Abstract
Background
Menstrually-related migraine (MRM) is a subtype of migraine associated with the ovarian cycle that imposes a significant burden on female patients. Although MRM and non-menstrual migraine (NMM) differ in clinical presentation and treatment response, their distinct neural mechanisms remain unclear. Emerging evidence suggests that alterations in intrinsic functional connectivity (FC) within and between large-scale brain networks may underlie the phenotypic heterogeneity of migraine subtypes. This study investigated FC alterations between patients with MRM and NMM, explored their correlations with clinical characteristics, and assessed the preliminary utility of FC in subtype differentiation.
Methods
Resting-state functional magnetic resonance imaging (MRI) with independent component analysis was used to examine whole-brain FC in 50 patients with MRM, 50 with NMM and 50 age-balanced healthy controls (HC). We analyzed within- and between-network connectivity across major resting-state networks, including the frontoparietal, default mode, salience and dorsal attention networks, and applied logistic regression to test whether FC values could classify migraine subtypes. Correlation analyses were further performed between FC measures and clinical indices, including disease duration, headache frequency, visual analog scale scores and Headache Impact Test (HIT-6) scores.
Results
Both MRM and NMM groups showed weaker within-network connectivity compared to HCs, primarily in the right frontoparietal, default mode and salience networks. Compared with NMM, the MRM group exhibited significantly stronger connectivity in the left frontoparietal network and weaker between-network connectivity between the dorsal attention and default mode networks. In the women with migraine, FC within the dorsal attention network (DAN) was negatively correlated with disease duration (r = −0.200, p = 0.046) and HIT-6 score (r = −0.183, p = 0.049). Furthermore, FC between the DAN and auditory network was inversely associated with disease duration (r = −0.225, p = 0.025). The logistic regression model achieved an area under the receiver operating characteristic curve of 0.73 (sensitivity = 0.70; specificity = 0.64) in distinguishing MRM from NMM.
Conclusions
Our findings reveal both shared and distinct alterations in large-scale brain networks in MRM and NMM, potentially explaining differences in clinical presentation and treatment response. This enhanced understanding of migraine pathophysiology supports the development of subtype-specific diagnostic tools and targeted therapies and underscores the value of resting-state fMRI as a non-invasive tool for migraine phenotyping and personalized care.
Registration Number
ChiCTR2200065586.
Keywords
Introduction
Migraine is the second leading cause of disability worldwide and disproportionately affects women, occurring two to three times more frequently in females than in males (1,2). Among women of reproductive age, migraine is the leading cause of years lived with disability globally (1). Notably, approximately one-quarter of female patients with migraine report that their symptoms are associated with menstruation (3). Menstrual migraine attacks are typically longer in duration and accompanied by more severe symptoms, such as nausea (4). Compared with non-menstrual migraine (NMM), menstrually-related migraine (MRM) is associated with greater headache impact and a higher overall burden on daily functioning (3,5). Although current treatment strategies – primarily involving triptans and non-steroidal anti-inflammatory drugs – are similar across subtypes, MRM has been reported to exhibit greater resistance to standard pharmacologic interventions (6). These clinical disparities highlight the critical need to elucidate the underlying neural mechanisms that differentiate MRM from NMM, which may ultimately inform more targeted and effective treatment strategies.
Functional brain network analysis provides a powerful framework for investigating the distributed neural dysfunction associated with migraine. Rather than focusing on localized abnormalities, this approach enables the identification of large-scale network reorganization and dysregulation that may underlie distinct migraine phenotypes (7,8). Neuroimaging techniques offer an objective means to investigate the intrinsic properties of the brain network by measuring blood oxygen level-dependent signals (9). Previous neuroimaging studies have identified regional differences between MRM and NMM; for example, altered nodal centrality in the inferior frontal gyrus and thalamus (10) and changes in effective connectivity involving the right insula (11). However, brain dysfunction associated with migraine may extend beyond isolated regions and involve alterations in distributed brain networks (12). To date, no study has investigated the large-scale brain functional network abnormalities of MRM in comparison to those of NMM.
In the present study, we employed resting-state functional magnetic resonance imaging (MRI) combined with independent component analysis (ICA) – a data-driven approach free from a priori assumptions (13). The aim of this study was to identify within- and between-network functional connectivity differences between MRM and NMM, to establish a predictive model using logistic regression with cross-validation for subtype classification, and to examine network alterations associated with clinical characteristics such as duration, headache frequency and headache-related disability.
