Abstract
Objective
To investigate potential causality between genetic liability to migraine and Alzheimer’s disease, intelligence, and brain volume using two-sample Mendelian randomization.
Methods
The exposure consisted of independent genetic variants associated with migraine in the largest (59,674 cases/316,078 controls) published genome-wide association study. Outcomes included Alzheimer’s disease (71,880 cases/383,378 controls), a measure of general intelligence (n = 269,867), intracranial volume (n = 11,373), and seven subcortical brain volumes (n ∼ 13,000), all with available genome-wide association study summary statistics. Mendelian randomization effects were estimated using inverse-variance weighted analysis.
Results
Genetic liability to migraine did not associate with Alzheimer’s disease (odds ratio [95% confidence interval] 1.01 [1.00–1.02], p = 0.07), intelligence (standardized beta [95% confidence interval] 0.01 [0.00–0.02], p = 0.13), or any brain volume measures (all p > 0.05). No individual migraine variant associated with any of the outcomes at genome-wide significance.
Conclusions
These data do not support a causal effect of migraine liability on Alzheimer’s disease, intelligence, or brain volume.
Introduction
Cognitive impairment during episodes of migraine headache is a well-established phenomenon; however, whether migraine influences interictal cognitive function and dementia risk is controversial (1). Plausibility for this link is supported by observations of a higher burden of clinically silent brain lesions (1) and smaller regional brain volumes (2) in patients with migraine. Additionally, a recent prospective cohort study (3) related migraine history to increased Alzheimer’s disease (AD) risk. In contrast, prospective population-based studies have not linked migraine to cognitive decline (1). Given the prevalence of migraine, establishing a causal link with poorer cognition would have implications for many patients, and could support migraine treatment as a strategy to slow cognitive decline.
Determining causality in observational research is limited by potential for residual confounding and reverse causality (4). These limitations may be overcome by using Mendelian randomization (MR), an analytic approach that uses genetic variants as instrumental variables for exposures to estimate causal effects on outcomes (4). Causal inference in MR relies upon the random assortment of genetic variants at gametogenesis, which reduces confounding and establishes temporal sequencing of exposure to outcome. We therefore leveraged MR to examine evidence for a causal effect of genetic liability to migraine on measures of AD risk, general intelligence, and regional brain volumes.
Methods
Exposure GWAS
We utilized data from the largest published genome-wide association study (GWAS) of migraine (5) to construct an instrumental genetic proxy for migraine liability (59,674 migraine cases and 316,078 controls, all of European ancestry). Participants were drawn from 27 population-based cohorts and six tertiary headache clinics. Cases were ascertained using physician diagnosis and/or self-report using validated questionnaires. As a validation dataset for the genetic instrument, we utilized publicly available (see URLs) data from UK Biobank (UKB), a population-based prospective cohort study that does not overlap with the migraine GWAS. Here, migraine was self-reported in a standardized interview with a nurse (10,007 cases/327,152 controls).
Outcome GWAS
We examined three cognition-related outcomes through available GWAS summary statistics. For the Alzheimer’s disease (AD) outcome, we used the largest publicly available meta-analysis of GWAS of AD, aggregating three GWAS (PGC-ALZ, IGAP, ADSP) and UKB, for a total of 71,880 cases and 383,378 controls, all of European ancestry (6). This GWAS utilized novel methods to combine clinically-diagnosed AD cases and cases defined using family history from UKB (6). In secondary analyses, we utilized an AD GWAS limited to diagnosed cases (21,982 cases/41,944 controls) (7). For the general intelligence measure, we used GWAS summary statistics from a meta-analysis of 14 cohorts, including UKB (n = 269,867) (8). Each cohort analyzed a “common latent g factor” derived as a sum score, mean score, or factor score from a multidimensional set of available cognitive tests. The betas used for the present analysis were standardized to mean zero and variance 1. For subcortical and intracranial volume (ICV), we used summary statistics from a GWAS of MRI-derived brain volume conducted through meta-analysis by the ENIGMA consortium, combining 50 cohorts (n up to 13,193 for the present analysis) (9). The reported effect sizes are in cubic millimetres, but were transformed to cubic decimetres for ICV and cubic centimetres for subcortical volumes to facilitate plotting with the other phenotypes in MR analysis.
Alcohol consumption was selected as a positive control for an outcome causally related to migraine, since patients with migraine avoid alcohol to minimize risk of attacks (11). These analyses used the alcohol consumption (units/week) variable in the UK Biobank (n = 112,117) (10).
