Abstract
Observational studies have linked migraine to a wide range of risk factors and diseases, although the causality of these associations is uncertain. Mendelian randomization (MR) is an analytic paradigm that exploits germline genetic variants as natural experiments, providing a framework for causal inference from observational data. In this narrative review, we summarize the core principles and assumptions of MR and highlight methodological considerations unique to its application to migraine research. We review key MR findings across several domains of migraine research, noting areas of consistent evidence, as well as conflicting results. We then discuss limitations of MR in the context of migraine research, emphasizing that MR is not a panacea for causal inference. We conclude by underscoring the need for interdisciplinary collaboration between methodologists and domain experts to prioritize patient-relevant questions, refine analytic approaches and translate genetic insights into more effective therapies.
This is a visual representation of the abstract.
Introduction
Migraine is a prevalent and leading cause of disability worldwide (1). To mitigate this disease burden, epidemiologic research has sought to identify associations between risk factors and migraine, including lifestyle behaviors (2), circulating biomarkers (3) and other medical conditions (4). Conversely, migraine itself has been examined as a risk factor for other adverse clinical outcomes. Among the most prominent of these are associations with greater risk of cardiovascular disease (5) to the extent that migraine is now included in the QRISK3 cardiovascular risk prediction algorithm (6).
Although numerous risk factors for migraine have been identified in observational studies, their causal contributions to disease risk remain uncertain. This uncertainty stems from the fact that, although traditional observational methods can identify associations, they are subject to inherent limitations that limit causal inference. Consequently, therapeutic strategies informed by observational studies are often not validated by subsequent randomized controlled trials, which constitute the gold standard for establishing causality (7). Residual confounding and reverse causality are among the most important biases that impact reliable inference in observational studies (Figure 1). Residual confounding arises when an unmeasured or inaccurately measured variable influences both the exposure and the outcome, leading to biased effect estimates. Reverse causality occurs when an apparent link association of an exposure with an outcome is actually driven by an unrecognized effect of the outcome on the exposure. This is particularly problematic for investigations of migraine, where the disorder itself alters behaviors such as diet, sleep and physical activity. Prospective, controlled studies can partially mitigate biases arising from confounding and reverse causality, although they cannot eliminate them entirely. These challenges highlight the need for alternative study designs for causal inference that avoid these biases.

Directed acyclic graph demonstrating the relationships investigated in a Mendelian randomization (MR) analysis. The β symbols refer to the quantitative measure of association: βO is the association of exposure with outcome in observational epidemiology; βGE is the association of the genetic instrument with the exposure; βGO is the association of the genetic instrument with the outcome. Solid arrows denote direct relationships. Direct relationships between the instrument and the outcome are biologically possible, but are considered pleiotropic and violate the assumptions of MR. Instead, the validity of MR requires that the effect of the instrument on the outcome should be mediated solely by the effect of the instrument on the exposure, The dashed line indicates the typical lack of confounding and reverse causation in genetic association that accounts for population structure, as emphasized by the “X” symbol. GWAS = genome-wide association studies; SNP = single nucleotide polymorphism.
Natural experiments have been applied across multiple research domains to strengthen causal inference in observational settings (8). These designs leverage scenarios in which exposure to a risk factor or intervention is driven by an external factor that results in approximate random allocation. By mimicking certain features of randomized controlled trials, natural experiments can identify effects that are less susceptible to conventional biases of observational research(9).
Human genetic variation provides a naturally randomized source of biological variation that can be leveraged under the natural experiment framework (10). Germline genetic variants are randomly assorted at gametogenesis and hence, under certain assumptions, are immune to environmental confounding. Moreover, germline genetic variants cannot be modified by disease processes, and therefore remain nearly unaffected by reverse causation. Mendelian randomization (MR) is an analytic technique that capitalizes on these properties by using genetic variants as proxies for exposures to infer causal effects from observational data (10,11). MR has been increasingly applied to investigate causal relationships in neurological diseases, including migraine (12). The first such application in migraine, published in 2017, evaluated the effect of serum calcium levels on migraine risk (13). Since then, the field has expanded rapidly: the number of published manuscripts using MR increased from 7 in 2020 to 57 in 2024 alone. However, the MR methodology is often inappropriately applied or interpreted, resulting in confusing and inconsistent findings (14). This highlights the importance of rigorous methodology and cautious interpretation to avoid unwarranted causal claims from MR analyses.
