Abstract
Background
Previous studies suggest the perimenstrual phase increases migraine without aura risk, attributed to the decline of estrogen in this phase. However, the link to the decline of estrogen in the post-ovulatory phase remains unclear.
Methods
We conducted a longitudinal cohort study among 563 women with migraine and a regular natural menstrual cycle. Participants completed a validated e-headache diary also including menstrual cycle tracking. Of these women, 31 (who participated in our Women Hormones Attacks and Treatment (WHAT)-hormone study) performed urine luteinizing hormone (LH) surge tests during two menstrual cycles to validate the estimation of the post-ovulatory estrogen decline. The association between the post-ovulatory estrogen decline and migraine incidence was assessed using a mixed logistic model with patient as random effect, and the post-ovulatory and menstrual window as fixed effects. Secondary analyses included a case-crossover model and a self-controlled case series (SCCS) model to strengthen robustness and control for time-invariant confounders.
Results
E-diary data from 2627 menstrual cycles, including 58 from the WHAT-hormone subgroup, were analyzed. Across all three statistical models, the post-ovulatory window showed no higher incidence of migraine (with or without aura) than other menstrual cycle days (mixed logistic model: OR 0.95, 95% CI 0.89–1.02; WHAT-hormone subgroup: OR 0.68, 95% CI 0.38–1.16). The perimenstrual window had the highest migraine incidence (SCCS model with luteal phase as reference: OR 1.67, 95% CI 1.60–1.75), followed by the follicular phase (OR 1.31, 95% CI 1.26–1.37).
Discussion
In contrast to what many women with migraine believe, there is no increased incidence of migraine attacks (with or without aura) during the post-ovulatory window, but a higher incidence during the follicular phase compared to the luteal phase was confirmed.
This is a visual representation of the abstract.
Background
Migraine is most burdensome for women aged 15 to 50 years, with its prevalence being three times higher in women than in men.1,2 Prepubertally, migraine prevalence is equal in boys and girls, while after menarche, migraine incidence in girls increases. 1 Although menstruation and other hormonal milestones are well-known to impact the incidence of migraine, the precise mechanisms by which sex hormones influence migraine susceptibility remain incompletely understood.3–6 It has been suggested that a drop in estrogen levels may increase attack susceptibility.7–9 This hypothesis is supported by previous clinical studies, which show that the odds ratio (OR) for migraine occurrence approximately doubles around menstruation in women.10–12 Migraine attacks that occur between days −2 to +3 of the menstruation are classified as perimenstrual attacks.12–14 These perimenstrual attacks last longer, have a higher recurrence risk with more frequent triptan use, and increased severity of symptoms compared to non-perimenstrual attacks.3,4 These attacks have migraine without aura characteristics as attacks with aura seem not to be affected by menstruation.4,12
In a normal menstrual cycle, there are two key phases of estrogen decline: one after ovulation, immediately after the luteinizing hormone (LH) surge (post-ovulatory), and another late luteal, preceding the onset of menstruation. 15 During menstruation, the decline in estrogen is of longer duration compared to the post-ovulatory drop, and accompanied by a decline in progesterone. 15 One study examined these two phases of estrogen decline by measuring urinary estradiol metabolites and relating them to migraine incidence across 120 menstrual cycles in 38 women with migraine. 13 Migraine incidence was highest during the late luteal estrogen decline, lowest during phases of rising estrogen, and not different during the post-ovulatory estrogen decline. 13 A second study in 81 women with migraine also suggested no association between ovulation and migraine incidence. 10 A third diary-based study of 55 women completing migraine and menstrual diaries over three cycles also reported no clear association between migraine attacks and ovulation. 16 However, evidence remains limited, as these studies involved small sample sizes, and ovulation timing was not biochemically confirmed in all cases. To accurately pinpoint ovulation, it seems crucial to either include only women with regular menstrual cycles (e.g., validated by an e-diary) or women who are closely monitored through measurements of the luteinizing hormone (LH) surge. The length of a menstrual cycle is defined as the number of days between the onset of menses in one cycle and the onset of menses in the next cycle. Menstrual cycles shorter than 24 days or longer than 38 days are considered abnormal.15,17 The luteal phase of the cycle is relatively constant in all women, lasting approximately 14 days. The variability in cycle length is typically due to differences in the follicular phase, which can range from 10 to 16 days and precedes the ovulation day. 15
The aim of our study was to conduct a large longitudinal cohort study in women with migraine and regular menstrual cycles, using our validated Leiden e-headache diary, which also includes daily menstrual cycle tracking. We hypothesized that the post-ovulatory decline in estrogen, similar to the premenstrual estrogen decline, increases the susceptibility to migraine attacks. A subgroup of women completed urine LH surge tests across two menstrual cycles to confirm accurate cycle timing.
