Abstract
Geopolymers (GPs) have been effectively used as an alternative to traditional calcium-based stabilizers for treating problematic expansive soils over the last decade. However, the high cost of precursors and limited availability of traditionally manufactured GPs pose significant challenges for commercial implementation. Utilizing locally available sources to manufacture GPs could address such limitations and promote sustainable development. A research study was designed and implemented to evaluate the efficacy of locally available calcined clay-based GP stabilizers to improve the performance of expansive soils and the results were compared with lime-treated and untreated soils. An array of laboratory studies was conducted using two natural clayey soils and two types of GPs to assess improvements in several engineering properties, including unconfined compressive strength before and after capillary soaking, resilient moduli, and moisture-induced strains. Supplemental microstructural studies using X-ray diffraction, optical microscopy, and scanning electron microscope imaging were conducted to identify the mineralogical changes and detect the precipitation of new reaction products. The engineering studies demonstrated that the application of GP significantly enhanced the strength, durability, and stiffness while reducing the swelling and shrinkage strains in the GP-treated soils. Microstructural analysis revealed that the precipitated GP gels effectively coated the soil particles and provided a uniformly bonded GP–soil matrix that enhanced the engineering performance. Overall, this study provides a comprehensive understanding with respect to the potential of using locally available calcined clay-based GPs as a sustainable and durable alternative in enhancing the engineering properties of expansive soils for the long-term performance of transportation infrastructures.
Transportation and other lightweight infrastructures built on expansive soils undergo significant distress caused by the low strength and high swelling and shrinkage characteristics of the underlying soils ( 1 ). Several traditional and non-traditional chemical stabilization techniques have been successfully implemented over the past several decades to address some of these inherent challenges ( 2 , 3 ). Among the conventional methods, calcium (Ca)-based stabilizers, such as cement and lime, have been predominantly used owing to the low cost of applications and substantial enhancement in the mechanical strength and durability characteristics of treated soil layers ( 4 , 5 ). However, the excessive reliance on these calcium-based stabilizers could engender notable environmental concerns, primarily associated with the considerable energy consumption during the manufacturing processes and consequent emissions of CO2 and other greenhouse gases ( 6 ). As a result, the research community seeks various environmentally friendly stabilizers that could be implemented as sustainable alternatives, aiming to efficiently mitigate the issues with traditional stabilizers ( 7 – 9 ).
Geopolymer (GP) has garnered significant attention within the academic and scientific communities as an environmentally friendly material owing to its remarkable capacity to reduce CO2 emissions ( 10 , 11 ). Research studies have indicated that GP production exhibits lower energy consumption compared to Portland cement, with energy savings of approximately 40% ( 12 ). In addition, the utilization of GP offers the potential to promote recycling by incorporating various industrial waste materials into its production process ( 13 , 14 ). Chemically, GPs are amorphous inorganic polymers synthesized in the temperature range of 20–90°C. This synthesis involves the mixing of aluminosilicate sources, such as clay, metakaolin (MK), and fly ash, with an alkaline activator solution (AAS) ( 15 , 16 ). The AAS comprises alkali metal cations such as KOH and NaOH, along with water and silica. In general, aluminosilicate-rich materials such as MK, fly ash, ground granulated blast furnace slag (GGBFS), and rice husk ash are used for alkali activation, resulting in the formation of inorganic cementitious products ( 17 , 18 ). The process of GP formation, known as “geopolymerization,” occurs when aluminosilicate solids, known as binders, react with concentrated liquid alkaline solutions, referred to as activators. Geopolymerization is characterized as an exothermic process that advances through multiple stages, including dissolution, reorientation, and solidification ( 19 ). Geopolymerization results in the formation of cementitious binding reaction products that have the ability to improve geomaterial properties and therefore have a huge potential for civil infrastructure applications ( 20 , 21 ).
GP has been successfully implemented in a wide range of applications, including fire resistance, nuclear waste immobilization, water purification, and construction materials ( 22 ). In the clayey soil stabilization aspect, GP treatment induces the flocculation of clay particles on precipitation of gel-like reaction products ( 23 ). Consequently, GP exhibits the ability to alter the behavior of clayey soil, transforming it from a plastic to a non-plastic state and subsequently mitigating moisture-induced swelling and shrinkage strains ( 24 , 25 ). Furthermore, the introduction of a GP binder enables the formation of a denser microstructure within the soil, enhancing both cohesion and internal friction ( 26 ). As a result, a significant improvement is observed in the mechanical properties of the treated soil, including unconfined compressive strength (UCS), shear strength, and wet–dry durability, among others ( 27 – 29 ). Several studies have indicated that GPs have the potential to be used as a versatile soil stabilizer for shallow-depth applications, including pavement base/subbase, embankment construction, shallow foundations, airport infrastructure, retaining walls, and deep soil mixing techniques ( 27 – 30 ). However, there exists a certain degree of hesitancy within the community with respect to the implementation. This hesitation can be attributed to factors such as the high cost associated with GP utilization, a limited understanding of the stabilization mechanism, and a lack of consistency among published studies caused by variations in GP synthesis recipes adopted by different research groups ( 8 , 31 ).
The utilization of aluminosilicate sources is essentially a fundamental requirement in GP production. Fly ash, GGBFS, and rice husk ash have been used in GP production; however, their significant impurities, non-stable chemical composition, and low silica and alumina content have posed challenges for researchers in controlling the influencing parameters and achieving the desired outcomes ( 32 , 33 ). Therefore, MK, obtained through the calcination process of kaolin, is widely favored because of its high purity and abundant silica and alumina content ( 34 ). However, the limited availability and high cost of MK in local areas can cause significant deterrence against its usage on an industrial scale ( 35 ). Therefore, it was hypothesized that the utilization of locally available soils, rich in kaolinite, as a potential alternative aluminosilicate source to develop GPs and subsequently treat problematic expansive soils could address some of the shortcomings of GP-based soil stabilization.
A research study was designed and conducted to evaluate the efficacy of a locally sourced calcined clay-based GP as a soil stabilizer for problematic expansive soils. Two different locally available clays were used to develop the GPs and act as aluminosilicate sources. Subsequently, the two novel GPs were used to treat two different expansive soils from Region-6. An array of engineering tests, including strength, stiffness, moisture-induced strains, and microstructural and mineralogical characterizations, such as X-ray diffraction (XRD) studies, optical microscopy (OM), and field emission scanning electron microscopy (FESEM) imaging, were performed to develop a comprehensive understanding of the effective stabilization of expansive soils. In addition, the results of GP treatment were compared with both untreated and conventional lime-treated soils. Overall, this study is expected to have an enormous potential to reduce construction costs while promoting sustainable and resilient development. By capitalizing on the abundance of local resources, the dependence on expensive imported materials could be minimized, thereby enhancing the economic feasibility of soil stabilization projects. The following section discusses the materials used and the engineering and microstructural test performed under the current research.
Materials and Methods
Geopolymer Synthesis and Optimum Geopolymer Composition
The synthesis of GPs required an aluminosilicate source and an alkaline activator solution (AAS), as illustrated in Figure 1. To assess the feasibility of locally available clay sources for aluminosilicate precursors, two types of local calcined clays, namely UPF (calcined at 700°C or 1292°F for 90 min) and TNKY (calcined at 800°C or 1472°F for 90 min), were utilized. Results from quantitative XRD studies indicated that UPF was primarily composed of 91.0% kaolinite, 2.2 % anatase, and 6.8% muscovite, while TNKY predominantly consisted of 74.9% kaolinite, 24.2% quartz, and 0.9% anatase. The AAS was synthesized by combining potassium hydroxide (KOH), deionized water, and amorphous-fumed silicon (IV) oxide (silica fume). Firstly, KOH flakes were dissolved in deionized water, resulting in the separation of K+ and OH- ions and an increase in the alkalinity (pH > 13.0). Then a target dosage of silica fume was added to the solution to achieve the desired Si:Al molar ratio (silica: 53.0% and aluminum: 43.8%) after mixing with the calcined clay. The AAS, containing KOH and silica fumes, was blended using a magnetic stirrer for a minimum of 48 h at room temperature to synthesize a homogeneous solution. To minimize contamination from atmospheric carbonation, the AAS was promptly stored in sealed containers after mixing. Subsequently, the calcined clays and the AAS were thoroughly mixed for at least 3 min to create a uniform solution to the desired Si:Al molar ratio. Through preliminary trials, the ratio of key GP components, such as SiO2/Al2O3 = 3, water/solid = 3, and K/Al = 1, was deemed suitable for stabilization. This optimized GP composition was referred to as K331 (where “K” represents the alkali source, that is, KOH, and the numbers denote the ratios of the various components mentioned) for the subsequent study.
