Abstract
Background: Migraine is a disorder of central information processing which may be characterized by increased amplitudes and reduced habituation of evoked and event-related potentials. In this pilot study, special behavioural training of habituation to aversive stimuli (MIPAS-Family = Migraine Patient Seminar for Families) was developed and proven effective in children suffering from migraine without aura.
Methods: 13 children with migraine participated in the MIPAS-Family programme and 13 other children with migraine were treated with biofeedback. The influence of both treatments on abnormal cortical information processing in migraine was assessed using recordings of the contingent negative variation (CNV), an event-related slow cortical potential.
Results: Both MIPAS training and biofeedback caused an equal reduction of migraine frequency and severity. However, MIPAS treatment was associated with a significant increase in iCNV habituation. Changes in the clinical course of migraine correlated positively with normalization of habituation: the greater the reduction in headache frequency, the greater the increase in CNV habituation was. These effects were not observed in the biofeedback group.
Conclusions: This study demonstrates that the specific treatment programme which was evolved from knowledge of pathogenetic mechanisms of migraine influences central information processing and leads to a clinical effect.
Introduction
There is a general agreement that migraine is associated with abnormal central information processing and deficient regulation of cortical excitability (1,2). Among other methods, these abnormalities may be characterized by using the contingent negative variation (CNV) – a slow cortical, event-related potential (3). Migraine patients demostrate increased amplitudes and reduced habituation of the CNV (4–8). These abnormalities were more pronounced several days before a migraine attack, demonstrating attack anticipation and increased susceptibility of the brain to migraine provoking and precipitating factors (9–14). One may suggest that normalization of the CNV in migraine would contribute to improvement of its clinical course. Indeed, clinically effective migraine prophylactic agents such as β-blockers, calcium antagonists, and anti-epileptic drugs lead to normalization of CNV amplitude and its habituation (15–18). Moreover, direct voluntary influence on CNV during a successful self-regulation of slow cortical potentials using neurofeedback training resulted in significant reduction of CNV amplitude and an increase in its habituation (19). It can be suggested that effective behavioural treatment programmes would cause a normalization of the cortical information processing in migraine, especially in children who are typically more receptive to behavioural treatments (20–22).
For this pilot study, we developed a special behavioural training programme which focused on confrontation with aversive stimuli (telephone ring tones, unpleasant odours, flash stimulation, etc.) for the purpose of relaxation-guided habituation (23–25). We hypothesized that this special behavioural treatment could normalize amplitude and habituation of the CNV. To assess specificity of training effects, a control group of migraine patients was treated with biofeedback, the treatment of first choice in children with migraine (20–22).
Subjects and methods
Subjects
Demographic and clinical characteristics of the patient groups

n.s., non-significant.
Scores for evaluation of migraine frequency: 1, daily; 2, several times a week; 3, weekly; 4, one to three times in a month; 5, less than monthly.
Scores for assessment of migraine intensity from 1 ‘no headache’ to 10 ‘very severe headache’.
Number of patients with a combination of migraine and episodic tension-type headache.
The study was permitted by the Ethics Committee of the Faculty of Medicine, University of Kiel, Germany. All participants and their parents were instructed about the study, and written informed consent according to the Declaration of Helsinki (current version, 1996) on biomedical research involving human subjects (Tokyo amendment) was obtained.
Description of the therapy
A detailed description of the behavioural programme MIPAS-Family (Migraine Patient Seminar for Families) and the evaluation of the clinical efficacy for the whole sample is presented in Gerber et al. (23–25). In short, MIPAS-Family consists of the education of, and a behavioural treatment programme for, children (8 sessions of 90 min) as well as an education programme for parents (4 sessions of 120 min) including three modules (diagnosis, education, and behavioural therapy). The aim of the children’s training was for them to learn habituation by aversive stimuli (for a detailed description, see Gerber and Gerber-von Muller (27)) and effective coping skills for stress and pain (28). The habituation was learned using confrontation with aversive stimuli (telephone ring tones, unpleasant odours, flash stimulation, etc.). The children learned to attenuate their perception of aversive stimuli and to control and reduce unpleasant autonomic sensations during stimulation. The confrontation with stimuli was repeated until the child said that he/she was no longer annoyed by the stimulus. The aim of the parent’s education was to teach parents how to perform MIPAS training at home. Previous studies have demonstrated a substantial influence of parent–child interactions and parental behaviour on the clinical course of paediatric migraine (see, for example, Siniatchkin et al. (29)). Therefore, integration of parents into treatment programmes of children with migraine may increase efficacy of therapeutic interventions.