Methods
Study population
In this cohort study we prospectively collected data from August 2018 to October 2024 at the Leiden Headache Centre (LHC). 18 This study is reported in accordance with the STROBE guidelines for observational studies. Patients diagnosed with migraine filled out e-questionnaires and e-headache diaries as part of research studies or clinical care at the Leiden University Medical Centre (LUMC). 19 Women aged ≥ 18 years with a migraine frequency of at least one attack per month and a regular menstrual cycle, were considered eligible. Participants were excluded if two consecutive menstrual cycles differed by seven days or more according to the STRAW 10+ criteria, and individual cycles were removed if the cycle length was <24 days or >38 days according to FIGO System 1.17,20 Patients were not allowed to use exogenous hormones. Diaries of at least two menstrual cycles per patient were required and each individual cycle had to have at least 80% adherence to the e-diary.
Women with chronic migraine (CM) (≥15 monthly headache days, including ≥8 migraine days) were excluded as detecting a clear pattern throughout the menstrual cycle would be challenging. To address the effect of excluding these patients, we also conducted a complete case analysis that included those with CM.
As part of the Women Hormones Attacks and Treatment (WHAT)-hormone study, a subgroup of 31 women with menstrual migraine measured their LH surge during one or two menstrual cycles between January 2022 and September 2024. Recruitment for this subgroup was conducted through newsletters, social media and the website www.whatstudy.nl/en, where participants could fill out a registration form. Interested participants were screened for additional inclusion criteria via a telephone call. Women were excluded if they smoked, had a body mass index (BMI) > 31 kg/m2, were pregnant or breastfeeding, used sex hormone-containing treatment and/or corticosteroids, or had any medical condition that could influence sex hormone levels such as polycystic ovary syndrome.
Data collection
Participants were monitored using validated headache e-diaries embedded in the Patient Journey App for at least two menstrual cycles.19,21 Each morning at 8 a.m. participants received a notification to complete the questionnaire referring to the previous 24 h (midnight to midnight). The questionnaire included 6–31 questions, depending on the responses, covering the presence of headache and/or visual aura symptoms and their characteristics, use of acute medication, changes in prophylactic medication or use of hormones, and menstruation. Menstrual bleeding was recorded daily in the e-diary, enabling identification of menstrual onset and cycle phase for each participant. If the e-diary was not completed by afternoon, a reminder was sent at 6 p.m. Once submitted, entries could not be edited, and questionnaires were time-locked after two days.19,21 An automated algorithm calculated for each day if it was classified as a migraine day according to the ICHD-3 criteria.22,23
In the WHAT-hormone subgroup, participants conducted daily ovulation tests (Clearblue Digital Ovulation Test, SPD Development Company, Bedford, UK) each morning at home, beginning on the 5th day after the onset of menstruation and continuing until a positive result was obtained. The LH surge was used as a proxy for ovulation, as it typically precedes follicular rupture by approximately 0.3–0.6 days on average. 24 In a minority of cycles (≈23%), the urinary LH peak may occur slightly before or after ovulation (ranging from 2.5 days before to 0.5 days after). 24 Despite this variability, urinary LH testing is widely used in both research and clinical practice as a non-invasive and reliable method for estimating the timing of ovulation, and was therefore selected for this study. These WHAT-hormone subgroup participants sent their positive test results of the LH surge to the researchers via WhatsApp, and this information was then registered for each participant.
Data analyses and statistics
The primary outcome was migraine attack incidence, defined as the first day of a migraine attack, in the ovulatory window, in comparison to outside the ovulatory window. The day of the LH surge for each individual cycle was calculated as 14 days before the onset of the following menstruation. For the WHAT-hormone subgroup the precise measured day of the LH surge was used. The ovulatory window was then defined as the day of the LH surge until the 5th day post-LH surge. This window was carefully selected with the estrogen-withdrawal theory in mind, as the decline in estrogen starts on the day of the LH surge, unlike the perimenstrual window, where the decline starts two days before menstruation.13,24,25 The period duration of five days was aligned with the perimenstrual window, which has consistently been defined as five days in prior studies.3,11,13,14 To assess potential variation in the estimated ovulatory window, we conducted two sensitivity analyses in which the window was shifted one and two days earlier, respectively.
