Abstract
Background
Stress is a major trigger for migraine attacks. Stress activates the hypothalamic-pituitary-adrenal (HPA) axis, releasing glucocorticoids (GCs) to maintain homeostasis, and migraine attacks may occur as an adverse effect of this response. We previously demonstrated in a mouse model that inhibiting corticosterone (CORT) synthesis by administering metyrapone before stress prevented stress-induced migraine-like behaviors. Given the unpredictable nature of stressors and their onset or termination, it is critical to better understand the adaptive and maladaptive effects of the HPA stress response. Here, we aimed to evaluate the effects of HPA axis modulation following the end of stress exposure.
Methods
Repeated stress induces migraine-like behaviors and priming to sodium nitroprusside (SNP) in mice. Metyrapone (to inhibit CORT synthesis), CORT (to evaluate its effects after exogenous administration), and adrenocorticotropic hormone (ACTH) (to test the effects of a hormone upstream to CORT) were administered post-stress. Additionally, α-melanocyte-stimulating hormone (α-MSH, an ACTH cleavage product) and tetrahydroisoquinoline (THIQ), a melanocortin 4 receptor (MC4R) agonist, were administered to examine melanocortin receptor involvement. Facial hypersensitivity was assessed via von Frey testing and grimace scoring was used to evaluate non-evoked pain. Serum CORT levels were measured in both control and stressed mice following ACTH administration.
Results
We examined post-stress HPA axis modulation on stress-induced facial hypersensitivity. Metyrapone reduced acute-phase hypersensitivity and reduced priming to SNP, suggesting sustained synthesis of CORT after stress plays a role in development of migraine-like behavior. Surprisingly, both CORT and ACTH treatments at 1- and 24-h post-stress alleviated stress-induced behaviors and priming. To determine if ACTH effects were mediated by an elevation in circulating CORT, metyrapone was administered before the ACTH injection. Metyrapone increased the ACTH reversal of stress effects on facial hypersensitivity. Furthermore, post-stress ACTH injections significantly increased serum CORT levels within 30 min. In addition to ACTH effects on CORT levels, ACTH effects could be mediated by the melanocortin system. Post-stress administration of α-MSH or the MC4R agonist THIQ, reduced migraine-like behaviors.
Conclusions
There is a complex relationship between stress, the HPA axis, and melanocortin signaling, in the effects of repeated stress exposure on migraine-like behaviors. In the early post-stress response phase, there are contributions from both CORT and MC4R signaling in the maintenance of behavioral effects. These findings suggest that targeting the HPA axis and MC4R after stress may be a potential therapeutic approach for stress-induced migraine attacks.
This is a visual representation of the abstract.
Introduction
Migraine can be triggered by various factors, but stress is the most commonly reported trigger (1). High levels of stress are particularly prevalent among individuals suffering from chronic daily migraines (2–4). Whether this relationship is due to stress as a cause or consequence of chronic migraine remains to be determined. To better understand this relationship, we investigated the effects of stress-related hormones on migraine-like behavior in mice. The hypothalamic-pituitary-adrenal (HPA) axis plays a crucial role in stress adaptation and homeostasis (5). The process starts with corticotropin-releasing hormone (CRH) released from hypothalamic paraventricular nucleus (PVN) neurons. CRH binds to Corticotropin-releasing hormone receptor 1 (CRHR1) on the pituitary gland, stimulating adrenocorticotropic hormone (ACTH) secretion from the anterior pituitary. ACTH then binds to melanocortin 2 receptor (MC2R) on the adrenal glands, triggering glucocorticoid release (6). Glucocorticoids exert genomic and non-genomic effects via the glucocorticoid receptor, regulating metabolism, immunity, and stress response (7,8). Their negative feedback through glucocorticoid (GR) and mineralocorticoid (MR) receptors is crucial for terminating the HPA axis stress response (9,10). Maladaptive changes in these circuits from repeated stress may contribute to migraine.
Our previous study showed that glucocorticoid signaling drives stress-induced migraine-like behavior in mice (11). Inhibiting glucocorticoid synthesis prevents this behavior and disrupts feedback, affecting CRH, ACTH, and HPA axis function. Functional imaging studies highlight the role of the hypothalamus in acute and chronic migraine (12–15). Using nitric oxide as a premonitory phase model revealed increased hypothalamus activity before headache onset (16). During the prodrome phase, hypothalamic activity was coupled with activation of the spinal trigeminal nuclei, and in the headache phase, with activation of the dorsal rostral pons (17). These distinct network changes in hypothalamic connectivity may play a key role in triggering migraines. The clinical features of migraines, such as cyclic attacks, prodromal symptoms, autonomic symptoms, and triggers like hormones and stress, further emphasize hypothalamic involvement (14). Moreover, chronic migraine patients show abnormal hypothalamic hormone patterns, including a decreased nocturnal prolactin peak, elevated cortisol, and a delayed melatonin peak (18), and more recent evidence suggests that endogenous glucocorticoids may serve as biomarkers for migraine progression and treatment response (19). Furthermore, corticosteroid treatments, such as a single-dose intravenous dexamethasone, have been shown to reduce the severity of recurrent headaches and improve responsiveness to nonsteroidal therapies (20). These findings suggest the HPA axis plays a key role in stress-induced migraine attacks.
In addition to the HPA axis, the hypothalamic melanocortin system also modulates stress responses (21–23). Melanocortins, derived from proopiomelanocortin (POMC), include ACTH and various MSH forms (α-, β-, γ-, δ-MSH). Melanocortin receptors (MC1R–MC5R) regulate functions like learning, thermoregulation, pain modulation, stress responses, and inflammation (24,25). MC4R, expressed in both central and peripheral nervous systems (26–28), is activated by the endogenous agonist α-MSH and pharmacological agents to reduce caloric intake, increase energy expenditure, and affect glucose and lipid metabolism (29,30). A previous study found that loss of MC4R function reduces HPA axis responses to stress, suggesting its role in HPA dysregulation (31). Additionally, MC4R is involved in nociception (32–34).
Cortisol is the primary glucocorticoid in humans, while corticosterone (CORT) is the primary glucocorticoid in rodents. Previous studies have shown that repetitive restraint stress induces migraine-like behaviors in a preclinical mouse model (35). Additionally, we previously showed that pretreatment with metyrapone, which inhibits CORT, prevents stress-induced migraine-like behaviors in mice, suggesting a role for glucocorticoid signaling in the development of migraine-like behaviors (11). Here, we sought to determine what role HPA axis signaling plays in the early post-stress phase of behavior that is contributing to the maintenance of stress responses. We administered CORT or inhibited its synthesis post-stress to evaluate what role CORT plays in post-stress behavior. Additionally, we proposed that elevation of the upstream hormone ACTH, along with its cleavage product α-MSH, may contribute to post-stress responses through the melanocortin receptor MC4R. By exploring these pathways, we aim to enhance our understanding of the mechanisms underlying stress-induced migraine attacks and identify potential therapeutic targets for rapid clinical translation given the current availability of many drugs targeting HPA axis function.