Methods
Study design and patients
The study was approved by the Medical Ethics Committee of West China Hospital of Sichuan University, registered on the Chinese Clinical Trial Registry and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants. All patients with migraine without aura were prospectively and consecutively recruited from the outpatient Neurology Department of West China Hospital between February 2023 and August 2024. Age-balanced female healthy controls (HCs) were recruited during the same period through poster advertisements.
As defined by the International Classification of Headache Disorders, 3rd editon (ICHD-3) beta criteria (14), MRM was diagnosed when migraine without aura occurred during the perimenstrual period (from two days before menstruation to three days after the onset of menstruation) in at least two of three consecutive menstrual cycles, with additional attacks permitted at other times of the cycle. Women experiencing migraine without aura exclusively outside the menstrual cycle were classified as having NMM. All patients with migraine were required to meet the following inclusion criteria: (i) female, 18–50 years of age, right-handed; (2) diagnosis of migraine without aura according to the ICHD-III beta criteria; (3) regular menstrual cycles of 28 ± 7 days; and (4) no use of migraine preventive therapy for at least one month and no intake of acute medication within 24 hours prior to the fMRI scan. Exclusion criteria were: (1) claustrophobia or other contraindications to MRI; (2) macroscopic brain lesions on MRI; (3) additional neurological or psychiatric disorders; (4) alcohol or drug abuse; (5) pregnancy or breastfeeding; (6) migraine attacks occurring within 72 hours before or after the MRI scan, as documented in headache diaries; and (7) left-handedness.
HCs were recruited through poster advertisements and were required to meet the following inclusion criteria: (1) female, 18–50 years of age, right-handed; (2) no personal or family history of migraine or other headache disorders; and (3) regular menstrual cycles of 28 ± 7 days. All HCs underwent structured clinical interviews and routine neurological examinations conducted independently by two experienced neurologists, and none reported a history of major medical, neurological or psychiatric disorders. In addition, the Generalized Anxiety Disorder scale and the Patient Health Questionnaire were administered to exclude potential psychiatric conditions. Exclusion criteria for HCs were identical to those applied to women with migraine.
Migraine assessment
The MRM and NMM groups completed an additional systematic questionnaire that assessed headache duration, frequency, pain severity and headache-related disability using the Headache Impact Test-6 (HIT-6) (15), which evaluates the adverse impact of headaches on daily life. Pain severity was measured using a 10-cm visual analogue scale (VAS), anchored at the left end with “no pain” and at the right end with pain as bad as it could possibly be”. The MRM and NMM groups were asked to mark the point on the line that best represented the severity of their worst headache pain during the preceding two weeks.
MRI acquisition
To eliminate the possible brain activity variations due to menstruation, all participants underwent MRI scanning 14 ± 2 days prior to the expected onset of their next menstruation (peri-ovulatory phase), as determined from prospective menstrual diaries maintained for at least three consecutive cycles.. All participants underwent imaging on the same 3.0 T MRI scanner (Trio; Siemens Medical Systems) with a 32-channel head coil. The scanning protocols followed the Human Connectome Project-recommended settings.
fMRI images were acquired using a multiband echo-planar imaging with two-dimensional acquisition and blood-oxygen-level-dependent contrast sequence with the following parameters: repetition time (TR) = 700 milliseconds, echo time (TE) = 37.8 milliseconds, flip angle = 52°, field of view (FOV) = 210 × 177 mm2, slice thickness = 2.1 mm, 64 axial slices and multiband factor = 8. Two resting-state runs were obtained, each consisting of 415 volumes (approximately 5 minutes per run), with both left-to-right and right-to-left phase encoding orientations to accelerate acquisition and reduce susceptibility-related distortions (16).
High resolution T1 weighted volumes were acquired by a magnetization-prepared rapid gradient-echo sequence: inversion time = 1000 milliseconds, TR = 2400 milliseconds, TE = 2.01 milliseconds, flip angle = 8°, 208 sagittal slices, matrix size = 320 × 320, FOV = 256 × 256 mm2, voxel size: 0.8 mm isotropic. High resolution T2 weighted volumes were acquired by a 3D turbo spin-echo sequence with variable flip-angle echo trains: TR = 3200 milliseconds, TE = 565 milliseconds. The geometric parameters are same with T1 weighted scan protocol.
Participants lay supine with head motion minimized using a molded liner. During fMRI acquisition, they were instructed to keep their eyes closed, remain awake and avoid directed thoughts (which was confirmed by self-reports immediately after scans).
