Abstract
Amplitudes of contingent negative variation (CNV) in 40 migraine patients were studied during the pain-free period and compared with healthy controls (n = 24). The early component (initial CNV) is especially more negative in migraine patients. Within the migraine group, 14 children with migraine without aura (mean age 13.6 years) were compared with 11 healthy children (mean age 12.4 years). Migraine children differ from healthy children in the same manner that adult migraine patients differ from healthy adults. The CNV of five healthy children did not differ from the CNV of their siblings suffering from migraine. We suggest that there is a family-related cortical hypersensitivity which does not necessarily lead to the development of migraine.
There are some neurophysiological abnormalities in adult migraineurs. When recording evoked potentials, these patients produce a pronounced stimulus sensitivity compared with healthy controls, even during the interictal interval (1, 2). The contingent negative variation (CNV), a slow event-related cortical potential recorded between a warning and an imperative stimulus, shows pronounced negative amplitudes and reduced habituation characteristics. Although these differ from normal during the interictal interval, there are no differences from normal during a migraine attack (3). CNV negativity rises to a maximum the day before the next attack, rendering a predictive value for the next oncoming migraine attack (4). During the interictal period, both adults and children suffering from migraine produce more pronounced CNV negativity than healthy controls. The most significant difference appears in the early CNV component (initial CNV, iCNV). iCNVis defined as the maximum amplitude between 550 and 750 ms after presentation of the warning stimulus (S1) and can be used as an indicator of expectancy and attention, modulated by the noradrenergic system. High initial amplitudes in adult migraine patients reflect a distinct cognitive expectancy and resource mobilization, which is associated with both an inherited cortical hyperactivity and the avoidance of aversive failure (5). In addition, CNV in migraine patients exhibits less habituation compared with healthy controls. This inherent mechanism of habituation protects the cortex against overstimulation. Reduced habituation in migraine patients refers to information-attention dysfunction between two migraine attacks (3). Besken et al. (6) recorded a higher mean CNV in children suffering from migraine compared with those suffering from tension-type headache and healthy controls. The authors explained this negativity on the basis of a retarded habituation in migraine children during the CNV recording session. Eversetal. (7) recorded a highly significant loss of cortical habituation in migraine children recorded by a P300 oddball paradigm and they characterize the underlying process as highly specific for migraine. They conclude that this specific cognitive processing seems to develop during adolescence. There is some evidence that children and adult migraine patients reveal a cortical hypersensitivity which may be shaped by social learning processes (5). This implies that a psychological stress reaction as well as a biological predisposition may be necessary to trigger a migraine attack. What about family members not suffering from migraine? Is there any evidence of a striking CNV amplitude? The aim of the present study was to obtain CNV data from family members concerning their migraine disposition and to elucidate possible changes in cortical processing.
Methods
Patients and subjects
Forty migraine patients and 24 healthy subjects were recruited. Headache diagnosis was made in accordance with the criteria of the International Headache Society (IHS, 8). All patients fulfilled the criteria of migraine without aura. In the migraine group, 14 children (mean age 13.6 years) and 26 adults (mean age 41.5 years) took part in the study. The healthy subjects were 11 children (mean age 12.4 years) and 13 adults (mean age47.8 years). In addition, in a later analysis of the subgroup of healthy children, five siblings of the migraine children (mean age 12.7 years) not suffering from migraine were examined (Table 1). These siblings had no experience of migraine or any other head pain.
Demographic data of all subjects. Figures in parentheses: standard deviation. Intensity was rated by a visual analogue scale from “1” (weakest pain) to “9” (strongest pain).