The biofeedback training was based on well-established procedures (for a detailed description see elsewhere (30,31)) and carried out using a routine biofeedback system Soft®med device (Insight Instruments, Vienna, Austria). The training aimed at improving the perception of physiological processes and control over body functions in order to learn how to relax the body as well as new relaxation-based strategies to cope with stress. The biofeedback was performed simultaneously using muscle activity (electromyography feedback from the frontalis muscle) and skin temperature (thermal feedback from the right hand). The training lasted 50 min and took place once a week. The children were rewarded for success of self-regulation.
Task
All participants were seated with eyes open in an armchair located in an electrically shielded, sound-proof room. The subjects were asked not to close, move, or blink their eyes. All stimuli were presented using E-prime (Psychology Software Co., USA). The auditory warning (S1) and imperative stimulus (S2) were produced by a loudspeaker located behind the subject with an intensity of 75 dB. The interval between S1 and S2 was 3 s. A CNV session consisted of 60 trials in which the subject was to react immediately to the imperative stimulus. The warning stimulus (S1) had a frequency of 1000 Hz and lasted 100 ms. The imperative stimulus (S2) had a frequency of 2500 Hz, lasted a maximum of 1500 ms and was de-activated by pressing a button with the dominant right hand. Reaction time was defined as the period between the onset of S2 and the pressing of the button. S1 and S2 pairs were offered at random intervals of 10–15 s. The duration of the recorded epoch was 6 s (the recording began 1 s before S1 and ended 2 s after S2). The period between recording onset and S1 was taken as the baseline for all measurements.
Recordings
The EEG was continuously recorded from 30 scalp sites (10–20 system plus FC1, FC2, CP1, CP2, FC5, FC6, CP5, CP6, TP9, TP10) with a reference located between Fz and Cz. Sintered Ag/AgCl ring electrodes were attached using the ‘BrainCap’ (Falk-Minow Services, Herrsching-Breitbrunn, Germany) which is the part of the EEG recording system ‘BrainAmp-MR’ (Brainproducts Co., Munich, Germany). Electrode impedance was kept below 5 kOhms. Data were transmitted from the high-input impedance amplifier (5000 Hz sampling rate, 250 Hz low-pass filter, 10 s time constant, 16-bit resolution, dynamic range 16–38 mV) to a computer via a fibre-optic cable. Vertical and horizontal eye movement artefacts were excluded via parallel recording of the electrooculogram (EOG) using electrodes positioned 5 cm above and below the right eye (VEOG) as well as next to the outer canthi (HEOG).
Data processing
After recording, the data were transformed to an averaged reference. Then, EEG signals were digitally filtered (30 Hz high cut-off) and segmented into epochs of 6 s. They were additionally corrected automatically for DC-drift by subtraction of a linear function calculated by regression analysis over the whole segment and corrected for eye movements and blinks using the algorithm described by Gratton et al. (32) Artefacts were rejected automatically if the signal amplitude exceeded 150 µV. This procedure was confirmed by visual inspection and any remaining smaller artefacts were removed. Overall, no more than 20 trials were removed in each of the patients and in each condition. In order to achieve consistency of analysed trials, only 40 artefact-free trials were included in the further analysis. All steps of off-line processing were performed using BrainVision Analyser v2.0 (Brainproducts Co.).