Preprocessing
The preprocessing procedure followed the Human Connectome Project Minimal Preprocessing Pipeline (17) and consisted of five main peplines as follows. Firstly, HCP PreFreeSurfer, FreeSurfer and PostFreeSurfer pipelines were successively conducted on the structural images. This step included an alignment between the Tl and T2 images, bias field and gradient distortion corrections, registration of the data to the standard Montreal Neurological Institute (MNI) space, and producing tissue maps and surface files for pia and white matter for each participant, followed by down-sampling and registering surface files. Once structural preprocessing was complete, the HCP pipelines fMRIVolume and fMRISurface were used on the functional MRI images. This process included removing spatial distortions, realigning volumes to compensate for participant motion, registering the fMRI data to the structural information, reducing the bias field, normalizing the 4D image to a global mean, masking the data and transferring the time series from the volume into the CIFTI standard space.
All preprocessed images were further checked for head motion using interframe shift (18) to ensure data quality. The mean interframe shift for all participants in all data sets was <0.2 mm. Single-person spatial ICA with automatic dimensionality estimation was performed in the FSL (the FMRIB Software Library), followed by ICA-based denoising. Linear regression was then applied to the full mixture matrix estimated by ICA to remove variance assigned to artifact components (19,20). For classification, we employed the trained HCP_hp2000.RData dataset, which has been validated in prior studies and demonstrated a classification accuracy of up to 99% (16).
Analysis of resting-state data
The analysis pipeline is depicted in Figure 1. The MELODIC tool within FSL was also utilized to perform spatial group-level ICA. Pre-processed, registered to MNI space and band-pass filtered BOLD (i.e. blood oxygenation level dependent) images of the whole group of participants were used as input. Resting-state networks (RSNs) were identified from group-level independent component (IC) maps derived through ICA. Two experienced neurologists independently performed visual inspection of the IC maps. Components reflecting motor artifacts, white matter, cerebrospinal fluid or physiological noise were excluded from further analysis. The remaining ICs were classified as nine canonical RSNs based on previous reports (13,21). Anatomical locations were determined based on the Harvard-Oxford cortical and subcortical structural atlases (22).

Postprocessing pipeline of resting-state fMRI data. RSN = resting-state networks; FC = functional connectivity. Group-level independent component analysis was performed on pre-processed functional magnetic resonance imaging (fMRI) data using the MELODIC tool in FSL to identify RSNs. Noise components were excluded through visual inspection by two experienced neurologists. Dual regression was then applied to obtain subject-specific spatial maps. FC values showing significant group differences were extracted from both within- and between-network analyses. These FC features were subsequently entered into a logistic regression model, followed by leave-one-out cross-validation and permutation testing for model performance evaluation.
Subject-specific spatial maps were obtained from the dual regression analysis (23) and were further compared. One-way analysis of variance (ANOVA) analysis was performed to compare the differences in resting-state functional connectivity (rsFC) after controlling for the influence of age. Then for brain regions that showed significant differences in covariance analysis, we compared each patient group with the control group and the two patient groups between them by applying unpaired t-test. Group differences between MRM, NMM and HC were assessed using FSL's randomize function with 5000 permutations and threshold-free clustering enhancement (TFCE) to correct for multiple comparisons (24).
The FSLNets package (http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/FSLNets) on the MATLAB platform (MathWorks Inc.) was used to determine the connectivity between the RSNs. Partial correlations were computed to assess the association between two RSNs while statistically controlling for the influence of all other RSNs, thereby providing a measure of more direct functional interactions (25). Partial correlation coefficients derived from all RSN pairs were transformed into z-scores for further analysis. In total, 5000 permutations were performed for each comparison. The results were family-wise error (FWE) corrected for multiple comparisons and considered significant at the p < 0.05 threshold.
FC values showing significant between-group differences in both within- and between-network analyses above were extracted as candidate features. These FC values were then entered into a logistic regression model with a logit link function for binary classification between MRM and NMM. The model was implemented in MATLAB using the glmfit and glmval functions. To ensure robust performance estimation, leave-one-out cross-validation (LOOCV) was applied. Within each fold, features were normalized to the [0, 1] range using MATLAB's mapminmax function. To evaluate the statistical significance of the classification, a permutation test with 5000 iterations was performed. In each iteration, participant labels were randomly shuffled and the entire LOOCV procedure was repeated. The empirical p-value was calculated as the proportion of permutations yielding performance metrics equal to or greater than those of the true model. Model performance was assessed using accuracy, sensitivity, specificity, and area under the receiver operating characteristic curve (AUC). Additionally, the consistency of feature weights across folds was examined to identify stable neurobiological markers.
Statistical analysis
ANOVA was used to compare demographic characteristics between the three groups. Two-sample t-test and chi-squared tests were used to compare the clinical characteristics of the patients (including disease duration, attack frequency, VAS score and HIT-6 scores) between the two groups (p < 0.05 was considered statistically significant). In the migraine patient group, Pearson's correlation was used to assess potential correlations between FC (mean connectivity within clusters with significant differences from dual regression and between-network rsFC for connections with significant intergroup differences from FSLNets) and clinical characteristic of migraine patient groups.
