Abstract
In this review, the evidence for trapezius muscle activity as a releasing factor for shoulder and neck pain is considered, mainly on the basis of studies in our laboratory. Two lines of evidence are produced, (i) vocational studies in an occupational setting, where muscle activity pattern is recorded by surface EMG and a clinical examination of the shoulder region of the subjects performed; and (ii) laboratory studies where muscle activity patterns and pain development are recorded in an experimental situation with mental stress and minimal physical activity. The vocational studies demonstrate pain development in the shoulder and neck despite very low muscle activity recorded, making it very difficult to assume muscular involvement for all cases with such complaints. However, the hypothesis of pain development through overexertion of a subpopulation of low-threshold motor units also makes it difficult to draw a firm negative conclusion. The laboratory experiments, on the other hand, show that trapezius activity patterns in response to stress have many features that would be expected if muscle activation induces pain symptoms. It is further noted that the trapezius is the only muscle with activity patterns that show these features. Possibly, we observe the effects of parallel physiological phenomena, e.g., a systemic autonomic activation that induces pain symptoms and also facilitates the motor response of some muscles. Evidence of autonomic activation of trapezius is presented by the observation of low-level, rhythmic EMG activity during sleep. However, this is not firm evidence for the above hypothesis, which at present best serves as a basis for further experimentation.
It is well known that heavy muscle exercise can induce muscle pain. If the intensity of exercise is reduced and duration at the same time extended, pain may still develop. No well-defined lower limit to the level of muscle activity, where the association between sustained muscle activity and pain induction is no longer valid, can be found. It is therefore tempting to assume that muscle activity is an etiological factor when pain symptoms with a perceived muscular location develop, even though no overt muscle exertion is observed. A number of diagnostic conditions with muscle pain involve shoulder and neck muscles, including conditions with symptoms primarily localized to the shoulder and neck (e.g., trapezius myalgia), conditions with widespread pain (e.g., fibromyalgia), and conditions where the pain symptoms are primarily localized to neighboring body regions (e.g., tension headache). Many studies have been carried out to demonstrate an association between muscle activity and pain, with mixed results. In some diagnostic conditions dual etiological mechanisms are envisioned (e.g., tension headache with and without muscular involvement) (1, 2). Other scientists consider the variation in results mainly due to the mixed scientific quality of studies; and studies of good quality indicate relatively unequivocally that low-level muscle activity is more prominently present for patients with, e.g., tension headache, than for controls (3).
For many years we have been interested in trapezius muscle activity as a releasing factor for the development of trapezius myalgia (4, 5). This interest has mainly been motivated by the need to understand and control shoulder pain as an occupational problem, but also to obtain insight into mechanisms initiating pain symptoms of muscular origin. The occupational setting of our field studies has the advantage of providing the opportunity of a detailed description of muscle activity patterns that repeat between workdays and in the longer-term may cause pain symptoms in the shoulder and neck. In this work, we realize that trapezius myalgia is only one of several medical conditions that cause pain in the shoulder and neck. However, palpation, which allows a specific localization of pain symptoms and trigger points, can infer trapezius involvement. Thus, in our studies we are not dealing with “trapezius myalgia” as an exclusive diagnosis, but with a condition of shoulder and neck pain where muscular involvement, including the trapezius, is likely. The aim of this paper is to review this work, with a view to presenting relevant information on the putative association between trapezius muscle activity and pain symptoms in the shoulder and neck.
Risk factors for shoulder and neck pain
In an occupational setting, sustained postures with elevated arms or repetitive arm work are well-established risk factors for shoulder and neck pain (6). These risk factors indicate a sustained activity pattern in the trapezius (and other shoulder muscles). Recording of surface EMG under these conditions shows that median activity levels can be as low as a few percent of the EMG activity at maximal voluntary contraction (% EMGmax), and as high as 20–30% EMGmax. This evaluation considers the full-wave rectified and averaged surface EMG signal, over time intervals of 0.2 sec. Static EMG activity level (i.e., representing the lower 10% point on the cumulative EMG distribution curve) (7) can likewise vary between virtually no static activity (<0.2% EMGmax) and more than 10% EMGmax. These values are low by conventional standards for evaluation of workload. Higher activity levels can be observed, but only for short periods. The within-group scatter and the differences in mean group values are notable and are discussed in the next section, with the aim of identifying the best data reduction procedures suitable for assessment of risk for complaint development. Subjectively, workers with high workload consistently attribute their complaints to working conditions, while many are uncertain of the cause of their pain symptoms when the physical workload is light.
