Abstract
Aim
The TACHIS study (from the ancient Greek adjective “tachýs” meaning rapid) aimed to evaluate eptinezumab effectiveness and tolerability in routine clinical practice, integrating patient-reported outcomes and use of International Headache Society (IHS)-endorsed categories of migraine control by treatment.
Background
Eptinezumab is the only intravenous anti-calcitonin gene related peptide (CGRP) monoclonal antibody (mAb) approved for migraine prevention. While its efficacy has been demonstrated in RCTs, real-world evidence in patients with prior preventive treatment failures is still limited.
Methods
TACHIS is a prospective, multicenter, observational study conducted in Italy. Adults with episodic or chronic migraine initiating eptinezumab were followed for 24 weeks. Primary outcomes included change from baseline in monthly migraine days (MMDs) and ≥50% responder rate. Secondary outcomes included changes from baseline in acute medication use, Migraine Disability Assessment (MIDAS) and Headache Impact Test-6 (HIT-6) and IHS-defined residual burden categories. Logistic regression identified factors of response status.
Results
A total of 128 patients were included (82% female; 82% chronic migraine). MMDs decreased overall by 5.7 days (95% CI: −7.2 to −4.3) at week 12 and 6.9 (95% CI: −8.5 to −5.2) at week 24 (p < 0.001). A ≥ 50% response was achieved in 43.8% and 48.2% of patients at weeks 12 and 24, respectively. Over 40% of patients achieved optimal or modest migraine control. CGRP targeted therapy-naïve patients experienced significant greater benefit, though non-naïve patients also improved. Female sex and chronic migraine diagnosis were independently associated with response at 12 weeks. Adverse events were infrequent (4.7%) and mild, with no discontinuations due to safety concerns.
Conclusions
Eptinezumab demonstrated effectiveness and tolerability in a real-world population of patients with migraine and prior preventive treatment failures. The integration of migraine control metrics provides a comprehensive evaluation of therapeutic impact and supports eptinezumab use in routine care.
Trial Registration
The TACHIS study was preregistered on clinicaltrial.gov, NCT06409845.
This is a visual representation of the abstract.
Introduction
The recent update of the Global Burden of Disease study on headache disorders confirmed migraine as a leading cause of years lived with disability worldwide (1). In Italy, as in other countries, migraine substantially impact patients and public health (2). Anti-calcitonin gene-related peptide (CGRP) pathway drugs, including monoclonal antibodies (mAbs) and small molecules (gepants), have rapidly become the mainstay of the preventive treatment for both episodic (EM) and chronic migraine (CM) (3). The recent guidelines of the International Headache Society (IHS) (4,5) together with the Practice Recommendations (6) now provide strong support for the use of anti-CGRP drugs for migraine prevention and even as a first-line treatment option. Their efficacy and tolerability have been consistently demonstrated and confirmed across randomized controlled trials (RCTs) as well as observational real-world evidence studies (7–9).
Over the past years, the availability of different anti-CGRP drugs has expanded. Following the introduction of the first subcutaneous anti-CGRP or CGRP receptor mAbs (anti-CGRP/R mAbs; erenumab, galcanezumab, and fremanezumab), other drugs have been available as preventive treatments in the European market, including two small molecules (gepants, rimegepant and atogepant) and an intravenous anti-CGRP mAb (eptinezumab) (3,10,11).
In clinical practice, these novel therapies have created an unprecedented scenario. On one hand, clinicians now have a broader range of migraine-specific treatments promoting a redefinition of resistant and refractory migraine (12). Importantly, efficacy with one anti-CGRP drug may still be achieved after the failure of another, supporting sequential use (i.e., switching) (5,13). On the other hand, patients who switch between anti-CGRP therapies have usually failed at least three preventive therapies, representing a difficult-to-treat population, generally presenting lower response rates to subsequent treatments (13). Indeed, patients with treatment-refractory or difficult-to-treat migraine continue to experience substantial disability despite multiple preventive therapies, highlighting a persistent unmet clinical need. This distinction is crucial when comparing novel real-world evidence to earlier studies, as more recent studies increasingly include patients who switched from prior anti-CGRP therapies, and who were not present in earlier clinical practice (and therefore observational studies) and excluded from RCTs (3).
At the same time, anti-CGRP therapies, along with increased awareness and optimization of migraine management, have promoted a more ambitious approach in migraine prevention. A recent position statement by the IHS (14) advocated raising standards in migraine prevention, proposing the adoption of ambitious, but still pragmatic, treatment goals aimed at achieving optimal outcomes, such as migraine freedom or less than four days with migraine or moderate/severe headache per month, focusing on the residual burden more than on the percentage reductions from baseline (14).
In this evolving landscape, new real-world studies can provide evidence about the actual possibility of pursuing the ambitious goal of migraine freedom in difficult-to-treat patients who switched from prior ineffective anti-CGRP therapies. Despite the increasing availability of anti-CGRP mAbs, evidence regarding the effectiveness of intravenous eptinezumab in patients switching from prior subcutaneous anti-CGRP therapies remains limited.
This is the first prospective study applying the IHS residual burden categories to evaluate eptinezumab effectiveness along with traditional outcomes (i.e., reduction from baseline in different variables). We report the findings of a pre-registered, multicentric, prospective real-world study, adhering to the IHS recommendation for designing real-world studies (15). Our primary aim is to evaluate the effectiveness and tolerability of eptinezumab, the latest anti-CGRP mAb approved and the only one available in intravenous formulation, in a difficult-to-treat population, including patients who had previously failed subcutaneous anti-CGRP mAbs.
Methods
Study design
TACHIS is a real-world, observational, prospective, multicenter (including 23 centers for this analysis), investigator-initiated, and independent Italian study, considering all consecutive outpatients treated with intravenous eptinezumab 100 or 300 mg for EM or CM. The study has a pre-planned two-year follow-up. In this study, we included all patients with potential 24 weeks of follow-up, who received at least one infusion of eptinezumab starting from October 2024 to December 2024. Thus, patients who discontinued treatment but with available data were included in the analysis (see Figure 1). The study was pre-registered on clinicaltrials.gov (NCT06409845).