Results
Demographics and clinical characteristics
Fifty patients with clinically MRM and fifty patients with clinically NMM were enrolled in the study. Fifty age-balanced female HC with no history of neurological or psychiatric illness were enrolled as a control group. There were no significant differences in age distribution among the three groups (p > 0.05) (Table 1). Moreover, the clinical symptoms and disease duration were not significantly different between the two migraine groups (all p > 0.05). Nevertheless, the average HIT-6 score of the MRM group was significantly greater than that of the NMM group (p = 0.026, 95% confidence interval (CI) = 0.488–7.552) (Table 1).
Demographic and clinical characteristics.

MRM = menstrually-related migraine; NMM = non-menstrual migraine; HC = healthy control; VAS = visual analog scale; HIT-6 = Headache Impact Test-6.
Identification of resting-state brain networks using ICA
Nine components were considered to represent neuronal signals, including the right frontoparietal network (RFPN), left frontoparietal network (LFPN), dorsal attention network (DAN), default mode network (DMN), lateral visual network (LVN), medial visual network (MVN), salience network (SN), sensorimotor network (SMN) and auditory network (AN) (spatial maps and locations of the 9 RSNs are shown in the supplementary material (Figure S1 and Table S1).
Within-network rsFC
Within-network rsFC was significantly reduced in both MRM and NMM compared to HC within the RFPN and DAN, whereas only NMM showed lower rsFC in the SN relative to HCs (pFWE−corrected < 0.05) (Figure 2 and Table 2). Comparisons between MRM and NMM revealed significantly higher rsFC in the LFPN in