Methods
Animals
Adult male and female ICR (CD-1) mice at 6–8 weeks old were purchased from Charles River Laboratories and housed under a 12-h light/dark cycle at the University of Texas at Dallas. Mice were randomly assigned to groups, and investigators were blinded to treatments. The total sample size analyzed in this study consisted of 295 female mice and 281 male mice. The average weight of male mice ranged from 33 to 36 g, while the average weight of female mice ranged from 28 to 32 g. Male and female mice were tested separately throughout the entire experiment. All procedures were approved by the Institutional Animal Care and Use Committee and followed ARRIVE guidelines, the policies of the International Association for the Study of Pain, and the National Institutes of Health guidelines for animal research.
Habituation
Each animal was handled for 5 min on the first day, then habituated in 4 oz cups (Choice) within the von Frey chamber for 2 h daily over 3–5 days, as described previously (35). Baseline pain sensitivity was tested by von Frey filaments and facial expression was observed by mouse grimace score.
Restraint stress paradigm
The preclinical stress model was based on our previous study (35). Briefly, animals were restrained in cylindrical tail access devices (Stoelting 51338) for 2 h daily over 3 consecutive days, starting after baseline measurements. The restraint took place from 10:00 am to 12:00 pm, preventing interference with the natural rise in corticosterone levels (36). During restraint, control animals were kept in their home cages without food or water. After restraint, both groups were returned to their cages with food and water. Stress and control mice were housed separately for the duration of the experiments.
Facial mechanical hypersensitivity
We previously demonstrated that restraint stress induces facial mechanical hypersensitivity lasting for a duration of 14 days (35). Briefly, baseline sensitivity was determined by habituation to the 0.6 g filament, with testing starting at 0.07 g and adjusting force accordingly (increasing to 0.6 g if no response was observed or decreasing to 0.008 g if a response occurred). Animals that did not respond to the 0.6 g filament were considered at baseline. Hypersensitivity was assessed using von Frey filaments (0.07–0.6 g) applied to the periorbital areas on 1, 3, 5, 7, 10 and 14 days after stress. The animals were randomly allocated to either the control group or the treatment group. By day 14, animals returned to baseline sensitivity. Nitric oxide donor sodium nitroprusside (SNP) was injected to test for priming. The von Frey testing was performed at 1, 3, 24, 48, and 72 h post-injection.
Spontaneous pain
Mouse grimace score (MGS), a standardized behavioral coding system for assessing spontaneous pain via facial expression change (37), was evaluated prior to von Frey testing to prevent mechanical stimulation influencing results. Animals were habituated in the chamber for 10–20 min, after which facial expression changes were assessed using five indicators, and scores were assigned directly. Five indicators, orbital tightening, nose bulging, cheek bulging, flattening of ears and flattening of whiskers, were scored from 0 (no grimace) to 2 (maximal grimace). Mice, randomly placed in testing chambers, were recorded for 3 min using camera (Logitech HD webcam C270). The facial expression changes were evaluated directly using these five indicators, with scores assigned in real time. Video recordings were primarily used as a reference to confirm scores, allowing for double-checking in cases of uncertainty. The recordings served as supplementary material for verification. Experimenters were blinded to the treatments or groups when measuring grimace scores.
Drugs
Metyrapone (50 mg/kg (38), Tocris Bioscience, USA) was prepared in 20% propylene glycol and administered subcutaneously twice: 20 min after the final stress session and 40 min prior to 24-h post-stress time point. A low dose of CORT (2 mg/kg (39), Sigma-Aldrich, St Louis, MO) was initially dissolved in 2% dimethyl sulfoxide (DMSO), diluted in peanut oil, and administered via intraperitoneal injection. ACTH (2 mg/kg, MedChemExpress, Monmouth Junction, NJ) and α-MSH (2 mg/kg, Sigma Aldrich, St Louis, MO) were prepared in 0.9% saline and injected intraperitoneally. MC4R was activated by an i.p injection of tetrahydroisoquinoline (THIQ) (2 mg/kg in 0.9% saline, Tocris Bioscience, USA). Drugs or vehicle injections (200 µl) were given one hour after removal from restraint on stress Day 3, and 24 h later. The initial behavioral test was conducted 24 h post-stress. The second injection was administered immediately after the behavioral test. In the ACTH with metyrapone experiment, metyrapone or vehicle was injected subcutaneously 20 min after the final stress session, followed by ACTH (2 mg/kg) or vehicle 40 min later. Second doses of metyrapone and ACTH were administered 24 h post-stress. SNP (0.1 mg/kg (35); Sigma Aldrich, St Louis, MO) was dissolved in 1X PBS and administered via intraperitoneal injection at a total volume of 150 μl.
Serum corticosterone measurement
The blood sample collection and serum CORT measurements were performed according to previously published methods (11). Serum CORT levels were measured using the DetectX® Corticosterone Enzyme Immunoassay Kit (Arbor Assays, MI, USA) following the manufacturer's protocol. A separate cohort of mice, distinct from those used in behavioral experiments. A total of 80 female mice and 70 male mice were used in these experiments. Animals underwent three consecutive days of restraint stress, followed by a 2 mg/kg ACTH injection 1 h after the final stress session on Day 3 (13:00). Blood samples were collected at the following time points: pre-ACTH injection (13:00 on Day 3), 30 min post-ACTH (13:30 on Day 3), 1 h post-ACTH (14:00 on Day 3), and 24 h post-stress (12:00 on Day 4). A second 2 mg/kg ACTH injection was administered 24 h later (12:00), and blood samples were collected at 30 min (12:30 on Day 4), 1 h (13:00 on Day 4), and 24 h (12:00 on Day 5) post-injection. To reduce stress from repeated blood draws, separate groups of animals were used for each sampling time point.