Procedure
After receiving informed consent from the parents, CNV recordings of all children and other subjects were carried out. In the case of migraine patients care was taken not to record CNV 2 days after an attack, because CNV amplitudes differ significantly within this period (4). If a migraine attack occurred in the 2 days following the examination (information obtained by a telephone call) the CNV record was not analyzed. Patients were not on prophylactic treatment for migraine, but were allowed to treat the attacks with medication.
A CNV session consisted of 32 trials in which the patients and the healthy subjects reacted to the target stimulus (GO response). Additionally, eight trials were randomly presented where no reaction was expected (NO-GO responses). The warning stimulus, S1, for the GO response had a frequency of 1,000 Hz and lasted 100 ms with an intensity of 75 dB(a). The S1 for the NO-GO response had a frequency of 200 Hz. The target stimulus S2 had a frequency of 2500 Hz, lasted a maximum of 1500 ms, and was terminated by pressing a key. All subjects were instructed to push the key as quickly as possible. Reaction time was defined as the period between the onset of S2 and pressing of the key. CNV was recorded over Cz with linked mastoids as reference. The electrode resistance was approximately 6 kOhms. The EEG was amplified with a bandpass of 0.03 to 35 Hz and digitized with a sampling rate of 100 Hz for each channel. The length of one recording was 6 s, with a randomized interval of 6 to 10 s between trials. Recording began 1 s before S1 and ended 2 s following the onset of S2. The interstimulus interval between S1 and S2 lasted t = 3 s. NO-GO trials were not analysed. The vertical electrooculogram (EOG) was recorded, too. CNV recordings containing eye blinks or artefacts were discarded. A protocol listed the number of rejected trials for each session. The period between recording onset and S1 was taken as the baseline for all measurements. The CNV session was conducted after 40 artefact-free trials (32 GO/8 NO-GO trials). The grand average of 32 GO trials of each CNV session for all patients was obtained. Total CNV amplitude, early component, late component, and reaction time were determined for each session. Total CNV amplitude was the mean amplitude between S1 and S2. The early component was calculated according to procedures developed by Böcker et al. (9): The maximum negative amplitude of all 32 GO trials between 550 ms and 750 ms after S1 was calculated and the latency of this maximum was used as the midpoint of a window of 200 ms duration. The mean amplitude of this individual window was defined as the early component. The late CNV amplitude was the mean amplitude during the 200 ms preceding S2. All statistical tests were performed with non-parametric Mann-Witney tests.
Results
Children vs adults
There were no significant differences in CNV parameters between migraine children and migraine adults (Table 2). The comparison of healthy children and healthy adults also revealed no statistical differences. The comparison between migraine children and healthy children showed significant differences in iCNV, with migraine children producing more negative amplitudes. The same results were seen in the comparison between adult migraineurs and healthy adults (Figs 1, 2).

Comparison of CNV parameters between migraine children and healthy children (*p < 0.05).
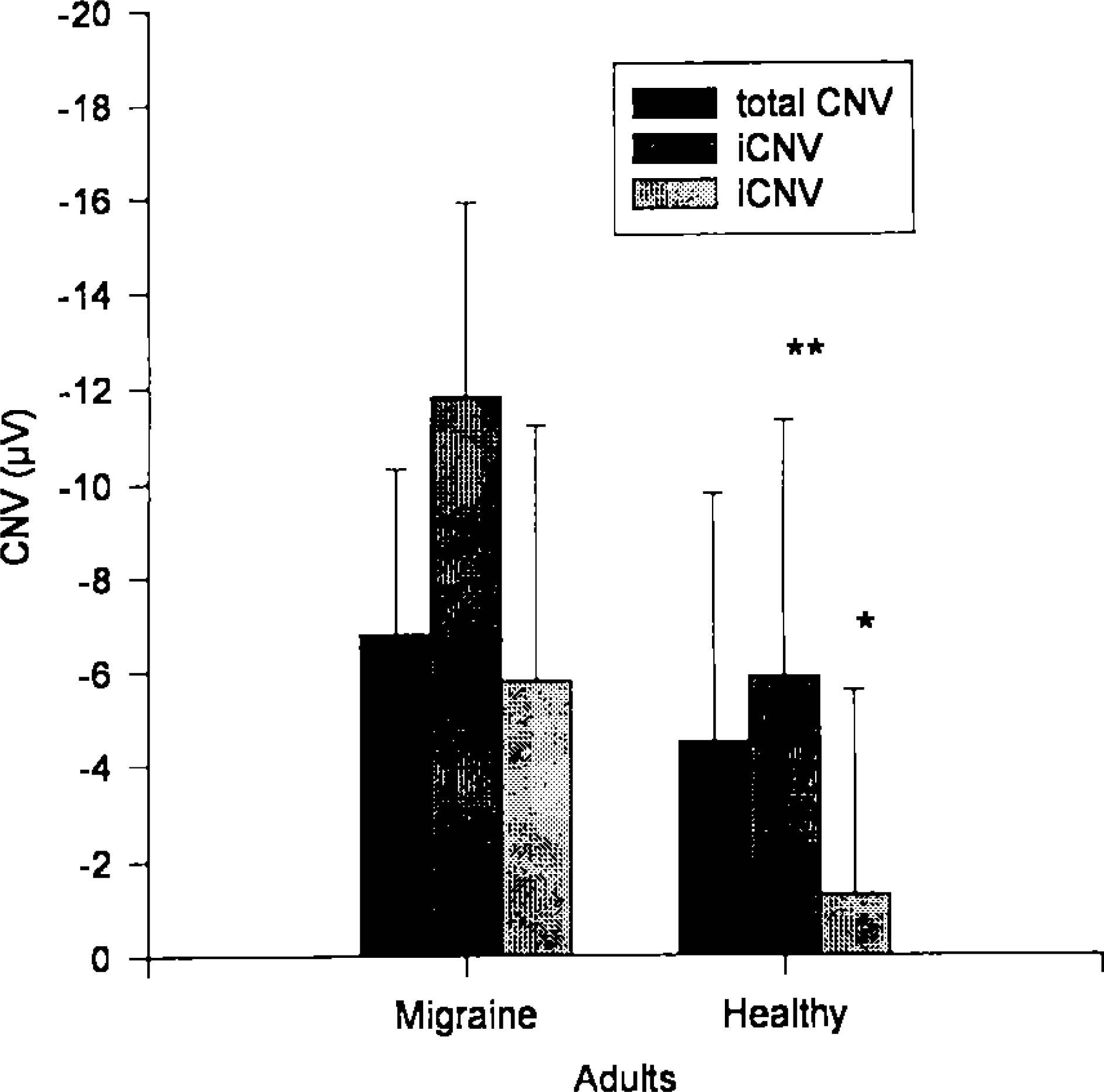
Comparison of CNV parameters between migraine adults and healthy adults (*p < 0.05; **p < 0.01).
Averaged CNV amplitudes in uV of 32 GO trials and the overall reaction time (rt) in ms for all migraine and control groups. Figures in parentheses: standard deviation. Statistics were performed with Mann-Whitney U-tests.