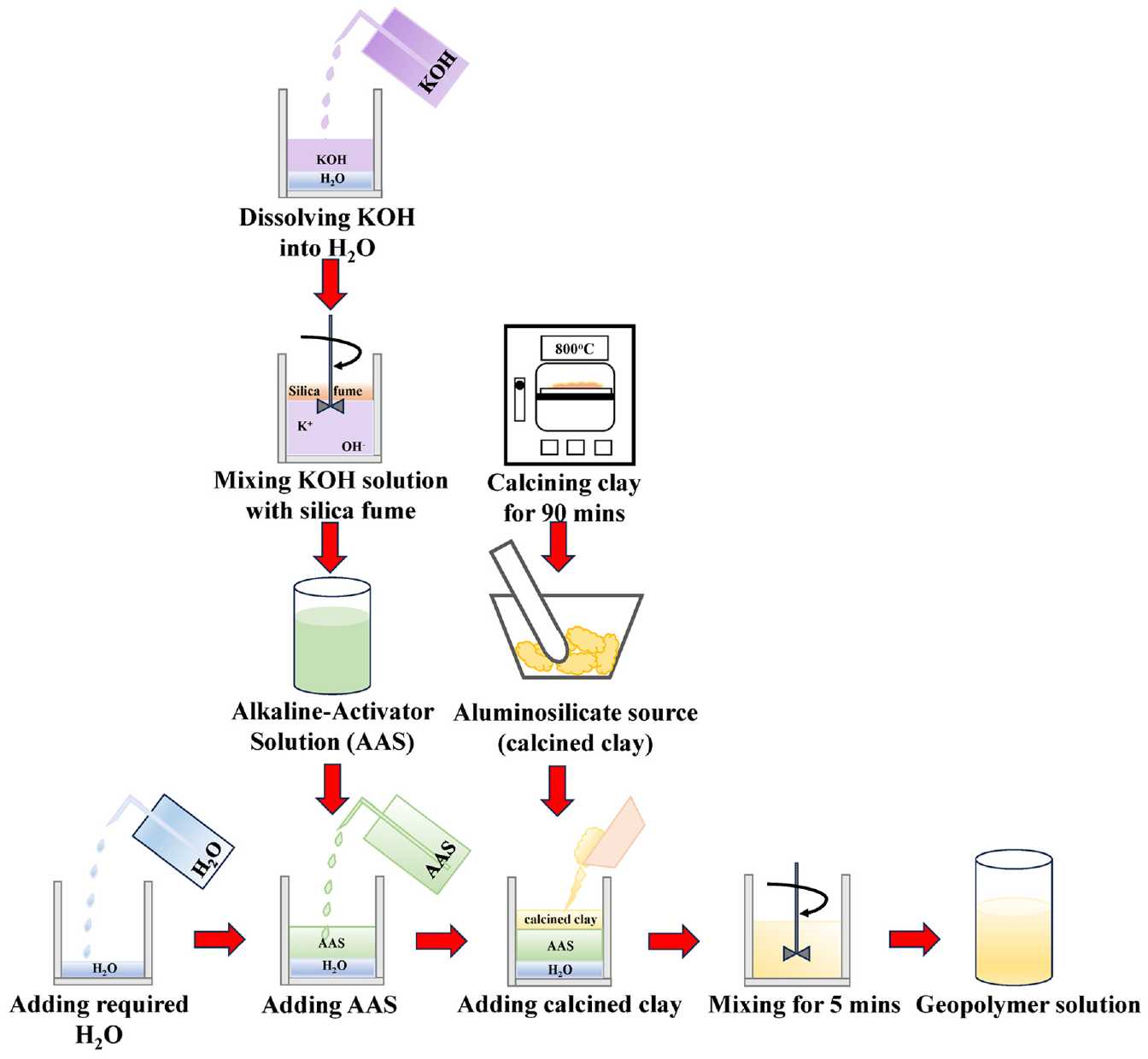
Schematic process for geopolymer preparation.
Geomaterials
Two natural expansive soils, predominantly found in Region-6, were collected from pavement sites located near the Dallas-Fort Worth Metropolitan area in Texas. Table 1 presents the basic engineering characterization results obtained in accordance with the respective American Standard for Testing and Materials (ASTM) and Texas Department of Transportation (TxDOT) standards. Figure 2 illustrates the particle size distribution of both natural soils. Soils S1 and S2 have been classified as low plasticity clay (CL) according to the Unified Soil Classification System (USCS). Soils S1 and S2 exhibited plasticity indices (PIs) of 19.4 and 16.7, free swell strains of 18.0% and 11.1%, and linear shrinkage of 10.3% and 9.0%, respectively, indicating their expansive behavior ( 36 ). In addition, mineralogical studies using quantitative XRD indicated that soil S1 was composed of 17.3% illite, 38.4% quartz, and 43% calcite. Similarly, soil S2 exhibited a composition of 12.8% illite, 24.4% quartz, and 62.8% calcite.
Basic Characterization Results of Natural Soils
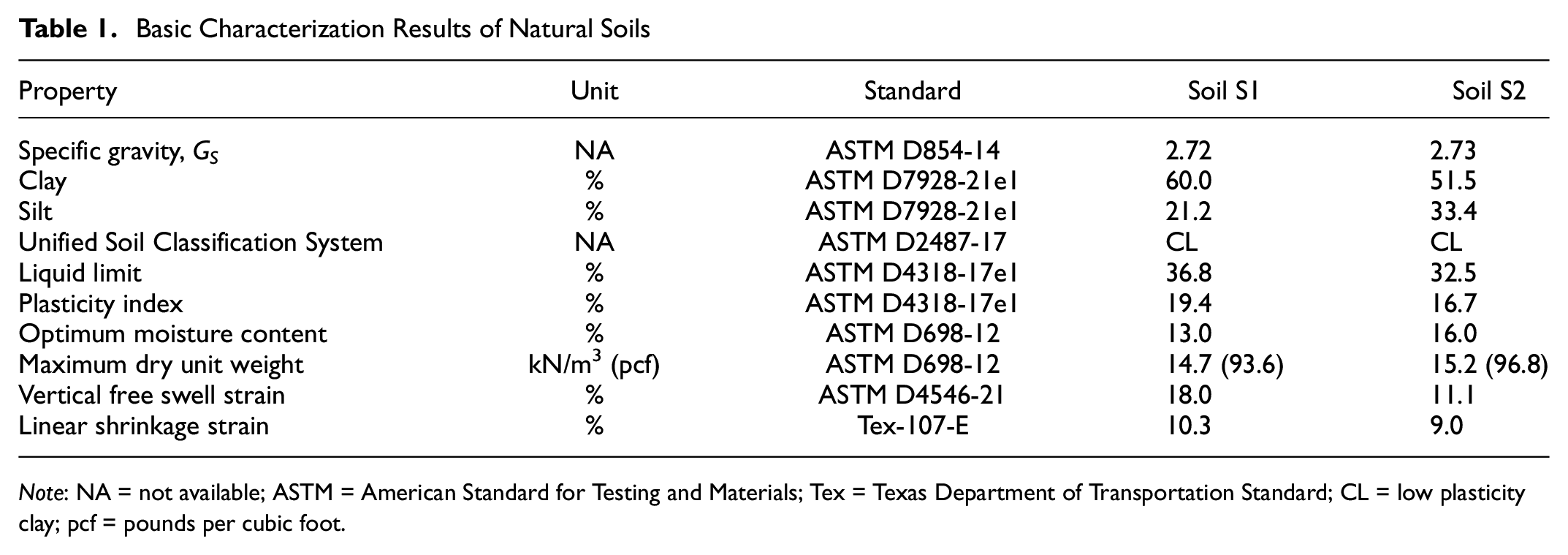
Note: NA = not available; ASTM = American Standard for Testing and Materials; Tex = Texas Department of Transportation Standard; CL = low plasticity clay; pcf = pounds per cubic foot.
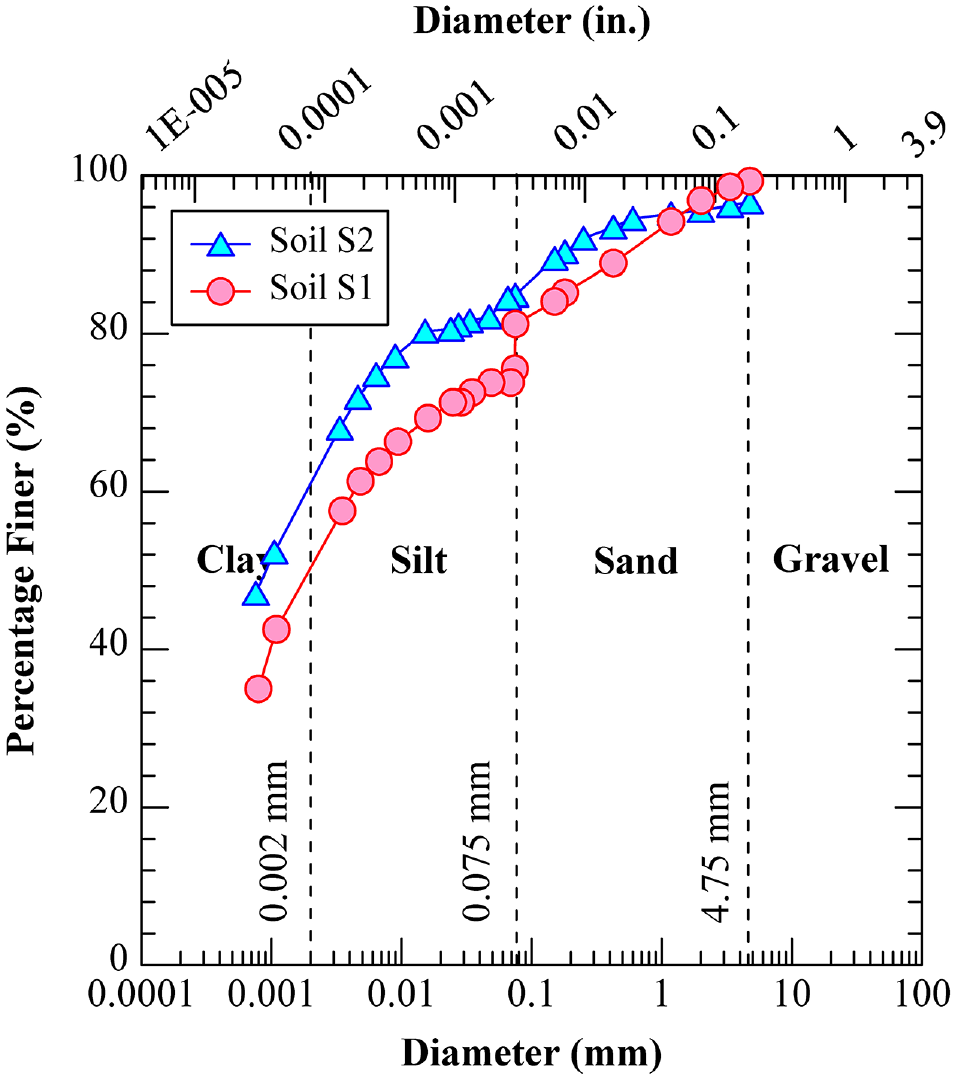
Grain size distribution of natural soils.