Mendelian randomization analysis
To identify independent variants for analysis, we first extracted from each outcome dataset all variants that were genome-wide significant (p < 5 × 10−08) for migraine. This cutoff corresponds to an F-statistic of 30, which minimizes weak instrument bias (4) in MR. For each outcome dataset, we then removed variants that could not be harmonized according to variant name, allele identity, and allele frequency by the TwoSampleMR software (12). Finally, we clumped the harmonized dataset at an R2 of 0.10 (using the 1000G European reference panel integrated in the TwoSampleMR package) to limit analysis to linkage disequilibrium-independent variants (12). The total number of SNPs used for each analysis is shown in Figure 1.

Causal diagram displaying the MR analysis, and its relative robustness to reverse causality and confounding.
This MR analysis can be conceptualized as a life-long natural experiment whereby participants are randomly allocated to more or less genetic liability to migraine, which is typically manifest early in life. Cognitive outcomes are then measured later in life as a function of the genetic liability to migraine. To determine the magnitude and direction of this relationship, we regressed the effects of the genetic variants on the outcome (e.g. variant effect on AD) on their effects on the exposure (i.e. variant effect on migraine), applying inverse variance weights of the variant-outcome association with intercept constrained to zero (12). Heterogeneity was determined using Cochran’s Q, as is standard in MR analyses. MR effects from analyses utilizing binary exposure (such as migraine) are most appropriately interpreted as reflecting the consequences of a lifelong increase in liability to the exposure (13). To facilitate interpretation, we transformed estimates to represent the effect of a doubling of the odds of migraine on the odds of the outcome (by multiplying betas and standard errors by ln(2) = 0.693), as previously recommended (13). Statistical analyses were conducted in R Version 3.5.3 using the TwoSampleMR v0.4.22 (12) package.
Results
Up to 42 SNPs were available for analysis in the outcome datasets (Figure 1). The genetic instrument strongly associated with migraine in the independent UKB sample (OR [95% CI] 2.16 [1.96–2.39], p = 5.60 × 10−53). Positive control analysis confirmed a causal effect of migraine liability on reduced alcohol consumption, further justifying use of the genetic instrument in MR analysis (beta alcohol units/week [95% CI] −0.02 [−0.03 to −0.01], p = 1.71 × 10−06).
The odds ratio of AD per doubling in the odds of migraine was consistent with the null (OR [95% CI] 1.01 [0.999–1.02], p = 0.07). This effect was similar when using GWAS restricted to clinically diagnosed AD cases (OR [95% CI] 0.96 [0.90–1.01], p = 0.13). Similarly, no effect was seen of migraine liability on general intelligence (standardized beta [95% CI] 0.01 [−0.003 to 0.02], p = 0.13), intracranial volume (beta [95% CI] 0.00 decimeter3 [−0.01 to 0.01], p = 0.86), or any of the seven MRI-defined subcortical volumes (all p > 0.05; Figure 2). Heterogeneity was not detected for the majority of the outcomes (p > 0.05), with the exception of hippocampal volume (Q = 72 on 38 degrees of freedom, p = 0.0008) and general intelligence (Q = 61 on 41 degrees of freedom, p = 0.02). No migraine risk variants were associated with the outcomes at genome-wide significance.