The trials were averaged and the amplitudes of the iCNV and lCNV components as well as the PINV were calculated. The iCNV was defined as the mean amplitude in a window of 200 ms around the maximal amplitude of the expectancy wave between 550–750 ms after S1 (4). The lCNV was the mean amplitude during the 200 ms preceding S2. PINV was the mean amplitude of CNV between 500–2000 ms following S2. Each recording was divided into four blocks of 10 consecutive trials to determine the course of habituation and trends in the early CNV amplitudes. Habituation was indicated by a negative slope, whereas loss of habituation was marked by a positive slope, as calculated by linear regression (y = ax + b, where a is the slope of habituation and b the intercept of linear regression) (33). Because only habituation of the iCNV shows relevance in the pathogenesis of migraine, only habituation of the iCNV was considered in this study (5,7,9,11,12). Three areas of interest were defined according to Gomez et al. (34) and Bender et al. (35,36): left and right ‘posterior parietal areas’ were formed by Pz, P3, P4, P7, P8; ‘central areas’ included leads Cz, C3, C4, CP1, CP2, CP5, CP6, FC1, FC2, FC5, FC6; ‘frontal areas’ consisted of sites Fz, F3, F4, F7, F8, Fp1, Fp2.
Assessment of the clinical course of migraine
Due to insufficient compliance, the headache diaries were kept only during the baseline (8 weeks before treatment) and the treatment phases. After both treatment programmes, only some of the children documented their headaches in a diary. Therefore, for assessment of the clinical course of migraine, headache questionnaires were used which had been filled out by the children before and after both treatments. The headache intensity was assessed using a visual analogue scale between 0 (no headache) and 10 (very severe headache). The headache duration was given in hours. The frequency of headache was estimated for the time window of 1 month (1, daily; 2, several times in a week; 3, weekly; 4, one to three times a month; 5, less than monthly).
Statistical analysis
All data were normally distributed (Kolmogoroff–Smirnoff-test) and characterized by homogeneous variances (F-test). For clinical variables (frequency, duration and intensity of headaches), we conducted analysis of variance with a between-subjects factor Group (MIPAS vs biofeedback) and a within-subjects factor Treatment (before vs after the treatment). For neurophysiological data (CNV amplitudes and coefficient of linear regression as a measure of habituation), we performed analysis of variance with the same between-subjects factor and within-subjects factors Parameter (iCNV vs lCNV vs PINV vs RT vs habituation), Location (frontal vs central vs parietal) and Treatment (before vs after treatment). Differences between the groups before and after treatment were proven using two-sample, two-tailed t-tests. In order to characterize the effect of each particular treatment in each group separately, we performed paired sample, two-tailed t-tests within each group for variables obtained before and after therapy. To describe the relationship between clinical (effect of the therapy: frequency after treatment – frequency before treatment) and neurophysiological (amplitude and habituation of iCNV before treatment – amplitude and habituation of iCNV after treatment) data, Pearson product moment correlations were calculated. For all calculations, the significance level was kept at P < 0.05. Bonferroni alpha adjustment was performed to correct for multiple comparisons.
Results
Clinical course of migraine
Table 1 demonstrates the clinical course of migraine before and after either the MIPAS-Family programme or biofeedback training. Before any treatment, patients from the MIPAS-Family group were characterized by less frequent and relatively short headaches compared to the biofeedback group, although the differences between the groups were non-significant. In both groups, migraine patients demonstrated a significant reduction in migraine frequency and intensity. ANOVA revealed a significant main effect Treatment for migraine frequency (F1,24 = 10.074; P = 0.004), intensity of headaches (F1,24 = 4.512; P = 0.044), and a tendency toward significance for migraine duration (F1,24 = 3.474; P = 0.075). Moreover, the main effect Group was significant for migraine frequency (F1,24 = 3.992; P = 0.05) and duration (F1,24 = 5.355; P = 0.03). However, the interaction Treatment × Group was non-significant for all clinical variables. It seems likely that both treatments programmes were associated with comparable efficacy such that neither was superior to the other.
Contingent negative variation
Figure 1 shows topographic distribution of the iCNV for each condition (Fig. 1A), grand averages of CNV amplitudes (Fig. 1B) before and after both treatments as well as curves (Fig. 1C) representing t-statistics which demonstrate differences between CNV amplitudes recorded before and after a particular treatment. Figure 2 represents amplitudes of different CNV components. Figures 3–5 demonstrate changes of CNV habituation before and after both treatment programmes.
