Abstract
Aim
To evaluate rimegepant, a calcitonin gene-related peptide receptor antagonist, for short-term prevention of fasting-triggered headache during Ramadan.
Methods
Participants, aged 18–65 years and diagnosed with migraine or headache attributed to fasting, were randomized to open-label once-daily (QD) rimegepant 75 mg orally disintegrating tablet (ODT) from weeks 1–4 of the fast (Immediate Start arm; n = 52) or weeks 2–4 of the fast (Staggered Start arm; n = 53). The primary endpoint was the difference in number of headache days of any intensity between the Immediate Start and Staggered Start arms during week 1. Other endpoints included headache days during weeks 2–4, moderate-to-severe headache days during weeks 1–4, headache duration (any intensity and moderate-to-severe) during weeks 1–4, rescue medication use during weeks 1–4 and treatment satisfaction. Treatment comparisons utilized nominal p values. Safety endpoints included adverse events (AEs), serious AEs and discontinuations due to AEs.
Results
All participants (n = 105) had a diagnosis of migraine and a history of headaches during fasting. The number of headache days of any intensity during week 1 (primary endpoint) was lower in the Immediate Start arm vs. the Staggered Start arm (LS mean (95% confidence interval) = 1.74 (1.16–2.31) days vs. 2.92 (2.34–3.49) days; p = 0.005). There were no significant differences between arms during weeks 2–4. Total duration of headaches of any intensity was shorter in the Immediate Start arm vs. the Staggered Start arm during week 1 (LS mean (95% confidence interval) = 10.1 (5.9–17.5) hours vs. 20.0 (13.0–28.3) hours; p = 0.041) and week 4 (LS mean (95% confidence interval) = 0.9 (0.2–3.7) hours vs. 4.6 (2.2–15.5) hours; p = 0.035). The number and duration of moderate-to-severe headaches was significantly (p < 0.05) lower in the Immediate Start arm vs. the Staggered Start arm during week 1. For the overall 4-week study period, there was a significant reduction in moderate-to-severe headache days per week in the Immediate Start arm vs. the Staggered Start arm (LS mean (95% confidence interval) = 0.60 (0.34–0.86) days vs. 1.00 (0.75–1.26) days; p = 0.038) and a trend towards reduction in headache days per week of any severity (LS mean (95% confidence interval) = 1.05 (0.72–1.39) days vs. 1.50 (1.17–1.84) days; p = 0.070). Use of rescue medication trended lower in the Immediate Start arm vs. the Staggered Start arm at all study weeks; all p > 0.05. Most participants (82.4%) reported being satisfied, very satisfied, or extremely satisfied with rimegepant at end of treatment. No AEs were reported.
Conclusions
QD rimegepant 75 mg ODT may be effective and well tolerated for short-term prevention of fasting-triggered headache in individuals with migraine.
This is a visual representation of the abstract.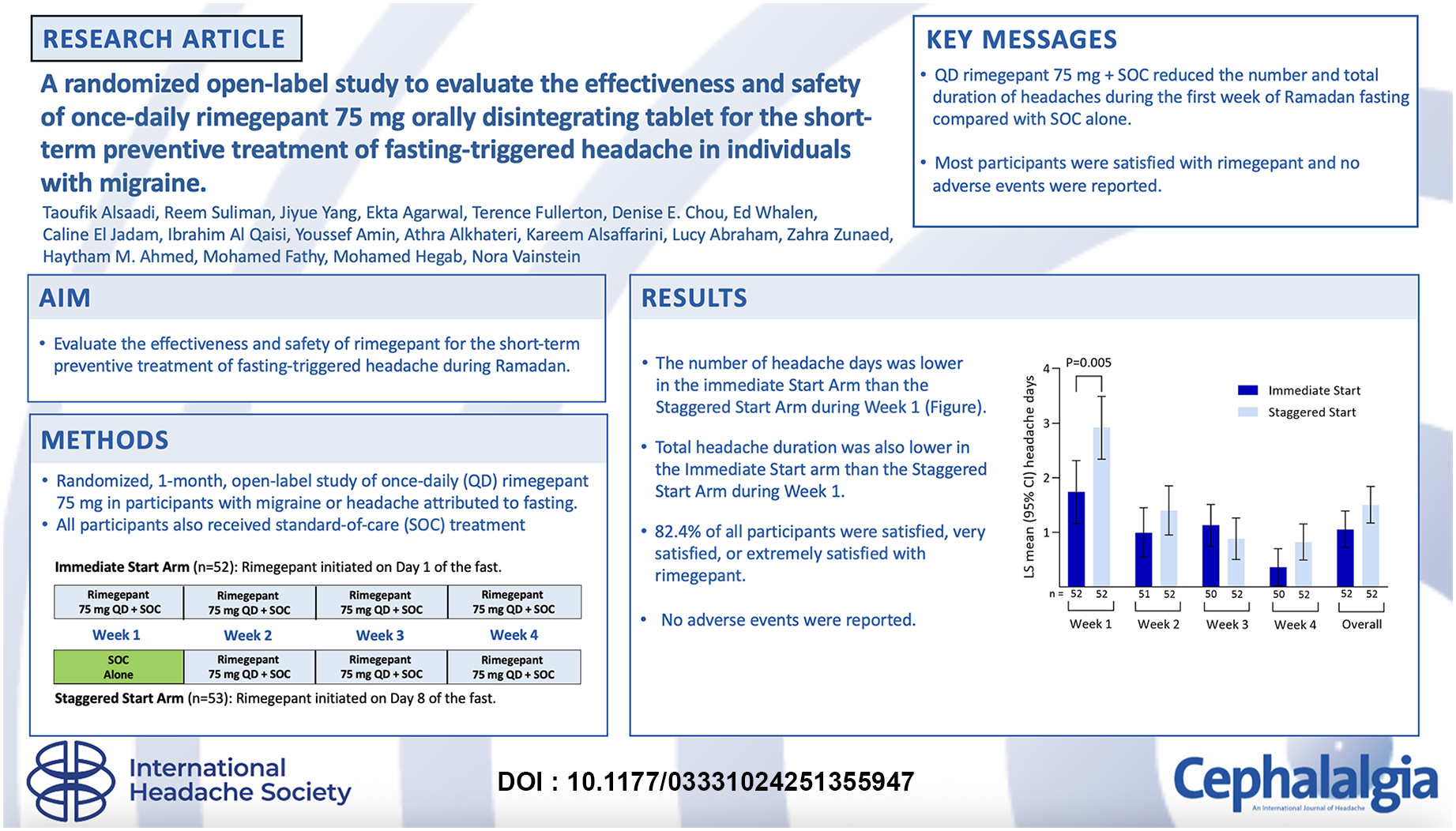
Keywords
Introduction
Fasting is a well-recognized headache trigger and headaches during fasting are significantly more common in people with a history of primary headache disorders such as migraine (1). Although headache attributed to fasting can trigger or resemble migraine, it is a secondary headache disorder distinct from migraine and other primary headache disorders (1). In contrast to the unilateral pulsating pain associated with migraine, headache attributed to fasting is typically characterized by a diffuse, non-pulsating, mild-to-moderate headache that occurs during fasting and is relieved after eating (1). The likelihood of developing such headaches increases with longer duration of fasting (2).