Preparation of Chemically Treated Soils
The collected soils were oven-dried at 105 ± 5°C (221 ± 9°F) for 24 h, crushed, and subsequently pulverized. Untreated soils were prepared by evenly mixing the dry natural soils with the optimum moisture content (Figure 3). For GP-treated soils, the GP dosages were determined as a percentage of the total weight of the GP solution, which consists of calcined clay, KOH, silica fumes, and water relative to the dry weight of the natural soil. To assess the effectiveness of GP treatment on soil properties, two different GP dosages, 8% and 20%, were selected as initial trial dosages to be mixed with the natural soils. The optimum dosage of lime-treated soils used for comparative analysis was determined as 5% of the dry weight of the soil in accordance with the Eades and Grim pH testing method (ASTM D6276-19). GP- and lime-treated soils were prepared by uniformly mixing the target dosage of GP or lime with dry natural soils. Subsequently, the required optimum molding water was added to the stabilizer–soil mixture and mixed evenly to form a homogeneous soil mixture. The treated soils were equilibrated at room temperature for 30 min before molding into various specimens at respective target dry unit weights (Figure 3). The GP-treated specimens were cured for three durations, 6 h (0 day), 3 days, and 7 days, while the lime-treated specimens were cured only for 14 days. All specimens were cured at room temperature (23.5 ± 0.5°C or 74.3 ± 0.9°F) in a hermetically sealed chamber with approximately 100% relative humidity to ensure sufficient moisture during the curing stages. For subsequent analysis, the specimens were abbreviated as XY-S, where X is the percentage of dosage (5%, 8% and 20%), Y is the type of chemical stabilizer (U: UPF-GP, T: TNKY-GP, L: lime), and S is the type of soil (S1 and S2). For instance, 20U-S1 is 20% of UPF GP-treated soil S1.
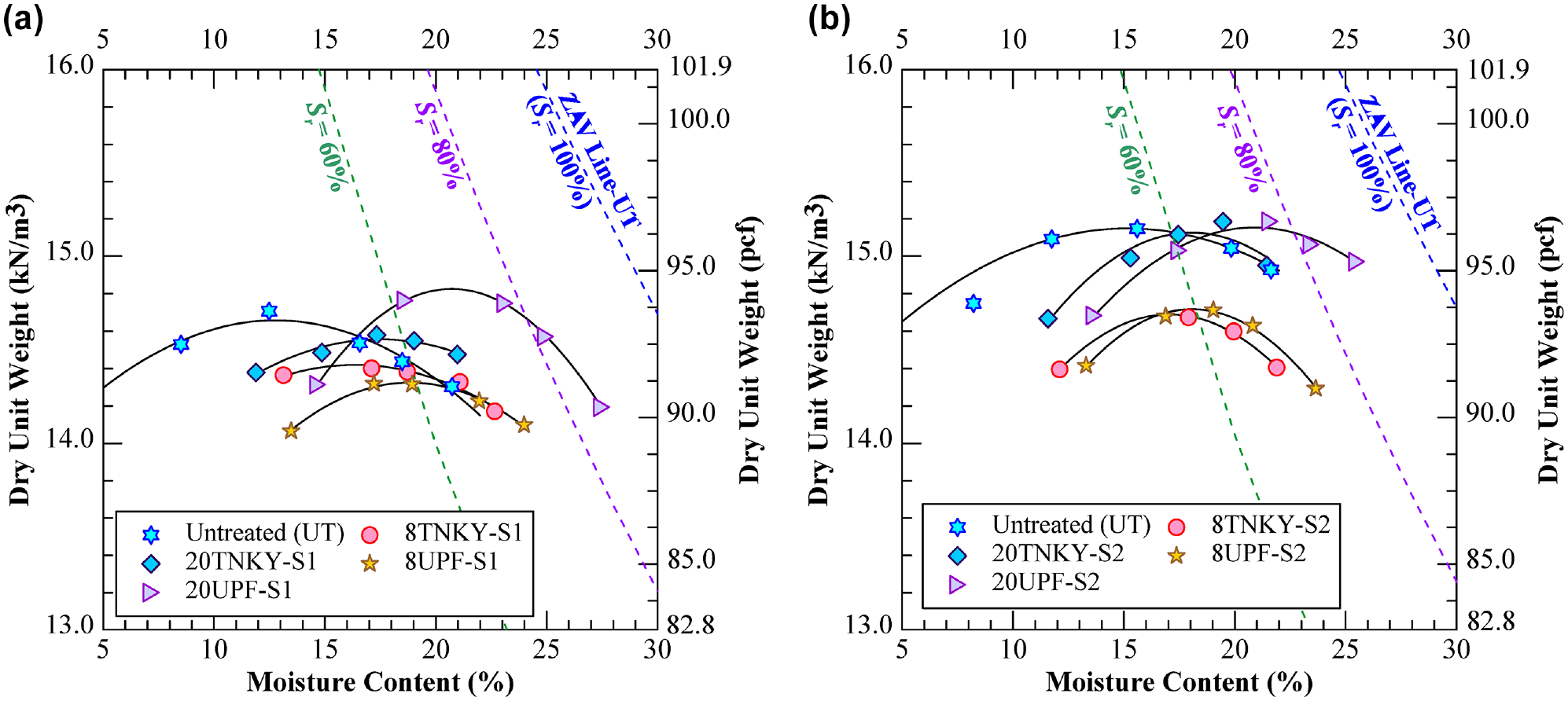
Dry unit weight–moisture content curves of different groups for (a) soil S1 and (b) soil S2.
Engineering Tests
UCS tests were performed to understand the strength improvement of treated soils caused by geopolymerization reactions. The specimens for UCS tests were compacted in three layers in cylindrical molds of diameter of 33 mm (1.3 in.) with an aspect ratio of 2:1 (height: diameter), at the target maximum unit weight (Figure 3). The moisture ingress was expected to have a significant impact on the mechanical strength of the soil and indirectly provide an indication of the durability of the chemical stabilizers. Therefore, the test was conducted on triplicate specimens for both untreated and treated soils at different curing periods, before and after capillary soaking for 48 h. The specimens were subjected to shearing at a constant strain rate of 1%/min as per ASTM D2166-06. The UCS values were determined by recording the average peak normal stress from the stress–strain curves of the triplicate specimens.
The resilient modulus (MR) is a ratio of deviatoric stress to the resilient or elastic strain experienced by subgrade material under repeated traffic loading conditions ( 37 , 38 ). It is a key input parameter for flexible pavement design in accordance with the Mechanistic-Empirical Pavement Design Guidelines ( 39 ). The application of chemical stabilization could enhance the resilient modulus and consequently improve the serviceability of subgrade layers to accommodate repeated traffic loads. Duplicate specimens for repeated load triaxial (RLT) tests were compacted using static compaction in three layers into a cylindrical mold with a diameter of 72 mm (2.8 in.) and a height of 148 mm (5.8 in.) at the target densities shown in Figure 3. To assess the influence of GP on the resilient moduli values, the RLT test was performed as per AASHTO T-307 on duplicate specimens and compared with the untreated and lime-treated soil groups. Before the test, specimens were preconditioned with 500 load cycles to remove the initial plastic strains. Thereafter, 15 loading sequences having 100 cycles with varying confining pressures (41.4, 27.6, and 13.8 kPa, or 6, 4, and 2 pounds per square inch [psi]) and deviatoric stresses (13.8, 27.6, 41.4, 55.2, and 68.9 kPa, or 2, 4, 6, 8, and 10 psi) were applied. The average value of the last five cycles in each sequence was reported as the resilient modulus of the particular stress state.
Specimens for vertical free swell were compacted using static compaction in a single layer into a mold with a diameter of 63.5 mm (2.5 in.) and a height of 25.4 mm (1.0 in.), at a similar compaction density of strength and stiffness specimens. The free swell test was performed in accordance with ASTM D4546-21 under constant vertical pressure of 1 kPa (0.15 psi). The dial gauge readings were monitored, and the tests were continued until constant dial gauge readings were noted for three consecutive days. The final reading of the dial gauge reading, indicating the highest value, was used to determine the free swell strain as a ratio between the final reading and the initial height of the soil specimens.
Linear shrinkage bar tests were conducted to investigate the shrinkage behavior of untreated and treated soils. For the untreated specimens, dry natural soils with particle sizes smaller than 0.425 mm or 0.0165 in. (U.S. Sieve #40) were mixed with distilled water until a consistency close to the liquid limit was achieved. Similarly, the chemically stabilized soils were prepared by mixing specimens for each combination, cured for 7 days (GP) or 14 days (lime), with distilled water to reach the same consistency. Once the specimens in each set exhibited a noticeable color change, they were placed in an oven and dried for 24 h at a controlled temperature of 105 ± 5°C (221 ± 9°F) in accordance with the Tex-107-E standard. After the drying process, the lengths of the specimens from each set were measured to determine the linear shrinkage strain of the soil groups.