First, we plotted the percentage of migraine on each day of the menstrual cycle for all women. Menstrual cycles were then divided into two segments based on the onset of menstruation and the ovulation dates and standardized to 28 days. Standardization was applied solely for visualization in graphs. The pre-ovulation segment was standardized to days 1–14 and the post-ovulation segment was standardized to days 15–28. Subsequently, for the graphs, the migraine attack incidence was calculated for each standardized day.
Additional plots were made following the same steps of standardization with migraine incidence subdivided into migraine attacks with aura and migraine attacks without aura, as a previous study has shown differing incidences across the menstrual cycle. 12 These plots were made using data of the whole cohort and another using only the data from women diagnosed with migraine with aura.
We conducted our analyses using three statistical approaches: (1) mixed-effects logistic regression, (2) case-crossover, and (3) self-controlled case series (SCCS).
The mixed-effects model was selected because it allows to account for patient intervariability by including individual participants as random effects, while ovulation and perimenstrual windows were included as fixed effects. The outcome was start of a migraine attack, including a differentiation between attacks with and without aura.
The self-controlled designs eliminate time-invariant confounders by allowing participants to serve as their own controls. In the case-crossover design, we stratified the data per patient, starting with the occurrence of migraine attacks and then examining whether these were preceded by days within the ovulatory window. Additionally, we conducted a sub-analysis excluding all days during the perimenstrual window, as this period is a known risk factor for migraine attacks. 13
The perimenstrual window is defined as days −2 to +3 relative to the onset of menstruation, with day 1 marking the start of menstruation and no day 0 included. 14 For the SCCS model, patient time was further segmented into four periods: the ovulatory window (days −14 to −9), the perimenstrual window (days −2 to +3), the luteal phase (following the ovulatory window and ending with the onset of perimenstrual window), and the follicular phase (following the perimenstrual window and ending before the ovulatory window). This method allows for the calculation of migraine risk across different phases of the menstrual cycle. In two separate analyses we used the follicular and luteal phases as reference periods. Calculations for the SCCS model were performed using conditional Poisson regression.
Our primary outcome was the onset of migraine attacks. We considered any missing diary entries as no migraine. If a patient missed one diary entry between two migraine days, we classified it as a migraine day, as it represented a single attack. All statistical analyses were performed in RStudio, version 4.4.0. For the mixed model, R package lme4 was used. For the case crossover model the survival package was used, and for the SCCS model the calculations were done with the base R stats package. P values < 0.05 were deemed statistically significant.
Subgroup and validation analyses
Subgroup analyses were performed for the WHAT-hormone subgroup using the same three models as previously mentioned, but the day of a positive LH-surge test was used to determine ovulation. An additional subgroup analysis was performed for participants that fulfilled ICHD-3 criteria of menstrual migraine, i.e., who had migraine attacks occurring in the perimenstrual window in at least two out of three consecutive cycles. 23 A diagnosis of menstrual migraine could not be determined in a small subset of women followed for fewer than three menstrual cycles, as these women experienced fewer than two cycles with perimenstrual migraine attacks and therefore could not meet the “two out of three cycles” criterion. A complete case analysis was performed with the mixed model and including all participants with chronic migraine that were excluded from the primary analyses. Additionally, we calculated the mean difference between the calculated ovulation date and the true ovulation date in the WHAT-hormone subgroup for validation purposes. Finally, we performed two sensitivity analyses in which the ovulatory window was shifted one and two days earlier, respectively, as well as an analysis in which the window was shortened from five to three days. The first sensitivity analysis addressed variability in the timing of estradiol decline around the LH surge, while the latter considered the possibility that the post-ovulatory decline in estrogen is shorter than that around menstruation.15,24
Ethical approval
Ethical approval for the anonymized use of headache e-diaries was obtained from the Medical Ethical committee of the Leiden University Medical Centre (LUMC). Participants did not have to provide additional informed consent. Patients in the LHC database that actively declared that they did not want to participate in medical research were not screened for eligibility. For the WHAT-hormone subgroup in which ovulation tests were performed, additional ethical approval was obtained by the Medical Ethical committee of the LUMC (NL74161.058.20). All these participants provided written informed consent.
Results
A total of 3992 patients in the LHC database were screened for study participation, of which 563 met inclusion criteria. A flowchart of participant selection is available in Figure 1. All women collectively completed daily headache e-diaries of 2627 menstrual cycles with a mean cycle length of 28.0 ± 2.7 days. The median follow-up time of all participants was 79 days with a median e-diary adherence of 99%. The WHAT-hormone subgroup contained 31 women with e-diaries of 58 menstrual cycles and a mean cycle length of 27.5 ± 3.4 days. In this subgroup, median follow-up was 52 days with median adherence of 100%. Baseline characteristics of the study population are shown in Table 1.

Flowchart of participant selection.