The global burden of fasting-triggered headache is substantial due to the large number of people who fast during religious holidays such as the Muslim month of Ramadan and the Jewish holiday of Yom Kippur. Approximately 21% of adults in the USA report that they fast at some point each year for religious reasons, including 80% of US Muslims (3). Globally, it is estimated that over 90% of the approximately two billion Muslims fast during Ramadan (4). In a large-scale observational study of 16,027 people fasting during Ramadan, over one-third reported experiencing headaches during fasting (5). Additionally, the number of people practicing intermittent fasting for medical or lifestyle reasons (e.g. weight loss and/or control of cholesterol, blood pressure or glucose) has grown rapidly in recent year (6,7). Headache, however, is among the most common adverse effects of intermittent fasting, affecting over 60% of individuals, and can interfere with daily activity and lead to individuals breaking their fast (8–10).
The month of Ramadan offers an ideal model for studying treatment approaches to fasting-triggered headache because of the prolonged duration of the fast (abstinence from food and drink from dawn to sunset for one month) and high frequency of headache, especially in the first week. Headaches occur in approximately 40–50% of fasting individuals on the first day of Ramadan and the frequency of headache is significantly lower following the initial 7–10 days (11–13).
“Short-term prevention” is a new, personalized treatment approach for conditions such as migraine and fasting-triggered headache in which patients pre-emptively take a medication prior to and during periods of known elevated risk, but not at other times when risk factors are absent. This approach can be beneficial for patients with predictable headache/migraine triggers, such as those who anticipate headaches during a planned fasting period or those who would like to fast for religious or health reasons but cannot fast due to the probability of an intolerable headache (14). Clinical research in fasting-triggered headache is sparse, however, and focuses on agents commonly used for the treatment of migraine; there are no treatments specifically indicated for fasting-triggered headache. In one study, short-term use of extended release paracetamol failed to significantly impact prevention of fasting-triggered headache (15). In contrast, short-term preventive use of frovatriptan (a long-acting triptan) decreased the incidence of fasting-triggered headache compared to placebo, although the differences between treatment groups were not statistically significant (16). Finally, two separate studies have demonstrated that use of the long-acting cyclooxygenase-2 (Cox-2) inhibitor etoricoxib is capable of reducing the incidence and severity of fasting-triggered headache (13,17).
Although the precise mechanisms underlying fasting-triggered headache are unknown, several factors have been proposed to play a possible role including dehydration, hypoglycemia, lack of sleep and caffeine withdrawal (2). Recently, it was demonstrated that levels of calcitonin gene-related peptide (CGRP; a neuropeptide of the trigeminovascular system that plays a key role in the pathogenesis of migraine pain)(18) are significantly elevated in participants who experienced headache on the first day of Ramadan compared to those who did not experience headache (19). Among participants experiencing headache (n = 21), 9.5% had a self-reported history of migraine and 52.4% had a self-reported history of non-specified headache (19). These preliminary findings suggest a role for CGRP in fasting-triggered headache and highlight the need for further investigation into this mechanism.
Oral CGRP receptor antagonists like rimegepant may be well suited for this approach due to their relatively fast tissue distribution, consistent steady state of drug exposure over the whole fasting period, and lack of wearing off toward the end of dosing interval. Oral CGRP receptor antagonists also have a rapid time to onset, short-to-intermediate half-life, and favorable safety profile (20).
Rimegepant is an oral small-molecule gepant that is currently the only agent approved for both the acute treatment of migraine and for the prevention of episodic migraine in adults (20). The recommended dose of rimegepant orally disintegrating tablet (ODT) is 75 mg (up to once a day) for acute treatment of a migraine attack and 75 mg every other day (EOD) for continuous preventive treatment of episodic migraine (20). These dosing regimens have been shown to be effective and well tolerated in a series of double-blind, randomized, placebo-controlled, phase 2/3 and phase 3 trials (21–24). Two long-term studies (12-week/52-week) have also demonstrated that dosing of rimegepant 75 mg up to once a day (QD) is well tolerated, with an adverse event profile comparable to placebo (25,26).
The present study therefore aimed to evaluate the effectiveness and safety of QD rimegepant 75 mg ODT for the short-term preventive treatment of fasting-triggered headache during the one-month period of Ramadan in the United Arab Emirates (UAE). QD dosing was assessed in this study because it offers more constant rimegepant exposure (reduced peak to trough variation) compared to EOD dosing, which may be critical for the prevention of fasting-triggered headache, particularly during the initial days of fasting.