the MRM group (pFWE corrected < 0.05) (Figure 2 and Table 2). In patients with migraine, rsFC within the DAN was negatively correlated with the disease duration (r = − 0.200, p = 0.046) and the HIT-6 score (r = − 0.183, p = 0.049) (Figure 3). Subgroup correlation analyses for women with MRM and NMM are provided in the supplementary material (Figure S2).
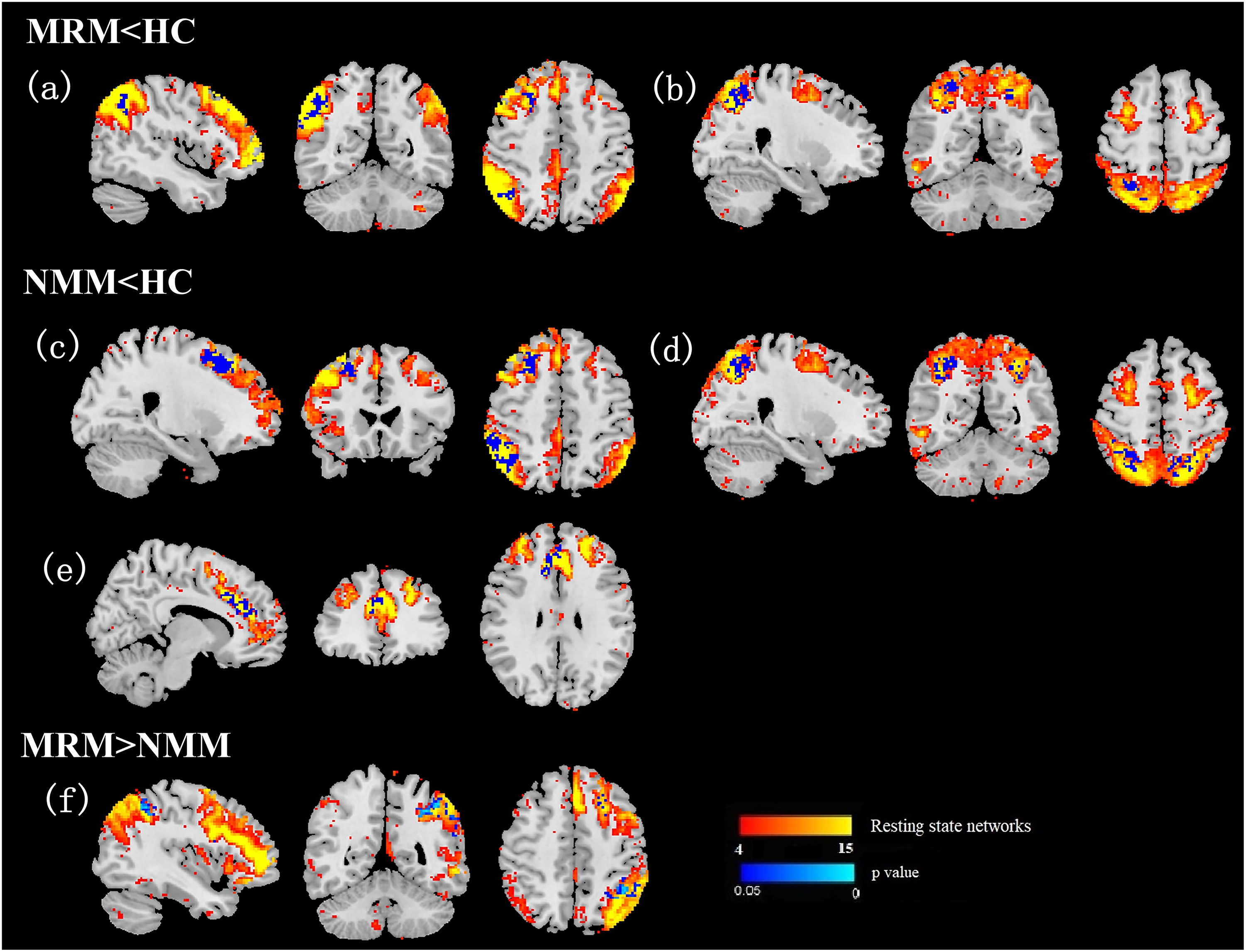
Within -network functional connectivity difference. MRM, menstrually-related migraine; NMM = non-menstrual migraine; HC = healthy control. (a, c) Right frontoparietal network. (b, d) Dorsal attention network. (e) Salience network. (f) Left frontoparietal network. Visualization was performed using FSLeyes (FMRIB Software Library, University of Oxford).
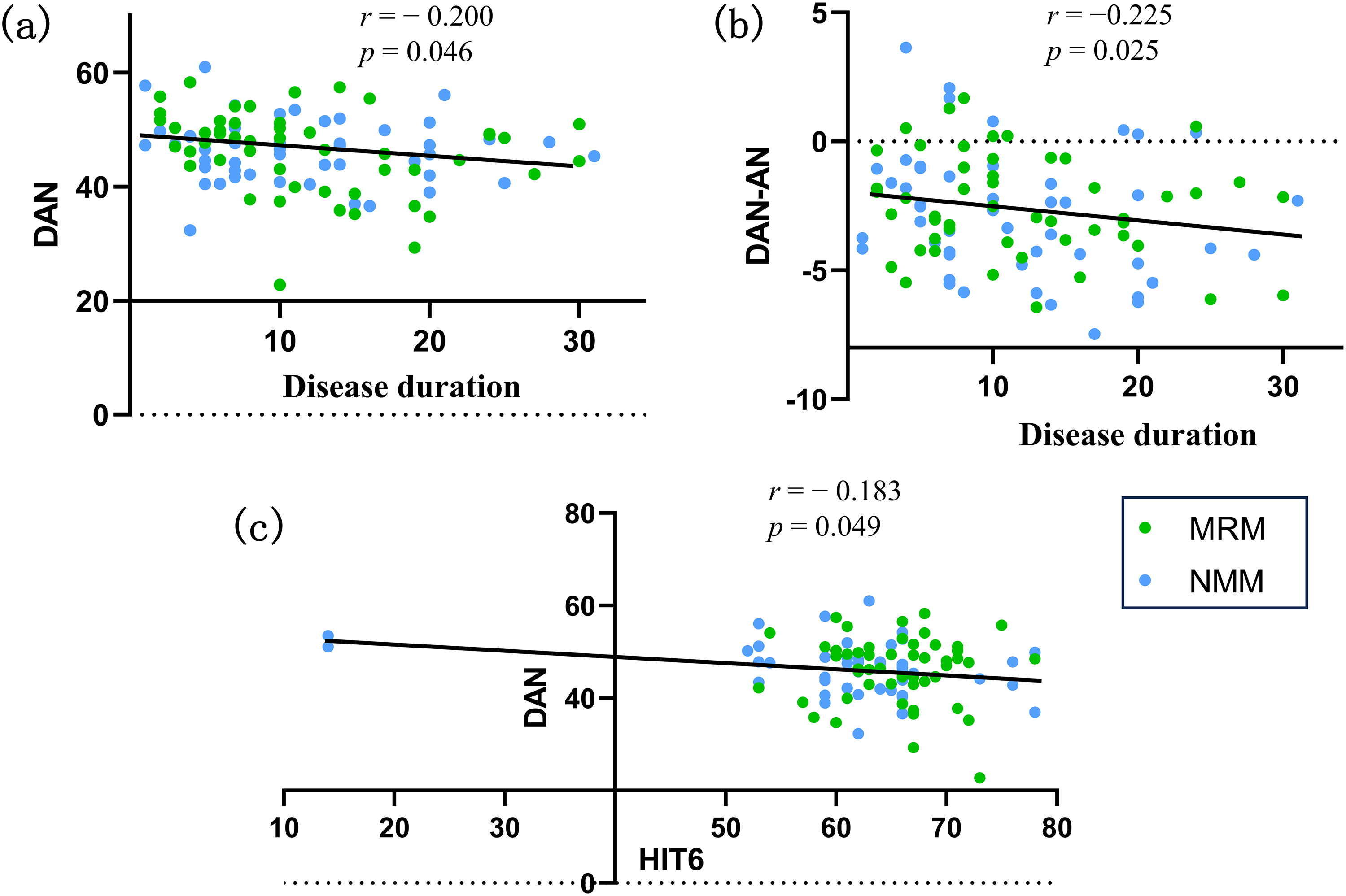
Scatter plot visual representations of correlation analyses. MRM = menstrually-related migraine; NMM = non-menstrual migraine; DAN = dorsal attention network; AN = auditory network; HIT-6 = Headache Impact Test. (a) Correlation between disease duration and within-network resting-state functional connectivity (rsFC) in the DAN. (b) Correlation between disease duration and rsFC between the DAN and AN. (c) Correlation between HIT-6 scores and within-network rsFC in the DAN.
Within-network functional connectivity differences.

MRM = menstrually-related migraine; NMM = non-menstrual migraine; HC = healthy control. Threshold-free clustering enhancement correction, p < 0.05, cluster size > 50 voxels; MNI = Montreal Neurological Institute coordinates.
Between-network connectivity