Microstructural and Mineralogical Characterization
Microstructural and mineralogical analyses were conducted on select soil and GP dosages after 7 days of curing using XRD, OM, and FESEM to study the mineralogical changes and visually detect the morphology of natural and GP-treated specimens. For XRD studies, the dry samples were pulverized and sieved through a 75 μm sieve. The powder samples were prepared in a 51.5 mm (2.03 in.) diameter polymethyl methacrylate (PMMA) holder with a height of 8.5 mm (0.33 in.) using the backloading technique. The PMMA holder with the powder sample was loaded onto the XRD instrument and subjected to a Cu-Kα radiation source at 10 mA and 30 kV, with a range of 2θ from 10° to 60° and a step size of 0.02°. Relative changes in the characteristic peaks and the formation of new peaks were used to identify the potential chemical reactions. In addition, an optical microscope, equipped with a versatile optical system enabling seamless transition from macro-to-micro viewing, was employed to investigate and compare the morphological changes between untreated and GP-treated specimens at different dosages. Because of the limitation on magnification and resolution of the OM, additional high-resolution image analyses were performed using FESEM to visually identify the formation of cementitious binding GP gels after geopolymerization and compared with untreated soil. These studies were conducted using a high-resolution scanning electron microscope equipped with a cold field emission gun, with a maximum resolution of approximately 1 nm on the dry specimens. The next sections provide comprehensive analysis and discussion on the engineering and microstructural studies performed on different soil groups.
Analyses and Discussion
Strength and Durability Studies
Figure 4 illustrates the unsoaked UCS values, as the average of the peak stress–strain curve obtained from triplicate specimens, along with the standard deviations (S.D.), for untreated and GP-treated soils with different treatment dosages and curing periods. The UCS values of the untreated and lime-treated soil S1 were 250 kPa (36.3 psi) and 900 kPa (130.5 psi), respectively. On subjecting the soil to GP treatment, the UCS of the stabilized soil increased with both curing period and treatment dosages (Figure 4a). Specifically, the 20U-S1 specimen showed a 26% increase in UCS after 6 h of curing and further increased to approximately 2.8 times higher than that of the untreated specimens. Similarly, the UCS of the 20T-S1 specimen was measured as 373 kPa after 6 h, and it increased to a 2.3 times higher value than the untreated specimens at the end of 7 days. This significant improvement can be attributed to the uniform binding resulting from strong physical and chemical bonds between GP and soil particles. It was also observed that the UCSs of the 8U-S1 and 8T-S1 specimens at 6-h curing were similar to those of the untreated specimens. This lower dosage did not result in a significant improvement in strength because of the presence of weak physical and chemical bonding caused by non-uniform binding. After 7 days of curing, the UCS values of 8% treated specimens improved marginally (35% increase) as compared to untreated specimens, indicating the inefficacy of a low dosage for treatment.
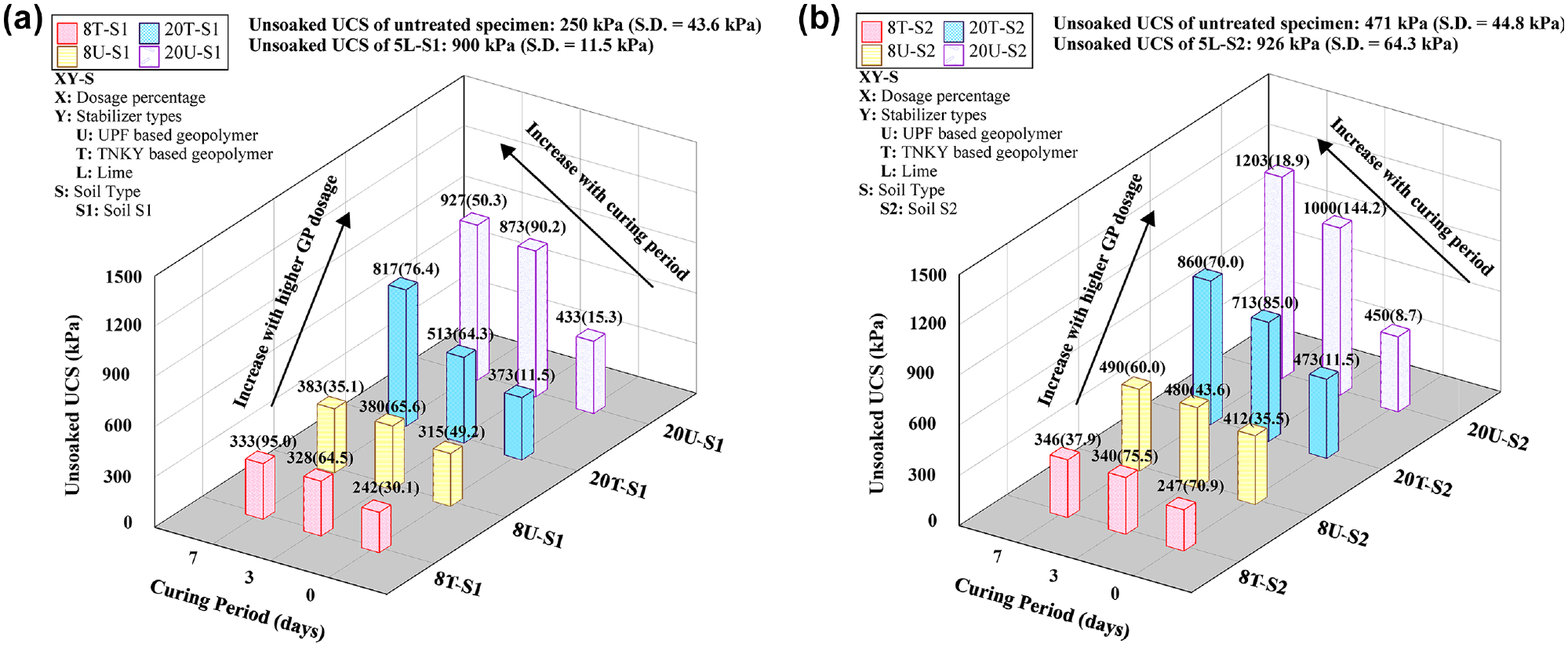
Unconfined compressive strength before capillary soaking results: (a) soil S1 group and (b) soil S2 group.
A similar trend was also observed in the S2 soil group (Figure 4b), where the UCS values increased with curing periods and treatment dosages. The UCS values of the untreated soil S2 and lime-treated soil were noted as 472 kPa (68.5 psi) and 926 kPa (134.3 psi), respectively. Both 20T-S2 and 20U-S2 did not exhibit any improvement after a 6-h curing period. However, after 7 days, the UCS of 20T-S2 revealed an approximate 82% increase compared to the untreated specimens. As for 20U-S2, a UCS of 1000 kPa (145.0 psi) was observed at 3 days, which further increased to 1200 kPa (174.0 psi) at 7 days. Similar to S1, 8% treatment showed no significant improvement in compressive strength. Sample 8T-S2 consistently showed lower strength compared to the untreated soil at all curing times and 8U-S2 exhibited relatively similar strength to the natural soil after 7 days of curing.
The stress–strain curves of unsoaked UCS tests for 7-day cured specimens indicated that the elastic modulus (initial tangent stiffness) of the treated specimens was higher than that of the natural soils (Figure 5). The elastic modulus of untreated soil S1 was measured at 35 MPa (5.1 kips per square inch [ksi]), which, after GP treatment, increased approximately six times to 224 MPa (32.5 ksi), 241 MPa (35.0 ksi), and 238 MPa (34.5 ksi) for the 20U-S1, 20T-S1, and 5L-S1 specimens, respectively, compared to the untreated soil. This was caused by the geopolymerization reactions, which effectively bind the soil particles inside the GP matrix and help to improve the compressive strength and elastic moduli values, resulting in similar or higher improvement in performance compared to the Ca-based treatment. Some of these reasons are verified with the help of microstructural analyses in the subsequent sections.