Methods
Study design and oversight
This was a single-center, two-arm, randomized, staggered start, open-label study conducted at the American Center for Psychiatry and Neurology (ACPN, Abu Dhabi, UAE) during the month of Ramadan (10 March to 9 April) in 2024. The study was conducted in accordance with all applicable UAE government regulations and the policies and procedures of the ACPN. The study protocol was reviewed and approved by Institutional Review Board of the ACPN (reference number ACPN_IRB_0061) and all study participants provided written informed consent before entering the study.
Participants
Participants aged 18–65 years with a diagnosis prior to age 50 years of migraine according to International Classification of Headache Disorders, 3rd edition (ICHD-3) 1.1 criteria or 1.2, or headache attributed to fasting based on ICHD-3 criteria 10.5, and with a history of migraine or headache related to fasting during the previous three years were eligible for inclusion. Key exclusion criteria included: a diagnosis of any primary headache disorder other than migraine; currently taking ubrogepant or atogepant for the acute or preventive treatment of migraine; injection of a preventive treatment for migraine (e.g. CGRP monoclonal antibodies or onabotulinumtoxinA) during the week prior to the first week of the fast and during the first week of the fast; stage 4 renal disease (creatinine clearance <30 ml/min); concomitant use of CYP3A4 inhibitors, CYP3A inducers or potent P-glycoprotein inhibitors; severe hepatic impairment; current evidence of uncontrolled, unstable or recently diagnosed cardiovascular disease, uncontrolled hypertension or uncontrolled diabetes; current diagnosis of major depressive disorder requiring treatment with atypical anti-psychotics, schizophrenia, bipolar disorder or borderline personality disorder; and history of use of narcotic or barbiturate containing medications on five or more days per month for three or more months prior to randomization.
Treatment
Participants were randomized in a 1:1 open-label manner to rimegepant 75 mg QD from day 1 (weeks 1–4) of the fast (“Immediate Start” arm) or to rimegepant 75 mg QD from day 8 (weeks 2–4) of the fast (“Staggered Start” arm) (Figure 1). Randomization was conducted using randomization cards. In total, 110 cards (55 for each treatment arm) were created. Cards were shuffled and participants were enrolled based on the card (arm) randomly selected. Participants took the first dose of rimegepant during the pre-dawn meal on day 1 (Immediate Start arm) or day 8 (Staggered Start arm) of the fast. Subsequent doses were taken daily, at the same time of day, throughout the 30-day fast. Participants could continue standard-of-care preventive treatment for migraine if the dosing regimen was stable for six or more months and expected to continue throughout the fast. Participants could use the following rescue medications for acute treatment of headache attacks at any time during the fast: aspirin, ibuprofen, acetaminophen (up to 1000 mg/day), non-steroidal anti-inflammatory drugs (NSAIDs), antiemetics, baclofen or short-acting triptans.

Study design. Abbreviations: QD = once daily; SOC = standard-of-care.
Assessments
Study visits occurred at baseline (in-person; three or more days before start of fast), at the end of week 1 (virtual; ± 3 days), at the end of week 2 (virtual; ± 3 days) and at follow-up (in-person; seven days after cessation of rimegepant treatment). Each day from the start of the fast until end of study (or study withdrawal), participants recorded data in clinical paper diary. Participants documented the occurrence and severity of headaches on a four-point scale (0 = no pain, 1 = mild pain, 2 = moderate pain, 3 = severe pain), rescue medication use and the occurrence of adverse events (severity and relationship to study treatment determined by the investigator). At the end of the study, participants also indicated how satisfied they were with rimegepant treatment on a seven-point scale from “Extremely Dissatisfied” to “Extremely Satisfied”.
Endpoints
The primary endpoint was the difference in the number of headache days of any intensity during week 1 between the Immediate Start arm (treated with rimegepant during week 1) and the Staggered Start arm (not treated with rimegepant during week 1). Other endpoints included the difference in the number of headache days of any intensity between treatment arms during weeks 2–4, difference in the number of moderate-to-severe headache days between treatment arms during weeks 1–4, the proportion of participants in each treatment arm with moderate-to-severe headaches during weeks 1–4, the proportion of participants in each treatment arm who used rescue medication during weeks 1–4, the proportion of the overall population that used rescue medications during week 2–4, the total duration of headaches of any intensity and of moderate-to-severe intensity in each treatment arm during weeks 1–4 and participant satisfaction with treatment in the overall population.
Additional subgroup analyses included the difference in headache days of any intensity, and of moderate-to-severe intensity, between treatment arms at weeks 1–4 for the following cohorts: those using preventive migraine medications at baseline, those not using preventive migraine medications at baseline, those with episodic migraine (based on historical diagnostic coding), those with chronic migraine (based on historical diagnostic coding), females and males.
Safety and tolerability endpoints included the proportion of participants experiencing an adverse event (AE), a serious AE or discontinuing treatment due to an AE.
Statistical analysis
Based on the sample size of convenience, up to approximately 100 participants were planned for enrollment in the study, targeting 50 or more participants in each treatment arm. No power analysis was performed.
Participant demographics, clinical characteristics and safety data were summarized descriptively for all randomized participants. Effectiveness was analyzed in all randomized participants who provided post-baseline data. For the primary endpoint, least-squares (LS) mean headache days were calculated using a repeated-measures model with an unstructured error covariance matrix adjusted for current use of background preventive migraine medications at baseline, diagnosed migraine type and gender. Endpoints comparing the proportion of participants who used rescue medications or who achieved treatment satisfaction between treatment arms were presented using descriptive statistics with p values calculated using Fisher's exact test. Endpoints comparing proportions of patients with headaches of moderate-to-severe intensity were presented using descriptive statistics. For endpoints comparing duration of headache, LS means were calculated from a zero-inflated negative binomial model adjusted for current use of background preventive migraine medications at baseline, diagnosed migraine type, and gender, with confidence intervals (CIs) and one-sided p values based on bootstrap sampling. For subgroup analyses comparing the number of headache days between treatment arms after categorization of participants by background preventive use (yes/no), migraine type (episodic/chronic) and gender (male/female), LS means were calculated from a repeated-measures model with an unstructured error covariance matrix adjusted for the two characteristics (background preventive use, migraine type and gender) not used for categorizing participants.