Flow chart with patients treated with eptinezumab at each follow up (a) and patients included in the effectiveness and safety analysis (b).
According to the IHS recommendations for the design of real-world studies in migraine (15), our study provides Data Quality Level 1 evidence. The checklist is reported in the Online Supplementary data.
The local Ethics committee approved the study as part of the Registro Italiano Cefalee (RICe) study (Studio RICe, 14591_oss CEAVC Studio RICe, 14591_oss and subsequent amendments). Other information on the RICe study is reported elsewhere (16,17). All patients signed a written informed consent before starting treatment with eptinezumab. The open online database Research Electronic Data Capture (REDCap) and the Empedocle electronic platform (developed for the RICe study) were used for data collection.
All patients received the drug according to clinical practice, Italian reimbursement rules and guided by physicians’ decision. No mandatory washout period was required between prior preventive treatments and eptinezumab initiation, reflecting routine clinical practice and potentially influencing early treatment effects.
Finally, dose selection (100 mg vs 300 mg) was based on treating physicians’ judgement according to clinical presentation, previous treatment history, and Italian reimbursement regulations.
Patient features
Participants were enrolled regardless of the number of preventive treatments stopped for ineffectiveness or poor tolerability (including other anti-CGRP drugs). Ineffectiveness was defined as no meaningful improvement in migraine-related variables after the administration of drugs for ≥6 weeks at the appropriate dose or intolerance to the drug according to the European Headache Federation (EHF) criteria and IHS guidelines (5,18).
Inclusion criteria were: i.) Individuals aged 18 years or older; ii.) diagnosis of migraine without aura, migraine with aura, or CM according to ICHD-3 (19); iii.) availability of data on migraine daysand other migraine related variables in the three months before starting eptinezumab; iv.) at least eight monthly migraine days (MMDs) in the three months before enrollment using paper diaries; v.) MIDAS score ≥11; vi.) good compliance to study procedures; vii.) clinical indication for prescription of eptinezumab.
Exclusion criteria were: i.) subjects with any contraindications to eptinezumab according to the EMA summary of product characteristics; ii.) concomitant diagnosis of medical diseases and/or comorbidities that could undermine the study according to clinicians; iii.) pregnancy and breastfeeding.
Collected variables
Clinicians diagnosed migraine and collected clinical and demographic features through a face-to-face interview, headache diary revision, and a semi-structured questionnaire: concomitant and previous preventive treatments, monthly headache days (MHDs), monthly migraine days (MMDs), number of monthly acute medications (AMNs), days with at least one use of acute medications (AMDs) before eptinezumab first infusion (i.e., baseline). To evaluate the overall severity of migraine, we evaluated both MHDs, defined as any day on which a patient recorded any type of headache, and MMDs, defined as any day of headache with migraine features or use of triptans.
We defined patients with medication overuse headache (MOH) as those patients with CM who satisfied the diagnostic criteria for MOH of the 3rd edition of the International Classification of Headache disorders (16). Other clinical variables, including comorbidities, were reported according to patients’ charts and outpatient interviews during clinical practice.
Finally, the Headache Impact Test (HIT-6) (20), the Migraine Disability Assessment (MIDAS) questionnaires (21) were administered at baseline and after 12 and 24 weeks of therapy. The Migraine Treatment Optimization Questionnaire-6 (mTOQ-6), the 12-item Allodynia Symptom Checklist (ASC-12), the Migraine-Specific Quality-of-Life Questionnaire (MSQ), the Migraine Interictal Burden Scale (MIBS-4), the Patient's Global Impression of Change (PGIC) and the Hamilton-A and -D questionnaires were collected but are not reported in the present study.
Adverse events (AEs) were collected during the entire duration of the study and were classified as treatment emergent adverse events (TEAEs) and severe adverse events (SAEs).
Outcomes and analysis
According to the guidelines of the IHS, the co-primary effectiveness outcomes of the TACHIS study were: i.) changes in MMDs at weeks 9–12 and 21–24 of treatment compared to baseline (four weeks prior eptinezumab administration); ii.) the percentage of responders (namely patients who presented a reduction of MMDs ≥50%, [RR50%], compared to baseline) at weeks 9–12 and 21–24 of treatment. We associated the assessment of the occurrence of TEAEs to evaluate the safety of the drug in a real-world population.
Secondary outcomes included from baseline (four weeks prior to eptinezumab administration) to weeks 9–12 and 21–24 of treatment:
The percentage of patients with MOH who were no longer over users; Changes in acute medication use (both AMNs and AMDs); Changes in HIT-6 questionnaire scores (36–78 scale); Changes in MIDAS questionnaire (0–270 scale); Percentage of adverse events (AE).
Exploratory
We also performed an exploratory, not pre-planned analysis to elevate the goals of migraine treatment and evaluate not only the percentage headache frequency reduction, but also the remaining burden of disease. We based this decision upon the recent IHS consensus statement (11), published after the start of the TACHIS study. The objective of this explorative analysis was to evaluate the residual burden of disease according to the cut-off defined by the consensus (i.e., migraine freedom [no migraine or moderate-to severe headache], optimal control [≤4 MMD], modest control [5–6 MMD] or insufficient control [>6 MMD]) in this population (14).
Furthermore, we evaluated the difference in effectiveness between patients non-naïve to anti-CGRP (defined as patients that failed at least one subcutaneous anti-CGRP mAb) and naïve to anti-CGRP (no prior subcutaneous anti-CGRP mab). Gepants as preventive were not included and not used in our cohort.