Baseline characteristics.

*Menstrual migraine was defined as women suffering from a perimenstrual migraine attack in two out of three menstrual cycles, and thus could not be determined in women who were followed during only two menstrual cycles. SD, standard deviation.
Figure 2 and 3 depict the probability of a migraine attack on each standardized day of the menstrual cycle for the total cohort and the WHAT-hormone subgroup. Visual interpretation shows migraine occurrence was highest during perimenstrual window and lowest during the ovulatory window and luteal phase.

Migraine incidence by day of the menstrual cycle with 95% confidence interval. Menstrual cycles were standardized to 28 days and ovulation was fixed to day 14. Data from 2627 menstrual cycles of 563 women are shown.
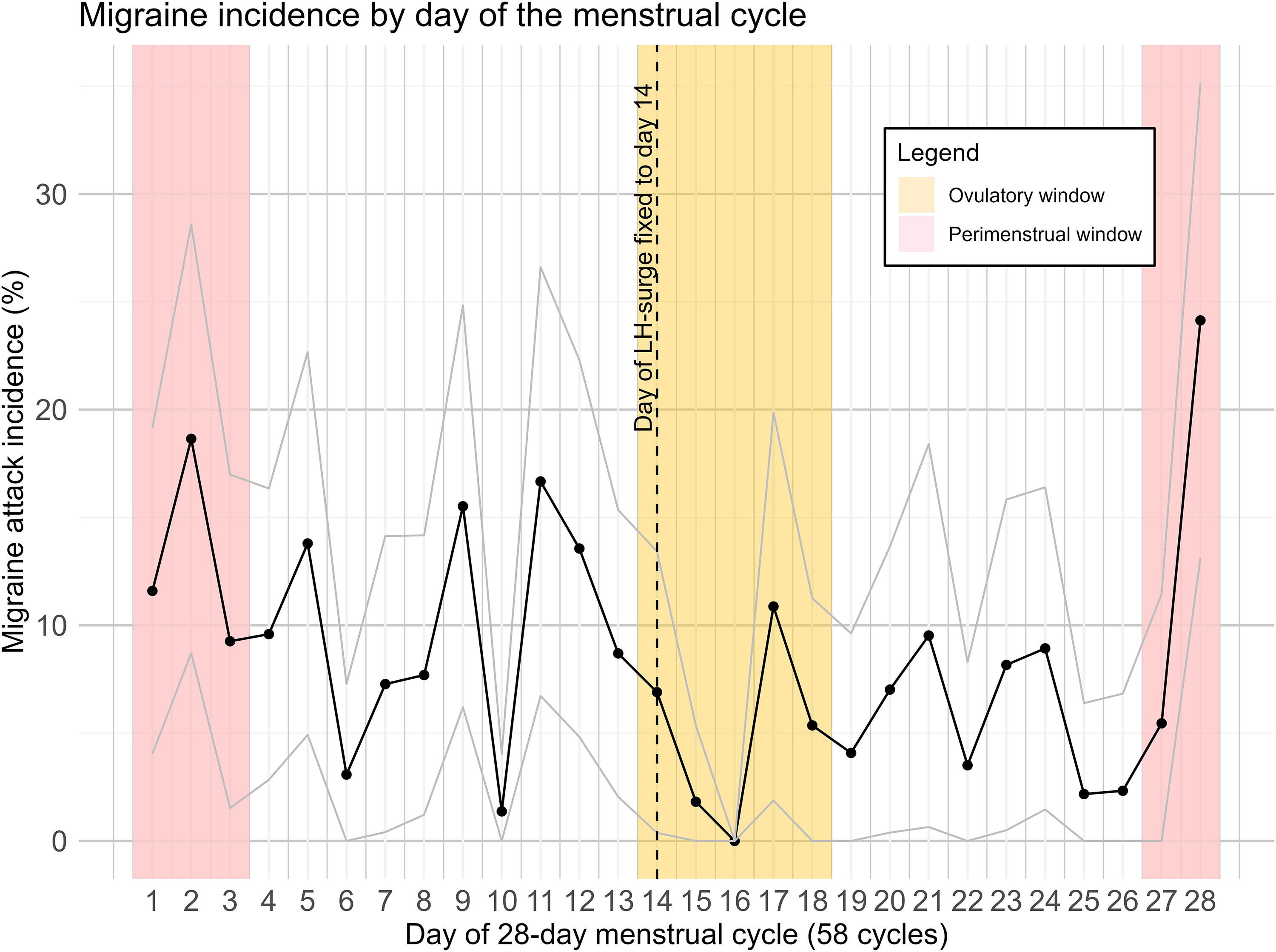
Migraine incidence in the WHAT-hormone subgroup by day of the menstrual cycle with 95% confidence interval. Datapoints were standardized to 28 days and ovulation is fixed to day 14. Ovulation was determined with urine LH-surge tests. Data from 58 menstrual cycles of 31 women are shown.