The statistical analysis plan was developed post hoc and, as such, all treatment comparisons are uncontrolled for type I error and all p values are nominal. Efficacy analyses are therefore considered exploratory.
Results
Study population
In total, 105 participants were randomized (Immediate Start arm = 52; Staggered Start arm = 53), 104 (99.0%) received rimegepant and 102 (97.1%) completed the study (Figure 2). Mean age of the study population was 38.4 years and 78.1% were female (Table 1). All participants had a diagnosis of migraine (episodic = 78.1%; chronic = 21.9%) and had a history of headaches during fasting; no participants were diagnosed with headache attributed to fasting. Mean Migraine Disability Assessment (MIDAS) score was 28.7, indicating severe disability. Almost half (48.6%) of participants used background preventive migraine medications at baseline including anti-CGRP pathway monoclonal antibodies (42.9%), antihypertensives (5.7%), anticonvulsants (1.0%), antidepressants (1.0%) and supplements (1.0%). Medications for acute migraine treatment were used by 21.9% of participants at baseline including triptans (19.0%), analgesics plus antiemetics (1.0%), antidepressants (1.0%), antihypertensives (1.0%) and NSAIDS (1.0%).

Participant disposition.
Baseline demographics and clinical characteristics of the study population.

MIDAS scores range from 0–270, with score representing little or no disability (0–5), mild disability (6–10), moderate disability (11–20), and severe disability (≥21).
Based on patient recall of headache days during the prior month.
Abbreviations: CGRP = calcitonin gene-related peptide; MIDAS = Migraine Disability Assessment.
The Immediate Start arm had more females (84.6% vs. 71.7%), more participants with chronic migraine (25.0% vs. 18.9%), a higher mean duration since diagnosis of migraine (11.1 vs. 7.8 years), a lower mean MIDAS score (24.2 vs. 33.2) and more participants using background preventive migraine therapies (67.3% vs. 30.2%) than the Staggered Start arm.
Effectiveness in the overall population
For the primary endpoint, the number of headache days of any intensity during week 1 was significantly lower in the Immediate Start arm compared to the Staggered Start arm (LS mean (95% CI) = 1.74 (1.16–2.31) days vs. 2.92 (2.34–3.49) days; p = 0.005) (Figure 3(a)). There were no significant differences between treatments arms during weeks 2–4. The number of moderate-to-severe headache days was also significantly lower in the Immediate Start arm compared to the Staggered Start arm at week 1 (LS mean (95% CI) = 1.04 (0.47–1.60) days vs. 2.19 (1.63–2.76) days; p = 0.005), with no differences between treatment arms during weeks 2–4 (Figure 3(b)). For the overall four-week treatment period, there was a significant reduction in the number of moderate-to-severe headache days per week in the Immediate Start arm compared to the Staggered Start arm (0.60 (0.34–0.86) days vs. 1.00 (0.75–1.26) days; p = 0.038) and a trend towards reduction in total headache days per week of any severity (LS mean (95% CI) = 1.05 (0.72–1.39) days vs. 1.50 (1.17–1.84) days; p = 0.070).
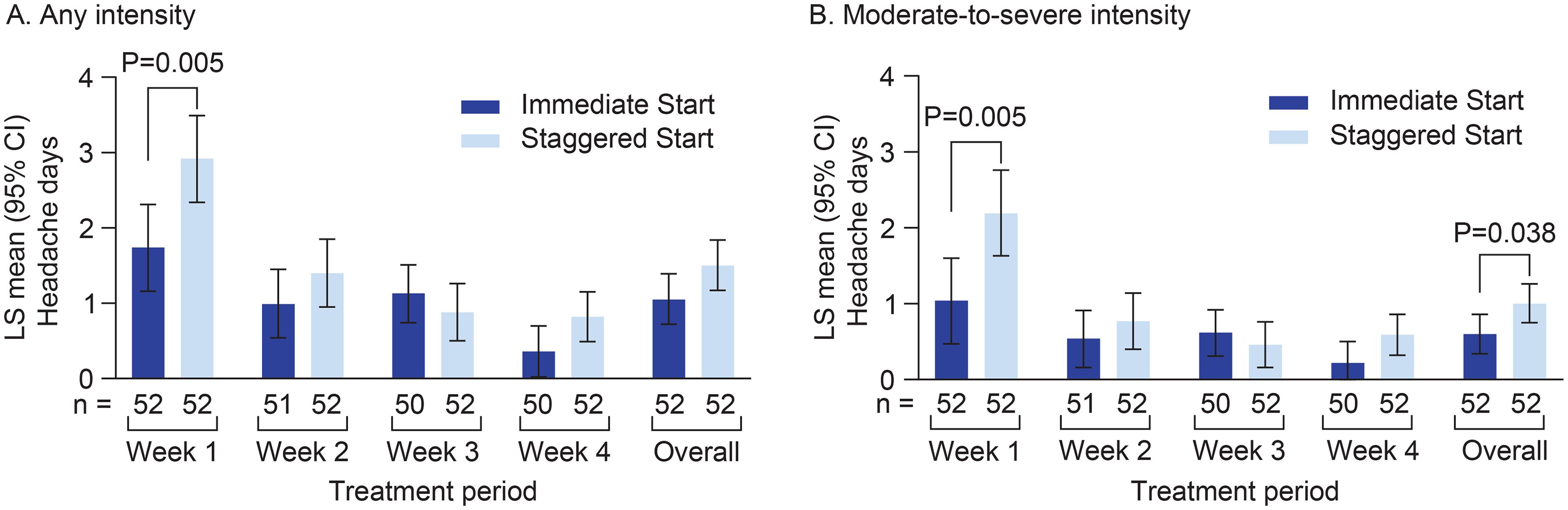
Analysis of the number of headache days of any intensity (a) or moderate-to-severe intensity (b) at each study week. LS means were calculated from a repeated-measures model with an unstructured error covariance matrix, adjusted for baseline characteristics of current use of background preventive migraine medications (yes/no), diagnosed migraine type (episodic/chronic), and gender (female/male). Overall represents the period from week 1 to the last week for which the subject provided headache data. LS means for Overall period were based on ‘main effect’ for each treatment arm. All P values are nominal. Abbreviations: CI = confidence interval; LS = least-squares; n = number of participants assessed.