Changes of the contingent negative variation after either MIPAS-Family programme or biofeedback therapy. (A) Topographic distribution of the amplitude of the iCNV component in both MIPAS and biofeedback groups. The voltage map was reconstructed for the amplitude of the iCNV in time window of +200 ms around the peak in the interval of 550–750 ms after the warning stimulus. The difference between voltage maps obtained before and after both treatments is shown in the upper part of the diagram (see ‘map of difference’). (B) Grand averages of CNV curves (shown only for Fz). These grand averages were used to define the iCNV amplitude for calculation of voltage maps. The warning stimulus occurred at the time point 0.0. (C) Diagram of t-values demonstrating differences between CNV amplitudes obtained before and after both treatment programmes for each sampling point. The most pronounced differences were observed in the MIPAS-Family group for frontal leads and iCNV component, as well as for PINV, although these differences did not survive Bonferroni alpha adjustment (see Results). Mean values and SD of CNV amplitude shown for all CNV components and all brain areas analyzed. Changes of CNV amplitudes (grand averages over all patients for Fz) from each block of CNV recording in the MIPAS-Family group before and after the therapy. It is obvious that there was a pronounced loss of habituation in the iCNV component before treatment (see the interval between 0.5 s and 1.0 s in the black curve characterizing the first block of recording which was substantially lower than the blue curve which represents the last block of CNV trials). After the treatment, the patients demonstrated a clear habituation of the iCNV (the black curve is larger than the blue one in the time window of the iCNV). Changes of CNV amplitudes (grand averages over all patients for Fz) from each block of CNV recording in the biofeedback group before and after therapy. There was a clear loss of habituation of the iCNV (see interval between 0.5 s and 1.0 s) before treatment (the black curve is lower than the blue curve) which persisted even after the treatment. Changes of the iCNV from each block of recordings represented as mean values and SD for both MIPAS-Family and biofeedback groups. Lines of linear regression as well as equations [y = ax + b, where a is a coefficient of linear regression shown as mean (SD) of all individual coefficients and b is an intercept also represented as a mean (SD)] demonstrate a pronounced loss of habituation in both MIPAS-Family and biofeedback groups before treatment. Habituation normalized, however, after treatment in the MIPAS-Family group but not in the biofeedback group. Note that positive regression coefficients in the equation indicate loss of habituation (i.e. reduced habituation), and negative coefficients illustrate increased habituation.