In this review, we provide a primer for understanding the application of MR to migraine research. We first outline the core assumptions and analytical framework of MR, and then summarize key findings from studies examining the causes and consequences of migraine. Finally, we discuss methodological challenges unique to migraine research and highlight priorities for future work.
Mendelian randomization methodology
Overview
In an MR analysis, genetic variants, typically common (allele frequency >1%) single nucleotide polymorphisms (SNPs), are used as proxies for an exposure to estimate its effect on an outcome. In the language of instrumental variable analysis, these variants are described as instruments for the exposure. Unlike studies focused on disease risk prediction or discovery of risk loci, MR uses these variants solely as tools for causal inference to support or refute a causal effect.
For valid causal inference in MR, the genetic variants used as instruments must meet three core assumptions. First, they must be strongly associated with the exposure. Investigators typically select variants that associate with the exposure at genome-wide statistical significance (p < 5 × 10−8), a threshold which controls for hypothesis testing across millions of variants. More lenient thresholds are sometimes used to increase the number of variants in an MR analysis, although this comes at the cost of including invalid instruments and introducing bias (15). These genetic associations are drawn from the summary statistics of genome-wide association studies (GWAS), which are now publicly available for thousands of traits. Summary-level data can also be used to obtain estimates of genetic associations with the outcome from a non-overlapping sample, in what is known as a two-sample MR design. By contrast, one-sample MR, which uses individual-level data from a single cohort, is less commonly applied due to limitations in data availability and potential bias from sample overlap (16).
The second MR assumption is that the association of the genetic variants with the outcome must be independent of confounders. An important source of confounding in this context is population stratification, in which genetic ancestry or population substructure correlate with variant allele frequencies and are in turn associated with the outcome. This potential bias may be mitigated through several analytic strategies, including restriction of analyses to homogeneous ancestry groups and application of statistical methods that adjust for genetic ancestry.
The final, and most stringent, analytic assumption is that the genetic variants used in the MR analysis impact the outcome solely through their effects on the exposure. Variants that exert effects on the outcome via other pathways are considered pleiotropic, and their inclusion in an MR analysis can bias results. The development of statistical methods to detect and mitigate this bias has been a central priority in methodological development for MR (17,18). Nevertheless, violations of the second and third MR assumptions are impossible to rule out entirely and represent inherent limitations of the approach.
Calculation of MR estimates
Statistical methods for MR are implemented in multiple publicly available software packages and are only briefly reviewed here (19,20). At the population level, a genetic instrument is interpreted as inducing differences in the mean level or risk of the exposure according to which alleles are inherited. Under a causal framework, this change in the exposure is expected to induce a consequent difference in the mean level or risk of the outcome. The Wald ratio quantifies this effect by dividing the variant's association with the outcome by its association with the exposure, providing an estimate of how much the outcome changes per unit difference in the exposure (Figure 1). In MR analyses involving multiple variants, individual Wald ratios may be combined using meta-analysis, with standard errors estimated using the delta method. Implicit in the interpretation of MR associations is the assumption that an exposure affects the outcome through a similar mechanism regardless of whether its variation arises from genetic or environmental factors (21).
GWAS data sources for migraine
Table 1 summarizes some GWAS data sources that have been used in two-sample MR analyses of migraine. The first migraine GWAS, published in 2010 by the International Headache Genetics Consortium (IHGC), included 5933 cases and identified a single genome-wide significant locus, although this association has not been replicated in subsequent studies (22). The following year, a second migraine GWAS reported three loci that have since been consistently replicated (23). Successive meta-analyses have rapidly increased case counts to 102,084 and identified 123 loci, including variants near genes that encode drug targets for migraine (e.g. HTR1F and CALCA/CALCB) (24,25).The most recent GWAS includes the largest sample sizes to date for the subtypes of migraine with aura (MA) and migraine without aura (MO), though statistical power remains limited for these outcomes. In parallel, the public release of summary statistics for a wide range of metabolic, lifestyle, cardiovascular and molecular traits has enabled efficient application of two-sample MR to investigate potential causal risk factors for migraine.
Genome-wide association study (GWAS) data sources for migraine research.