The total duration of headaches of any intensity was significantly shorter in the Immediate Start arm compared to the Staggered Start arm during week 1 (LS mean (95% CI) = 10.1 (5.9–17.5) hours vs. 20.0 (13.0–28.3) hours; p = 0.041) and week 4 (LS mean (95% CI) = 0.9 (0.2–3.7) hours vs. 4.6 (2.2–15.5) hours; p = 0.035) (Figure 4(a)). The total duration of moderate-to-severe headaches was also significantly shorter in the Immediate Start arm compared to the Staggered Start arm during week 1 (LS mean (95% CI) = 6.6 (2.2–12.2) hours vs. 17.7 (11.0–26.5) hours; p = 0.011), but not during weeks 2–4 (Figure 4(b)).

Analysis of the total duration of headaches of any intensity (a) or moderate-to-severe intensity (b) at each study week. LS means were calculated from a zero-inflated negative binomial model adjusted for baseline characteristics of current use of background preventive migraine medications (yes/no), diagnosed migraine type (episodic/chronic), and gender (female/male). CIs and 1-sided P values were based on bootstrap sampling. Overall represents the period from week 1 to the last week for which the subject provided headache data. All P values are nominal. Abbreviations: CI = confidence interval; LS = least-squares; n = number of participants assessed.
The proportion of participants who experienced moderate-to-severe headache days in the Immediate Start and Staggered Start arms, respectively, was 36.5% and 69.2% during week 1, 25.5% and 30.8% during week 2, 26.0% and 30.8% during week 3, and 16.0% and 32.7% during week 4 (Figure 5). The proportion of participants using rescue medications trended lower in the Immediate Start arm than the Staggered Start arm during week 1 (21.2% vs. 32.7%), week 2 (13.7% vs. 25.0%), week 3 (6.0% vs. 11.5%) and week 4 (8.0% vs. 13.5%), but differences were not significant (all p > 0.05) (Figure 6). The proportion of all study participants (both treatment arms combined) who used rescue medications during weeks 2, 3 and 4 were 19.4%, 8.8% and 10.8%, respectively. Paracetamol was the most frequently used rescue medication in both treatment arms during each week of the study (see supplementary material, Table S1).

Proportion of participants with a maximum headache intensity of moderate or severe at each study week. No statistical analysis was performed. Abbreviations: n = number of participants assessed.

Analysis of the proportion of participants using rescue medication during each treatment week. All nominal P values (from Fisher's exact test) were > 0.05. Abbreviations: n = number of participants assessed.
At the end of treatment, 82.4% of 102 participants who completed the satisfaction survey reported being either satisfied, very satisfied, or extremely satisfied with rimegepant (Immediate Start arm = 92.0%, Staggered Start arm = 73.1%) (Figure 7). The proportion of participants who reported being very satisfied (36.0% vs. 26.9%) or extremely satisfied (26.0% vs. 15.4%) with rimegepant tended to be higher in the Immediate Start arm than in the Staggered Start arm.
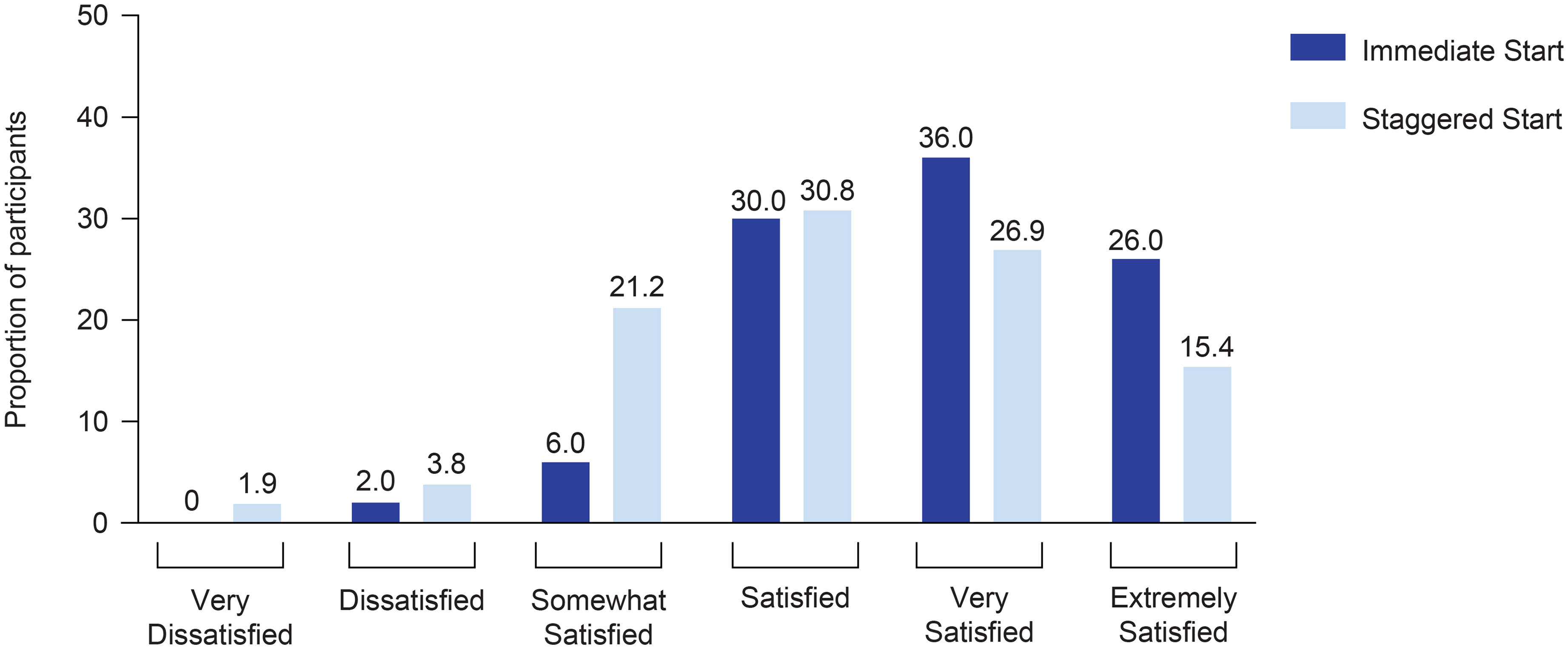
Participant satisfaction with rimegepant at end of treatment. In total, 102 (Immediate Start n = 50; Staggered Start n = 52) participants were assessed. Possible responses ranged from “Extremely Dissatisfied” to “Extremely Satisfied” (no participants had a response of “Extremely Dissatisfied”. P-value from an extension of Fisher's exact test was 0.157.