Primary outcomes
We found no increased incidence of migraine in the ovulatory window in any of the three statistical models. The mixed model yielded an OR of 0.95 for migraine in the ovulatory window (95% CI: 0.89–1.02, p = 0.13) (Table 2). The case-crossover model showed an OR of 0.86 (95% CI: 0.80–0.92, p < 0.001), indicating that the ovulatory window occurred less frequently just before a migraine attack compared to all other days of the menstrual cycle. When days within the perimenstrual window were removed from the analysis to investigate the effect of ovulation compared to non-perimenstrual days, the case-crossover model resulted in an OR of 0.95 (95% CI: 0.89–1.02, p = 0.17) (Table 3). In the SCCS model, the ovulatory window was associated with lower incidence of migraine when compared to the follicular phase (OR: 0.72, 95% CI: 0.69–0.75, p < 0.001), but not when compared to the luteal phase (OR: 0.95, 95% CI: 0.91–1.00, p = 0.68) (Table 4).
Results of mixed logistic regression model with first day of migraine attack as dependent variable and ovulatory window and perimenstrual window as fixed effects and the patient as random effect. Data of 2627 menstrual cycles from 563 patients with migraine are shown.

SE, standard error; OR, odds ratio; CI, confidence interval.
Results of case-crossover model with data stratified per patient, comparing odds for first day of migraine attack within the ovulatory window to outside of the ovulatory window. A second analysis was performed with exclusion of observations in the perimenstrual window. Data of 2627 menstrual cycles from 563 patients with migraine are shown.

SE, standard error; OR, odds ratio; CI, confidence interval.
Results of SCCS model. Data is stratified per patient and odds-ratios are calculated for first day of migraine attack within a certain period of the menstrual cycle, in comparison to follicular phase and luteal phase as reference periods. Data of 2627 menstrual cycles from 563 patients with migraine are shown.

OR, odds-ratio; CI, confidence interval; levels of significance: *** ≤ 0.001.
Only migraine attacks without aura were influenced by sex-hormone fluctuations during the menstrual cycle, whereas the incidence of migraine attacks with aura remained stable (Figure 4).
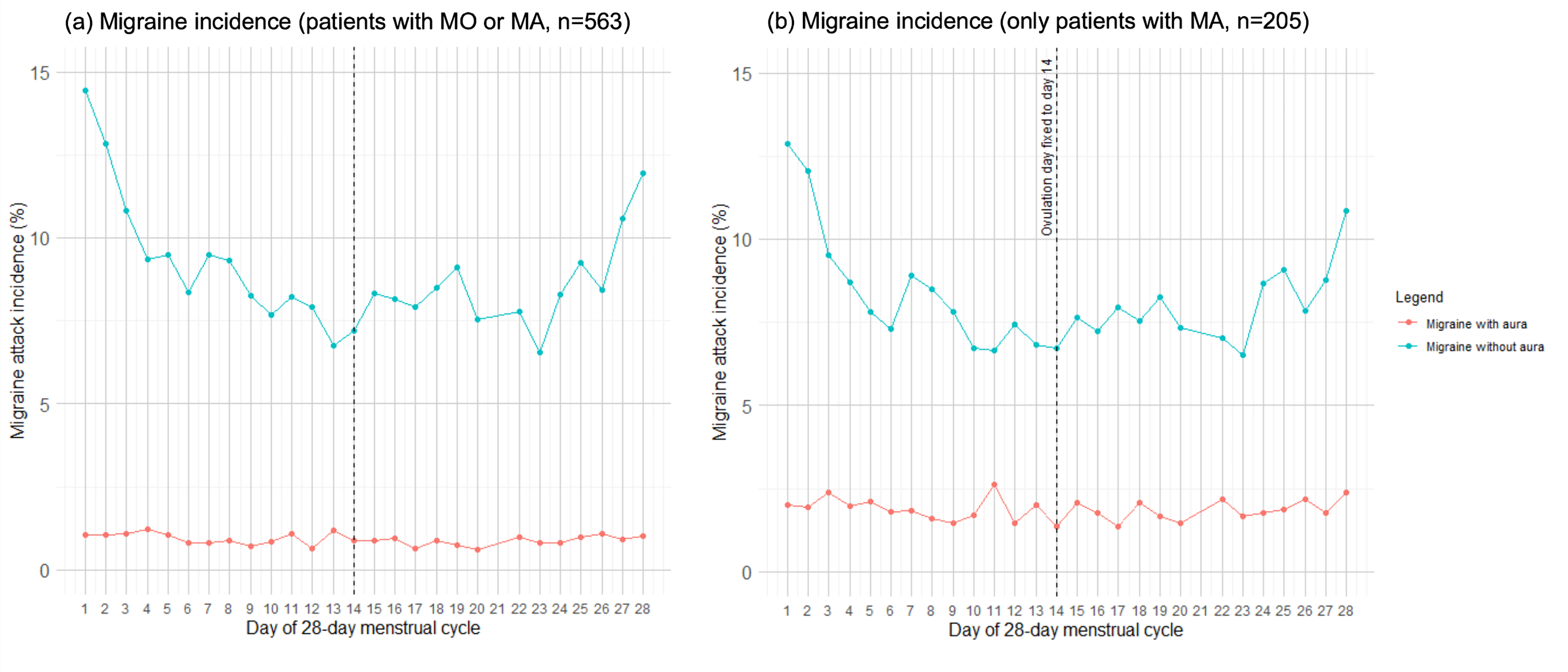
Migraine incidence by day of the menstrual cycle, standardized to 28 days and calculated day of ovulation fixed on day 14. The data is subdivided into migraine attacks with aura and migraine attacks without aura. (a) Patients with migraine without aura (MO) and migraine with aura (MA) pooled. Total of 2627 menstrual cycles from 563 patients (205 MA + 358 MO). (b) Only patients with an MA diagnosis. Total of 926 menstrual cycles from 205 patients with MA are shown.
Subgroup analyses – WHAT-hormone subgroup