Stress is another, generally accepted risk factor for shoulder and neck complaints (8), as it is for many pain syndromes localized to the head (e.g., 9) and (perhaps less prominently) low back (e.g., 10, 11). A problem when using stress as a risk indicator is that this variable is not well defined and is based entirely on subjective reports. In occupational field studies, stress may or may notbe associated with pain in the shoulder and neck (12), and it is difficult to know whether the inconsistent results are due to poor reliability or to a genuine variability between groups of workers. Attempts have been made to introduce stress-associated metavariables on theoretical grounds, e.g., psychological demands, decision latitude and social support, which have proven quite successful in explaining risk of cardiovascular disease (13), but no consistent pattern has emerged with respect to pain syndromes (14, 15). Nevertheless, the many positive reports of an association between stress and pain in the shoulder and neck leave little doubt that work stress and life stresses in general represent a risk factor for shoulder and neck complaints.
A third group of risk factors relates to individual susceptibility for shoulder and neck complaints. Individual susceptibility is indicated by, e.g., previous experience with similar symptoms (16). This is hardly surprising. Another risk indicator of possible etiological significance is a high level of subjectively perceived tension (“perceived general tension”), which has been associated with shoulder and neck complaints in all our field studies when included. This has been the case also when other subjective variables like work stress have not shown a statistical association with complaints (Vasseljen et al., submitted). This association is only observed for pain symptoms in the shoulder and neck, and not for symptoms in the lower trunk and extremities. A tendency to an association with unspecified headache is observed in some studies. In interviews, the perception of tension is often described as the sensation of elevating the shoulders (unpublished observations), pointing to activation of muscles that function as lifters of the shoulder girdle. The perception of tension can be a long-term effect of stress, thus possibly pointing to a physiological correlate of stress as a risk factor for shoulder and neck complaints.
Trapezius EMG activity and shoulder—neck pain
The previous section summarized results that can be interpreted as indirect evidence of trapezius muscle activity being a factor in the development of shoulder and neck complaints, even at low muscle activity levels. Muscle activity recording from the trapezius in these studies allows a more direct examination of this putative relationship, provided the recordings (usually of 30 to 60 min duration) are representative of the critical muscle activity patterns. This is generally secured by choosing work situations with little variation in work tasks over the workday. Another consideration is the choice of EMG variables to represent the risk of complaints. We calculate static and median EMG activity level as a proportion of the EMG activity at maximal voluntary contraction, representing generally accepted normalization and evaluation procedures (17).
A consistent between-group association between trapezius EMG level and subjectively reported shoulder and neck symptoms in the last 12 months is found in our studies both when static and median activity level are used to indicate workload. The individual, within-group variation is, however, large (Fig. 1). The variance in data accounted for by a linear regression analysis increased from 15% to 30% when excluding those who reported a positive indication of psychosocial stress. An even clearer pattern emerged when the data were ordered according to the static EMG level and mean symptom score calculated separately for workers with and without subjective stress symptoms (Fig. 2). A curvilinear association was found with consistently higher symptom score at the same static EMG activity level of those reporting stress symptoms or problems. This is particularly evident at low EMG activity levels: workers without stress problems reported few and minor complaints, while a considerable higher mean symptom score was found for those with such problems. This pattern was consistent for two groups of workers with very different work tasks, namely office work and light, repetitive production work (Fig. 3). If median activity level is used as the workload indicator, this dose-response association was no longer evident.

A. Scatter plot of symptom score for the shoulder and neck versus median trapezius activity, while performing representative work tasks. The symptom score is a composite score, representing both intensity and occurrence of complaints in the last 12 months (18). Four groups of workers are represented, performing work tasks as indicated in the box. A linear regression line is shown, indicating a highly significant but relatively weak association. B. The same data, now shown on a group basis, with median and SEM values for trapezius activity and symptom score.