Migraine children vs healthy brothers and sisters
In a subanalysis of 11 healthy controls, brothers and sisters of migraine children (n = 5) were observed who had not yet had experience of headache. Table 3 gives comparisons between migraine children (mc), healthy controls (hc), and healthy siblings of migraine children (bsm). There was a tendency for age in healthy controls to be lower than in the other two groups (p < 0.1). With this small number of observations, there were no statistical differences in CNV amplitudes between migraine children and their healthy brothers and sisters. The latter produced the same pronounced negativity as their migraine brothers and sisters (Fig. 3). Healthy children differed significantly in their iCNV from migraine children but not from the healthy siblings of migraine children. In this case, a difference could be observed in the total CNV, where healthy children produced less negative amplitudes.

CNV parameters of migraine children, healthy children, and healthy brothers and sisters of migraine children. According to Table 3, there were no significant differences between migraine children and their brothers and sisters (*p < 0.05).
Averaged CNV amplitudes in uV and reaction time in ms of 32 GO trials for migraine children (mc), healthy children (hc), and healthy brothers and sisters of migraine children (bsm). Figures in parentheses: standard deviation. Statistics were performed with Mann-Whitney U-tests.

Discussion
The data confirm previous findings of a more pronounced negativity in migraine patients compared with healthy controls. Even in childhood the same observation in iCNV amplitudes can be seen. Gerber and Schoenen (5) hypothesized a “possibly” genetic cortical hypersensitivity which is shaped by social learning processes. This abnormal information processing could be the structural basis on which a migraine attack might be triggered in the case of biological or psychological stress. The striking markers of a special cortical processing in migraine can be demonstrated in childhood and remain stable in the case of adult female migraine patients (7). Surprisingly, the healthy siblings of children suffering from migraine may seem to be characterized by the same abnormalities in CNV as migraineurs. This may mean that pronounced cortical negativity, which reflects the level of sensitivity and reactivity of cortical neurons and which can be recorded by CNV, may not be sufficient for the occurrence of migraine. However, this increased sensitivity and reactivity of the central nervous system could account for a possible predisposition. It is important to note that increased amplitude of iCNV is characteristic for the whole migraine family, including in part even healthy members of the family. This suggests a familial transmission of increased cortical negativity. Is cortical hypersensitivity in migraine patients determined by genetic factors, or is it a consequence of learning processes within the family? Further studies should elucidate the possible mode of inheritance for CNV characteristics on a large sample of migraine families. Why do some children develop migraine and some do not if they have the same biological predisposition? One reason why migraine does not appear inevitably in all family members may be individual variability and thresholds, as well as hormonal and social conditions of the potential “migraine” child. On the one hand, there is a possibility for healthy siblings of migraine children to develop migraine in later years. Such a development may be indicated by a pronounced negativity in CNV amplitudes before the first attack occurs. Absence of headaches in siblings could be explained by a lacking influence of different provoking agents (cortical overstimulation, chronic stress), which might cause the first migraine attack in their relatives. On the other hand, psychological and behavioral factors could predispose only some of the siblings for the development of a migraine. In a study with migraine families, it was demonstrated that parents interact differently with their migrainous child compared with his/her healthy brothers and sisters: there were significantly more verbal interruptions, activities inhibiting the child's independence, corrections, and punishments towards migraineurs (10, 11). We suggest that a maladaptive familial atmosphere can contribute to the manifestation and maintenance of the clinical course of migraine. Finally, the possibility cannot be excluded that there is a mechanism of protection against the triggering of a migraine attack lacking in a migraine child.
Hence CNV studies provide compelling evidence that the brain of children and adults suffering from migraine is characterized by hypersensitivity and hyperreactivity. This feature of the migrainous brain could be both inherited and acquired. It is obvious that the abnormal CNV amplitude may be discussed as one of numerous factors which predispose children to developing migraine. However, CNV abnormalities are not sufficient for the occurrence of this disease; other important factors, for example behavioral ones, have to be investigated as possibly contributing to the occurrence of migraine.