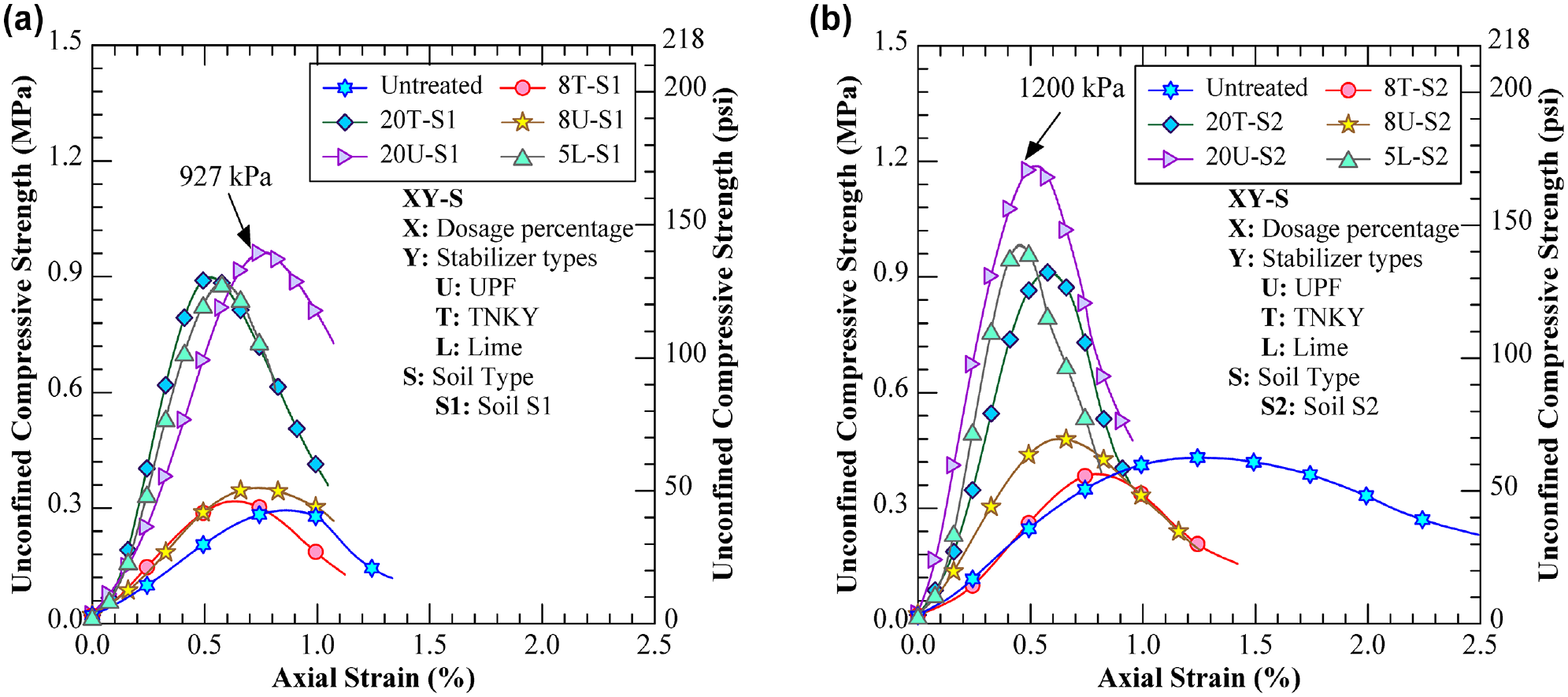
Stress–strain relationship of unsoaked specimens: (a) soil S1 group and (b) soil S2 group.
The activated UPF GP-treated specimens exhibited relatively higher strength as compared to the activated TNKY GP-treated groups. Furthermore, the UPF specimens attained the maximum strength rapidly, within 3 days, while the TNKY specimens continued to increase in strength over the 7-day curing period. This difference could be attributed to the higher kaolinite and lower quartz contents in UPF calcined clay compared to TNKY calcined clay. In the geopolymerization process, the presence of aluminosilicate sources is critical for the formation of cementitious materials. Kaolinite, with its composition of silica tetrahedral and alumina octahedral sheets, could serve as an aluminosilicate source. This enables kaolinite to actively participate in the polymerization process and contribute to the formation of cementitious materials, unlike quartz, which is a less reactive mineral. Therefore, UPF calcined clay allows for the formation of more gel-like cementitious materials, resulting in better binding of soil particles and higher strength.
It was also interesting to note that the S2 group illustrated higher strength than the S1 group for both untreated and treated specimens. This difference was attributed to the soils’ chemical composition, as XRD results showed that soil S2 was composed of a higher percentage of calcite minerals as compared to soil S1. The presence of an alkali source in the GP has the potential to dissolve calcite in natural soil to form secondary Ca(OH)2, which could eventually promote the precipitation of cementitious binding phases through pozzolanic reactions (Equation 1). The dissolution of calcite was also verified using an XRD test, as discussed in the subsequent section. As a result, soil S2 exhibited higher compressive strength as compared to S1 for all curing periods.
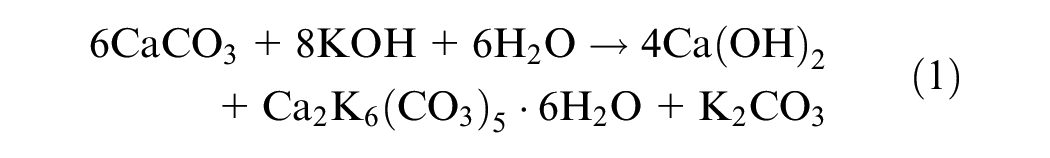
Results from UCS tests conducted on 48-h capillary soaked specimens to assess the improvement in durability of GP-treated specimens are shown in Figure 6. The strength retention parameter (RS), as presented in Equation 2, was calculated to indirectly evaluate the durability of GP-treated soils after exposure to moisture, in comparison to lime-treated soils:
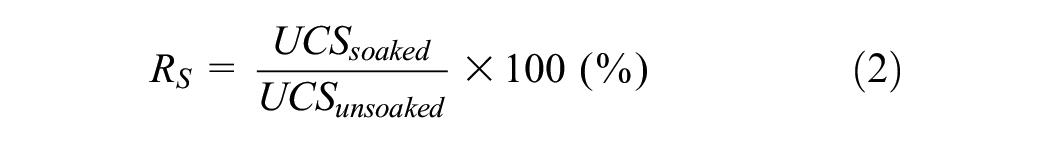
where UCSunsoaked represents the strength values of the specimens before capillary soaking and UCSsoaked represents the strength values of the specimens after capillary soaking for 48 h.
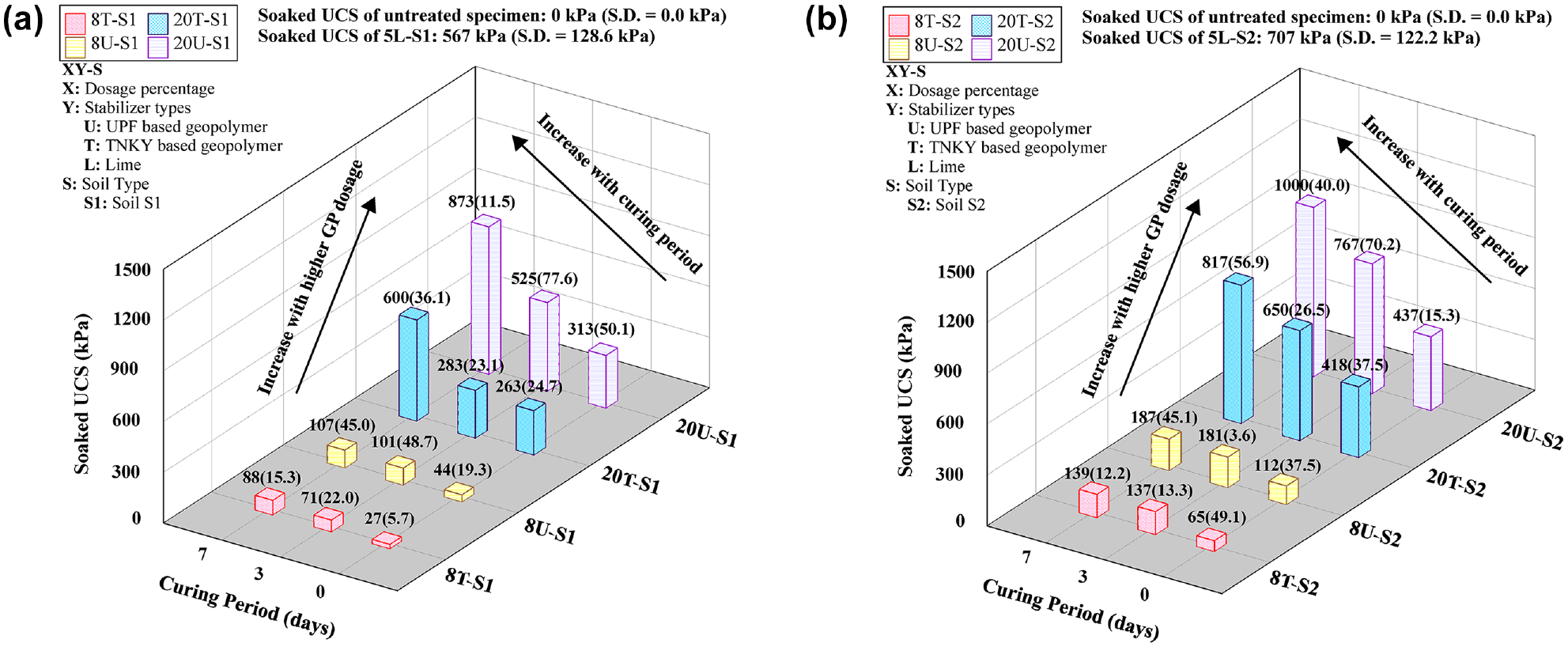
Unconfined compressive strength results after 48 h of capillary soaking: (a) soil S1 group and (b) soil S2 group.
The strength of both natural soils plummeted, and the specimens collapsed after the test, indicating the soils were poor in durability. Absorption of water by the clay minerals during capillary soaking resulted in volumetric swelling (>20%) caused by the formation of diffused double layers around clay particles. The repulsive forces exerted by water molecules on clay particles, coupled with a reduction in bulk density, contributed to the loss of strength. Treated specimens also underwent a reduction in strength caused by moisture intrusion during capillary soaking, from the weakening of bonds; however, treated specimens performed significantly better compared to natural soils after moisture conditioning. Similar to the unsoaked UCS, it could be noticed that the soaked UCS also increased with the curing period and treated dosages. Similar to unsoaked UCS values, after moisture conditioning, significant retention of strength was observed in the GP-treated specimens only beyond 3 days of curing. This indicated that GP required a sufficient amount of time to form both physical and chemical bonds, which increased over time, resulting in the enhancement of compressive strength and mitigation of the detrimental impacts from moisture intrusion.