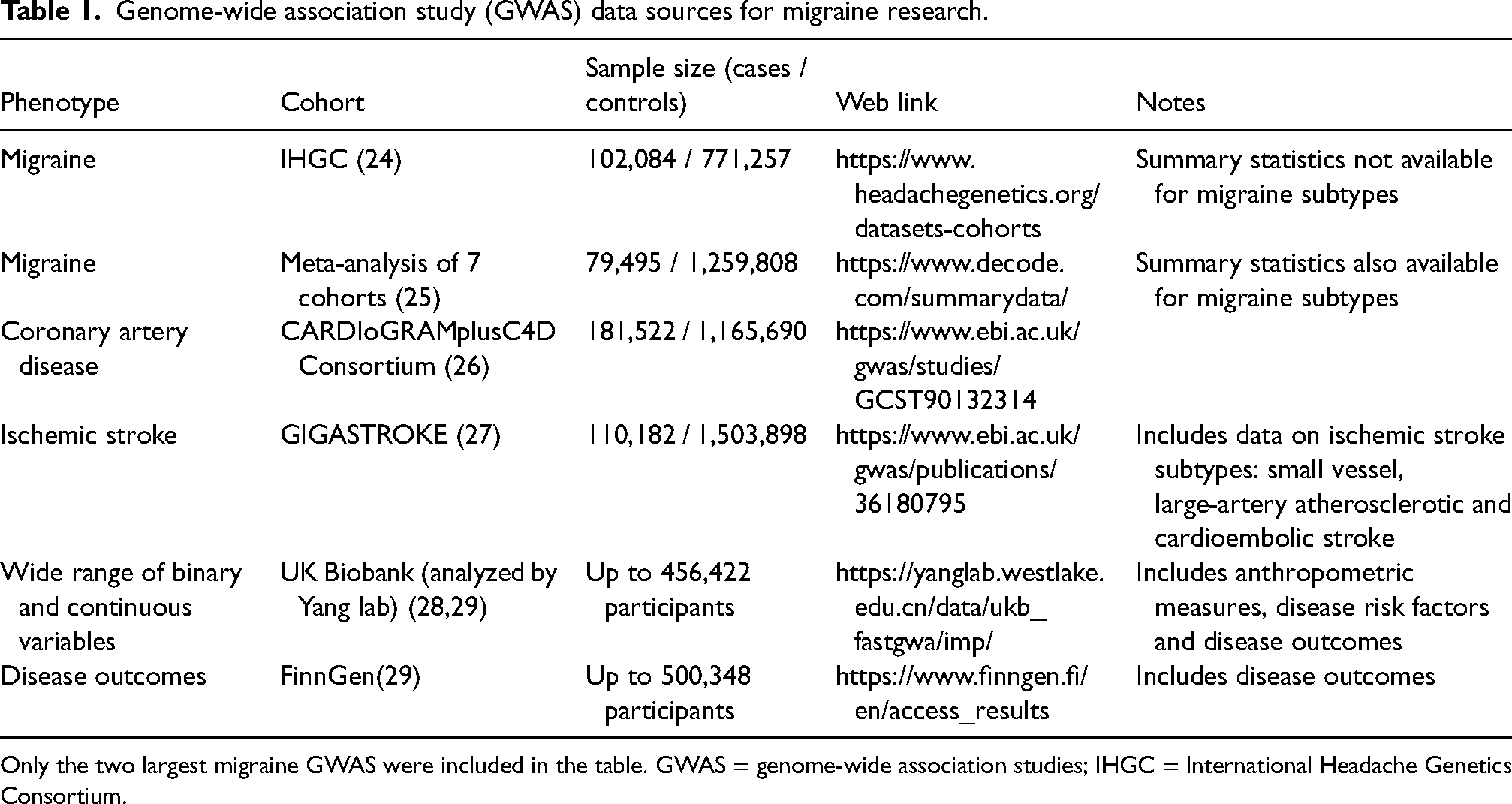
Only the two largest migraine GWAS were included in the table. GWAS = genome-wide association studies; IHGC = International Headache Genetics Consortium.
Mendelian randomization studies of the causes and consequences of migraine
We now provide a non-exhaustive overview of MR analyses that have leveraged these GWAS data sources to investigate the causes and consequences of migraine. In view of the large number of tested hypotheses, we focus on findings that surpassed study-specific statistical significance thresholds. Throughout, we highlight consistencies in findings at the same time as noting limitations that merit further investigation.