Statistical analysis
All data shown in the figures are mean ± SEM. These behavior data were analyzed for each time point using two-way repeated measures ANOVA and then followed by Bonferroni post hoc analysis. Serum CORT levels from the control group and stress group were analyzed using one-way ANOVA followed by Bonferroni post hoc analysis. Male and female data were analyzed separately. Additional analysis was also done comparing serum levels between male and female mice using a one-way ANOVA followed by Bonferroni post hoc analysis. This approach may reduce power for detecting small effects. While Bonferroni post hoc analysis is conservative, it ensures robustness against false positives. Data were analyzed using Prism 10 (GraphPad Software). Significance was set at P < 0.05 for all analyses. Following testing of baselines to confirm mice had no responses to the maximum filament forces prior to stress, mice were randomly assigned to different groups. The experimenter was blinded to the drug treatments, ensuring they were unaware of which animal received which drug. G power analysis was employed to determine the required sample size for each experiment. The expected effect size was determined based on our previously published data in this animal model (35,40). For periorbital von Frey tests, all sample sizes met or exceeded the recommended minimum of 5, as calculated using G Power to achieve a desired power of 0.8. An a priori power analysis was conducted with an effect size of 1.8, an alpha level of 0.05, and the number of measurements used was 5. The von Frey test was the primary behavioral readout, as it provides a quantitative measure of facial mechanical hypersensitivity. Grimace scoring was included as a complementary measure to assess non-evoked pain, providing additional insight into the pain state of the animals. Therefore, the primary power calculation was based on von Frey testing, as it was the main behavioral outcome measure. Variation in group sizes was due to factors like animal availability and occasional exclusions for health issues or abnormal baseline pain sensitivity. However, no animals were excluded after the baseline assessments, and no data points were omitted from the study. All illustrations were created using Biorender.
Results
Post-stress metyrapone administration reduced stress-induced facial mechanical hypersensitivity in mice
Our previous studies demonstrated that modulating the HPA axis through metyrapone pre-administration prior to a stress session effectively prevents stress-induced facial hypersensitivity and priming in both male and female mice (11). This finding suggests that inhibiting stress-induced glucocorticoid fluctuations can block the development of migraine-like behaviors. Given that migraines typically occur after stress resolution, this study first examined whether post-stress metyrapone could mitigate or prevent stress-induced hypersensitivity. Metyrapone was administered twice, first at 20 min after the final (3rd) stress session and again 40 min before the 24 h post-stress time point (Figure 1(a)). Stressed female mice given vehicle injections exhibited significantly increased facial hypersensitivity compared to control mice treated with the vehicle (represented as decreased withdrawal thresholds). In contrast, stressed female mice treated with metyrapone showed a significant reduction in stress-induced facial hypersensitivity during the post-stress phase compared to their vehicle-treated counterparts. During the post-SNP phase, metyrapone treatment provided only partial relief from stress-induced hypersensitivity in stressed females (Figure 1(b)). The effects of metyrapone during the post-SNP phase appear to be indirect and are likely due to its ability to mitigate stress-induced hypersensitivity during the acute phase. Grimace score analysis revealed no significant difference between stressed female mice treated with the vehicle and those treated with metyrapone (Figure 1(c)). Similarly, in male mice, metyrapone treatment significantly reduced stress-induced facial hypersensitivity in the post-stress phase (Figure 1(d)). Grimace score data also showed no significant difference between stressed male mice treated with the vehicle and those treated with metyrapone (Figure 1(e)). These findings suggest that post-stress inhibition of CORT synthesis via metyrapone alleviates stress-induced hypersensitivity.

Post-stress administration of metyrapone alleviates restraint stress-induced facial hypersensitivity in female and male mice. (a) The experimental timeline is shown in the scheme. Mice were exposed to restraint stress for three consecutive days, then injected with either 50 mg/kg metyrapone or vehicle 1 h after stress on the last day and again 24 h later. Mice were divided into four groups: control with Veh (n = 8 females, n = 9 males), control with MET (n = 8 females, n = 9 males), stress with Veh (n = 8 females, n = 10 males) and stress with MET (n = 8 females, n = 10 males). On Day 14, after returning to baseline, all mice received 0.1 mg/kg SNP. (b and d) Facial withdrawal thresholds to periorbital stimuli applied to the face and (c and e) the grimace scores were measured in female and male mice during the acute phase (post-stress) and priming phase (post-SNP). A two-way ANOVA followed by Bonferroni multiple comparison analysis was used to compare the groups. * indicated significant differences between stressed with vehicle and control with vehicle groups. # indicated significant differences between stressed with vehicle and stressed with MET groups. Data are represented as mean ± SEM. *, #P < 0.05, **, ##P < 0.01, ***P < 0.001, ****, ####P < 0.0001. Abbreviations: MET: metyrapone, Veh: vehicle. See Supplementary Table 1 for a complete statistical analysis.
Post-stress CORT administration reduced stress-induced facial mechanical hypersensitivity in mice
Our previous study showed that pretreatment with metyrapone, which blocks CORT synthesis, prevents stress-induced hypersensitivity, highlighting the key role of CORT in the development of migraine-like behavior in mice (11). To explore the clinical relevance of modulating the HPA axis by altering CORT levels, we investigated whether post-stress CORT administration could alleviate or exacerbate stress-induced facial hypersensitivity. A low dose of CORT was administered at two timepoints: 1 h after the final stress session and 24 h post-stress (Figure 2(a)). Mice were assessed for mechanical sensitivity 24 h after stress, followed immediately by the second CORT injection. Stressed female mice showed significant hypersensitivity compared to controls. While no significant difference was observed between vehicle- and CORT-treated stressed females, those receiving CORT injections reduced hypersensitivity in the post-stress phase (Figure 2(b)). Stressed male mice treated with CORT showed a reduction in hypersensitivity during the post-stress phase (Figure 2(d)). During the post-SNP injection phase, CORT treatment significantly reduced mechanical hypersensitivity in both stressed female and male mice compared to controls (Figure 2(b) and (d)). Stressed female mice showed significant grimacing, which was reduced significantly with CORT treatment (Figure 2(c)), while there was no significant difference in grimace scores was observed overall in male mice (Figure 2(e)). These data suggest that administering a low dose of CORT after stress alleviated stress-induced facial mechanical hypersensitivity in both female and male mice.