The strength retention factor (Rs) of all specimens for various curing periods is illustrated in Figure 7. A higher dosage (20%) of GP provided better resistance to moisture intrusion as compared to an 8% dosage. At a lower dosage, there is insufficient GP availability and, subsequently, the geopolymerization fails to uniformly bind the soil particles together. However, for the higher dosage, the uniform coating of soil particles and the formation of strong bonds could prevent moisture intrusion into the soil, resulting in higher strength retention. The morphological changes with different dosage percentages were verified using an optical microscope in the later sections. For instance, after soaking, 8U-S2 retained about 32% of the unsoaked UCS for a 7-day curing period. However, 20U-S2 exhibited approximately 83% of the unsoaked UCS. Consequently, the results indicate that the UCS of GP-treated soils improves with longer curing periods and higher GP dosages. The 20% treatment of both GP types demonstrated a significant increase in strength, highlighting the effectiveness of GP stabilization in enhancing the mechanical strength properties of the soil.
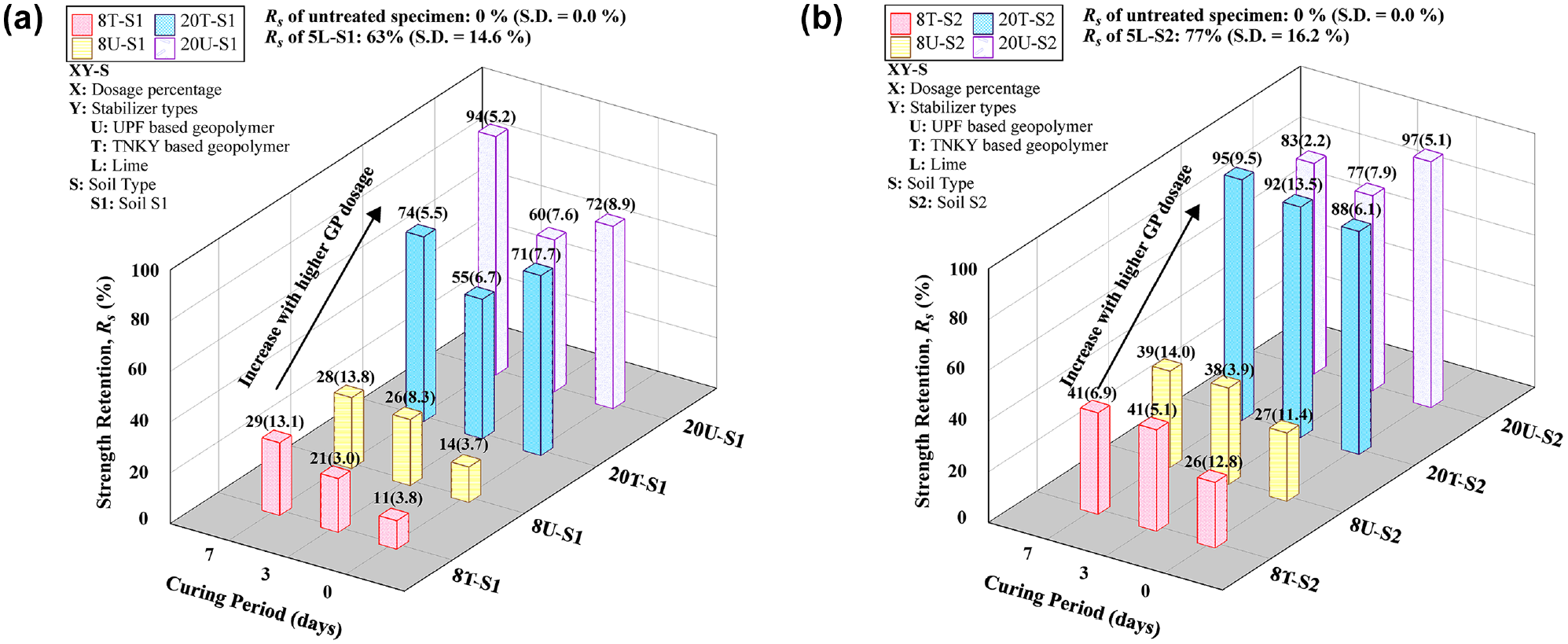
Strength retention, Rs, after capillary soaking: (a) soil S1 group and (b) soil S2 group.
Stiffness Properties
To evaluate the influence of GP on soil stiffness property, resilient moduli values of various soil groups were modeled using a universal model, which is recommended by NCHRP project 1-37A for pavement design (Equation 3):
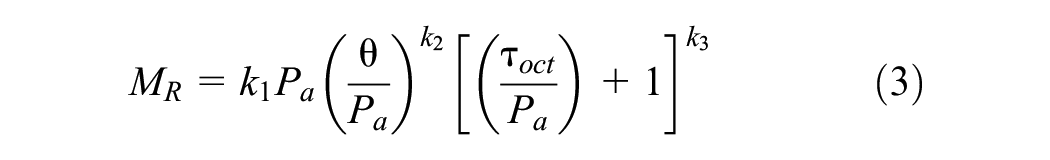
where MR is the resilient modulus (MPa), θ is bulk stress (kPa), Pa is atmospheric pressure (kPa), τoct is octahedral shear stress (kPa), and k1, k2, k3 are model constants. Here, k1 is directly related to the elastic modulus of the specimens, whereas k2 reflects the impact of bulk stress on moduli values and k3 is influenced by the deviatoric stresses on the specimens.
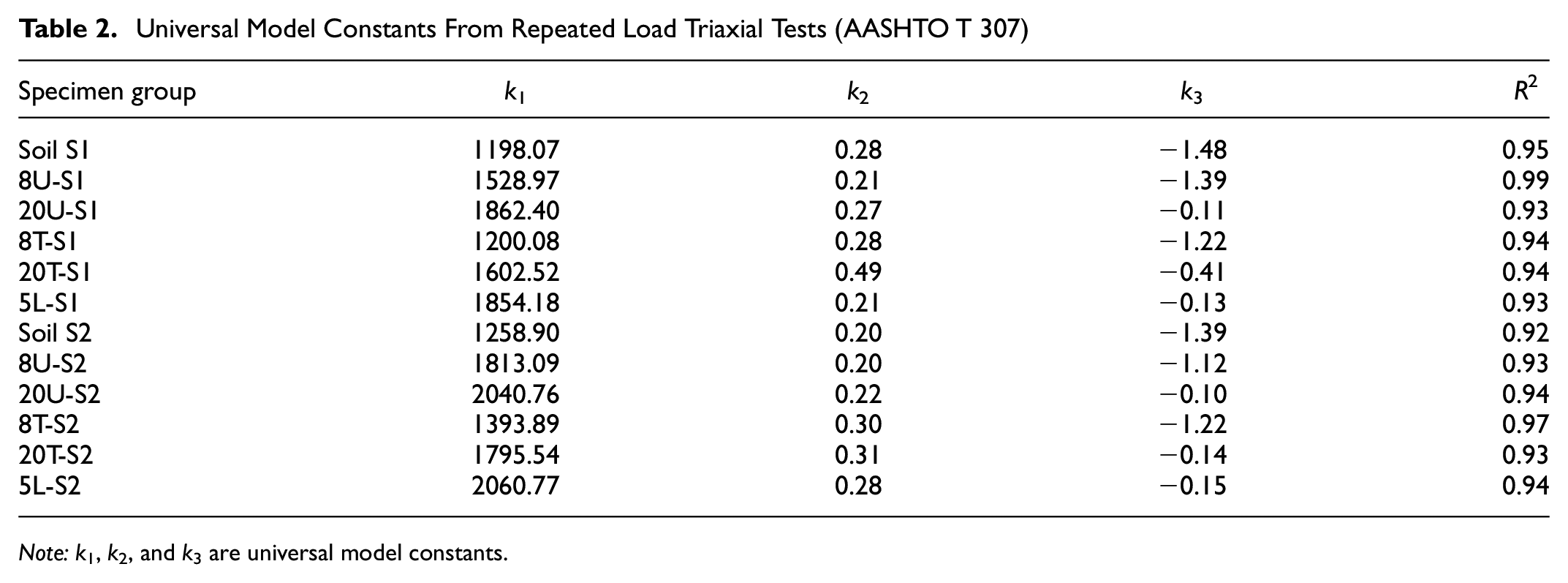
Table 2 presents the universal model constants for both the untreated and treated soil groups under unsoaked conditions. The negative value of k3 indicates the softening behavior as exhibited by the untreated soil S1 and S2 when subjected to increasing deviatoric stresses. This suggests that these soils have low shear strength and are prone to failure under repeated traffic loading. The application of GP stabilization resulted in an improvement in elastic moduli, as evidenced by the increased values of k1. While the improvement in k1 values for 8T-S1 and 8T-S2 compared to the untreated specimens was not significant, a substantial increase of over 80% was observed for 20U-S1 and 20U-S2, as well as approximately 60% for 20T-S1 and 20T-S2. Notably, the k1 values for 20U-S1 and 20U-S2 were found to be comparable to those of 5L-S1 and 5L-S2, respectively. This indicates that the chemical and physical bonding between the soil particles and GP played a crucial role in enhancing the stiffness of the treated specimens. These findings underscore the potential of GP as a viable alternative to lime stabilizers for improving the stiffness of subgrade soils. Furthermore, the treated specimens exhibited higher k3 values, indicating a transition from softening to hardening behavior in response to deviatoric stresses. However, no noticeable trend was observed in the k2 parameter for either untreated or treated specimen groups.
Universal Model Constants From Repeated Load Triaxial Tests (AASHTO T 307)
Note: k 1, k2, and k3 are universal model constants.