Vascular disease
For several decades, migraine was thought to be primarily initiated by vasodilation of meningeal and cerebral arteries, although this “vascular theory” of migraine has since been challenged by experimental physiological evidence (30,31). An alternative approach to investigating a vascular contribution to migraine pathophysiology would be to demonstrate evidence for associations between migraine and cardiovascular disease risk. Indeed, several large epidemiological studies have demonstrated independent associations between migraine and increased risk of incident stroke, coronary artery disease (CAD) and atrial fibrillation (6,32,33). These associations have been particularly pronounced for the subtype of migraine with aura. Whether these relationships are causal remains uncertain.
We have performed several MR analyses testing the associations between migraine and vascular diseases (Figure 2) (34–37). In one of the earliest MR analyses applied to migraine, we investigated the association of genetic liability to migraine with risk of CAD and atrial fibrillation (36). In contrast to findings from observational studies, these analyses found a protective association between genetic liability to migraine and CAD risk, as well as no association with atrial fibrillation. These results raise the possibility that the observational association between migraine and increased CAD risk may be biased. Moreover, recent epidemiological studies have shown that individuals at high cardiovascular risk are less likely to have active migraine or develop incident migraine (38,39). The mechanisms underlying the protective effect warrant further investigation, and may involve developmental differences in the vasculature of patients with migraine, or differences in protective lifestyle behaviors in migraine patients such as avoidance of alcohol (36).

Flowchart for conduct of Mendelian randomization (MR) analysis. GWAS = genome-wide association studies.
In a subsequent MR analysis, we investigated the association of genetic liability to migraine with ischemic stroke subtypes and cervical artery dissection (35). We identified an inverse association between genetic liability to migraine and large-artery atherosclerotic stroke, a stroke subtype that shares pathophysiological features with CAD. This finding was replicated by an independent research group analyzing the same datasets (40). There was no evidence for association with cardioembolic stroke, nor was there an association with small vessel stroke. However, genetic liability to migraine was associated with increased risk of cervical artery dissection, which is consistent with findings from observational research (41).
As another form of cerebrovascular disease, we investigated genetic liability to migraine in relation to intracranial aneurysm risk and found an association with increased risk of intracranial aneurysm and subarachnoid hemorrhage (34). Parallel analyses of abdominal and thoracic aortic aneurysms yielded similar associations, further supporting an influence of migraine liability on both intracranial and extracranial vasculature (34). Reverse MR analyses showed no association of genetically proxied intracranial aneurysm risk with migraine. This suggests that, on average, intracranial aneurysms do not cause headache, although we cannot exclude effects of aneurysm subtypes that are poorly represented in the studied populations (e.g. giant or rapidly growing aneurysms). These findings collectively suggest that genetic liability to migraine protects against atherosclerotic disease while predisposing to diseases of arterial wall elasticity and structural integrity (Figure 2).
MR has also been applied to investigate causal relationships between cardiovascular risk factors and migraine. We found evidence for a causal effect of higher diastolic blood pressure (DBP) adjusted for systolic blood pressure (SBP) on increased risk of migraine (37). This association was also reported in two independent MR analyses (42). Conversely, we found that genetically proxied higher pulse pressure and higher SBP adjusted for DBP were associated with reduced risk of migraine. In the reverse causal direction, genetic liability to migraine was associated with lower SBP and lower pulse pressure, but not with DBP. These findings provided further evidence supporting a vascular contribution to migraine risk.
In contrast, several cardiovascular risk factors have not been found to relate to migraine risk in MR analyses. Our group found no evidence from MR analyses for causal associations between migraine and lipid subfractions (43). In unidirectional MR analyses, Siewert et al. (42) also found no evidence for an association between genetically proxied low-density lipoprotein cholesterol or type 2 diabetes and risk of migraine. In a subsequent MR analysis that leveraged a larger migraine GWAS, Islam et al. (44) also detected no significant causal effects of glycemic traits on migraine risk, nor any reverse effects.
Taken together, these genetic findings support a vascular contribution to migraine pathophysiology. An emerging hypothesis is that migraine may serve as an indicator of arterial health, reflecting a tradeoff between increased susceptibility to disorders of vessel wall elasticity and structural integrity and reduced risk of atherosclerotic disease. Further research aimed at elucidating these mechanisms may reveal new therapeutic opportunities for both migraine and vascular disease.
Gastrointestinal diseases
Gastrointestinal symptoms are common in migraine, with emesis and nausea included in the International Classification of Headache Disorders criteria for migraine (45). Additionally, gastrointestinal diseases have long been recognized as comorbid with migraine (46). Several MR analyses have been conducted to investigate whether these associations may be causal. Welander et al. (47) investigated bidirectional relationships between genetic liability to migraine and risks of inflammatory bowel disease and celiac disease. They found no convincing evidence for causal associations in either direction. There was weak, nominal evidence suggesting an effect of genetic liability to celiac disease on migraine subtypes, although these findings did not survive correction for multiple testing (Figure 3).