The results of ANOVA among the three groups indicated significant differences in rsFC between the DAN and DMN, between the LVN and SMN, between the DAN and AN, and between the LVN and AN after controlling for age. Post-hoc pairwise comparisons demonstrated that the MRM group exhibited significantly reduced rsFC between the DAN and DMN compared to both the HC and NMM groups (pFWE corrected = 0.043) (Figure 4). Additionally, MRM patients showed lower rsFC between the LVN and SMN relative to HCs (PFWE corrected = 0.041) (Figure 4). In contrast, NMM patients displayed weaker rsFC between the DAN and AN (pFWE corrected = 0.008) (Figure 4), but stronger rsFC between the LVN and AN (pFWE corrected = 0.01) (Figure 4), compared to HCs. In patients with migraine, rsFC between the DAN and AN was negatively correlated with the disease duration (r = −0.225, p = 0.025) (Figure 3). Subgroup correlation analyses for women with MRM and NMM are provided in the supplementary material (Figure S2).

The significant differences in between-network functional connectivity (rsFC). MRM = menstrually-related migraine; NMM = non-menstrual migraine; HC = healthy control. (a) Significantly different rsFC between the three groups after ANOVA. (b) The results of post-hoc two-sample t-tests (p < 0.05, family-wise error corrected).
Classification via logistic regression
The logistic regression model achieved an AUC of 0.73, with a sensitivity of 0.70 and a specificity of 0.64, in distinguishing patients with MRM from those with NMM (Figure 5). Permutation testing validated the statistical significance of the model's performance (sensitivity: p = 0.0008; specificity: p = 0.04; AUC: p < 0.0002). The relative contribution of each candidate feature to the classification performance is detailed in the supplementary material (Table S2).