Effectiveness in subgroups of interest
Female/male subgroups
The number of headache days of any intensity and of moderate-to-severe intensity were significantly (p < 0.05) lower in the Immediate Start arm than in the Staggered Start arm during week 1 in participants who were using concomitant preventive migraine medications at baseline and in participants who were not using concomitant preventives. No significant differences between treatment arms were noted in either subgroup during weeks 2–4 (see supplementary material, Figure S1).
Episodic/chronic subgroups
The number of headache days of any intensity and of moderate-to-severe intensity were significantly (p < 0.05) lower in the Immediate Start arm than in the Staggered Start arm during week 1 in participants with episodic migraine. No significant differences between treatment arms were noted during weeks 2–4 in the episodic subgroup or during any week in those with chronic migraine (see supplementary material, Figure S2).
Preventives/non-preventives subgroups
The number of headache days of any intensity and of moderate-to-severe intensity were significantly lower in the Immediate Start arm than in the Staggered Start arm during week 1 and week 4 (moderate-to-severe intensity only) in female participants. No significant differences between treatment arms were noted at weeks 2–4 in the female subgroup or at any week in male participants (see supplementary material, Figure S3).
Safety in the overall population
No AEs, serious AEs or discontinuations due to AEs were reported. Two participants in the Immediate Start arm discontinued treatment for other reasons; one after one week of treatment and one after two weeks of treatment. These discontinuations were participant withdrawals due to not wanting to travel or commit to study assessments during Ramadan and were not due to safety/tolerability concerns or lack of efficacy.
Discussion
While the burden of fasting-triggered headache is growing, clinical assessment of potential therapies is sparse. The present study is the first to assess an agent that targets the CGRP pathway (rimegepant, a CGRP receptor antagonist) for the short-term preventive treatment of fasting-triggered headache. Although the study population allowed for inclusion of participants with migraine triggered by fasting or headache attributed to fasting, all participants in the present study had a diagnosis of migraine and reported experiencing headaches during Ramadan fasting in the past 1–3 years. Therefore, it is likely that the study population consisted of those with migraine triggered by fasting instead of headache attributed to fasting as defined by ICHD-3 criteria 10.5. It is not unexpected that all study participants had a diagnosis of migraine because fasting-triggered headache is common in participants with primary headache disorders and the study was conducted in a specialty clinic for psychiatric and neurological diseases.
The findings of the present study showed that QD dosing of rimegepant 75 mg ODT (in addition to standard-of-care acute and preventive treatments) had a clinical benefit that was evidenced by a reduction in the number of headache days, the severity of headaches and the duration of headaches compared to participants using standard-of-care alone during the first week of Ramadan. Rimegepant was also associated with a trend towards reduction in rescue medication use during week 1, a favorable safety and tolerability profile, and overall high levels of treatment satisfaction during the study.
Generally, the frequency and severity of headaches was similar in weeks 2–4 of the fast because all participants were taking rimegepant at this time and, additionally, the frequency of headaches is highest during the first week of the fast but typically decreases over time (12,13,15,16). Although the frequency of headache typically decreases over time in most individuals, headaches still persist at later times and some patients do not develop headaches until later in the fast (5,11–13,15). The safety and tolerability of rimegepant doses greater than 75 mg were explored early during clinical development, including single doses up to 1500 mg, multiple-dose regimens up to 600 mg QD for 14 days and 300 mg twice daily for up to 14 days. Across these studies, rimegepant was found to be generally safe and well tolerated with no dose-limiting toxicities (27). The basis for treating current study participants for the full month of Ramadan, rather than just the first week, was that headaches persist at later points in the fast and rimegepant has demonstrated a favorable long-term safety profile in previous trials. This approach allowed for a more comprehensive assessment of rimegepant effectiveness during a prolonged fasting period. In addition, to optimize coverage for short-term prevention of fasting-triggered headaches in this study, the QD regimen was explored with the aim of providing more sustained rimegepant exposure and CGRP receptor inhibition over the treatment period, therefore minimizing breakthrough headache (28,29). Further research would be required to compare the efficacy of EOD dosing relative to QD dosing on fasting-triggered headaches.