Post-stress administration of CORT reversed stress-induced facial hypersensitivity in female and male mice. (a) The experimental timeline is shown in the scheme. Mice were exposed to restraint stress for three consecutive days, then injected with either 2 mg/kg CORT or vehicle 1 h after stress on the last day and again 24 h later. Mice were divided into four groups: control with vehicle (n = 7 females, n = 7 males), control with CORT (n = 7 females, n = 7 males), stress with vehicle (n = 8 females, n = 8 males), and stress with CORT (n = 8 females, n = 8 males). On Day 14, after returning to baseline, all mice received 0.1 mg/kg SNP. (b and d) Facial withdrawal thresholds to periorbital stimuli applied to the face and (c and e) the grimace scores were measured in female and male mice during the acute phase (post-stress) and priming phase (post-SNP). A two-way ANOVA followed by Bonferroni multiple comparison analysis was used to compare the groups. * indicated significant differences between stressed with vehicle and control with vehicle groups. # indicated significant differences between stressed with vehicle and stressed with CORT groups. Data are represented as mean ± SEM. *, #P < 0.05, **, ##P < 0.01, ***, ###P < 0.001, ****, ####P < 0.0001. Abbreviations: CORT: corticosterone, NO: nitric oxide, SNP: sodium nitroprusside. See Supplementary Table 1 for a complete statistical analysis.
Post-stress ACTH administration attenuated stress-induced facial mechanical hypersensitivity in mice
Figure 1 showed that post-stress metyrapone treatment alleviated stress-induced hypersensitivity in both female and male mice during the post-stress phase. However, administration of metyrapone post-stress inhibits CORT synthesis, disrupting the negative feedback loop on the ACTH secretion and leading to elevated ACTH levels. To explore whether the effect of metyrapone on stress-induced hypersensitivity was due to the reduction of CORT, increased ACTH, or both, we administered ACTH injections at 1 h and 24 h after stress (Figure 3(a)). No significant differences were observed between vehicle- and ACTH-treated control female mice post-stress. Stressed mice treated with vehicle became hypersensitive, while ACTH injections reduced stress-induced facial hypersensitivity during both the post-stress and post-SNP injection phases in females, similar to males (Figure 3(b) and (d)). Grimace score data showed a reduction between vehicle- and ACTH-treated stressed mice in both sexes, but no significant differences were observed (Figure 3(c) and (e)). These findings suggest that ACTH may play a role in stress-induced behavioral effects in both male and female mice.

Administration of ACTH attenuates stress-induced facial hypersensitivity in female and male mice. (a) The experimental timeline is shown in the scheme. Mice were exposed to restraint stress for three consecutive days, then injected with either 2 mg/kg ACTH or vehicle 1 h after stress on the last day and again 24 h later. Mice were divided into four groups: control with vehicle (n = 10 females, n = 9 males), control with ACTH (n = 10 females, n = 10 males), stress with vehicle (n = 9 females, n = 11 males) and stress with ACTH (n = 9 females, n = 11 males). On Day 14, after returning to baseline, all mice received 0.1 mg/kg SNP. (b and d) Facial withdrawal thresholds to periorbital stimuli applied to the face and (c and e) the grimace scores were measured in female and male mice during the acute phase (post-stress) and priming phase (post-SNP). A two-way ANOVA followed by Bonferroni multiple comparison analysis was used to compare the groups. * indicated significant differences between stressed with vehicle and control with vehicle groups. # indicated significant differences between stressed with vehicle and stressed with ACTH groups. Data are represented as mean ± SEM. *, #P < 0.05, **, ##P < 0.01, ***P < 0.001, ****P < 0.0001. Abbreviations: ACTH: adrenocorticotropic hormone, NO: nitric oxide, SNP: sodium nitroprusside. See Supplementary Table 1 for a complete statistical analysis.
Alterations in serum CORT levels following two post-stress ACTH injections in mice
Exogenous ACTH injection was expected to raise serum CORT levels (41–43). We next conducted experiments similar to those in Figure 3, but here we measured serum CORT levels following two ACTH injections (Figure 4(a)) in female and male mice. We were interested in determining whether the effects of ACTH on behavior in female and male mice were due to the direct action of ACTH or its effect on increasing CORT levels in combination with CORT influence on behavior. Blood samples were collected at the specific time points shown in Figure 4(a) from both control and stressed groups. It is important to note that the CORT measurements were different from the mice in the behavior study, with each data point representing a new mouse. As shown in Figure 4(b), serum CORT levels in control female mice remained unchanged following ACTH injection. However, in stressed female mice, serum CORT significantly increased 30 min after the first ACTH injection, then returned to baseline by 1 h. This response pattern was consistent following the second ACTH injection (Figure 4(c)). In control male mice, serum CORT levels showed a significant rise 30 min after the first ACTH injection, followed by a return to baseline, but remained stable with no change after the second injection (Figure 4(d)). In stressed male mice, we observed a non-significant upward trend in serum CORT levels at 30 min after the first ACTH injection, with a significant increase following the second injection at the same time point (Figure 4(e)).
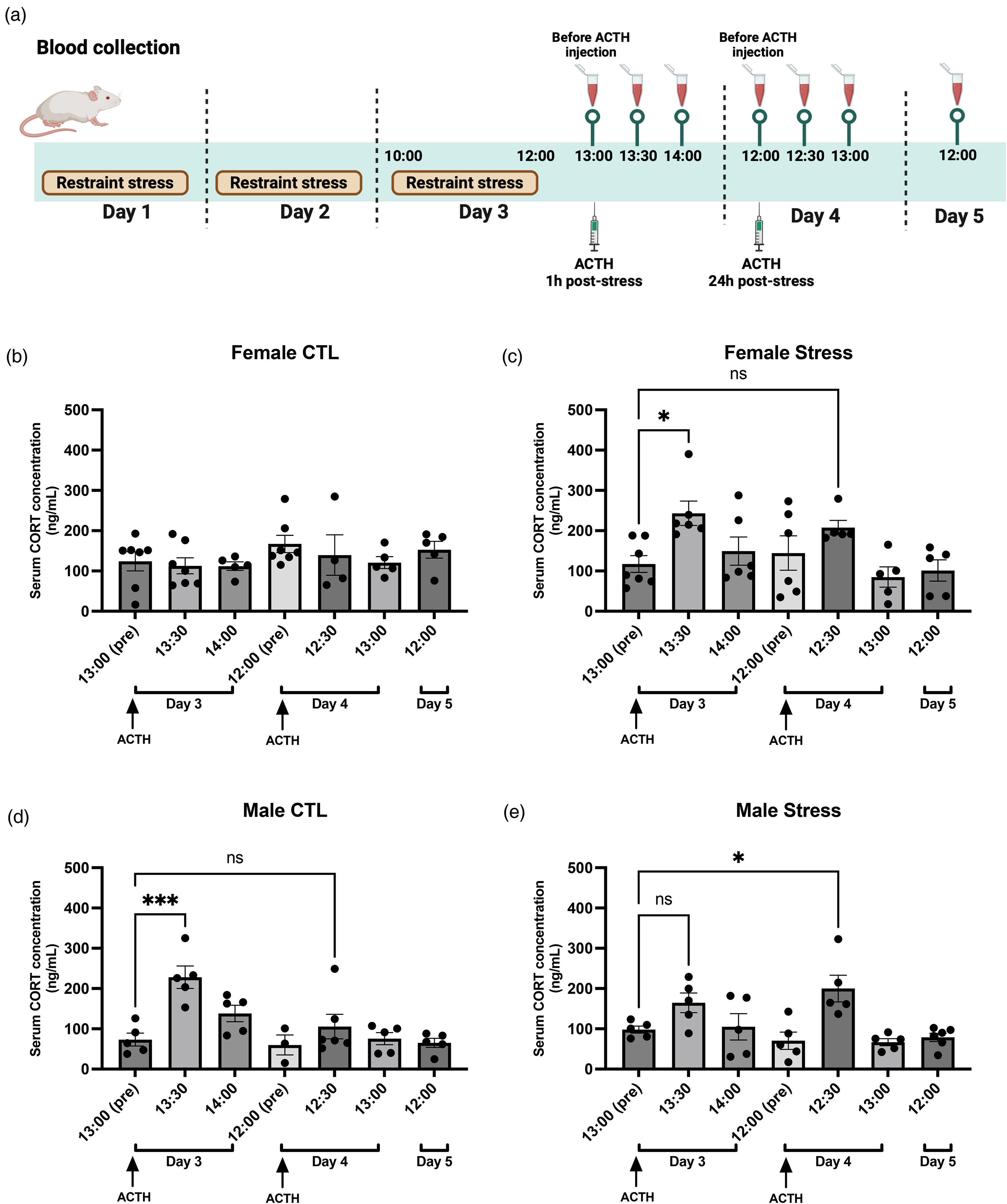