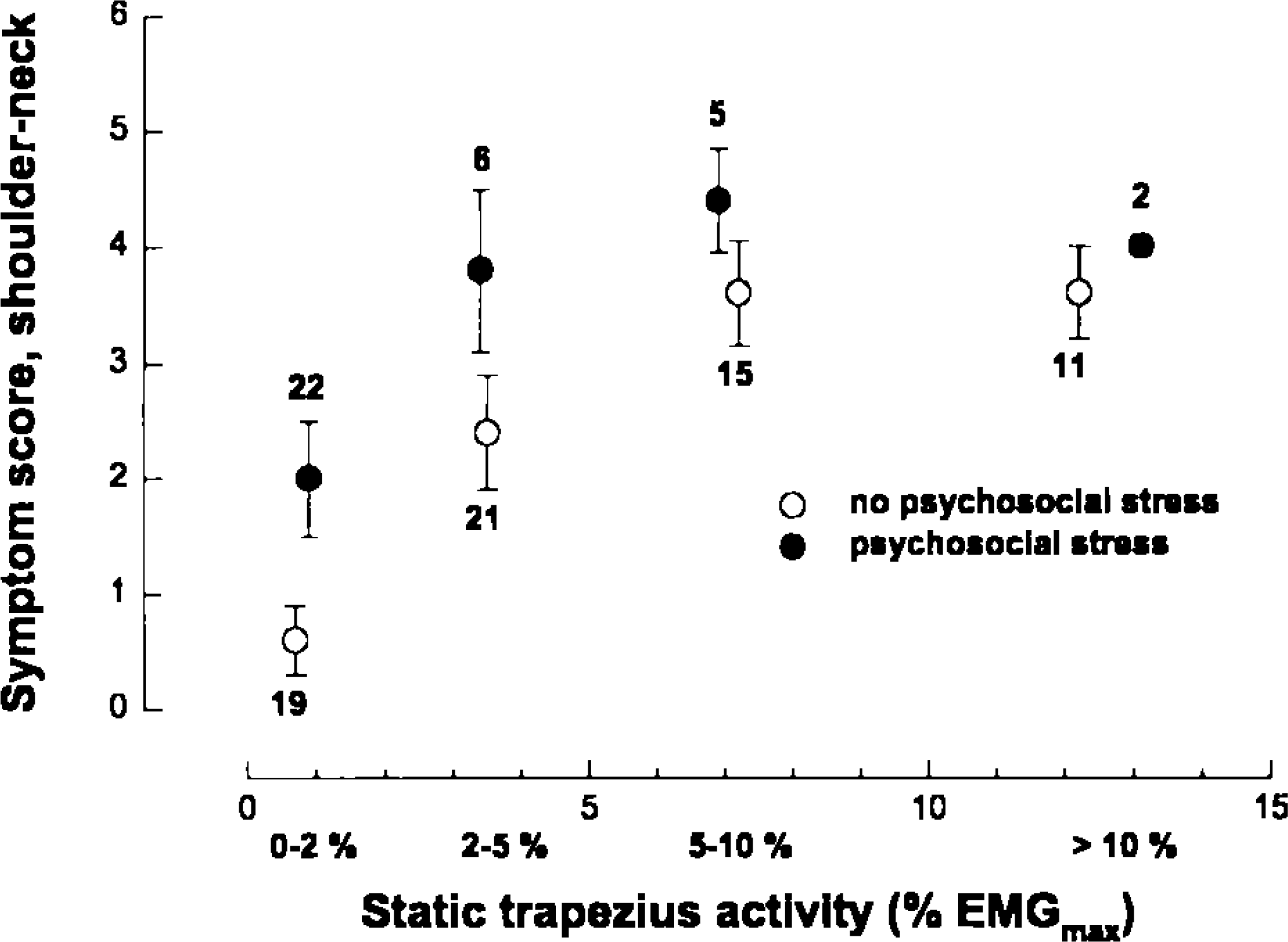
Symptom score of the same subjects (mean, SEM), but using static trapezius activity to indicate physical load. Numbers above each data point indicate number of subjects in each group. The subjects are stratified according to whether they have indicated psychosocial stress or not. The static EMG activity data are grouped as indicated on the x-axis.