In the WHAT-hormone subgroup the ovulatory window did not show an increased migraine incidence (OR: 0.68, 95% CI: 0.38–1.16, p = 0.17) (Table 5). The case-crossover design yielded an OR of 0.56 (95% CI: 0.33–0.96, p = 0.03). When days within the perimenstrual window were removed from the analysis to solely investigate the effect of ovulation compared to non-perimenstrual days, the OR attenuated to 0.66 (95% CI: 0.38–1.15, p = 0.28) (Online Supplementary Table e-1). In the SCCS model, migraine incidence was also not significantly higher during the ovulatory window when compared to the follicular phase (OR: 0.60, 95% CI: 0.34–1.03, p = 0.07), or to the luteal phase (OR: 1.00, 95% CI: 0.52- 1.94, p = 0.99) (Online Supplementary Table e-2).
Results of mixed logistic regression model in the WHAT-hormone subgroup (n = 31, 58 menstrual cycles) with the

SE, standard error; OR, odds ratio; CI, confidence interval.
Subgroup analyses – menstrual migraine group
An additional subgroup analyses was conducted for all women who met the criteria for menstrual migraine. 23 Of the 563 participants, 238 women (42%) met the criteria for menstrual migraine and 46 women (8%) could not be diagnosed due to fewer than three cycles of e-diary data. Within the menstrual migraine subgroup, the ovulatory window was also not associated with an increased migraine incidence (Online Supplementary Table e-3). In the menstrual migraine subgroup the mixed model yielded an OR of 0.98 (95% CI: 0.89–1.07, p = 0.64) which is similar to the results found in the entire cohort (OR: 0.95, 95% CI: 0.89–1.02, p = 0.13).
Sensitivity and validation analyses
To evaluate the reliability of the calculated ovulation day, we determined the mean difference between the calculated ovulation day (the 14th day before the start of menses), and the day of positive LH-surge urine test as gold standard in the WHAT-hormone subgroup. This difference was on average 0.28 days (95% CI: −0.17–0.73).
A complete case analysis was conducted by including the 105 participants initially excluded due to chronic migraine, resulting in a final cohort of 668 participants. Within this group, the ovulatory window was also not associated with migraine occurrence (OR: 0.97, 95% CI: 0.91–1.03, p = 0.31) (Online Supplementary Table e-4), which is similar to the results obtained in the primary analysis (OR: 0.95, 95% CI: 0.89–1.02, p = 0.13).
In the sensitivity analyses, where the ovulatory window was shifted one or two days earlier, the association between migraine and the ovulatory window remained consistent with the primary analysis. When shifting the window by one day (day −15 to −10), the mixed model yielded an OR of 0.93 (95% CI: 0.87–0.99, p = 0.03). When shifting the window by two days (day −16 to −11), the mixed model yielded an OR of 0.91 (95% CI: 0.85–0.98, p = 0.01). When the ovulatory window was shortened from five to three days, the OR was 0.96 for migraine during the ovulatory window (95% CI: 0.88–1.04, p = 0.34), which was also consistent with the primary analysis.