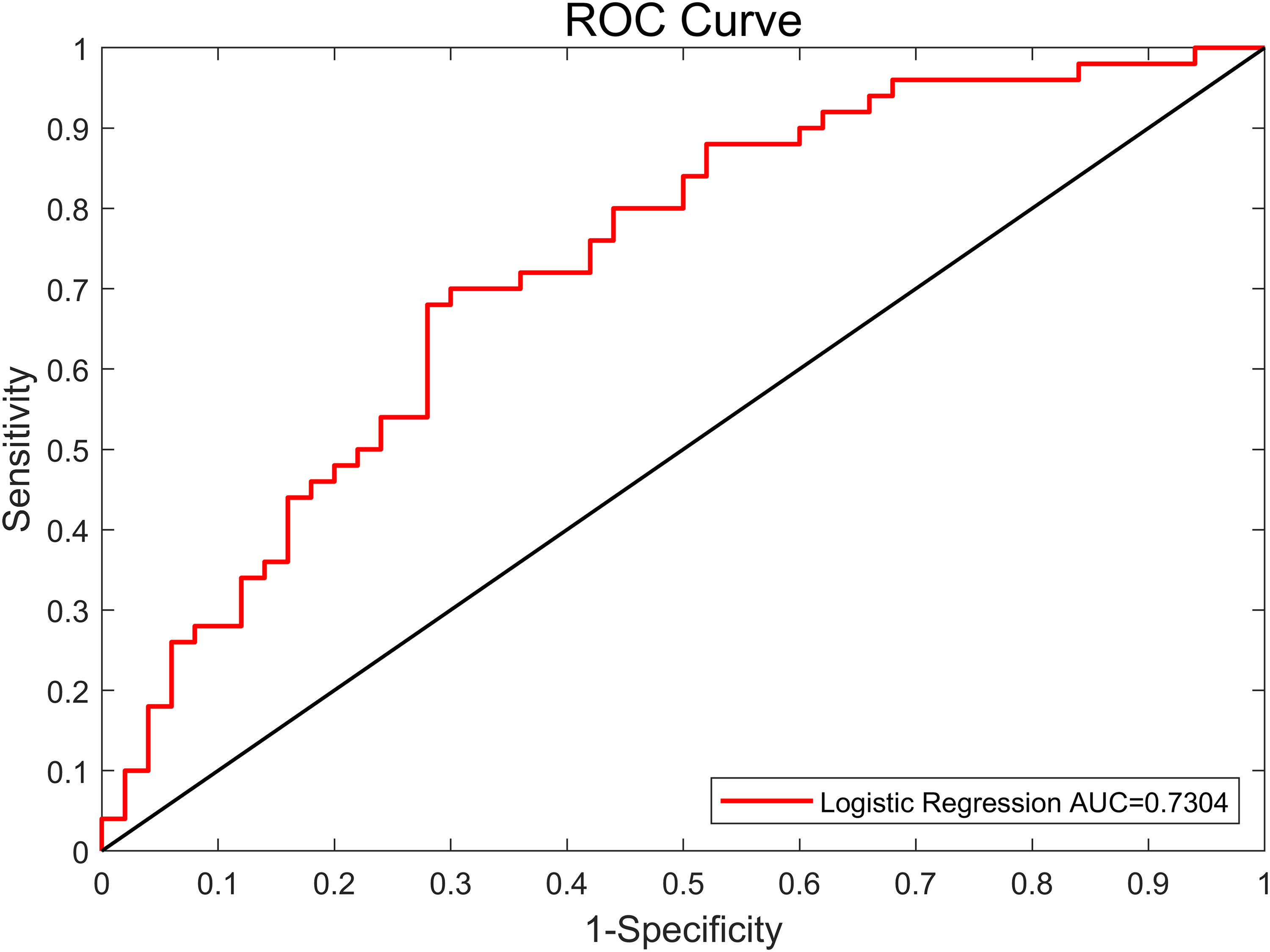
Classification accuracy of intergroup functional connectivity differences in distinguishing patients with menstrually-related migraine from non-menstrual migraine.
Discussion
In the present study, we investigated within- and between-network rsFC changes using the ICA approach and our findings revealed both shared and distinct patterns of within- and between-network connectivity changes among MRM, NMM and HC. The LFPN, DAN and DMN may play important roles in the specific pathophysiology of MRM, providing novel insights into the pathophysiological mechanisms underlying migraine subtypes in women.
Consistent with previous studies, we found that both MRM and NMM groups exhibited weaker within-network rsFC in RFPN and DAN compared to healthy controls. These networks are critically involved in attentional regulation and top-down executive functions, which are frequently disrupted in patients with migraine (26–28). Importantly, rsFC within the DAN in women with migraine was negatively correlated with disease duration, suggesting that longer disease duration may lead to progressive disruption of attentional networks. The lower within-network rsFC, which has also been described in other neurological diseases, might be a maladaptive and unspecific compensatory response to brain damage (29). Furthermore, weaker rsFC within the DAN showed a weak negative correlation with HIT-6 scores, reinforcing the potential clinical relevance of attentional network dysfunction (30). Interestingly, we observed a stronger within-network rsFC in the LFPN in MRM compared to NMM. This stronger rsFC may represent an adaptive response aimed at maintaining goal-directed cognitive control in the face of broader network dysfunction. The LFPN is critically involved in executive functioning and is influenced by hormonal fluctuations, particularly estrogen, which has been shown to modulate prefrontal cortical activity and large-scale network connectivity (31,32). Fluctuations in estrogen levels – particularly the rapid decline observed in the late luteal phase – have been linked to increased migraine severity in MRM (33). Prior studies further suggest that estrogen-dependent modulation of prefrontal and network function may selectively affect LFPN activity in MRM (34). Such interactions may help explain why MRM attacks cluster around specific phases of the menstrual cycle, whereas NMM is less hormonally dependent.