Divergent vascular effects of genetic liability to migraine. Red, pointed arrows indicate pathways that confer increased risk. Black arrows with a flathead indicate pathways that confer reduced risk. The “X” indicates the absence of evidence for a causal effect in either direction. Generated using the BioRender software ( https://www.biorender.com).
In a subsequent analysis using distinct analytic approaches, Chasman et al. (48) examined the bidirectional associations between genetic liability to migraine and gastrointestinal diseases. This study replicated the null association between genetic liability to migraine and inflammatory bowel disease. Although migraine and irritable bowel syndrome frequently co-occur, there was no evidence to support a causal relationship in either direction, suggesting that their comorbidity reflects shared etiological factors. By contrast, there was evidence for associations between genetic liability to several non-inflammatory gastrointestinal disorders (peptic ulcer disease, gastroesophageal reflux disease and diverticular disease) and migraine. However, in a random-effects inverse-variance weighted sensitivity analysis aiming to account for heterogeneity in SNP-specific MR estimates, only the association of diverticular disease with migraine remained statistically significant. Although brain–gut pathways may provide a broad conceptual framework for this finding, there is no strong a priori mechanistic explanation, particularly given the distinct tissue types involved in the pathogenesis of these disorders.
Cognition, dementia and neuroimaging phenotypes
Epidemiologic studies have reported associations between migraine and incident dementia, raising interest in a potential causal link (49). To evaluate this relationship, we conducted MR analyses examining the association of genetic liability to migraine with Alzheimer's disease (AD) and cognitive performance (50). We found no evidence in these analyses for a causal association. By contrast, subsequent MR analyses reported positive associations between genetic liability to migraine and AD risk (51–53). However, methodological limitations complicate the interpretation of these findings. Several of these studies relied on migraine GWAS summary statistics from a single cohort (e.g. UK Biobank) to identify genetic proxies for migraine, capturing as few as 9% of the genetic instruments available in more recent, large-scale GWAS (52,53). Furthermore, these three studies utilized a GWAS of AD that included proxy-defined cases obtained using family history. This approach has been shown to be biased and can lead to misleading MR estimates (54). These limitations underscore the need for updated MR studies leveraging comprehensive genetic instruments and rigorously phenotyped AD cases.
The first MR investigation of magnetic resonance imaging (MRI)-derived brain volume phenotypes found no evidence for association (50). Three subsequent MR studies have investigated this relationship and yielded mixed results (Table 2). One of these studies reported no significant associations after applying Bonferroni correction for multiple comparisons (Table 2) (55). Zhang et al. (56) identified a protective association between thickness of the right superior parietal gyrus and reduced risk of migraine. Mitchell et al. (57) reported that increased size of the ventral diencephalon was associated with reduced migraine risk, and that genetic liability to migraine was linked to increased amygdala volume. Drawing definitive conclusions from these studies is challenging due to the lack of consistent or replicated findings, and methodological limitations in the application of MR to investigations of MRI-derived phenotypes (see Discussion). These results should therefore only be regarded as hypothesis-generating.
Comparison of Mendelian randomization studies examining the association of genetic liability to migraine with MRI-derived brain volume metrics.

The table only includes MR associations that were statistically significant after correction for multiple comparisons. GWAS = genome-wide association studies; IHGC = International Headache Genetics Consortium; MRI = magnetic resonance imaging.
Health-related behaviors
MR has been applied to investigate bidirectional relationships between migraine and health-related behaviors. Yuan et al. (64) investigated the bidirectional associations of genetically proxied alcohol consumption, smoking and coffee consumption on risk of migraine in the UK Biobank and FinnGen cohorts. Genetic liability to smoking initiation was positively associated with migraine risk, whereas coffee consumption showed a protective association. We extended this approach to sleep traits, finding significant evidence for an association between genetically proxied difficulty awakening (as a proxy for sleep inertia) with increased risk of migraine, and nominal evidence for an association between genetic liability to insomnia (65). This observation aligns with emerging clinical evidence that treatment of insomnia may reduce migraine frequency (66).