Statistical analysis
Due to the real-world setting design, we did not perform a structured sample size calculation, and all consecutive outpatients treated with eptinezumab were enrolled. The patients included have the baseline data for the primary outcomes and at least one dose of eptinezumab performed. All patients received infusions at 12-week intervals according to label; deviations were recorded.
Normality test by means of the Shapiro-Wilk test proved the non-normality of several variables. Thus, statistical analysis was conducted with non-parametric tests. We reported mean [95% confidence interval or interquartile range (IQr) or mean plus standard deviation (SD) as appropriate] for continuous variables and number (percentage) for categorical data. No imputation was made for missing data, which are reported in tables, figures and in the text as appropriate. The study aimed to capture real-world clinical variability, and missingness was judged to be non-systematic. Pre-post treatment differences within groups for quantitative variables were compared using the Wilcoxon signed-rank test, while the exact McNemar's test was applied for proportions in paired samples. A Mann-Whitney U test was conducted to assess differences between two independent groups for continuous variables.
To explore potential factors of clinical response, both univariate and multivariate logistic regression analyses were performed. The dependent variable was binary and defined as achieving a ≥ 50% reduction in MMDs from baseline, assessed at three months and six months. Independent variables were selected a priori based on clinical relevance. Univariate logistic regression was first conducted for each variable separately, using response status at six months as the dependent variable. Odds ratios (Exp(B)), 95% confidence intervals (CIs), and p-values were reported. Variables with p < 0.1 in univariate models, or of clinical interest, were considered for inclusion in the multivariate analysis.
Multivariate logistic regression was then performed to identify independent predictors of response at 12 and 24 weeks. Multicollinearity was assessed prior to inclusion. Model fit was evaluated using the omnibus chi-square test and Nagelkerke R2 statistics.
A two-tailed p-value <0.05 was considered significant for all variables, with a Bonferroni's correction where appropriate. All data were analyzed using SPSS software version 29.0 (IBM Corp. SPSS Statistics, Armonk, NY, USA) and graphs designed using GraphPad Prism version 10.00 (La Jolla, USA).
Results
Baseline characteristics
A total of 128 patients were included in the final analysis (female 82.0%, mean age 46.9 years [SD 12.3]). CM was present in 105 patients (82.0%) and it was comorbid with MOH in 79 (61.7%) participants. At baseline, most patients received 100 mg of eptinezumab (95.3%), with only 4.7% receiving 300 mg. Clinical, demographic features and migraine-related questionnaire scores on disability and quality of life are fully detailed in Table 1. Of these patients 39.8% were receiving concomitant migraine preventive treatment.
Clinical and demographic features of the overall population.

Percentages are expressed on column total. MOH, medication overuse headache; BMI, body mass index; Significant values are in bold. Percentages are expressed on column total.; MHDs, monthly headache days; AMNs, number analgesics per month; AMD; days with at least one analgesics use per month; ASC-12: Allodynia Symptom Checklist; HIT6, headache impact test; MIBS-4: Migraine Interictal Burden Scale; MIDAS, Migraine Disability Assessment questionnaire; MSQ, Migraine-Specific Quality of Life Questionnaire; SD, Standard deviation
The median number of previously ineffective preventive treatments was 4.0 (median 95%CI: 4.2–4.8). Details on prior migraine preventive treatments are detailed in Online Supplementary Table 1.
Eptinezumab was prescribed for one or more of the following reasons: ineffectiveness (86.7%), partial effectiveness (19.5%), poor tolerability (4.7%), adverse events (2.3%) of previous therapies, or patient preference (3.1%).
Main outcomes and co-primary outcomes
Effectiveness at weeks 9–12 and 21–24 of treatment
In the overall population, all main outcomes demonstrated significant reductions at week 12 and week 24, as detailed in Table 2. For the co-primary outcome (changes in MMDs), MMDs decreased by 5.7 days (95% CI: −7.2 to −4.3) at week 12 and by 6.9 days (95% CI: −8.5 to −5.2) at week 24 (both p < 0.001). MHDs decreased from baseline 21.6 (SD 7.0) to 13.6 (SD 9.2) at week 12 (mean change −8.0 days; 95%CI: −9.5 to −6.5; p < 0.001) and to 12.9 (SD 9.3) at weeks 21–24 (−8.5 days; 95% CI: −10.2 to −6.9; p < 0.001). AMNs changed by 8.5 (95% CI: −12.4 to −4.7) and 10.1 (95% CI: −14.2 to −6.0) at weeks 9–12 and 12–24, respectively (p < 0.001), and AMDs were reduced by 7.0 (95% CI: −8.5 to −5.5) and 7.1 (95% CI: −9.0 to −5.2) days (p < 0.001 for both) (Figure 2).
Migraine-related variables and disability questionnaires at baseline and after 12- and 24-weeks with eptinezumab in the overall, episodic and chronic populations.

Significant values are in bold. Percentages are expressed on column total.; MHDs, monthly headache days; AMNs, number analgesics per month; AMD; days with at least one analgesics use per month; SD, Standard deviation. Bonferroni for multiple correction was applied (p < 0.0167).
Subgroup analysis showed significant improvements for both CM and EM patients. In CM, MHDs decreased from baseline 23.6 (SD 5.8) to 15.2 (9.1) at week 12 and 14.3 (9.5) at week 24, with a total mean reduction of 9.2 days (p < 0.001). Similarly, EM patients improved from 12.7 (SD 4.5) to 6.2 (SD 4.5) and 6.5 (SD 3.9), with a mean reduction of 5.9 days (p < 0.001). Reductions in MMDs, AMNs, and AMDs followed a comparable pattern (Table 2).
Response rates
At 9–12 weeks, a ≥ 50% reduction in MMDs (RR50%) (co-primary outcome) was observed in 43.8% (56/128) of the overall population, increasing to 48.2% (52/108) at 21–24 weeks. A ≥ 75% response was achieved by 14.1% (18/128) and 21.3% (23/108) of patients at weeks 9–12 and 21–24, respectively. Similar trends were observed for MHDs response. Details for EM and CM, as well as MHDs response are reported in Table 3 and Figure 3.