Between-network alterations further distinguished MRM from NMM. MRM patients showed reduced connectivity between the DAN and DMN compared to both NMM and HC groups. This disconnection is notable because effective anticorrelation between DAN and DMN is essential for flexible cognitive control and attentional engagement. Disrupted interactions between these networks may underlie the greater disability burden observed in MRM, as supported by the significantly higher HIT-6 scores in the MRM group. Additionally, MRM patients showed weaker connectivity between the LVN and SMN, suggesting impaired sensory integration. Such disruption may reduce the brain's ability to filter and regulate multimodal inputs, thereby contributing to the heightened sensitivity to visual, auditory or somatosensory stimuli, providing a plausible neural basis for the hypersensitivity frequently reported during menstrual migraine attacks (3,35). In contrast, NMM patients showed distinct connectivity disruptions between the DAN and AN, as well as stronger connectivity between LVN and AN. These changes may reflect distinct sensory processing profiles and possibly different compensatory mechanisms compared to MRM. While both MRM and NMM share some disruptions in core cognitive and sensory networks, the specific connectivity alterations observed in MRM may contribute to its greater clinical burden and resistance to treatment (36).
Several limitations should be acknowledged. First, as a cross-sectional study, causal inferences regarding the directionality of the observed brain–behavior associations cannot be made. Longitudinal studies are warranted to examine the temporal dynamics of network changes in relation to clinical progression. Second, though the number of participants was relatively larger than those of the existing research, studies with larger sample size are needed to increase the statistical power. Third, all participants were recruited from a single center, which may restrict the generalizability of the findings. Replication in larger, multicenter cohorts should be evaluated in future studies to enhance the robustness of these network-level biomarkers.
In conclusion, our findings demonstrate both overlapping and distinct alterations in large-scale functional brain networks in MRM and NMM. While both subtypes exhibit reduced connectivity in key cognitive networks, MRM is characterized by more pronounced decoupling between attentional and default mode systems, as well as increased LFPN connectivity that may represent an adaptive response to broader network dysfunction. These network-specific alterations may underlie differences in clinical presentation and treatment response, emphasizing the need for migraine subtype-specific diagnostic markers and therapeutic approaches. Future work should integrate advanced neuroimaging with artificial intelligence and predictive modeling to refine migraine subtype-specific phenotyping and forecast clinical trajectories. Such approaches may enable the identification of reproducible neurobiological markers, support patient stratification, and ultimately guide individualized diagnostic and therapeutic strategies in precision headache care.
Article highlights
Altered brain network connectivity found in both MRM and NMM patients.
MRM shows stronger connectivity in the left frontoparietal network vs. NMM.
Weaker dorsal attention–default mode connectivity unique to MRM
Supplemental Material
sj-docx-1-cep-10.1177_03331024251396102 - Supplemental material for Divergent functional connectivity patterns in menstrually-related and non-menstrual migraine: A large-scale resting-state fMRI study
Supplemental material, sj-docx-1-cep-10.1177_03331024251396102 for Divergent functional connectivity patterns in menstrually-related and non-menstrual migraine: A large-scale resting-state fMRI study by Yusha Tang, Hua Li, Bosi Dong, Leihao Sha, Ruiqi Yang and Lei Chen in Cephalalgia
Footnotes
Author contributions
Yusha Tang: formal analysis, data curation, investigation, visualization, writing-original draft, writing review & editing, funding acquisition. Hua Li: data curation, investigation. bosi dong: data curation, investigation. Leihao Sha: data curation, investigation. ruiqi yang: data curation, investigation. Lei Chen: conceptualization, methodology, project administration, supervision, writing-review & editing. All authors reviewed and approved the final version of the manuscript submitted for publication.
Consent to participate
Written informed consent was obtained from all participants.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical considerations
This study involves human participants and was approved by the Ethics Committee on Biomedical Research of West China Hospital of Sichuan University (20221528) and registered on the Chinese Clinical Trial Registry (Registration name: Study of the pathogenesis of menstrual migraine based on multimodal data; Number: ChiCTR2200065586). All procedures performed in studies were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The manuscript has been prepared in compliance with the STROBE guidelines for observational studies.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Sichuan Province (grant number 24NSFSC1652), 1·3·5 project for disciplines of excellence-Clinical Research Incubation Project, West China Hospital, Sichuan University (grant number 23HXFH008) and Sichuan CREDIT Pharmaceutical Special Fund (grant number 312200692).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