Molecular factors
Several targeted MR analyses have investigated the association of molecular factors with migraine risk. The first published MR study of migraine investigated the association of serum calcium levels with migraine risk, finding evidence that increased serum calcium increases migraine risk (13). However, this association was non-significant when examined in a subsequent, larger iteration of the IHGC migraine GWAS (42,67). Several circulating proteins have been investigated in isolation for their association with migraine, including insulin-like growth factor 1 (protective) (68), hypoxia-inducible factor 1-α (null) (69) and soluble Trem1 (nominal but non-significant after multiple comparisons adjustment) (70).
Several hypothesis-free MR investigations of molecular factors and migraine have been undertaken. In an analysis of 316 unique metabolites, Tanha et al. (71) identified potential causal effects between lower levels of X-11315, higher levels of LPE(20:4) and shorter fatty acid length on risk of migraine. They triangulated this finding with preliminary evidence from the migraine clinical literature suggesting benefit from supplementation with fish oil rich in very long-chain omega-3 fatty acids (71). Tanha et al. (72) also examined the association of 10 common laboratory-based tests, including lipids, iron status and liver enzyme tests, with migraine. The only association surpassing the Bonferroni-adjusted threshold was gamma-glutamyl transferase, although sensitivity analyses indicated the association was likely biased by horizontal pleiotropy.
Guo et al. (73) investigated twelve coagulation cascade-related measures and found that higher levels of coagulation factor VIII, von Willebrand factor levels and phosphorylated fibrinopeptide A levels were associated with increased risk of migraine. Stratified analyses suggested more pronounced effects for MA. Sensitivity analyses showed that the association of coagulation factor VIII with migraine was driven by genetic instruments near the pleiotropic ABO locus, and that independent associations between factor VIII and von Willebrand factor could not be distinguished (73). Although these findings hint at a connection between coagulation function and migraine, they rely on trans-acting variants located far from the genes encoding the relevant proteins, which makes pleiotropy more likely than with cis-acting variants situated near those genes (74,75). Of the proteins significantly associated with migraine in the primary analysis, only von Willebrand factor could be tested in sensitivity analyses restricted to cis-acting variants, which yielded effect estimates similar to the primary analysis, although with attenuated significance potentially due to reduced power (73).
Challenges and future directions
Conventional limitations of MR
All MR analyses are susceptible to bias from pleiotropic effects of variants that influence the outcome through pathways independent of the exposure (10). Although sensitivity analyses can provide some reassurance, they cannot fully exclude this potential bias. Additionally, genetic variants typically explain a modest proportion of variation in the exposure, which can limit statistical power. This limitation may be evaluated through power calculations or by examining the width of confidence intervals around MR estimates (76).
Interpreting effect estimates from MR analyses also requires attention to several additional assumptions and limitations (10). MR analyses estimate the impact of lifelong changes in an exposure on the outcome. In the case of the DBP analysis (37), the association represents the effect of a lifelong increase in DBP on migraine. This contrasts with randomized controlled trials which typically evaluate short-term effects of interventions. For exposures that exert a cumulative or time-dependent effect on an outcome, the MR estimate may be larger than the effect observed in interventional studies, although the effect directionality should be preserved. This may also impact the interpretation of biological effects that are age-dependent or emerge only during specific developmental windows, such as for MRI-derived brain volume measures. Additionally, conventional MR analyses assume linearity in the exposure-outcome relationship (77), which precludes assessment of non-linear effects and inference about extreme exposure levels. For example, findings from the MR analysis of DBP and migraine may not generalize to severe diastolic hypertension or hypotension.
In addition, the interpretation of MR analyses of binary exposures (e.g. migraine) is not straightforward. Such analyses do not estimate the effect of transitioning from the unexposed (e.g. migraine-free) to the exposed state (e.g. with migraine) but instead estimate the effect of genetic liability: a latent, continuous predisposition that influences the likelihood of developing the exposure. As an example of how this is expressed, in the MR analysis of migraine and subarachnoid hemorrhage, the MR estimate is reported as the odds ratio of subarachnoid hemorrhage per doubling in migraine risk (34). Point estimates from these analyses should not be directly compared with corresponding observational epidemiological estimates that use migraine status as an exposure in epidemiological studies. For these reasons, it has been proposed that MR estimates be interpreted primarily as a test of the causal null hypothesis, providing evidence about directionality rather than reliable estimates of effect size (21).