Serum CORT levels following ACTH injection in control and stressed female and male mice. (a) Mice were subjected to three consecutive days of 2-h restraint stress. One hour after the final stress session, 2 mg/kg of ACTH was administered to the animals, and blood samples were collected at specific time intervals for serum corticosterone (CORT) measurement. The sampling schedule was as follows: pre-ACTH injection (13:00, Day 3), 30 min post-ACTH injection (13:30, Day 3), 1 h post-ACTH injection (14:00, Day 3), and 24 h post-stress (12:00, Day 4). ACTH (2 mg/kg) was administered to the mice 24 h after the final stress exposure, with blood samples taken at the following time points: 30 min post-ACTH injection (12:30, Day 4), 1 h post-ACTH injection (13:00, Day 4), and 24 h post-ACTH injection (12:00, Day 5). (b) Control female mice injected with ACTH 1 h post-stress did not exhibit significant changes in serum CORT levels at any of the measured time points (n = 4–7 per group). (c) In contrast, stressed female mice injected with ACTH showed a significant increase in serum CORT levels at 30 min post-injection on the final day of stress (13:30, Day 3, n = 6). Additionally, there was a trend toward increased CORT at 30 min post-ACTH injection on the following day (12:30, Day 4, n = 5) compared to pre-injection levels (13:00, Day 3, n = 7). (d) Control male mice administered ACTH 1 h post-stress also exhibited a significant increase in serum CORT levels at 30 min post-injection (13:30, Day 3, n = 5), relative to pre-injection levels (13:00, Day 3, n = 5). However, no further changes in serum CORT were observed at subsequent time points (n = 4–6). (e) Stressed male mice showed a trend toward increased CORT levels at 30 min post-injection on the final stress day (13:30, Day 3, n = 5). A significant increase was observed at 30 min post-injection on the following day (12:30, Day 4, n = 5), when compared to pre-injection levels (13:00, Day 3, n = 5). One-way ANOVA followed by Bonferroni multiple comparison analyses were used. Data are represented as mean ± SEM. * indicate pre-ACTH injection vs. other time points in females and males. * P < 0.05, *** P < 0.001. Abbreviations: ACTH: adrenocorticotropic hormone, CTL: control. See Supplementary Table 1 for a complete statistical analysis.
Alleviation of stress-induced hypersensitivity by ACTH does not depend on CORT
While ACTH affects behavior, it did not produce a significant increase in CORT beyond what is observed with stress alone. This suggests that the effects of ACTH on behavior may not be driven by increased CORT levels. To determine if the reduction in hypersensitivity from post-stress ACTH injections was due to the direct effect of ACTH only, stressed and control mice received metyrapone 40 min before each ACTH injection to block CORT synthesis after three days of restraint stress (Figure 5(a)). Stressed female mice treated with the vehicle showed significant hypersensitivity, while ACTH with metyrapone treatment in stressed mice led to a significant reduction in hypersensitivity (Figure 5(b)). Similarly, ACTH with metyrapone-treated males exhibited significant reductions in stress-induced facial hypersensitivity during both the post-stress and post-SNP injection phases (Figure 5(d)). Grimace scores were significantly higher in vehicle-treated stressed female mice compared to controls post-stress. However, ACTH with metyrapone-treated females showed a reduction in scores (Figure 5(c)). Similarly, vehicle-treated stressed male mice had higher grimace scores than controls, while ACTH with metyrapone-treated stressed male mice significantly reduced scores post-stress (Figure 5(e)). The results suggest that ACTH treatment alleviated stress-induced hypersensitivity in both male and female mice, even when CORT synthesis was blocked with metyrapone, highlighting the independence of mechanisms in HPA-axis modulation of ACTH effects in the hypersensitivity response to stress. We also compared the stress treated with ACTH group to the ACTH with metyrapone-treated stressed group in both female and male mice, as shown in Online Supplementary Figure 3. In females, no significant differences were observed between groups across time points. In males, while a significant difference was noted on Day 1 post-stress, the overall effects on alleviating stress-induced hypersensitivity were similar between groups.

Administration of metyrapone prior to post-stress ACTH injection alleviated stress-induced facial hypersensitivity and grimace score in female and male mice. (a) The experimental timeline is shown in the scheme. Mice were exposed to restraint stress for three consecutive days, then injected with either 50 mg/kg metyrapone + 2 mg/kg ACTH (MET/ACTH) or vehicle + vehicle (Veh/Veh) 1 h after stress on the last day and again 24 h later. Mice were divided into four groups: control with Veh/Veh (n = 7 females, n = 8 males), control with MET/ACTH (n = 7 females, n = 8 males), stress with Veh/Veh (n = 9 females, n = 9 males) and stress with MET/ACTH (n = 9 females, n = 9 males.) On Day 14, after returning to baseline, all mice received 0.1 mg/kg SNP. (b and d) Facial withdrawal thresholds to periorbital stimuli applied to the face and (c and e) the grimace scores were measured in female and male mice during the acute phase (post-stress) and priming phase (post-SNP). A two-way ANOVA followed by Bonferroni multiple comparison analysis was used to compare the groups. * indicated significant differences between stressed with Veh/Veh and control with Veh/Veh groups. # indicated significant differences between stressed with Veh/Veh and stressed with MET/ACTH groups. Data are represented as mean ± SEM. #P < 0.05, **, ##P < 0.01, ***, ###P < 0.001, ****, ####P < 0.0001. Abbreviations: MET: metyrapone, Veh: vehicle, ACTH: adrenocorticotropic hormone. See Supplementary Table 1 for a complete statistical analysis.