Swelling and Shrinkage Strains
The natural soils exhibited significant free swell percentages of 18% and 11% for soil groups S1 and S2, respectively (Figure 8). This swelling phenomenon can be attributed to the formation of diffused double layers around clay particles when water infiltrates the soil. The majority of the swelling occurred within the initial day of testing. After lime stabilization, the free swelling strain was reduced significantly to 1.2%, which was caused by the formation of cementitious materials to bind soil particles together and prevent moisture intrusion. Similar to lime treatment, GP also effectively mitigated the free swelling. The 8% GP-treated specimens recorded a final free swell strain of less than 2%, while the 20% treatment resulted in a reduction to below 1%. This significant reduction can be attributed to the polymerization process within the GP solution. This mechanism led to the formation of a solid gel-like material that effectively bound the soil particles together, consequently reducing their potential for water absorption and subsequent swelling. Overall, it was observed that 20% GP treatment was more effective than lime stabilization for both soils and GP types.
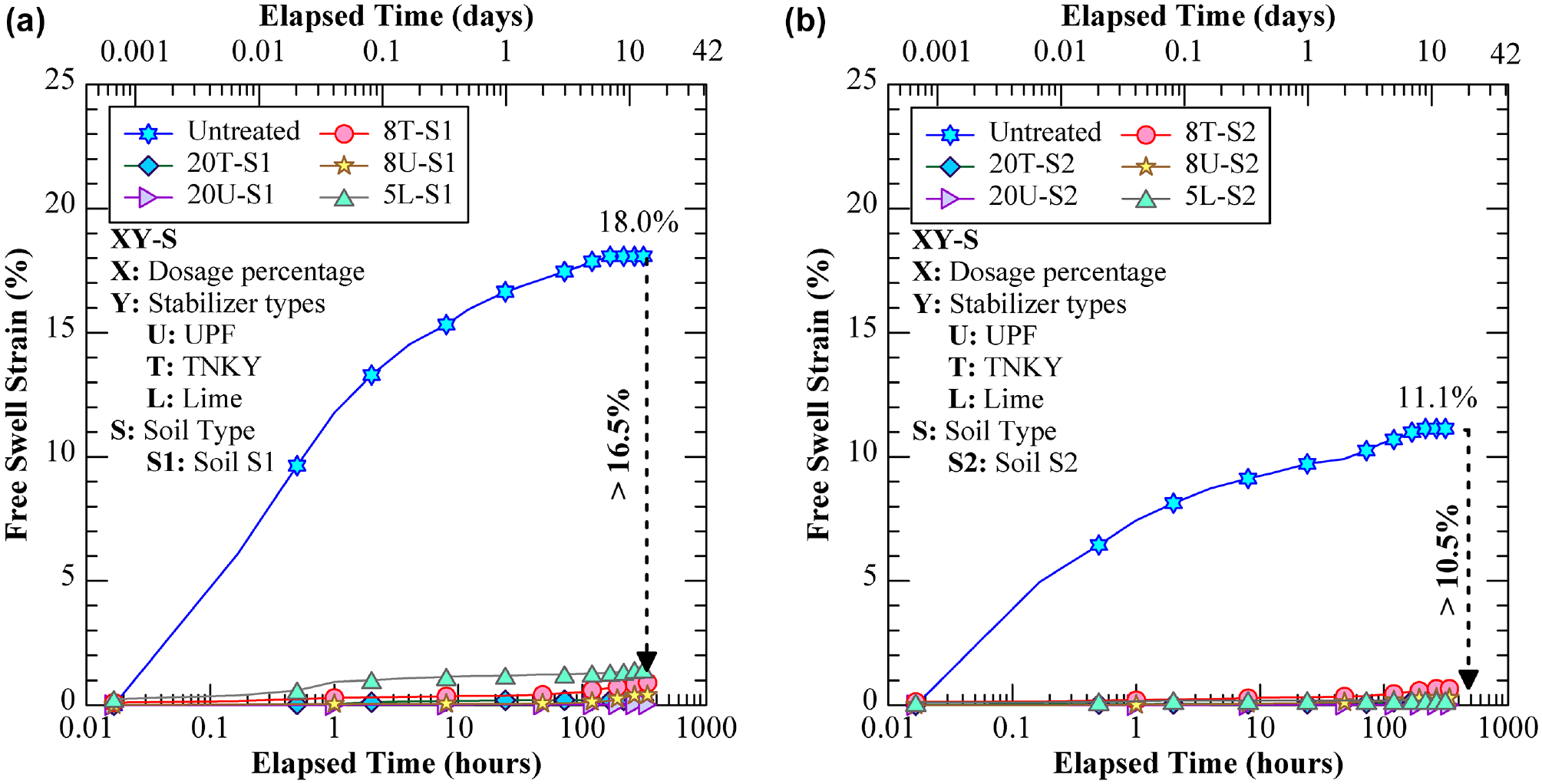
Vertical free swell strain values for untreated and treated specimens: (a) soil S1 group and (b) soil S2 group.
Figure 9 illustrates the shrinkage strain values of both untreated and treated specimens. Linear shrinkage strains of 10.3% and 9.0% were observed in the natural soil S1 and S2, respectively. Significant reductions in the linear shrinkage strain were observed following chemical stabilization. This notable decrease can be attributed to the solid gel-like material formed as a result of the geopolymerization process. This material effectively binds the soil particles together, causing them to agglomerate and leading to changes in texture, plasticity, and reduced water-holding characteristics. For example, specimens 8T-S1 and 8U-S1 exhibited a linear shrinkage of 3%, which was comparable to the 5L-S1 specimen. Moreover, specimens 20T-S1 and 20U-S1 showed no noticeable shrinkage after a 7-day curing period. This can be attributed to the formation of a fully formed network of GP gels that uniformly coated the soil particles. Therefore, from the above discussion, a higher GP dosage demonstrates proficiency in reducing soil instability caused by swelling and shrinkage during seasonal moisture ingress and egress. Overall, the engineering tests showed the efficacy of GP in enhancing the performance of expansive soils. The next section discusses the results from select microstructural and mineralogical studies to support some of the observations noted during the engineering tests.
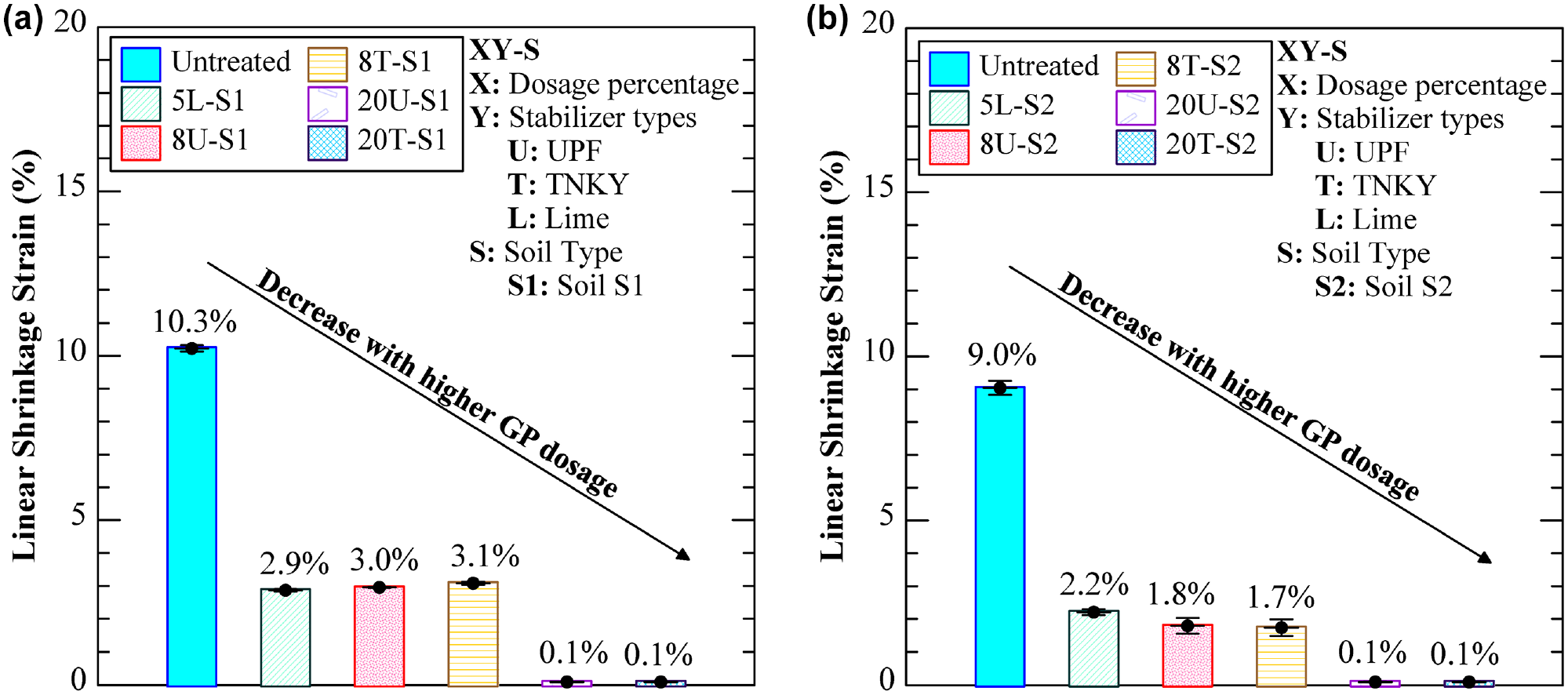
Linear shrinkage strain for untreated and treated specimens: (a) soil S1 group and (b) soil S2 group.