Response rate (reduction in MMDs or MHDs, reported as percentage) after 12- and 24-weeks with eptinezumab.

Percentages are not cumulative (nested subsets). MMDs, monthly headache days; MHDs, monthly headache days.
Secondary and exploratory outcomes
Level of control of the disease
According to the IHS consensus framework, at week 12–24, 15.6% of the overall cohort achieved optimal control, and 18.8% achieved modest control, while 65.6% remained in the insufficient control category. No patient reached migraine freedom at this timepoint. At week 24 (n = 108), migraine freedom was observed in two patients (1.9%), optimal control in 14.8%, modest control in 25.9%, and insufficient control in 57.4% (Table 4 and Figure 4).
Effectiveness assessment according to IHS consensus statement during 12 and 24 weeks of treatment with eptinezumab.

Percentages are expressed on column total if not otherwise specified. IHS, international headache society.
Subgroup analysis showed that patients with EM were more likely to reach better outcomes. At week 12, 47.8% of EM patients achieved optimal control compared to 8.6% of CM patients. By week 24, optimal control was maintained in 20.0% of EM and 13.6% of CM patients, while migraine freedom was achieved by only two patients in the CM group (2.3%). The proportion of patients with insufficient control remained higher among CM patients at both timepoints (73.3% at week 12 and 64.8% at week 24), compared to EM (30.4% and 25.0%, respectively) (Table 4). Therefore, patients with EM demonstrated higher proportions of optimal or modest control compared with those with CM.
Disability questionnaires
At 12 and 24 weeks, MIDAS and HIT-6 questionnaires in the overall population as well as in EM and CM patients demonstrated significant improvements. Details are reported in Table 2 and Online Supplementary Table 2.
Anti-CGRP mAbs naïve vs switch (CGRP non-naïve) cohorts
Seventy patients (70/128, 54.7%) were anti-CGRP naïve and 58 (45.3%) had previously been treated with at least one subcutaneous anti-CGRP mAb. As expected, baseline migraine burden was higher in the non-naïve group across all variables: MHDs, analgesics used per month (both AMNs and AMDs,) (p < 0.001). Differences between the two groups are shown in Online Supplementary Tables 3 and 4.
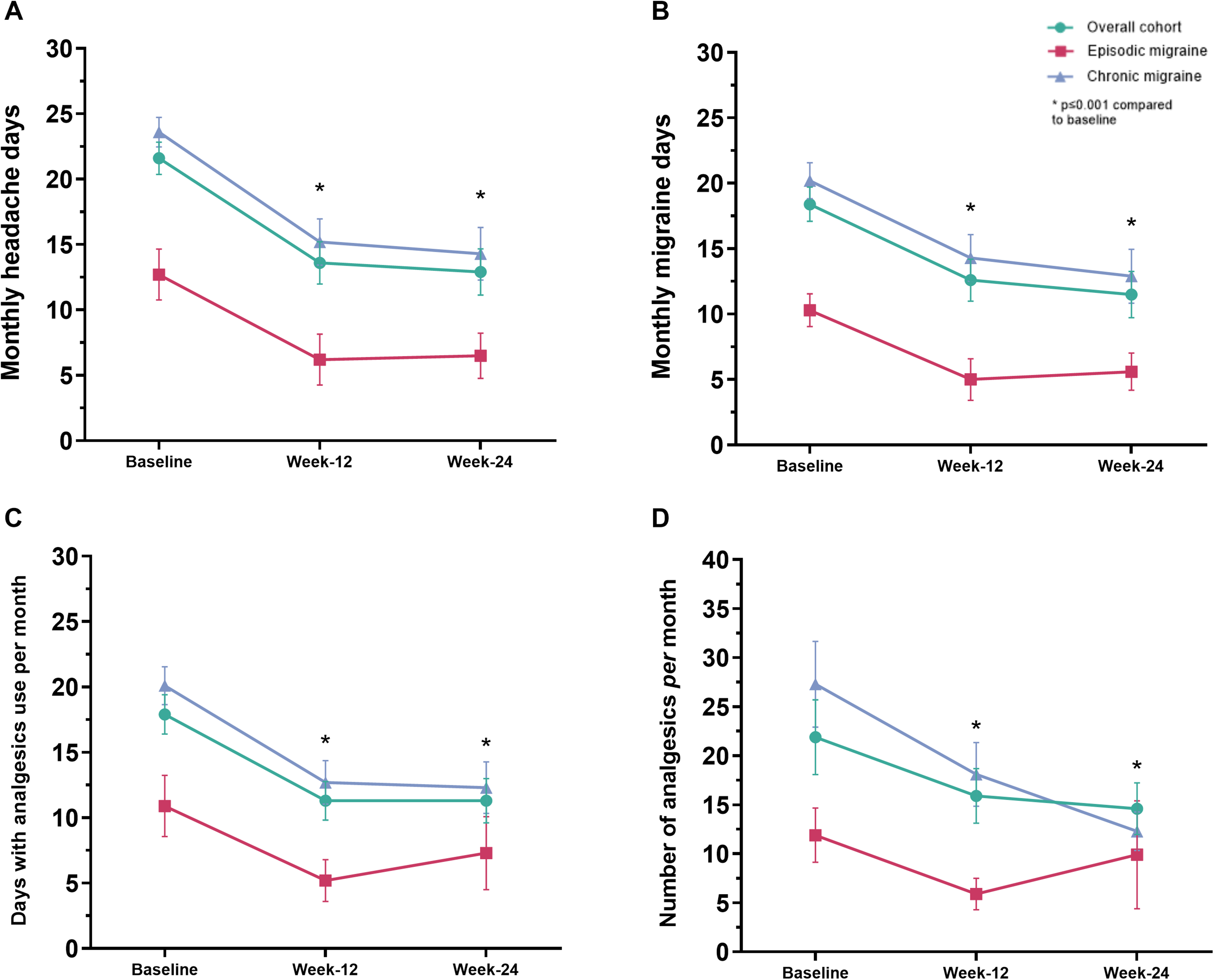
Migraine-related variables (MHDs (a), MMDs (b), AMDs (c) and AMNs (d)) at baseline and across 9–12 and 21–24 weeks with eptinezumab in the overall cohort, episodic and chronic migraine. P values compared to baseline assessed with Wilcoxon rank test and corrected with Bonferroni. No statistical comparison among group was performed.