Post-stress administration of α-MSH relieved stress-induced facial mechanical hypersensitivity in mice
We further investigated whether α-MSH, a cleavage product of ACTH, contributes to stress-induced migraine-like behaviors. After three days of restraint stress, mice received α-MSH injections 1 h after the final stress session and again 24 h later, following pain sensitivity assessment (Figure 6(a)). In female mice, the von Frey test data showed no difference between control mice treated with a vehicle and those treated with α-MSH. Stressed mice treated with vehicle showed significant hypersensitivity during the post-stress and post-SNP injection phases, while α-MSH treatment significantly reduced hypersensitivity during both phases (Figure 6(b)). Similar results were observed in male mice. Stressed mice treated with α-MSH significantly reduced the stress-induced hypersensitivity during both the post-stress and the post-SNP injection phases (Figure 6(d)). Mouse grimace scores showed no differences between control groups treated with a vehicle or α-MSH, with no spontaneous pain behaviors observed. Stressed female and male mice treated with a vehicle had higher grimace scores than controls. While α-MSH treatment reduced grimace scores in both sexes compared to their vehicle-treated stressed groups, the difference was not significant (Figure 6(c) and (e)). These findings suggest that the effect of ACTH on stress-induced hypersensitivity may be due to its breakdown product, α-MSH.

Post-stress α-MSH injection attenuated restraint stress-induced facial hypersensitivity in female and male mice. (a) The experimental timeline is shown in the scheme. Mice were exposed to restraint stress for three consecutive days, then injected with either 2 mg/kg α-MSH or vehicle 1 h after stress on the last day and again 24 h later. Mice were divided into four groups: control with vehicle (n = 8 females, n = 9 males), control with α-MSH (n = 8 females, n = 9 males), stress with vehicle (n = 11 females, n = 10 males) and stress with α-MSH (n = 10 females, n = 10 males.) On Day 14, after returning to baseline, all mice received 0.1 mg/kg SNP. (b and d) Facial withdrawal thresholds to periorbital stimuli applied to the face and (c and e) the grimace scores were measured in female and male mice during the acute phase (post-stress) and priming phase (post-SNP). A two-way ANOVA followed by Bonferroni multiple comparison analysis was used to compare the groups. * Indicated significant differences between stressed with vehicle and control with vehicle groups. # indicated significant differences between stressed with vehicle and stressed with α-MSH groups. Data are represented as mean ± SEM. *, #P < 0.05, **, ##P < 0.01, ***, ###P < 0.001, ****, ####P < 0.0001. Abbreviations: α-MSH: alpha-melanocyte stimulating hormone. See Supplementary Table 1 for a complete statistical analysis.
Post-stress THIQ administration alleviated stress-induced facial mechanical hypersensitivity in mice
Figures 3 and 6 show that ACTH or α-MSH administration post-stress alleviated stress-induced hypersensitivity, suggesting that ACTH may exert its effects through α-MSH. To explore the mechanism of α-MSH, we examined the role of the α-MSH receptor MC4R using the agonist THIQ. Mice received two THIQ injections, at 1 h and 24 h after the final stress session, following the same timeline as the ACTH and α-MSH experiments (Figure 7(a)). Stressed female mice treated with vehicle showed a significant hypersensitivity, consistent with the ACTH or α-MSH studies. However, stressed mice treated with THIQ reduced facial hypersensitivity during the post-stress phase and the post-SNP injection phase (Figure 7(b)). In stressed male mice, THIQ-treated mice significantly reduced facial hypersensitivity compared to vehicle-treated mice during the post-stress phase and post-SNP injection phase (Figure 7(d)). Stressed female mice showed increased grimace scores post-stress, but THIQ treatment significantly reduced scores compared to vehicle (Figure 7(c)). Similarly, stressed male mice treated with THIQ significantly reduced grimace scores compared to those treated with vehicle (Figure 7(e)). These results suggest that direct activation of MC4R can attenuate post-stress behavioral responses, highlighting a receptor through which α-MSH or ACTH produces its effects.