Challenges with MR analyses of neuroimaging features
MR studies of migraine and radiological outcomes have substantial analytic flexibility, including in the choice of GWAS sources for both migraine and brain volume traits. Because this flexibility allows many different analytic combinations and implicit hypothesis tests, it increases the risk of reporting false positive findings. Additionally, some volumetric measures encompass anatomically and functionally distinct subregions (e.g. thalamic volume), making it difficult to determine which components are specifically affected by migraine. Moreover, GWAS of neuroimaging features are often measured at a single time point per participant, which precludes determination of when during brain development a certain region may be affected by liability to migraine. Systematic approaches that rigorously control for false-positive rates and include independent replication would enhance the reliability of these findings.
Phenotyping challenges
GWAS of migraine subtypes have had limited sample sizes. MR analyses applied to these data are consequently underpowered, precluding assessment of subtype-specific causal effects. This limitation is particularly salient for MR analyses of vascular outcomes because MA has been selectively associated with cardiovascular disease in several epidemiological studies (78–80). There may also be differences in causal effects of migraine on vascular disease according to age and sex, and delineating these differential effects requires stratified, individual-level analyses. However, the recent expansion of migraine GWAS sample sizes may soon permit well-powered MR investigations of subtype, age and sex-stratified effects (24).
Addressing questions that inform therapeutic development and clinical decision-making
To date, MR investigations of migraine have not yielded insights that have meaningfully improved patient care. Although this may reflect the relative nascency of the field, several study design-related factors may also contribute to the limited clinical impact. First, all studies referenced in this review relied on GWAS of migraine case–control status, and therefore primarily inform the biology of migraine susceptibility. While the pathophysiology underlying susceptibility is likely relevant to therapeutic strategies for individuals with established migraine, the genetic determinants of migraine severity may differ from those that drive disease onset (81–84). MR analyses leveraging such outcomes may more directly generate insights that translate into therapeutic advances (85). Second, therapeutic insights may be accelerated through targeted application of the MR drug target paradigm to migraine. In this framework, the exposure is defined as perturbation of a protein drug target, allowing inferences that more directly inform drug development. This approach can be applied both to identify opportunities for drug repurposing and to evaluate the likely efficacy and safety of emerging targets (e.g. through phenome-wide MR analyses). Finally, confidence in MR findings can be strengthened through independent replication, which is becoming increasingly feasible with the widespread availability of GWAS data, particularly across diverse ancestries. Confidence can be further increased through triangulation using orthogonal forms of genetic analyses (e.g. rare variant analyses), as well as omics-based methodologies (e.g. proteomics).
Peer review
A thorough evaluation of findings from an MR analysis of migraine requires expertise in the disease process, genetic epidemiology, and the clinical context needed to prioritize relevant research questions. Identifying reviewers with this breadth of expertise is challenging, particularly with the increasing demands on the time of referees. While published recommendations and guidelines can assist in manuscript assessment, close collaboration between journals and experts in the field is also necessary to ensure the continued publication of high-quality studies (14). Taken together, these limitations highlight the importance of triangulating MR results with complementary methodologies, distinct data sources and the published literature (86).
Conclusions
MR has provided insights into the causes and consequences of migraine, although cautious interpretation of findings is essential. Cross-domain collaboration will be key to advancing the field and translating findings into tangible improvements in patient care.
Article highlights
Observational studies have linked migraine to a wide range of risk factors and diseases, though the causality of these associations is uncertain. MR is an analytic paradigm which exploits germline genetic variants as natural experiments, providing a framework for causal inference from observational data, and has been increasingly applied to investigate migraine and related disorders.
In this narrative review, we first summarize the core principles of MR and highlight methodological considerations unique to migraine research. We then synthesize key MR findings across several domains, noting areas of consistent evidence, as well as conflicting results. Finally, we discuss key limitations of MR in the context of migraine research, emphasizing that MR is not a panacea for causal inference.
We conclude by calling for closer collaboration between methodologists and disease domain experts to prioritize research questions, refine analytic approaches, and translate genetic insights into improved therapeutic strategies.
Footnotes
Author contributions
Iyas Daghlas: conceptualization, Investigation, Writing – original draft preparation.
Daniel I. Chasman: conceptualization, investigation, supervision, writing – original draft preparation, writing – review & editing.
Data availability
No new data were generated or analyzed in support of this research.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
Some of the research described in this review was supported by 1R21NS104398 from National Institute of Neurological Disorders and Stroke (NINDS)/NIH to DIC.