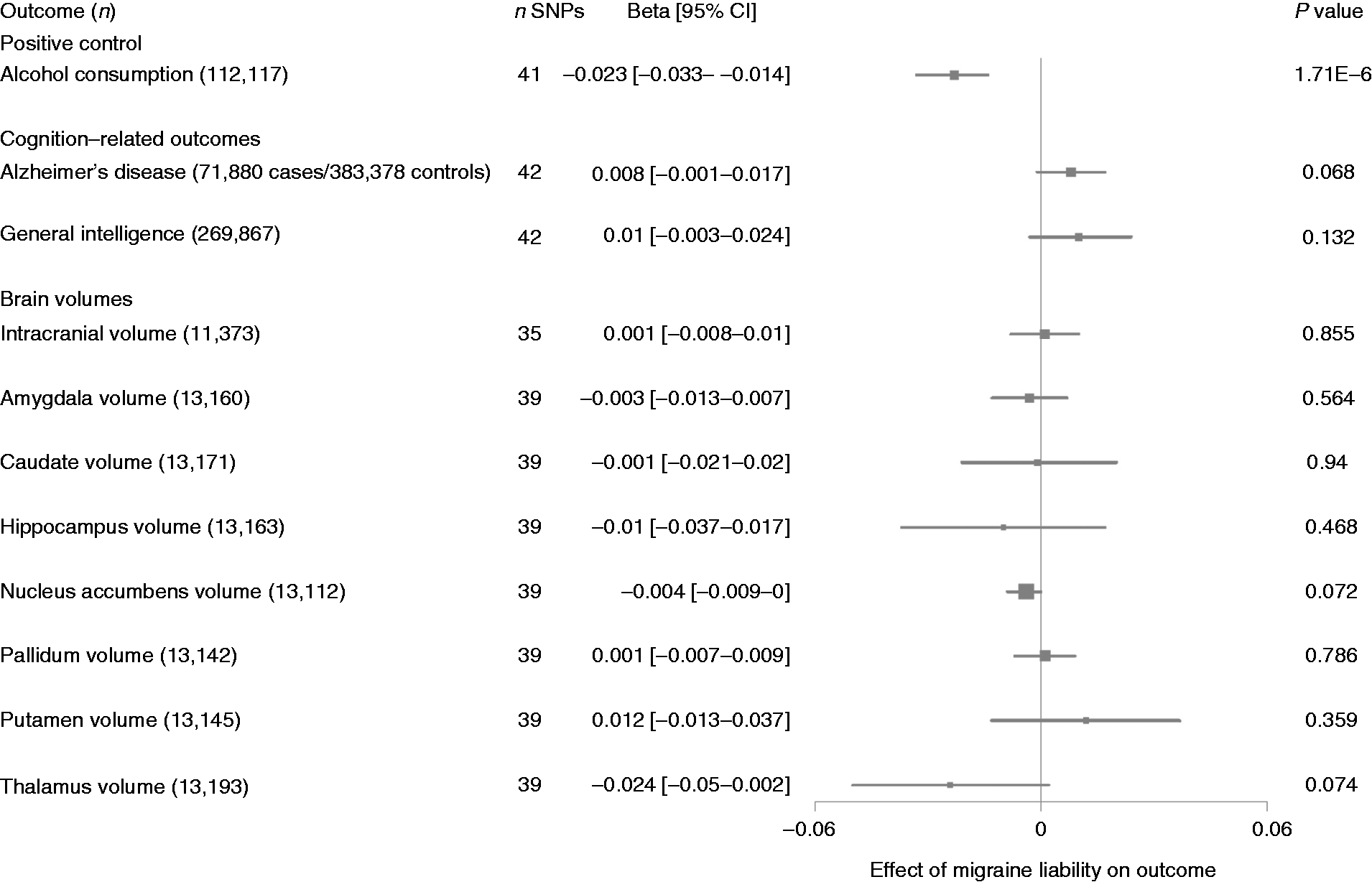
Two-sample Mendelian randomization effects of migraine liability on cognition-related and positive control outcomes. Alcohol consumption is measured in units consumed per week. Effects on AD are presented as log-odds. General intelligence is on the standardized beta scale. ICV is presented in dm3, and subcortical volumes are presented in cm3.
Discussion
This two-sample MR study did not support a causal effect of migraine liability on AD risk, general intelligence, or a range of MRI-defined brain volumes. The confidence intervals were narrow and large causal effects are therefore unlikely. These null findings are consistent with multiple cohort studies, which did not identify an effect of migraine on rates of cognitive decline (1). However, a recent cohort study showed a positive association between migraine and AD (3). Residual confounding is a potential explanation for the divergence of our results from these findings. For example, solely controlling for educational attainment does not capture the multidimensional nature of socioeconomic status and may be susceptible to measurement error (3). In contrast, genetic variants carry the advantage of random assortment at gametogenesis and are thus generally not associated with these environmental confounders.
Strengths of this study include the two-sample MR design and the assessment of several cognition-related outcomes including AD, intelligence, and brain volumes. There are also limitations to consider. The null associations may be due to insufficient power. Despite widespread use of the methodology, techniques for power calculation in MR analyses utilizing a binary exposure have not been developed. We instead i) validated the instrument in an independent dataset, ii) utilized a positive control, iii) leveraged the largest available GWAS, and iv) focused on the precision of effects as reflected in the confidence intervals, which did not contain clinically meaningful effect sizes. The migraine GWAS identified no common variants specific to migraine with aura (6,332 cases/144,883 controls), and all variants associated with migraine without aura (8,348 cases/139,622 controls) were already identified in the primary meta-analysis (5). This therefore precludes reliable MR investigations by migraine subtype. However, we note that our results are applicable to the majority of patients with migraine who do not experience aura. Finally, we were unable to examine sex-specific effects of migraine due to lack of available sex-specific GWAS.
In summary, this large-scale MR study does not support a causal influence of migraine liability on various measures related to cognitive function.
Public health relevance
Patients are unlikely to be at elevated risk for Alzheimer’s disease or cognitive decline due to migraine. Treating or preventing migraine is unlikely to impact the population burden of dementia.
Footnotes
Ethics or IRB approval
This study used anonymized, summary-level association statistics and therefore did not require ethical or IRB approval.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DIC was supported by grants from the NINDS/NIH (R21NS09296 and R21NS104398). PMR is supported by a grant from the NHLBI/NIH (K01 HL128791).