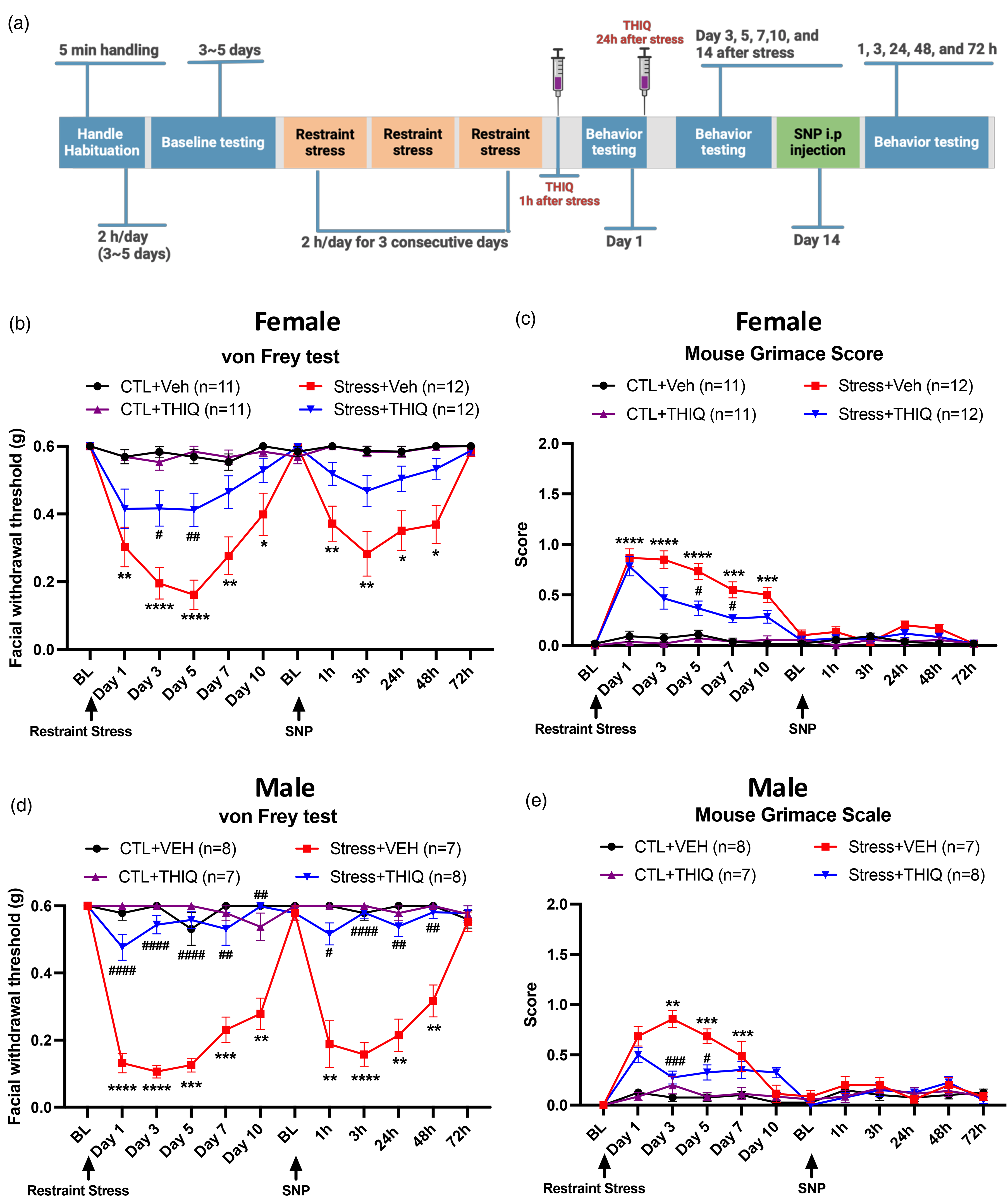
Post-stress THIQ injection reversed restraint stress-induced facial hypersensitivity and grimace score in female and male mice. (a) The experimental timeline is shown in the scheme. Mice were exposed to restraint stress for three consecutive days, then injected with either 2 mg/kg THIQ or vehicle 1 h after stress on the last day and again 24 h later. Mice were divided into four groups: control with vehicle (n = 11 females, n = 8 males), control with THIQ (n = 11 females, n = 7 males), stress with vehicle (n = 12 females, n = 7 males) and stress with THIQ (n = 12 females, n = 8 males.) On Day 14, after returning to baseline, all mice received 0.1 mg/kg SNP. (b and d) Facial withdrawal thresholds to periorbital stimuli applied to the face and (c and e) the grimace scores were measured in female and male mice during the acute phase (post-stress) and priming phase (post-SNP). A two-way ANOVA followed by Bonferroni multiple comparison analysis was used to compare the groups. * indicated significant differences between stressed with vehicle and control with vehicle groups. # indicated significant differences between stressed with vehicle and stressed with THIQ groups. Data are represented as mean ± SEM. *, #P < 0.05, **, ##P < 0.01, ***, ###P < 0.001, ****, ####P < 0.0001. Abbreviations: THIQ: tetrahydroisoquinoline, melanocortin 4 receptor agonist. See Supplementary Table 1 for a complete statistical analysis.
Discussion
The HPA axis likely contributes to migraine attacks triggered by stress. During stress, CRH stimulates the release of ACTH from the pituitary gland, which in turn promotes glucocorticoid secretion (44,45). These glucocorticoids provide negative feedback on both CRH and ACTH, highlighting the pivotal role of CRH in regulating stress responses and its involvement in stress-induced migraine (46). Previous findings show the involvement of the hypothalamus in headache pathogenesis (12–15), with abnormal hormone patterns like cortisol and nocturnal prolactin in chronic migraine (18). Elevated CRH levels along with neuroendocrine disruptions are seen in the cerebrospinal fluid of chronic migraine and medication overuse headache patients (47), further implicating CRH in migraine (48). Taken together, this evidence highlights the critical role of the HPA axis and its neuroendocrine factors in migraine mechanisms.
We previously demonstrated that CORT is essential for development of stress-induced behavioral response in mice, as blocking or inhibiting CORT signaling can prevent migraine-like behaviors (11). In this study, post-stress metyrapone reduced hypersensitivity in females and males (Figure 8). Moreover, we found that a low dose of CORT administered after stress alleviated migraine-like behaviors, highlighting the importance of timing and dosing in influencing stress responses (Figure 8). Consistent with our findings, previous studies have demonstrated a biphasic cortisol response: higher CORT levels suppress LPS-induced IL-1β, while lower levels promote IL-1β expression, explaining the dual pro- and anti-inflammatory role of CORT (49–51). Another possible explanation for the effects observed here is that administering a low dose of CORT post-stress may stabilize CORT fluctuations following stress-induced elevations, thereby alleviating hypersensitivity. Stress-induced migraine attacks tend to develop after the termination of stress (52) and separately, corticosteroid therapy is tapered rather than abruptly stopped to avoid secondary adrenal insufficiency or recurrence of underlying conditions. Accordingly, the precipitous decline in CORT levels after three consecutive days of stress could have maladaptive consequences. However, why administration of metyrapone post-stress is able to reduce facial hypersensitivity is not clear given this should further reduce CORT levels. It is likely that the exact level of CORT in the post-stress phase needs to be tightly regulated in order to prevent any maladaptive consequences.