Symptom score (mean, SEM) of office and light manual workers, grouped according to static muscle activity level and stratified on psychosocial stress. Numbers above each data point indicate number of subjects in each group. (◯ = Office, no psychosocial stress; • = office, psychosocial stress; □ = chocolate packing, no psychosocial stress; ▪ = chocolate packing, psychosocial stress).
A puzzle with this pattern of results is the pain development at very low EMG activity levels. Other mechanisms are clearly operating than those that initiate muscle pain in physical exercise. A hypothesis that has received considerable attention in recent years is pain induction through selective overexertion of low-threshold motor units (19): single-motor units are presumed damaged by sustained activity patterns and cause pain symptoms in a situation where the large majority of muscle fibers are inactive. The hypothesis is based on the Henneman size principle (20) of a fixed recruitment order of motor units, and has indirect experimental support in the finding of “ragged red” fibers, which are prominent in the descending parts of the trapezius muscle (21). These muscle fibers have a fragmented morphologic appearance and stains for NADH-tetrazolium reductase, indicating mitochondrial dysfunction and disturbed metabolism. They are always type I, i.e., are among the first recruited, and have larger cross-sectional area than other type I fibers in the same muscle. The hypothesis is intriguing in that it represents an attempt to bridge the gap between the concept of muscle overload as a source of pain and the observed low EMG activity levels. It also presents a considerable experimental challenge, in that single motor unit activity patterns are not easily studied even by intramuscular electrodes, and can certainly not be monitored through surface EMG recordings.
We have attempted to establish indirect indicators of motor unit activity pattern, the so-called “EMG gap” analysis, where short periods of muscle inactivity in the surface EMG are identified (Fig. 4; 22). In a longitudinal study of healthy (i.e., normally pain-free) workers recruited to a work situation with repetitive work tasks and where some developed shoulder and neck complaints, EMG gaps were a good predictor of future pain symptoms (23). The interpretation of EMG gaps as an indicator of activity pattern in low-threshold motor units has received some support from laboratory studies with parallel recording of single-motor units and surface EMG. Several cases of motor unit substitution, i.e., the temporal replacement of a motor unit with motor units of initially higher recruitment threshold that coincided with EMG gaps, have been observed. This is, however, a statistical association: motor unit substitution may take place without EMG gaps (illustrated in Fig. 5), and gaps may happen without motor unit substitution.