The reduction in headache days, headache severity, and headache frequency observed with rimegepant (Immediate Start arm vs. Staggered Start arm) in the present study compares favorably with previous trials in individuals with headaches triggered by fasting. In a randomized, double-blind, placebo-controlled trial, use of a long-acting triptan (frovatriptan 5 mg) prior to the onset of a 20-hour fast in participants with a history of migraine triggered by fasting reduced the incidence of headache compared to placebo (36% vs. 53%), but this difference was not statistically significant (16). The odds of developing a moderate-to-severe headache were 40% lower in the frovatriptan group, but again, the difference was not statistically significant (16). There was also no difference in the use of rescue medications between treatment groups (16). Due to the observation of numerical improvements in headache frequency and severity, the lack of statistically significant benefits was attributed, in part, to the low number of participants in the frovatriptan (N = 33) and placebo (N = 34) groups (16). In a double-blind, randomized, placebo-controlled, crossover trial in participants with a history of headaches during fasting, use of the long-acting Cox-2 inhibitor etoricoxib (90 mg QD; N = 96) during the first 2 weeks of Ramadan reduced the incidence (21% vs. 46%) and severity of headache compared to placebo (N = 92) during the first 5 days of the fast (13). There was no difference in the incidence of headache in the second week as headache frequency dropped substantially over time in the placebo group (13). A single 120 mg dose of etoricoxib has also been shown to reduce headache incidence (36.4% vs. 67.7%) and severity compared to placebo when taken prior to the 25-hour fast of Yom Kippur (17).
While previous trials of fasting-triggered headache demonstrate effectiveness for etoricoxib and, possibly, frovatriptan, the safety profile of rimegepant (and other gepants) is more favorable. For example, due to its vasoconstrictive properties, frovatriptan is contraindicated in patients with ischemic coronary artery disease, history of stroke or transient ischemic attack, peripheral vascular disease, or uncontrolled hypertension (30). Etoricoxib is not currently approved in the USA and also has cardiovascular contraindications including congestive heart failure, uncontrolled hypertension, ischemic heart disease, peripheral arterial disease or cerebrovascular disease (31). In contrast, rimegepant is not contraindicated in these populations and several previous studies in migraine have demonstrated that rimegepant is generally well tolerated, including long-term dosing up to once a day (20,24–26). Frequent use of triptans (and NSAIDs) is also associated with medication overuse headache, which may make them unsuitable for intermittent preventive treatment (1,32). Gepants have not been associated with a risk of MOH in preclinical, clinical or real-world settings and may represent a more rational option for intermittent prevention (25,33,34). In the present study, no AEs were reported during the four-week study period. The lack of any AEs being reported may represent a cultural factor unique to the UAE study population since 12.6–17.1% of participants in previous short-term (single dose) trials of rimegepant conducted in the USA, China and South Korea reported experiencing one or more AE (21–23,35).
The trend towards reduction in use of rescue medication for the acute treatment of headache in the Immediate Start arm vs. the Staggered start arm observed in the present study, although not statistically significant, is notable because rimegepant has been associated with long-term reductions of other acute treatments of migraine in previous studies. In a long-term open-label safety study, for example, use of rimegepant (up to once per day as needed (PRN) for 52 week or EOD plus PRN for 12 weeks) for acute treatment of migraine was associated with significant and sustained reductions in the use of select analgesics (e.g. acetaminophen and NSAIDs) and antiemetics (36). In a recent nationwide registry study in Denmark, initiation of rimegepant (for acute or preventive treatment of migraine) was associated with reduction in acute headache medications including triptans, NSAIDs, opioids and paracetamol (37). Notably, the proportion of patients meeting overuse criteria for acute headache medications was 69% prior to rimegepant initiation and 51% following rimegepant initiation. A real-world commercial claims database analysis in the USA has also demonstrated that acute treatment of migraine with rimegepant reduces opioid use (38).
In addition to highlighting the potential of rimegepant for the management of fasting-triggered headache in individuals with migraine, the results of the present study support recent evidence that implicate a possible role of CGRP in fasting-triggered headache. This evidence comes from a randomized, open-label trial in participants (N = 61) fasting for Ramadan, demonstrating that CGRP was significantly elevated in participants who experienced headache on the first day of Ramadan compared to those who did not experience headache (median 126.1 vs. 105.8 pg/ml), regardless of whether participants had received preventive treatment with paracetamol earlier in the day (19). Elevated CGRP levels were also associated with an increased risk of fasting-triggered headache (odds ratio = 1.32; 95% CI = 1.06–1.22) (19). Elevated CGRP has been demonstrated in multiple headache disorders and plays a key role in migraine-related pain (39). As such, several agents targeting the CGRP pathway, including rimegepant, are approved for the acute or preventive treatment of migraine. The findings from the present study may also suggest a role for CGRP in fasting-triggered headaches and support the need for further investigation into this area.
Results from this study also demonstrate the utility of QD rimegepant for short-term headache prevention during periods of elevated risk. The efficacy and safety of rimegepant in this study was supported by the high treatment satisfaction ratings provided by participants, particularly those in the Immediate Start arm who initiated rimegepant treatment at the beginning of the fast. It is possible that these findings, where fasting was the risk factor for headache, may extend to other situations where short-term prevention may be applicable. Other possible headache triggers may include times of elevated stress, exercise, bright lights, caffeine withdrawal, weather and menstruation (14,40,41). It is possible rimegepant, or other gepants, may be beneficial for the prevention of headaches caused by these triggers, but further research is required. It should be noted, however, that there may be unknown risks associated with short-term preventive strategies with rimegepant. These include difficulty in predicting times of increased headache risk and unnecessary exposure to rimegepant, particularly long-term exposure despite the favorable safety profile of rimegepant demonstrated in clinical studies up to 52 weeks in duration (42).