Discussion
In this study we found no increased incidence of migraine attacks (with or without aura) during the post-ovulatory phase in women. Interestingly, we observed a relatively higher incidence of migraine attacks during the follicular phase compared to the luteal phase, possibly due to the residual effects of perimenstrual estrogen decline.
Previous studies on the relationship between migraine and the menstrual cycle were often limited by imprecise cycle timing, relying on cycle-day counts from menstruation onset. In contrast, we were able to estimate the post-ovulatory window with reasonable precision in women with regular menstrual cycles. This allowed us to specifically examine the role of post-ovulatory estrogen decline.
Visual representations of migraine attack frequencies in relation to the menstrual cycle are commonly standardized to a 28-day cycle, anchored to the first day of menstruation.12–14 While this approach allows for general comparisons, it may complicate the interpretation of migraine attack incidence during the follicular and ovulatory phases when analyses are based on forward-counting cycle days (e.g., day +14) without accounting for inter-individual variability in cycle length. In such cases, phases of the menstrual cycle may not align consistently across participants, potentially obscuring phase-specific effects such as the post-ovulatory estrogen decline. However, we were able to estimate the post-ovulatory window in women with a regular menstrual cycle with reasonable precision, as supported by those who measured the LH-surge. Importantly, our findings highlight the perimenstrual window as the key period of elevated migraine incidence, rather than the ovulatory phase as sometimes perceived by women with migraine. This aligns with our previous work showing that retrospective self-reports of (menstrual) migraine are often inaccurate, and that patients may find it challenging to reliably recall the timing and frequency of migraine attacks in relation to their menstrual cycle.14,21
Contrary to our initial hypothesis based on the estrogen-withdrawal theory, our study found no evidence of increased migraine incidence during the post-ovulatory window. Since only perimenstrual declining estrogen and not ovulatory declining estrogen seem to be linked to migraine, a possible explanation may be that a sustained period of high estrogen is necessary to prime the system, making the subsequent drop more likely to trigger migraine attacks.7,13 In contrast, the post-ovulatory estrogen decline occurs after a shorter period of estrogen exposure and may therefore be less likely to elicit this effect. Another possible explanation for the lower migraine incidence around ovulation may involve protective effects from progesterone or its derivative, allopregnanolone, both of which increase during ovulation but are low during the menstruation. 15 Both enhance GABAergic (gamma-aminobutyric acid) activity, stimulating antinociceptive effects, potentially counteracting migraine susceptibility during the post-ovulatory phase.26,27
A limitation of our study might be some missing e-diary data, although adherence was high (99% [IQR 96%–100%]), and imputing missing data as either migraine or non-migraine days had no impact on the results. It should be noted that menstrual migraine was diagnosed in 42% of women, a lower rate than previously reported. 14 This difference is probably due to the exclusion of hormonal contraceptive users, who often experience increased migraine attack incidence during the hormone-free interval, but were excluded for the current analyses. Our previous research has demonstrated that accurately diagnosing menstrual migraine requires the use of e-diaries. 14 Therefore, we are confident that the proportion of menstrual migraine reported in this study, and consequently our subgroup analysis, is valid. Further, this study included only female participants who self-identified as women and with a menstrual cycle, and our findings may not be generalizable to all women. Secondly, in the larger cohort, ovulation was estimated rather than directly measured using LH-surge testing, introducing the possibility of some misclassification. However, anovulatory cycles are relatively uncommon among regularly menstruating women, with prior studies reporting the prevalence of ovulatory cycles based on menstrual history to be 91.9–99.5%.28,29 Therefore, any residual misclassification of ovulation is expected to be limited. Thirdly, using multiple within-person analyses, we found no group-level increase in migraine during the post-ovulatory window. However, these methods might still miss small subgroups with distinct trigger patterns, so future longitudinal studies tracking multiple triggers (including hormonal changes) may be useful to fully exclude the possibility of a post-ovulatory effect in a (very) small subset of women. Finally, the estimation of both the perimenstrual and post-ovulatory windows remains somewhat arbitrary. The post-ovulatory estrogen decline is theoretically shorter in duration than the perimenstrual decline; however, sensitivity analyses restricting the post-ovulatory window to three rather than five days produced similar results. The definition of the perimenstrual window was originally proposed in 2000, based on evidence of increased migraine incidence during the two days preceding menstruation and the first two days of menses, 10 and has since been widely adopted. Our findings, however, suggest that migraine risk may persist into the early follicular phase, supporting the consideration of an extended perimenstrual window. This may also have consequences for the definition of menstrual migraine by the ICHD classification.