A section of surface EMG recording that illustrates the EMG gap analysis. The EMG recording system has an inherent noise level of 1.0 to 1.5 μV, representing about 0.1 to 0.2% EMGmax. This value is subtracted and the zero value corresponds to the lowest detected EMG level. The median rest level of normal subjects (0.13% EMGmax) and 95% confidence intervals are indicated. The chosen detection level for EMG gaps is twice the upper CI of rest level. Two gaps are shown during the period when the muscle is active.
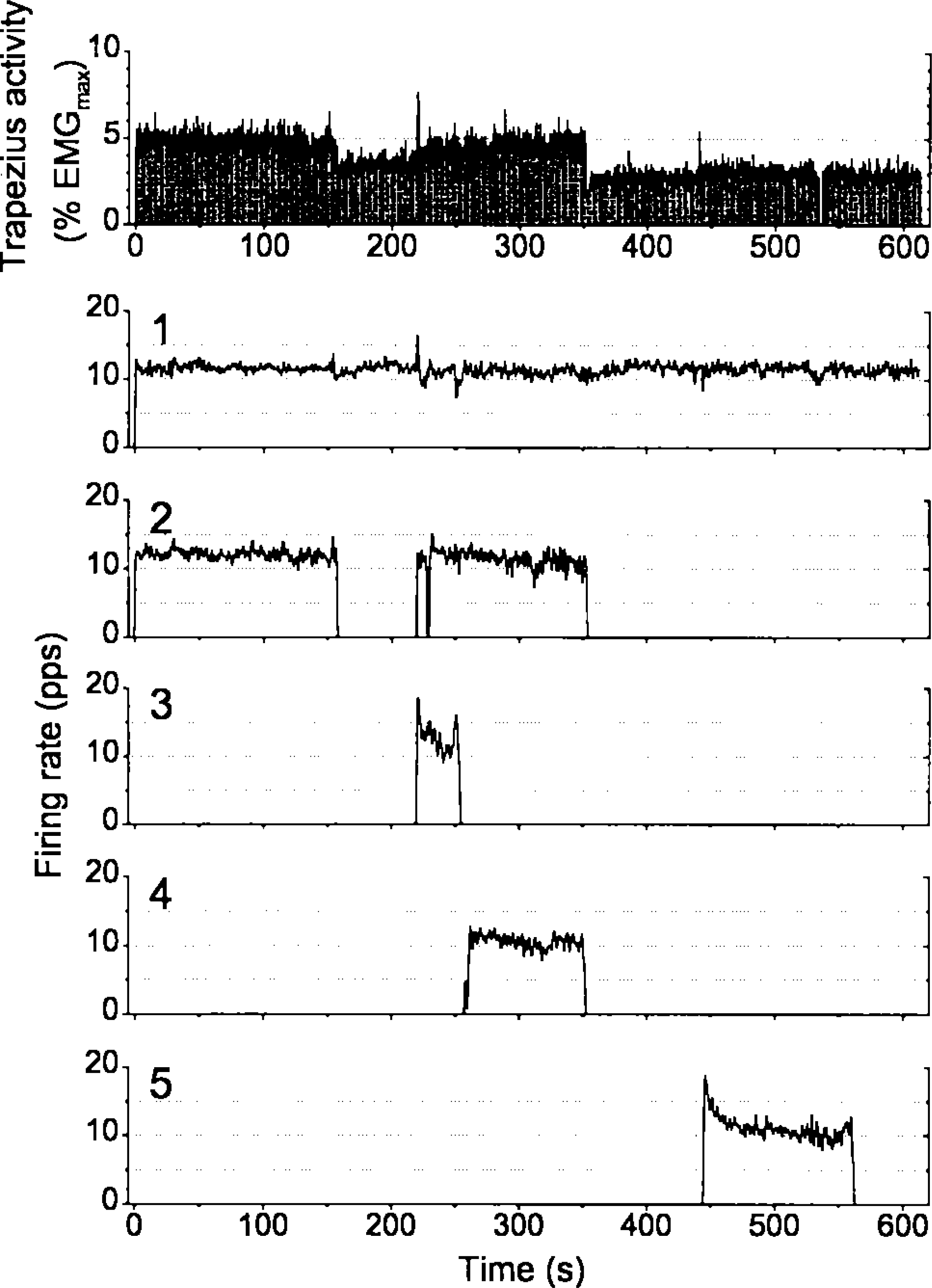
Surface EMG activity (upper panel) and firing rate (low-pass filtered at 0.5 Hz; five lower panels) of five motor units during a laboratory experiment with mental stress (Westgaard and De Luca, unpublished observations). The motor units are identified by use of a 3-channel fine-wire recording electrode, and using the Precision Decomposition Technique of De Luca (35). A low-level, sustained activity pattern is shown in the surface EMG during the first part of the test, derived from sustained firing of individual motor units. Motor unit no. 1 is active throughout the 10-min experiment, while other units are cycling in and out, depending on overall activity level and the activation history of individual units. The experiment demonstrates motor unit substitution, in that unit no. 2 is not active during the last part of the test. At this time the surface EMG level is reduced and a unit (no. 5) that was not active during the first part of the contraction is recruited. Other experiments have shown several examples of sustained motor unit firing throughout the 10-min test period (31). In other experiments motor unit substitution has coincided with an EMG gap in the surface EMG (e.g., 36).
The EMG gap analysis has also been carried out in other vocational studies, without finding the same association with complaints (5). Thus, EMG gaps (or other indicators of EMG variation pattern) cannot serve as a general risk indicator of shoulder and neck complaints, and we do not at present know when the variable is a valid risk indicator. We have further noted that in studies where trapezius EMG gaps distinguish workers with and without shoulder pain, those with pain symptoms also present a higher EMG signal in situations with nominal rest of the shoulder muscles (“uninstructed rest”) (24, 25). These observations can be taken as a further indication that sustained activation of low-threshold motor units in situations with nominal rest can be a risk factor for musculoskeletal complaints. However, the inconsistent results imply there is no easy way to verify this relationship, or exclude other physiological pathways to muscle pain.