Over the 24-week treatment period, both groups experienced significant improvements in migraine-related variables. In CGRP-naïve patients, MHDs decreased from 19.3 ± 6.6 at baseline to 10.4 ± 8.2 at week 12 (mean change: −8.9; 95% CI: −10.9 to −6.8, p < 0.001), and to 10.3 ± 8.3 at week 24 (mean change: −6.9; 95% CI: −9.1 to −4.8, p < 0.001). Parallel improvements were seen in MMDs (–6.0 and −6.2 days at weeks 12 and 24, respectively), AMNs (–6.6 and −7.8), and AMDs (–6.8 and −6.7), all with p < 0.001 (Figure 5 and Online Supplementary Table 5).
In non-naïve patients, MHDs declined from 24.4 ± 6.4 to 17.5 ± 8.9 at week 12 (mean change: −8.7; 95% CI: −10.9 to −6.4, p < 0.001), and to 16.2 ± 9.4 at week 24 (–8.4; 95% CI: −10.9 to −5.9, p < 0.001). MMDs and analgesics use (AMDs and AMNs) showed comparable reductions at both time points, all statistically significant (Figure 5).
Among naïve patients, 65.7% at week 12 and 68.9% at week 24 achieved a ≥ 30% reduction in MMDs, compared to 44.8% and 48.9% of non-naïve patients, respectively. An RR50% in MMDs was observed in 45.7% and 42.6% of naïve patients, and in 41.4% and 53.2% of non-naïve patients at weeks 12 and 24, respectively. Migraine freedom was achieved by only two naïve patients (3.3%) at week 24 (Figure 6). A similar pattern was seen for MHDs (Online Supplementary Table 6).

Response rate (reduction in MMDs) in the overall population (a) and 50% response rate in the overall cohort, episodic and chronic migraine subgroups (b).
Therefore, CGRP-naïve patients showed greater reductions in MMDs and higher responder rates than non-naïve patients across all timepoints.
Finally, based on the IHS consensus categories, CGRP-naïve patients achieved higher rates of optimal or modest migraine control compared to non-naïve patients, particularly at week 12. Indeed, at week 12, 24.3% of CGRP-naïve patients reached optimal control versus 5.2% of non-naïve patients. Modest control was achieved by 21.4% of naïve and 15.5% of non-naïve patients. The proportion of patients with insufficient control was notably higher in the non-naïve group (79.3% vs 54.3%).
At 24 weeks, migraine freedom was achieved by two patients (3.3%) in the CGRP-naïve group. Rates of optimal control remained limited in both groups (13.1% in naïve vs 17.0% in non-naïve), whereas modest control was more common among naïve patients (39.3% vs 8.5%). Several patients continued to report >6 migraine days per month, particularly in the non-naïve group (44.3% vs 74.5%), reflecting a higher residual disease burden in those with prior anti-CGRP exposure (Online Supplementary Table 7).
Factors associated with 50% response status at 12 and 24 weeks
Considering the overall population, in the univariate logistic regression analysis, using 12-week 50% response as the dependent variable (n = 128), several baseline clinical features were evaluated as potential factors, selected based on previous studies. The number of baseline AMDs and CM diagnosis were significantly associated with response at 12 weeks, with higher AMDs and CM diagnosis correlating with a reduced likelihood of achieving a ≥ 50% reduction in monthly migraine days (AMDs, OR = 0.939; 95% CI: 0.895–0.986; p = 0.012 and CM, OR = 0.269; 95%CI: 0.102–0.711; p = 0.008). Female gender showed a trend toward higher odds of response (OR = 2.576; 95% CI: 0.942–7.047; p = 0.065), contrariwise to AMNs (OR = 0.981; CI: 0.959–1.004; p = 0.099).

International Headache Society (IHS) outcomes during treatment with eptinezumab in the overall cohort (a) in patients with episodic migraine (b) or chronic migraine (c) and in patients who were anti-CGRP naïve (d) or non-naïve (e).
Other baseline characteristics, including age, BMI, psychiatric comorbidities, MOH, prior prophylactic failures, and prior exposure to anti-CGRP or botulinum toxin A, were not significantly associated with treatment response (Online Supplementary Table 8).
At the multivariate logistic regression model using variable with p > 0.01 in the univariate analysis (and corrected for sex and age), the female gender emerged as a statistically significant factor of response (OR = 3.380; 95% CI: 1.087–10.512; p = 0.035), whereas CM diagnosis was a negative factor of response (OR = 0.247; 95% CI: 0.073 −0.839; p = 0.025). Age, and AMNs were not significantly associated with response. The model demonstrated moderate fit (Nagelkerke R2 = 0.142) (Online Supplementary Table 9).
Using 24 weeks 50% response as dependent variable (n = 108), none of the investigated baseline variables reached statistical significance for predicting ≥50% reduction in MMDs at 24 weeks following eptinezumab treatment. However, prior failure to anti-CGRP mAbs showed a trend toward lower odds of response (OR = 0.493; 95% CI: 0.227–1.070; p = 0.074) as well as AMNs (OR = 0.979; 95%CI: 0.955–1.004; p = 0.092). Other demographic and clinical variables, including age, gender, BMI, psychiatric comorbidity, and CM diagnosis, were also not associated with treatment response (Online Supplementary Table 10).

