osteochondral autograft transplant has been used for small to medium-sized defects of the talus, with a rate of satisfaction up to 95% for primary procedures.41,67 Osteochondral autograft transplant has been used in revision settings as well, with considerable improvement of AOFAS and VAS scores.42,78 Allografts show a higher rate of failure and higher costs, greater risk of disease transmission, and limited availability, whereas the autologous techniques can entail donor site morbidity. 14 The effect of donor site morbidity on outcomes has not been fully clarified. Considering low-weightbearing, dimensional properties and surface curvature, only 3 harvest sites are available within the knee: the lateral side of the intercondylar notch, the superomedial trochlea, and the superolateral trochlea.27,36,63 In a systematic review by Bexkens et al 7 that included 190 patients, the rate of donor site morbidity was 7.8% (10/128 patients). Of those patients, 7% (9/128 patients) reported knee pain during activity and 0.8% (1/128 patients) reported locking episodes. Shimozono et al, 71 in a meta-analysis of 24 studies (915 procedures), estimated the rate of donor site morbidity to be from 6.7% to 10.8%. Nakagawa et al 53 investigated the clinical outcomes of donor sites in the knee after osteochondral autograft transplant. Those investigators found that 34 of 40 patients (85%) were asymptomatic at a mean follow-up of 43.1 months.
Despite the encouraging results with autografts, the use of allografts has become more prevalent to avoid donor site morbidity. Studies have shown that allografts achieve similar clinical outcomes as autografts and avoid donor site morbidity but have a greater rate of revision and failure.12,26 Two studies compared allografts versus autografts for osteochondral defects of the talus. Ahmad et al 3 conducted a prospective study comparing 16 autograft procedures with grafts from the ipsilateral femoral condyle versus 20 fresh size-matched allografts; the mean VAS scores improved similarly in both groups. Shimozono et al 70 retrospectively evaluated 25 patients treated with autografts versus 16 patients treated with allografts for talar defects and found that AOFAS and MOCART scores were better in the autograft group. Both of these studies found higher rates of failure and revision surgery in the allograft group.
The present study found a rate of failure of 14.7% (24/163 patients). This finding agrees with previous evidence of failure rates between 12% and 30%. Ahmad et al 3 found that 18.8% of allografts did not heal properly as shown on computed tomography scans or plain radiographs after 6 months. Gross et al 26 reported fragmentation and resorption of the allograft in one-third of patients (3/9). Haene et al 31 evaluated the failure rate of allografts in 16 patients at 48 months of follow-up. Those investigators evidenced failure in graft incorporation in 13% (2/16), osteolysis in 31% (5/16), and cysts in 50% (8/16) of patients at 4 years.
The higher rate of failure in allografts may derive from immune-mediated mechanisms. 54 Immune responses similar to those implicated in allogenic organ transplant rejections have been documented in human and animal osteochondral allograft transplant.17,60,72,73 Sirlin et al 72 demonstrated that 31% (11/36) of patients who underwent allograft implantation in the knee presented anti-human antibody. These patients had a statistically significant greater rate of edema, thicker interface, abnormal graft marrow, and surface collapse. 72 Pomajzl et al 61 demonstrated a substantial loss of sulfated glycosaminoglycans and osteocalcin in the graft-host interface, along with a high osteoclast activity. They further demonstrated the presence of T-helper, T-cytotoxic, and NK cells. Moreover, Pomajzl et al demonstrated the presence of tumor necrosis factor α throughout the allograft. These features discourage the use of osteochondral allograft transplant. If a suitable harvest site is unavailable, other techniques can be used, such as AMIC, mACI, and matrix-induced stem cell transplant (mAST); these are feasible and reliable options that exploit the regenerative potential of autologous tissue.5,6,8,25,62 Particulated juvenile articular cartilage (PJAC) transplant has been also used to manage osteochondral defects of the talus. However, given its limited evidence and controversial results, PJAC was not included in this analysis.11,40,43
This study has some limitations. The analyses were performed regardless of the surgical approach (arthroscopy, mini-arthrotomy, arthrotomy). Furthermore, the limited number of included articles and procedures may have negatively affected our results. The retrospective design of most of the studies represents an important limitation; however, the current literature lacks high-quality studies, and further investigations are required. Furthermore, given the lack of studies directly comparing the 2 types of graft, the meta-analysis collected data from only 2 trials.3,70 Thus, even if no heterogeneity was detected, the reliability of the results is questionable. Most authors did not clarify the type of allograft used and did not specify its provenance or any additional procedures (eg, sterilization) before transplant. Given the lack of data, primary and revision surgeries were often mixed, and some authors combined the surgeries with other procedures. This may have influenced the results, increasing the risk of selection bias. Given the lack of data, the analyses were performed regardless of the cause of the defect or the talar location of the defect (eg, medial vs lateral). Thus, our results must be interpreted within the limitations of the present investigation. Future studies are required to overcome these limitations.
Conclusion
Based on the main findings of the present systematic review, talar osteochondral transplant using allografts was associated with higher rates of failure and revision compared with autografts at midterm follow-up. However, given the limited published data, the strength of the conclusions is weak and further high-quality comparative studies are required.
Footnotes
Acknowledgements
The authors thank Mr Michael Berner for his support and motivation during the study conceptualization. They also thank Dr Andreas Bell and Dr Jens Schneider for their perpetual clinical support and education.
Submitted January 1, 2021; accepted April 9, 2021.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
An online CME course associated with this article is available for 1 AMA PRA Category 1 Credit™ at ![]() . In accordance with the standards of the Accreditation Council for Continuing Medical Education (ACCME), it is the policy of The American Orthopaedic Society for Sports Medicine that authors, editors, and planners disclose to the learners all financial relationships during the past 12 months with any commercial interest (A ‘commercial interest’ is any entity producing, marketing, re-selling, or distributing health care goods or services consumed by, or used on, patients). Any and all disclosures are provided in the online journal CME area which is provided to all participants before they actually take the CME activity. In accordance with AOSSM policy, authors, editors, and planners’ participation in this educational activity will be predicated upon timely submission and review of AOSSM disclosure. Noncompliance will result in an author/editor or planner to be stricken from participating in this CME activity.
. In accordance with the standards of the Accreditation Council for Continuing Medical Education (ACCME), it is the policy of The American Orthopaedic Society for Sports Medicine that authors, editors, and planners disclose to the learners all financial relationships during the past 12 months with any commercial interest (A ‘commercial interest’ is any entity producing, marketing, re-selling, or distributing health care goods or services consumed by, or used on, patients). Any and all disclosures are provided in the online journal CME area which is provided to all participants before they actually take the CME activity. In accordance with AOSSM policy, authors, editors, and planners’ participation in this educational activity will be predicated upon timely submission and review of AOSSM disclosure. Noncompliance will result in an author/editor or planner to be stricken from participating in this CME activity.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