Mental stress, EMG activity, and pain development: field studies
A conclusion of these early studies is that mental stress, as a risk indicator for shoulder and neck complaints, is not easily detected by the vocational EMG activity pattern. Mental stress further appears to be a more significant risk factor at low than at high physical workloads. We have therefore recently carried out studies among workers (shop assistants) with low physical workload of shoulder muscles. Their main task is to induce sales through customer relations. Their physical workload is light and varied, and no heavy or sustained physical activity is observed for the shoulder muscles. In this group, mental stress is likely to be the most significant risk factor for shoulder and neck pain, and an association with muscle activity pattern is less likely masked by EMG activity due to physical exertion (Westgaard et al., submitted).
About half the workforce reported complaints in the shoulder and neck, and about half of those with pain attributed the symptoms to stresses at work. The trapezius muscle activity was very low, with group mean value of the median activity level at 4% EMGmax, and 0.4% EMGmax for the static component. The EMG gap time, i.e., EMG activity levels lower than 0.5% EMG max, was on average 12% of the work time. There was no difference in the EMG activity level between those with and without pain symptoms in the shoulder and neck, but a clear difference in perceived tension was observed. This was the only distinguishing variable with respect to pain state. For these workers it is difficult to believe that sustained, low-threshold motor unit activity is a problem, considering the very low EMG activity levels and the high fraction of time without discernible EMG activity. The results strengthen the impression that sustained motor unit activity may not be an exclusive cause of shoulder pain symptoms, especially when stress is operating as a risk factor. No association between the EMG activity level or pattern and the subjective perception of tension was found in these recordings, and has not been found in any other study.
Mental stress, EMG activity, and pain development: laboratory studies
Motor activation by mental stress is a well-known phenomenon studied for decades in the laboratory (e.g., 26, 27). We have explored this phenomenon in a laboratory setting to examine whether this motor response is likely to cause musculoskeletal complaints due to stress. The laboratory setting allows a closer and better-controlled examination of the phenomenon, but the association between a motor response and the medical condition of the subjects may be difficult to ascertain.
We have shown for normally pain-free subjects that: (i) trapezius shows a clear motor response to cognitive stress, and is one of the most responsive muscles in our experimental setting (28), (ii) the motor response is dose-dependent, i.e., a higher level of stress leads to higher muscle activity levels (29), (iii) complex tasks with a higher level of information-processing associate with higher muscle activity levels, at least for some subjects (30), and (iv) the low-variation, mostly stable, activity pattern observed in surface EMG recordings reflects sustained motor unit activity (31 and Fig. 5).
Thus, the trapezius response to cognitive stress has many features we would expect if the response is to be associated with pain development in the shoulder and neck. This putative association was examined in a more direct manner by extending the cognitive test to 1 h and including more recording sites in the upper body region, i.e., bilateral recording from trapezius, splenius capitis, and the temporal and frontal muscles. Pain scores (bilateral in case of the trapezius and temporal sites) were made on VAS for these sites, and pain reports collected every 10 min (32). Experiments were carried out on normal, pain-free subjects and on patients with shoulder myalgia, fibromyalgia, tension headache, and cervicogenic headache.
For the normally pain-free subjects, about half reported significant pain development during the test, predominantly in the shoulder and neck. The pain response was at a low level and subsided quickly after the test (32). Subjects that during the test reported a pain increase in the shoulder of at least 10 VAS units responded with higher trapezius EMG activity than those without a pain response at this location. A linear correlation analysis showed a statistically significant, but weak association, as may be expected if sustained motor unit activity is the critical underlying feature causing the pain response. There was no association between muscle activity and pain development in underlying muscles for the other recording sites.