A few limitations of the present study should be noted, including the open-label design, which can introduce bias by impacting participant treatment expectations, lack of a placebo or blinded control group, and a post-hoc analysis plan that did not control for multiple comparisons and type I error. The study also had a relatively small sample size that was not powered for statistical analysis and was, therefore, exploratory in nature. A portion of participants (approximately 50%) were on background preventive therapy, including antibodies targeting the CGRP pathway. This may have affected the results by enriching the study population with individuals who are, at least somewhat, responsive to CGRP-directed therapies. There were also some imbalances in participant demographics and clinical characteristics (e.g. gender, migraine type, use of preventives) between treatment arms that could have had an unknown impact on the study results. In addition, assessment of non-pain symptoms (e.g. nausea, photophobia and phonophobia) and aspects that could potentially serve as a headache trigger or confounding factor during fasting (e.g. sleep patterns, hydration levels, caffeine withdrawal, menstrual cycles) were not assessed. Conclusions in the subgroups of interest may be limited by the low number of participants in some cohorts. Finally, as noted earlier, no participants were diagnosed with headache attributed to fasting based on strict ICHD-3 criteria (all participants had a migraine diagnosis) and thus treatment effects in participants without a historical migraine diagnosis are unknown. It should be noted, however, that all participants had headaches triggered by fasting based on historical recall of headache causation.
Conclusions
QD dosing of rimegepant 75 mg ODT was well tolerated and reduced the frequency, severity and duration of headache in fasting individuals who had a history of headaches during fasting and a diagnosis of migraine. Overall, these findings suggest that rimegepant may be a promising treatment for the short-term prevention of fasting-triggered headaches in individuals with migraine. Rimegepant is not currently indicated for the treatment of fasting-triggered headache and adequately powered, well-controlled, blinded studies would be required to confirm the clinical efficacy and safety/tolerability of CGRP pathway antagonism for the short-term preventive treatment of fasting-triggered headache.
Clinical implications
QD rimegepant 75 mg, in addition to standard-of-care, was associated with reduced number and total duration of headaches during the first week of Ramadan fasting compared to standard-of-care alone.
Most participants reported being satisfied, very satisfied or extremely satisfied with rimegepant at end of treatment and no adverse events were reported over three to four weeks of treatment.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251355947 - Supplemental material for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine
Supplemental material, sj-docx-1-cep-10.1177_03331024251355947 for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine by Taoufik Alsaadi, Reem Suliman, Jiyue Yang, Ekta Agarwal, Terence Fullerton, Denise E Chou, Ed Whalen, Caline El Jadam, Ibrahim Al Qaisi, Youssef Amin, Athra Alkhateri, Kareem Alsaffarini, Lucy Abraham, Zahra Zunaed, Haytham M Ahmed, Mohamed Fathy, Mohamed Hegab and Nora Vainstein in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024251355947 - Supplemental material for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine
Supplemental material, sj-pdf-2-cep-10.1177_03331024251355947 for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine by Taoufik Alsaadi, Reem Suliman, Jiyue Yang, Ekta Agarwal, Terence Fullerton, Denise E Chou, Ed Whalen, Caline El Jadam, Ibrahim Al Qaisi, Youssef Amin, Athra Alkhateri, Kareem Alsaffarini, Lucy Abraham, Zahra Zunaed, Haytham M Ahmed, Mohamed Fathy, Mohamed Hegab and Nora Vainstein in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024251355947 - Supplemental material for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine
Supplemental material, sj-pdf-3-cep-10.1177_03331024251355947 for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine by Taoufik Alsaadi, Reem Suliman, Jiyue Yang, Ekta Agarwal, Terence Fullerton, Denise E Chou, Ed Whalen, Caline El Jadam, Ibrahim Al Qaisi, Youssef Amin, Athra Alkhateri, Kareem Alsaffarini, Lucy Abraham, Zahra Zunaed, Haytham M Ahmed, Mohamed Fathy, Mohamed Hegab and Nora Vainstein in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_03331024251355947 - Supplemental material for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine
Supplemental material, sj-pdf-4-cep-10.1177_03331024251355947 for A randomized open-label study to evaluate the effectiveness and safety of once-daily rimegepant 75 mg orally disintegrating tablet for the short-term preventive treatment of fasting-triggered headache in individuals with migraine by Taoufik Alsaadi, Reem Suliman, Jiyue Yang, Ekta Agarwal, Terence Fullerton, Denise E Chou, Ed Whalen, Caline El Jadam, Ibrahim Al Qaisi, Youssef Amin, Athra Alkhateri, Kareem Alsaffarini, Lucy Abraham, Zahra Zunaed, Haytham M Ahmed, Mohamed Fathy, Mohamed Hegab and Nora Vainstein in Cephalalgia
Footnotes
Acknowledgements
Medical writing support was provided by David Wateridge, PhD, and Matt Soulsby, PhD, CMPP of Engage Scientific Solutions and was funded by Pfizer. Some of the data in this article were presented at the 18th European Headache Congress in December 2024 (Rotterdam, Netherlands) and at the 77th Annual Meeting of the American Academy of Neurology in April 2025 (San Diego, CA, USA).
ORCID iDs
Ethical statement
The study was conducted in accordance with all applicable UAE government regulations and the policies and procedures of the ACPN. The study protocol was reviewed and approved by Institutional Review Board of the ACPN (reference number ACPN_IRB_0061) and all study participants provided written informed consent before entering the study.
Funding
This study was conducted as a collaboration between the American Center for Psychiatry and Neurology (ACPN; Abu Dhabi, UAE) and Pfizer. The ACPN funded the study and is the study sponsor. Rimegepant was supplied by Pfizer.
Declaration of conflicting interests
TA, RS, CEJ, IAQ, YA, AA and KA declare no conflict of interest. JY, EA, TF, DEC, EW, LA, ZZ, HMA, MF, MH and NV are (or were at the time of the study) employees of Pfizer and may own stock/options in Pfizer.
Data availability
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