Migraine-related variables (MHDs (a), MMDs (b), AMDs (c) and AMNs (d)) at baseline and across 9–12 and 21–24 -weeks with eptinezumab in naïve and non-naïve to anti-CGRP mAbs. P values compared to baseline assessed with Wilcoxon rank test and corrected with Bonferroni. No statistical comparison among groups was performed.
In the multivariate logistic regression analysis, no variable reached statistical significance. However, prior failure to anti-CGRP monoclonal antibodies showed a trend toward an inverse association with treatment response (p = 0.074), with patients previously exposed to anti-CGRP mAbs demonstrating lower odds of achieving a clinical response (OR = 0.453; 95% CI: 0.190–1.081). Age, gender and AMNs at baseline were not independently associated with response. The overall model fit was modest (Nagelkerke R2 = 0.110) (Online Supplementary Table 11).
Overall tolerability, adverse events and dropouts
During the 24-week observation period, six out of 128 patients (4.7%) experienced at least one treatment-emergent adverse event, with a total of seven adverse events (5.5%) reported. The most common AEs were constipation (four patients, 3.1%), followed by fatigue (two patients, 1.6%), and weight gain (one patient, 0.8%). The majority of adverse events were classified as mild in intensity (6/7, 85.7%), while one was of moderate intensity (14.3%). No severe adverse events were reported. Additionally, only one patient (0.8%) experienced an issue related to the infusion procedure.
The overall dropout rate over 24 weeks was 21.1% (27/128 patients). Specifically, two patients (1.6%) discontinued treatment by week 12, while an additional 25 patients (19.8%) discontinued between weeks 12 and 24. Among the 27 dropouts, the most common reason was loss to follow-up (20/27, 74.1%), followed by lack of effectiveness (5/27, 18.5%), and non-compliance or adverse events (1/27 each, 3.7%) (Online Supplementary Table 12).
Discussion
In the present study, we reported the effectiveness and tolerability of eptinezumab in a real-world setting, enrolling patients usually excluded from RCTs and exploring the control of disease based on the residual migraine burden according to the IHS position statement, along with percentage reduction in migraine-related variables.
Our findings may be summarized as follows: i.) Eptinezumab reduced migraine frequency and acute medication use at 24 weeks. ii.) Overall, 48% of patients achieved ≥50% response, with higher response rates observed among CGRP-naïve and episodic migraine patients—particularly for ≥30% reduction in MMD and in achieving optimal or modest migraine control based on IHS criteria. iii.) Very low rates of mild adverse events were reported, with no serious safety concerns and no issues related to the infusion itself, supporting the safety profile established in previous RCTs.

Response rate 50% (reduction in MMDs) in naïve and non-naïve to anti-CGRP mAbs during treatment with eptinezumab.
Overall, our data strongly supports the effectiveness, safety, and tolerability of eptinezumab in a real-world setting for patients with both EM and CM, regardless of previous preventive treatments failures, including subcutaneous anti-CGRP therapies.
The response and effectiveness observed with eptinezumab confirms prior evidence from RCTs (7), such as PROMISE (22,23) and DELIVER (24), as well as real-world studies like the EMBRACE (25). However, the TACHIS study extends these findings by providing prospective evidence in a predominantly treatment-refractory cohort and by including the evaluation of control of migraine.
In the EMBRACE II study (25), an observational study with an overlapping design and patient population (i.e., a prospective, multicenter Italian study), 74 patients were enrolled and completed 24 weeks of follow-up. A 50% response in monthly migraine days at weeks 21–24 was achieved by 69% of patients. Among those with CM, the ≥50% responder rate was equal to 73%. These proportions are substantially higher compared to our study (n = 128), in which 48% of patients achieved a 50% response at 24 weeks, and 47% among those with CM. The difference is even more pronounced for the 75% response rate: 21% in our study versus 40% in EMBRACE II overall, and 43% among CM patients compared to 23%.
Some degree of difference could be attributed to the higher proportion of patients with MOH in our cohort (81.8%) compared to EMBRACE (66.1%), although this alone cannot account for the entire variability in response rates. Other clinically relevant variables, including age, sex, disability, and prior treatment failures (including anti-CGRP mAbs), were comparable between cohorts (25). As both cohorts were drawn from Italian centers, geographic or regulatory differences in prescribing practices are unlikely to explain the observed discrepancies.
Another important difference lies in the discontinuation rate, an indirect measure of effectiveness and tolerability and a common concern in real-world studies. In our study, 27 patients (21%) discontinued treatment for various reasons, primarily loss to follow-up. In contrast, only two patients discontinued treatment in the EMBRACE II study, one due to lack of efficacy and one due to relocation abroad. The discontinuation rate observed in our cohort is consistent with previous real-world studies involving subcutaneous anti-CGRP therapies and, in particular, with those evaluating switching strategies (9,13).
Briefly, compared with the EMBRACE II and DELIVER studies, our cohort showed markedly higher baseline migraine severity, with a greater burden of CM and MOH. These differences likely contributed to the comparatively lower responder rates observed in TACHIS.
These findings may reflect both the observational design of the study and the inclusion of patients with a higher baseline migraine burden.
Our results align with other real-world studies and RCTs. A study conducted in Singapore (26), which included 30 patients with only CM treated with eptinezumab 100 mg, reported that 47% and 52% of patients were ≥30% responders at months 3 and 6, respectively, while 20% and 28% were ≥50% responders. Notably, this cohort included patients with a history of multiple prior treatment failures: 57% had previously tried three or more preventive therapies, and 70% had been treated with other anti-CGRP mAbs before starting eptinezumab (26).
Regarding RCTs, the proportion of ≥50% and ≥75% responders in our real-world cohort appears consistent with the results of the DELIVER trial (24) which enrolled patients previously treated with two to four preventive therapies. In DELIVER (24) ≥ 50% response rates ranged from 52% to 59%, and ≥75% response rates from 21% to 28%, depending on whether patients received 100 mg or 300 mg of eptinezumab. It is important to note that our population had a significantly higher migraine burden, with a greater proportion of patients affected by CM (82% vs 46%) and MOH (62% vs 13%), as well as the use of at least three prior preventive treatment failures (100% vs 37%) (22).