The patient groups showed more marked pain development than the pain-free subjects. The pain response followed the diagnostic features of the patients, i.e., located to the shoulder and neck region for shoulder myalgia patients, widespread in the case of fibromyalgia patients and so on. All patient groups, including the headache patients, reported pain development in the shoulder region (one-sided for the cervicogenic headache patients). In contrast, the muscle response was stereotypic for all patient groups, with an enhanced activity level only in the trapezius and only for the first 10 to 20 min of the test, subsiding slowly thereafter. For the cervicogenic headache patients the trapezius response was only observed on the symptomatic side (33). Thus, in the case of shoulder pain and trapezius EMG activity, the results of the different series of experiments were supportive of muscle activity involvement in pain initiation, as the patient groups, which showed stronger pain development in the shoulder, also had the higher trapezius EMG response. This was not the case for the other recording sites. Within the fibromyalgia group, the amplitude of the trapezius response correlated not only with shoulder pain development, but also with pain development at other locations, despite a lack of correlation between the corresponding pain responses (Bansevicius et al., submitted). Thus, the trapezius EMG response stands out as a special case of stress-induced muscle activation. To the extent stress-induced muscle activity is related to pain development in a dose-response manner, this is apparently only the case for trapezius and shoulder pain among the sites and muscles we have examined.
Alternative hypotheses: autonomic activation of the motor system
One line of reasoning that may explain the conflicting results concerns parallel physiological effects of a general activation response resulting from exposure to stressful influences. Important responses to stress include release of neuropeptides and hormones like adrenaline, causing increased heart rate and effects on the peripheral cardiovascular system. The long-term effects of such responses are implicated in the development of cardiovascular diseases and may conceivably influence the musculoskeletal system with activation of nociceptive receptors. A problem with a hypothesis of pain initiation through stress-induced systemic effects is that these are assumed to be general in the body, while the stress-induced pain symptoms are predominantly localized to shoulder, neck and head. Even though a considerable topographic variation in the responses of the autonomic nervous system is recognized, we are not able to point to specific responses that match the bodily distribution of stress-induced pain symptoms. In contrast, the observed, stress-induced motor responses have a distribution that matches the pattern of pain symptoms (28).
Limbic regions of the brain, such as the hypothalamus, have strong inputs to the spinal motor system with segmental and proprioceptive inter-neurons, which again is a main source of input to the motoneurons. These brain regions also control the autonomic nervous system. The limbic component of the motor system is termed the “third motor system” by Holstege (34), and is concerned with gain setting of motoneurons and providing triggering mechanisms for rhythmic activity (respiration, shivering, cardiovascular regulation). The influence of the third motor system over the function of striated muscles varies from strong control over respiratory muscles like the diaphragm to minor influence over peripheral extremity (flexor) muscles involved in fine manipulative movements. It is conceivable that a muscle like trapezius receives relatively strong input from the limbic component of the motor system. The perception of elevated shoulders as a reaction to stress would suggest this, as do more anecdotal descriptions of shoulder movement taking part in respiration.
We have obtained more direct experimental evidence of such influences through 24-h recordings of muscle activity in the trapezius. During sleep, when the somatic component of the motor system is minimal, a sustained activity pattern similar to that observed during the cognitive stress test is observed in the trapezius (Fig. 6A). Our experience from these studies suggests that the muscle activity during sleep represents sustained activation of low-threshold motor units. The EMG signal contains rhythmic components reflecting respiratory rhythm, and the slower cardiovascular regulatory rhythm of 0.1 Hz may also be represented (Fig. 6B). The activity level during sleep is generally low, but sustained muscle activity as high as 10% EMGmax has been observed. This observation represents a so-far unrecognized strain on low-threshold motor units, and may contribute to the development of the ragged-red fibers previously mentioned.

A. Sustained trapezius activity recorded during the first 2 h of a sleep period. Part of this activity period is FFT analyzed, and shows rhythmic components corresponding to respiratory and cardiovascular regulatory rhythm (B).
The observation of a strong autonomic influence onto trapezius motoneurons is, however, not unequivocal evidence for the hypothesis of motor activity being an etiological factor in pain development. It can be a secondary phenomenon to parallel, but unknown, physiological processes of etiological significance. This would explain why other muscles with a similar susceptibility to develop pain symptoms do not show the same motor response. If, on the other hand, sustained single-motor unit activity is an etiological factor in pain development, this activity is (so far) only reliably detected in an experimental situation with cognitive stress and minimal physical activity, and only for the trapezius muscle. This observation also requires an explanation, and a main aim of current research projects is to untangle these seemingly contradictory results.