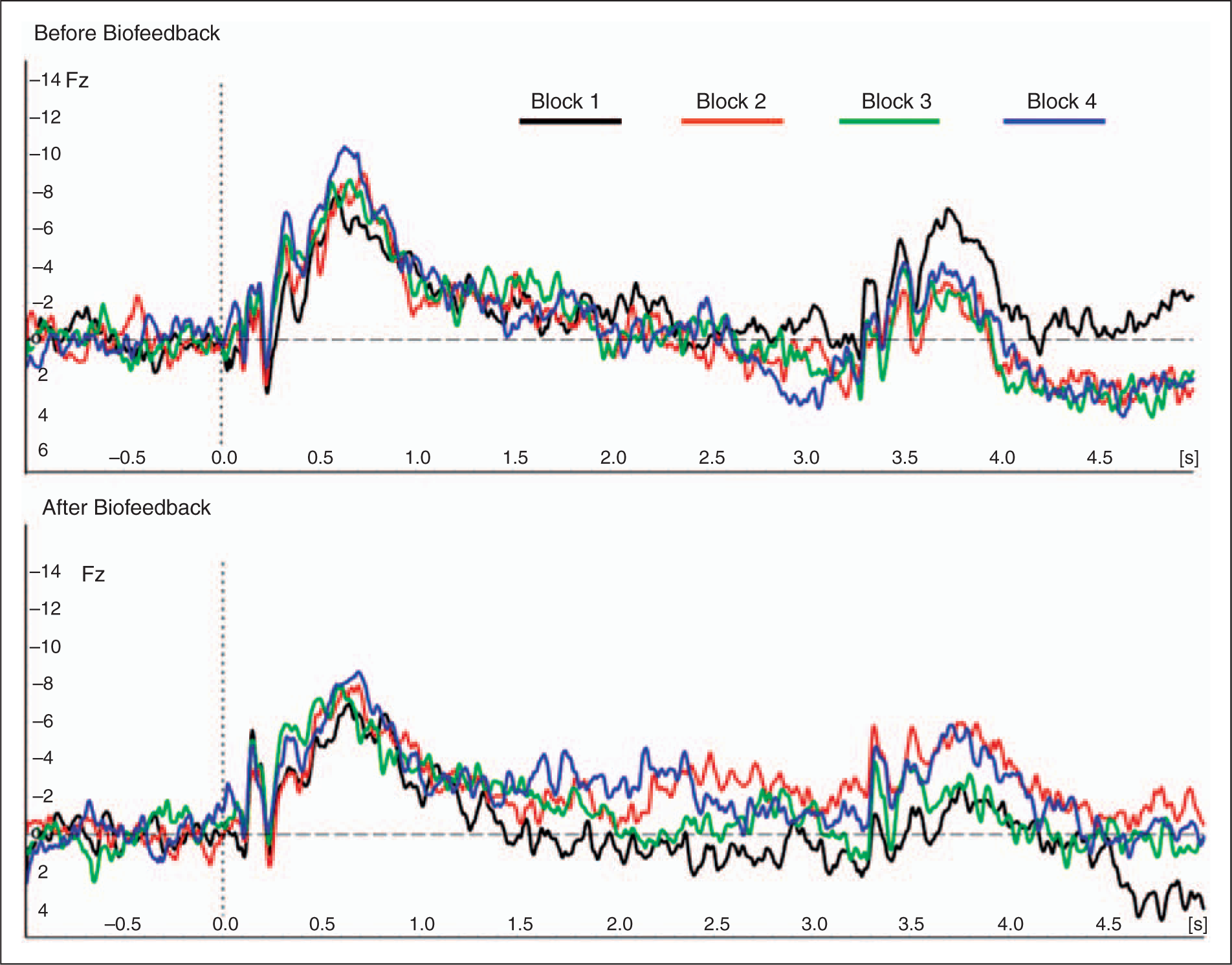

Analysis of variance revealed significant main effects Parameter (F3,72 = 11.399; P < 0.001) and Location (F2,48 = 16.424; P < 0.001) as well as significant interactions Treatment × Parameter × Group (F3,72 = 2.811; P = 0.045) and Parameter × Location (F6,144 = 33.162; P < 0.001). In the MIPAS group, post-hoc tests demonstrated that there was a clear reduction in the iCNV amplitude in the frontal region (t12 = −2.192; P = 0.049) and in the lCNV amplitude in the central region (t12 = −2.439; P = 0.03). However, both changes did not survive the Bonferroni alpha adjustment. The normalization of the iCNV habituation after the MIPAS treatment also remained significant after correction for multiple comparisons (for the frontal region: t12 = 5.933; P < 0.001 and for the central region: t12 = 4.844; P < 0.001). There were no significant changes in any of CNV parameters after treatment in the biofeedback group. The described effects may be well illustrated by curves of t-statistics (Fig. 2C): there was no substantial deviation in the curve of the biofeedback group but a significant change in t-coefficients in the time frame of the iCNV and PINV in the MIPAS group. However, only changes in iCNV amplitude were significant. Analysis of topographic changes (Fig. 1A) revealed a clear reduction in the dispersion of the iCNV after MIPAS-Family training in the prefrontal and frontal area. After biofeedback, there were no changes in topography of the iCNV component. No other topographic changes of any other CNV components were observed.
Relation between CNV parameters and clinical course of migraine
Figure 6 shows results of the correlation analysis between changes in the clinical course of migraine after a particular treatment programme and changes in iCNV amplitude and habituation after treatments. Significant correlation was found for the iCNV in the MIPAS-Family group but not in the biofeedback group. The greater the reduction in migraine frequency, the more pronounced the normalization of the iCNV habituation was after MIPAS-Family training.
Results of Pearson product moment correlations between changes of habituation (regression coefficients before treatment minus coefficients after treatment) and changes of migraine frequency (scores of migraine frequency before treatment minus scores after treatment) demonstrated for both MIPAS-Family and biofeedback groups. Note in the MIPAS group that the better the efficacy of the treatment was, the greater the reduction of regression coefficient was, i.e. the more pronounced the habituation was.