In our study, subgroup analyses showed that CGRP-naïve patients experienced better outcomes across nearly all endpoints, results that are in line with prior studies with anti-CGRP mAbs (13,25,26) and gepants (27,28). Moreover, the effectiveness of eptinezumab was numerically reduced in patients who had not previously responded to other CGRP mAbs. We could speculate that this result is due to a longer duration of disease, chronification time and a different journey of patients, as the number of previous treatments failed (3.0 for naive vs 6.0 for non-naive) showed or that patients have a different sensibility to CGRP pathway inhibition. Furthermore, patients previously treated with other anti-CGRP mAbs may exhibit persistent trigeminovascular activation, central sensitivity, altered CGRP-receptor-ligand dynamics, or partial downregulation of CGRP-related pathways following prolonged exposure. However, patients with prior exposure to anti-CGRP mAbs, who are typically excluded from RCTs and underrepresented in prior real-world (RW) studies, also achieved significant benefit from eptinezumab. These findings support the clinical relevance of anti-CGRP mAbs within class switching.
It is noteworthy that previous studies have relied solely on standard response metrics, such as percentage reductions in monthly migraine days. In contrast, our study applied, for the first time, both the categories of the IHS position statement on disease control by drugs (14) and the IHS framework for real world evidence design and quality assessment (15), providing a quality level 1. This approach shifts the focus from relative response rates to absolute measures of disease control based on the residual number MMD or days with moderate-to-severe headache, which may be more informative in real-world populations with high baseline headache days. In our cohort, while only 1.9% of patients achieved migraine freedom at 24 weeks, over 40% reached either optimal or modest control. These stratified outcomes offer a more detailed perspective on treatment benefit focusing on the patient’s well-being. In this context, whether increasing the eptinezumab dose from 100 mg to 300 mg and when could further reduce residual burden remains an open question.
Investigating sex differences is becoming increasingly important in the context of precision medicine, particularly in migraine research. Recent research has focused on the effectiveness of anti-CGRP therapies (both mAbs and gepants, as preventive and acute treatments) in the male population (29,30). A recent metanalysis (30) reported that anti-CGRP mAbs and gepants as preventives are effective in both male and female subgroups whereas, in the male subgroup, the efficacy of gepants for acute treatment did not differ significantly from placebo; however, the studies were not powered to detect sex-specific differences (30). To date, there is no consistent evidence of reduced efficacy in men for anti-CGRP mAbs, nor have RW studies or post hoc analyses of gepants/mAbs showed conclusive sex-related differences in treatment outcomes (7,31–33).
In our multivariate analysis, female sex was associated with higher odds of achieving treatment response. However, this should not be interpreted as evidence of ineffectiveness in the male subgroup. As previously noted, our study was not powered to evaluate sex differences, and the proportion of male patients was relatively small. Nevertheless, emerging evidence suggests that peripheral nociceptors exhibit sexual dimorphism, indicating that the underlying mechanisms of migraine pain may differ between men and women (29,34,35). This has important implications for both the understanding of migraine pathophysiology and the development of targeted treatments. Despite the higher prevalence of migraine in women, most studies have not stratified outcomes by sex, limiting our ability to fully assess potential differences in clinical presentation and therapeutic response in men.
Finally, regarding safety, eptinezumab was well tolerated over the 24-week observation period. A total of six out of 128 patients (4.7%) experienced at least one AE, with seven events reported overall (5.5%). No severe adverse events occurred, and no AEs led to treatment discontinuation. These results are in line with previous RW studies (7,25) but substantially lower than those reported in the DELIVER trial (41–42%) (24). This discrepancy may be attributed to differences in AE assessment between clinical trials and RW practice, where passive reporting and patient perception of tolerability can be very different. Notably, all patients in our cohort had failed at least three prior preventive treatments, which may have influenced their reporting thresholds, particularly for mild events.
The TACHIS study presents several strengths. First, it is one of the few prospective real-world studies of eptinezumab, with pre-planned data collection and follow-up extending to 24 weeks and including the larger sample size so far. Second, it includes a representative and difficult-to-treat population, with high rates of prior CGRP treatment failure, MOH, and CM. Third, the study applies multiple outcome assessments, including validated PROMs, allowing for a comprehensive evaluation of treatment impact on both migraine frequency and patient-centered variables. Furthermore, it is the first study so far that applied both the IHS-defined criteria for disease control and IHS recommendation for data quality reporting. Lastly, the use of electronic data collection platforms ensured consistent data acquisition across sites, reducing information bias.
Nonetheless, some limitations must be acknowledged. First, the absence of a control group does not allow evaluation of causal inference and may introduce regression to the mean. Second, observational design is susceptible to selection bias; including patients with more severe or refractory migraine. Third, the single-country design may limit generalizability to healthcare systems with different referral pathways or treatment access. Fourth, the absence of a formal sample size calculation reduces the statistical power of subgroup and regression analyses, mainly for sex-specific differences (82% of participants were female). Fifth, the short-term follow-up period (24 weeks) does not allow assessment of long-term sustainability of benefits, which is crucial for chronic conditions and considering the reimbursement rules in Italy, we could not evaluate the difference between 100–300 mg at this timepoint. Finally, although dropout rates were comparable to other real-world studies, loss to follow-up was the leading cause and may have influenced response estimates.
Conclusion
The TACHIS study confirms that eptinezumab is an effective and well-tolerated option for migraine prevention in routine clinical practice. Our study included subjects with at least three previous failures of preventive treatments, there was a large portion of CM patients and a relevant percentage of subjects who did not benefit from at least one subcutaneous CGRP mAb. The adoption of the IHS framework disease control and inclusion of multidimensional patient-reported outcomes represent significant methodological advances in real-world migraine research, and we strongly support their application in future observational studies.