This study has several strengths. We used three statistical models, each offering different advantages. The mixed-effects model accounts for individual-level variability by incorporating random effects, making it well-suited for longitudinal data and accommodating time-varying covariates. In contrast, both the case-crossover and self-controlled case series (SCCS) models effectively eliminate time-invariant confounders by using each participant as their own control, enhancing internal validity. Additionally, these self-controlled models are more efficient, reducing the need for large control groups. The SCCS model further allows for an overview of the relative risk of migraine across different phases of the menstrual cycle, as it is specifically designed to assess time-dependent risks within individuals. Our data are collected with the Leiden validated headache e-diary with an automated algorithm to evaluate whether each headache day met migraine criteria.21,23 Diaries were time-locked after 48 h, reducing recall bias. The large cohort of 563 participants with recordings of 2627 menstrual cycles improves generalizability, and diaries of participants were collected as part of research but also as part of real-world studies as well as standard clinical care, making the results more applicable to the overall migraine population. 19 An additional strength of the study is the use of LH-surge urine tests in the WHAT-hormone subgroup to determine cycle timing. In this subgroup, we confirmed that estimating the LH surge as the 14th day before menses is reasonably accurate in women with regular menstrual cycles. On average, the calculated LH surge deviated by only 0.28 days from the actual day.
In conclusion, our study found no increased incidence of migraine attacks (with or without aura) during the post-ovulatory phase in women, but a higher incidence during the follicular phase compared to the luteal phase. Thus, the common belief among women with migraine that the post-ovulatory phase is a high-risk period for attacks is not supported.
Key Findings
The post-ovulatory estrogen decline was not associated with increased migraine attacks.
The perimenstrual window showed the highest migraine incidence, consistent across all analytic models.
Ovulation timing estimated from menstruation was validated by LH-surge testing (mean difference 0.28 days).
Only migraine without aura followed hormonal patterns; attacks with aura remained stable.
Results challenge the common belief that ovulation increases migraine risk.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261436415 - Supplemental material for Migraine attack incidence in relation to the post-ovulatory estrogen decline: A prospective cohort study
Supplemental material, sj-docx-1-cep-10.1177_03331024261436415 for Migraine attack incidence in relation to the post-ovulatory estrogen decline: A prospective cohort study by Britt W.H. van der Arend, Sally Bakker, Daphne S. van Casteren, Iris E. Verhagen, Nadine Pelzer, Irene de Boer, Suzanne C. Cannegieter, Antoinette MaassenVanDenBrink and Gisela M. Terwindt in Cephalalgia
Footnotes
Ethical considerations
Ethical approval for the anonymized use of headache e-diaries was obtained from the Medical Ethical committee of the Leiden University Medical Centre (LUMC). Participants did not have to provide additional informed consent. Patients in the LHC database that actively declared that they did not want to participate in medical research were not screened for eligibility. For the WHAT-hormone subgroup in which ovulation tests were performed, additional ethical approval was obtained by the Medical Ethical committee of the LUMC (NL74161.058.20).
Consent to participate
All these participants provided written informed consent.
Author contributions
GMT, DSvC and AMvdB contributed to the funding acquisition. BWHvdA, SB, GMT and SCC contributed to the conceptualization, methodology and formal analysis. BWHvdA and SB contributed to the visualization, data curation and project administration. BWHvdA, SB and GMT wrote the original draft. All authors revised and edited the manuscript and approved the final version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by grants from the Dutch Research Council (849200007) and the Dutch Brain Foundation (HA2017.01.05).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BWHvdA, DSvC and IEV report grants from the Dutch Research Council and the Dutch Brain Foundation.
SCC and SB have no conflicts of interest.
IdB reports independent support from the Dutch Heart Foundation and the Clayco foundation.
AMvdB received research grants and/or consultation fees from AbbVie, Amgen/Novartis, Eli Lilly, Lundbeck, Manistee, Pfizer, Satsuma, Teva, and Tonix.
GMT and NP report grants or consultancy support from AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Teva, and independent support from Dutch Research Council, European Community, Dutch Heart Foundation, and Dutch Brain Foundation, Dioraphte, and the Clayco foundation.
Data availability statement
Anonymized individual participant data, along with a data dictionary that explains each field, will be available to others upon reasonable request after the planned publications have been finalized and a data sharing agreement has been signed. To submit a request, please contact GM Terwindt at g.m.terwindt@lumc.nl.
Supplemental material
Supplemental material for this article is available online