Discussion
Clinical efficacy of MIPAS-Family
MIPAS-Family is a new psychotherapeutic programme for the treatment of paediatric migraine which is based on current knowledge of pathogenetic mechanisms (23–25). Because reduced habituation to repetitive stimuli is one of the important features of the migrainous brain (1,2), behavioural strategies directed toward learning habituation constituted the main therapeutic procedure of the programme (24). MIPAS-Family led to a significant reduction in migraine frequency and intensity and substantially increased the children’s quality of life and daily functioning (23,25). The efficacy of the training was comparable to the efficacy of biofeedback training. Indeed, biofeedback has been shown as a treatment of first choice for paediatric headaches and caused a stable improvement of headaches also after a long period of follow-up, especially in patients with tension-type headache (20–22,37–39). However, MIPAS-Family provides some clear advantages compared to biofeedback. MIPAS-Family is a group intervention. First, the children have the opportunity to share their experience of coping with headaches and stress with other sufferers, they enjoy social support and reinforcement, and they enhance their group competence and self-esteem. These non-specific effects may explain the more pronounced improvement of quality of life in the MIPAS group compared with biofeedback group even though the clinical efficacy was equal (25). Second, due to the involvement of parents there was a better transfer of learned abilities into everyday life. MIPAS-Family was superior to biofeedback and contributed to more stable clinical efficacy after 6-month follow-up (23,25). The discrepancy with previous studies on biofeedback and paediatric headaches may be explained by a differential effect of the MIPAS programme: the most studies which have demonstrated long-lasting effect of biofeedback, have been performed in patients with tension-type headache (37–39). It seems likely that the biofeedback is less effective in migraine than in tension-type headache, especially by taking the long-term perspective. Finally, as a group intervention, MIPAS-Family is more economical and relies less on financial and therapeutic resources. Thus, in light of the increasing economic pressure in European and American healthcare systems, MIPAS-Family may have more chance to find its place in therapeutic programmes of general practitioners and behavioural therapists.
Normalization of CNV habituation after therapy
This study provides evidence that the treatment programme aimed at improving habituation is indeed associated with significant normalization of iCNV habituation and that changes in habituation are closely related to improvement in the clinical course of migraine. These relationships between clinical and neurophysiological parameters were obvious in children who took part in the MIPAS-Family programme, but not in children who underwent biofeedback treatment. The findings are in line with previous neurophysiological research. On the one hand, it has been demonstrated repeatedly that migraine patients are characterized by increased amplitude and reduced habituation of the iCNV and that these abnormalities are most pronounced several days before a migraine attack, representing attack anticipation and increased susceptibility to provoking agents (9–14). On the other hand, different pharmacological agents such as β-blockers, calcium antagonists and anti-epileptic drugs as well as behavioural interventions such as neurofeedback training of self-regulation of slow cortical potentials (i.e. CNV) have caused a significant reduction in migraine frequency and contributed to normalization of iCNV habituation (15–19). The current study demonstrates that behavioural intervention programmes such as MIPAS-Family may influence the clinical course of migraine through the same mechanisms as pharmacological treatment options and neurofeedback, i.e. through normalization of cortical information processing in migraine. It seems likely that the MIPAS-Family programme improves the ability of children with migraine to cope with stressful situations which may potentially influence abnormal habituation, to adjust to aversive stimuli in these situations and, therefore, to prevent further abnormalities of the central information processing which may lead to a migraine attack.
However, the normalization of the iCNV habituation seems to be not the only way in which different prophylactic treatments exert an influence on clinical symptoms in migraine. In the biofeedback group, we observed a clear reduction in headache frequency and intensity but no normalization of the iCNV habituation. We suggest that biofeedback may reduce autonomic and central reactivity (for example, during stress) and cortical excitability (40–42). Moreover, migraine patients are characterized by increased muscular activity in the head (43–46). The overactivity in peripheral muscles may be associated with sensitization of the trigeminal neurovascular system and may, therefore, increase susceptibility to migraine attacks (47). Muscular relaxation through biofeedback may normalize sensitivity of the trigeminal nucleus caudalis and increase the threshold for migraine attacks. Finally, there could be a non-specific cognitive effect which causes pronounced clinical improvement. Behavioural training such as biofeedback improves awareness to provoking agents, self-assurance and confidence in coping with stress and pain (48). In such a way, the clinical course of migraine may be influenced by different specific and non-specific mechanisms. Whether the normalization of iCNV habituation is an important prerequisite or predictor of a better clinical efficacy or stability of therapeutic effects in migraine has to be evaluated in future studies.