Further studies with larger sample sizes are needed to better understand the differences in eptinezumab effectiveness and discontinuation rates and to optimize patient selection and treatment management strategies in clinical practice.
Clinical implications:
Treatment with eptinezumab showed consistent and clinically meaningful reductions in both monthly migraine and headache days across episodic and chronic migraine patients, demonstrating real-world effectiveness in a refractory cohort.
According to the International Headache Society (IHS) categories, about 15–20% of patients achieved optimal control and 20–26% modest control after 24 weeks of treatment.
Naïve patients to anti-CGRP therapy had greater benefit, with higher proportions achieving optimal or modest control, whereas non-naïve patients had a higher residual burden despite improvements.
Safety was favorable, with only 4.7% of patients reporting mild adverse events, no severe events, and no discontinuations due to tolerability issues.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251414659 - Supplemental material for Levels of migraine controls following International Headache Society (IHS) recommendations with eptinezumab: Effectiveness and tolerability in a 24-week, prospective multicenter study (the TACHIS study)
Supplemental material, sj-docx-1-cep-10.1177_03331024251414659 for Levels of migraine controls following International Headache Society (IHS) recommendations with eptinezumab: Effectiveness and tolerability in a 24-week, prospective multicenter study (the TACHIS study) by Luigi Francesco Iannone, Elisa Maria Piella, Danilo Antonio Montisano, Carla Fasano, Gabriele Sebastianelli, Gianluca Coppola, Delfina Ferrandi, Claudia Lanni, Maria Pia Prudenzano, Marina de Tommaso, Paola Merlo, Francesco De Cesaris, Alberto Chiarugi, Antonio Munafò, Francesca Pistoia, Raffaele Ornello, Alberto Doretti, Licia Grazzi, Flavia Lo Castro, Roberto De Icco, Gloria Vaghi, Gianluca Avino, Marina Romozzi, Paolo Calabresi, Stefania Battistini, Alessandra Rufa, Maria Albanese, Michele Trimboli, Giovanna Carlucci, Marcello Silvestro, Antonio Russo, Innocenzo Rainero, Maria Rosaria Valente, Luisa Fofi, Marilena Marcosano, Pierangelo Geppetti, Claudia Altamura, Fabrizio Vernieri, Cristina Tassorelli, Simona Sacco, Simona Guerzoni and in Cephalalgia
Footnotes
Acknowledgments
The “Società Italiana per lo Studio delle Cefalee” (SISC) is acknowledged for the “Registro Italiano delle Cefalee (RICe)”.
ORCID iDs
Ethical considerations
The local Ethics committee approved the study as part of the Registro Italiano Cefalee (RICe) study (Studio RICe, 14591_oss CEAVC Studio RICe, 14591_oss and subsequent amendments; 2024).
Consent to participate
All patients signed a written informed consent before starting treatment with eptinezumab.
Author con tributions
LFI, GaSe, EMP, CF and DAM had full access to all of the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. LFI designed the study, performed statistical analysis and drafted the manuscript. GaSe, EMP, CF and DAM performed administrative and technical support. All Authors recruited patients, and critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LFI received financial support, consulting fees for the participation in advisory boards and support for attending meetings from: Teva, Eli Lilly, Lundbeck, Pfizer and AbbVie; he is Associate Editor for Frontiers in Neurology Headache and Neurogenic Pain section. GS received honoraria from AbbVie.
FV has received financial support from Allergan-AbbVie, Angelini and Lundbeck for investigator-initiated trials; consulting fees for the participation in advisory boards from AbbVie, Angelini, Eli Lilly, Lundbeck, Organon, Novartis, Pfizer, and Teva; honoraria for scientific lectures and presentations from AbbVie, Eli Lilly, Lundbeck, Novartis, Organon, Pfizer, and Teva; support for attending meetings from Abbvie, Amgen, Eli Lilly, Lundbeck, Pfizer, and Teva; he has been Principal Investigator in clinical trials sponsored by AbbVie, Eli Lilly, Lundbeck, Pfizer, and Teva; he is Co-Specialty Editor for Frontiers in Neurology Headache and Neurogenic Pain section.
RO reports personal fees from AbbVie, Bayer, Eli Lilly, Lundbeck, Novartis, Organon, Pfizer, and Teva.
CT has received, in the last 3 years, personal fees for participation in advisory boards or for speaking at sponsored symposia from AbbVie, Eli Lilly, Ipsen, Lundbeck, Medscape, Pfizer, Organon and Teva. Her research group has received research grants from AbbVie, EraNet Neuron, Migraine Research Foundation and competitive grant from the Italian Ministry of Health and Italian Ministry of research. Her institution has received payments for clinical trials from AbbVie, Biohaven, Chordate, Eli Lilly, Ipsen, Lundbeck, Pfizer and Teva. She is Associate Editor of Cephalalgia and member of the Classification Committee of the International Headache Society.
SS reports personal fees as speaker or advisor from Abbott, Allergan-Abbvie, AstraZeneca, Bayer, Boehringer, Eli Lilly, Lundbeck, Pfizer, and Teva; she received research grants from Novartis and Uriach; she is President of the European Stroke Organisation, Editor-in-Chief of Cephalalgia and Cephalalgia Reports, and Assistant Editor for Stroke.
MA received travel grant, honoraria as a speaker, or for participating in advisory boards from Abbvie, Teva, Organon, Pfizer, Eli Lilly and Lundbeck.
GV received personal fees from Lundbeck and TEVA.
RDI reports personal fees and non-financial support from Eli-Lilly, AbbVie, Pfizer, Lundbeck, and TEVA.
Other authors reported no conflicts of interest.
Data availability statement
Data supporting the findings in the present study are reported in the article and in the supplementary materials. The data collected and analyzed for the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