Microstructural and Mineralogical Characterization
To investigate the chemical reaction behavior between TNKY-based GP and natural soils, XRD tests were conducted on representative specimens of untreated and 20% TNKY GP-treated soil S2. As both GPs have kaolin as a key mineral it is expected to show similar behavior for soil treatment. Similarly, except for calcite concentration, the chemical composition of both soils is also similar, and therefore both are expected to show similar behavior on GP treatment. The diffractogram of the untreated specimen (S2) indicated the presence of illite, calcite, and quartz as major minerals in natural state (Figure 10a). After 20% GP treatment and 7 days of curing, no additional peak was detected from the GP because of the inherent amorphous nature of GP gels. However, the intensity of each natural mineral peak in the 20T-S2 specimen significantly reduced, particularly for the calcite mineral (from 39,500 to 14,000 cps; Figure 10b). This result suggested that at a high pH environment (pH > 13.0) from the GP stabilizer promoted a chemical reaction between calcite and KOH, as described in Equation 1. Parallelly, the dissolution of SiO2 and Al2O3 present in clay minerals and partial destruction of the silicate mineral structure from quartz potentially reacted with Ca2+, resulting in the formation of additional cementitious materials, such as calcium-silicate hydrate and calcium-aluminate hydrate ( 40 – 42 ). This supports the observation that soil S2, which has a higher calcite content (62.8%), exhibited greater compressive strength and stiffness compared to soil S1 in all tested combinations (Figures 4 and 6).
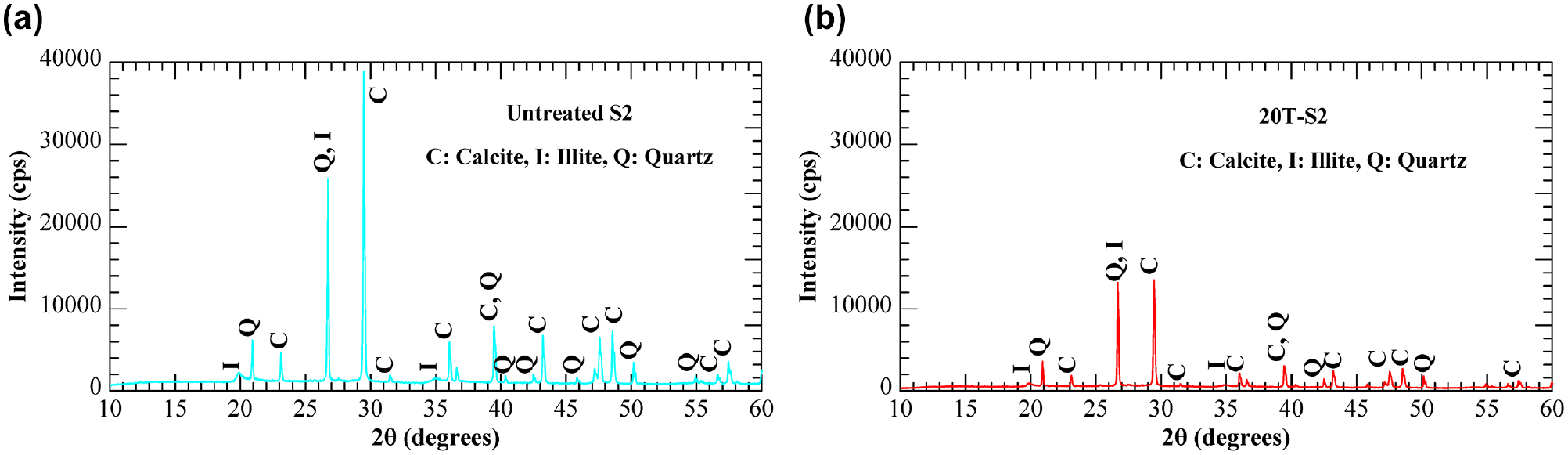
X-ray diffraction results of different specimens: (a) untreated (S2) and (b) 20T-S2.
OM was performed to investigate the macro-level distribution of different dosage of TNKY-GP within soil specimen S2, as shown in Figure 11. The untreated soil exhibited a highly heterogeneous morphology with minerals of various colors (Figure 11a). After treating with 8% GP, a noticeable non-uniform GP coating (distinct GP granules) was observed on the soil surface because this coating led to a reduction in the heterogeneous coloration of the natural soil minerals (Figure 11b). However, the soil treated with 20% GP dosage displayed a distinct layer of GP, with no distinct granules withing the observed matrix (Figure 11c). This showed that at higher GP dosage, the untreated soils were more uniformly coated with the 20% GP dosage as compared to the 8% GP dosage. The uniformly coated soil matrix resulted in enhanced compressive strength and stiffness, and reduction in moisture-induced strains (Figures 4–9). As a higher dosage of GP was found to be effective as compared to 8% treatment, to detect the precipitated GP gels in a higher resolution for 20% TNKY-treated S2, FESEM image analysis was performed and analyzed as follows.

Optical microscope images for different specimens: (a) untreated (14×), (b) 8T-S2 (14×), and (c) 20T-S2 (14×).
Figure 12, a and b, illustrates the untreated specimen, which primarily exhibited open pore structures with flaky particles. However, after curing for 7 days, the GP-treated specimens (20T-S2) displayed a remarkably different morphology. The clay particles were observed to be coated with a layer of GP matrix, resulting in reduced macro pores and a smoother surface (Figure 12d). These coatings provided physical and chemical bonding, effectively holding the loose clay particles together and contributing to a significant improvement in engineering properties. Therefore, this finding suggests that higher dosages of GP provided more cementitious material to bind the soil particles together, resulting in increased strength and reduced swelling–shrinkage strain (Figures 4–9). Overall, the microstructural studies provide enough evidence to validate the observed engineering performance and show the benefits of the locally available calcined clay-based GPs in effectively stabilizing the problematic clayey soils available in Region-6.

Field emission scanning electron microscopy images at different magnifications: (a) untreated (500×), (b) untreated (5000×), (c) 20T-S2 (500×), and (d) 20T-S2 (5000×).
Summary and Conclusions
A research study was conducted to investigate the effectiveness of local calcined clay-based GP in enhancing soil engineering properties. Two clayey soils from Region-6 were treated with two calcined clay-based GP (UPF and TNKY) to evaluate the improvements in strength, durability, stiffness, swelling, linear shrinkage, and microstructural characteristics. Based on the experimental results, some major findings are presented as follows.
The 20% GP treatment significantly improved UCS before and after capillary soaking, performing better than the 8% GP dosage or 5% lime treatment. Moreover, the activated UPF-based GP showed better results compared to the activated TNKY-based GP, primarily because of the higher composition of kaolinite and lower quartz. Soil S2, which consisted of a higher percentage of calcite minerals, exhibited greater strength than soil S1. This was attributed to the high alkaline environment from GP treatment, which resulted in the production of more cementitious materials.
The resilient moduli of the treated soils exhibited substantial improvements when compared to the untreated specimens. The specimens subjected to a 20% GP treatment demonstrated a hardening behavior, whereas the untreated and 8% treated specimens illustrated softening behaviors, indicating that the 20% GP dosage was effective in enhancing the shear strength of the soil. Notably, the performance of 20U-S1 and 20U-S2 was comparable to that of 5L-S1 and 5L-S2, respectively.
Untreated soils S1 and S2 exhibited significant moisture-induced strains. However, after GP treatment, both the swell and shrinkage behaviors were significantly reduced. The 20% GP dosages helped in maximum reduction in moisture susceptibility as compared to the 8% GP-treated or 5% lime-treated soils. This indicated the higher efficacy of GP as a stabilizer for high swell–shrink soils, as compared to traditional calcium-based stabilizers.
XRD studies showed that the reduction in intensities of calcite, quartz, and other clay mineral peaks helped in the formation of cementitious binding gels with the geopolymerization reactions that imparted additional benefits to the S2 soil groups as compared to S1 soil. Optical microcopy showed untreated soils had a polychromatic heterogeneous morphology, and 8% GP treatment fails to develop a uniformly coated surface on soils when compared to 20% treatment. Scanning electron microscopy results illustrated untreated specimens with open pore structures with flaky particles. After stabilization, the clay particles were observed to be coated with a layer of GP matrix, resulting in reduced macro pores and a smoother surface, which helped in enhancing the strength, stiffness, and moisture-induced strains.
Overall, this research provided significant insights into the potential use of locally sourced calcined clay-based GP as a solution for transportation infrastructures. These findings are expected to contribute to the development of sustainable stabilization guidelines and benefit transportation infrastructure practitioners by minimizing the costs of materials and construction. However, future investigations are needed to examine its long-term performance and sustainability across various soil types. A life-cycle sustainability assessment should be carried out to provide users with the necessary information for implementing this solution. In addition, it is crucial to conduct pavement test sections using GP-treated soils in field implementations. This will help bridge the gap between laboratory data and practical applications, ensuring a comprehensive understanding of the GP’s effectiveness and viability in transportation infrastructure projects.
Footnotes
Author Contributions
The authors confirm contribution to the paper as follows: study conception and design: N. Biswas, A.J. Puppala, S. Chou, O. Huang, M. Radovic; data collection: S. Chou, O. Huang; analysis and interpretation of results: S. Chou, N. Biswas, O. Huang, A.J. Puppala, M. Radovic; draft manuscript preparation: S. Chou, N. Biswas, A.J. Puppala, O. Huang, M. Radovic. All authors reviewed the results and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Transportation Consortium of South-Central States (Tran-SET)-Region 6’s University Transportation Center (Award #22GTTAMU40), the NSF Industry-University Cooperative Research Center (I/UCRC) program at the Center for Integration of Composites into Infrastructure (CICI) at Texas A&M University, NSF PD: Dr. Prakash Balan; Award #2017796.