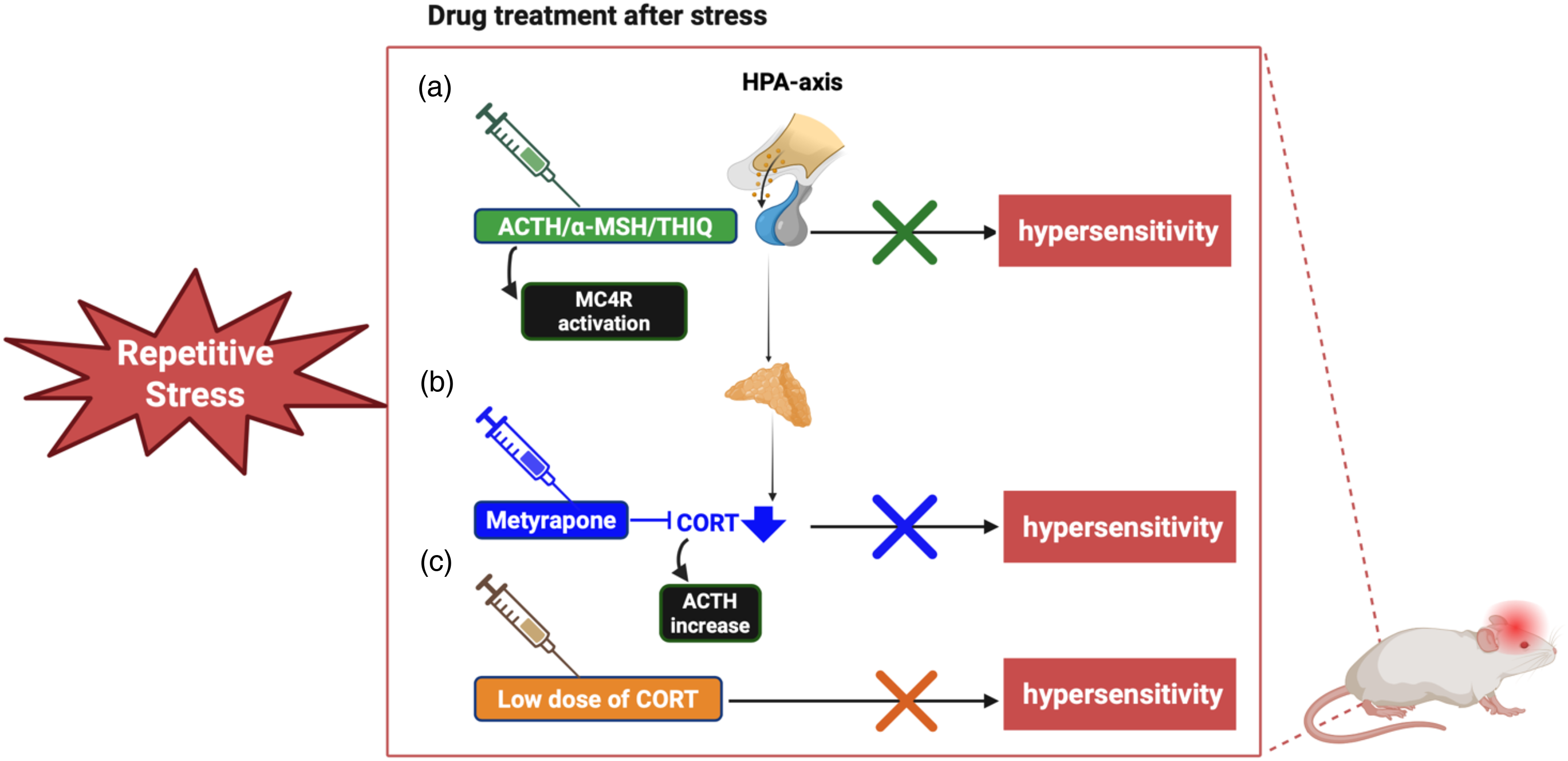
Modulation of the post-stress HPA axis or melanocortin system helps alleviate stress-induced hypersensitivity in mice. The schematic illustrates how repeated stress activates the HPA axis, leading to CRH release from the hypothalamus, which stimulates ACTH secretion from the pituitary and subsequently increases CORT release from the adrenal gland. These hormonal fluctuations can lead to maladaptive outcomes, such as migraine attacks. This study demonstrates that modulating hormone levels after stress can alleviate stress-induced hypersensitivity in mice. (a) First, post-stress administration of ACTH, α-MSH, or the MC4R agonist THIQ effectively reduced hypersensitivity, highlighting the involvement of the melanocortin system in stress-related behavioral responses. (b) In addition, post-stress treatment with metyrapone, which inhibits CORT synthesis and causes a feedback increase in ACTH, also alleviated hypersensitivity, suggesting that both ACTH and CORT play a role in mediating this response. (c) Finally, a low dose of CORT administered after stress exposure was sufficient to alleviate hypersensitivity, further supporting the modulatory influence of glucocorticoids in this process. This study suggested that targeting the HPA or melanocortin system with existing FDA-approved drugs may offer a fast-track approach to developing effective post-stress migraine treatments.
We demonstrated that post-stress treatment with ACTH, α-MSH or THIQ effectively reduced stress-induced hypersensitivity in both sexes, indicating a crucial role of MC4R in the connection between stress and migraine (Figure 8). It has been reported that MC4R plays a significant role in regulating pain processing across various experimental models of painful conditions (33,34,53). It is widely expressed throughout the nervous system, including regions associated with the autonomic nervous system (ANS) (54). Moreover, the receptor presence in rat trigeminal ganglia (TG) was confirmed by detecting MC4R transcripts and protein expression through Western blotting (55,56). These findings suggest that MC4R and its ligand α-MSH may play a pivotal role in linking stress to migraine pathophysiology.
Stress may contribute to migraine by promoting neurogenic inflammation in the dura mater, a process closely linked to migraine pathology (57,58). α-MSH has been shown to be a potent inhibitor of systemic inflammation by suppressing proinflammatory cytokines and non-cytokine regulators such as NO (59). The MC4R is known to mediate central anti-inflammatory effects of α-MSH, with studies suggesting that MC4R activation of anti-apoptotic pathways may partially explain the neuroprotective properties of α-MSH in a mouse model of traumatic brain injury (60). Beyond its interaction with MC4R, α-MSH also binds to other melanocortin receptors, including MC1R, MC3R, and MC5R, which are also implicated in anti-inflammatory effects. MC1R, expressed in endothelial cells, monocytes, macrophages, neutrophils, and mast cells, has been shown to attenuate IL-8 and TNF-α-mediated inflammatory responses (61). Moreover, α-MSH reduces cerebral inflammation via MC1R by lowering Iba-1 positive cell activation in the brain (60,62). MC3R is found in lymphocytes and macrophages, where it mediates strong anti-inflammatory effects of MC3R agonists (63–65). Additionally, MC5R has been implicated in the immunomodulatory functions of B and T lymphocytes and mast cells, further highlighting the diverse anti-inflammatory roles of α-MSH (66,67). While our data with THIQ support a role for MC4R, they do not fully explain the effects of α-MSH given this peptide can act through multiple receptors.
ACTH treatment combined with metyrapone treatment was effective at reversing hypersensitivity in males and in females, suggesting ACTH acts independently of CORT in both sexes. These findings highlight the complex roles of HPA axis signaling in stress-induced migraine-like behaviors. Additionally, serum CORT levels revealed that control females did not exhibit increased CORT following ACTH injection, while control males showed a significant rise at 30 min post-injection, indicating greater ACTH responsiveness in males under normal conditions. However, in females, ACTH only leads to elevated CORT in animals exposed to repeated stress while in males, ACTH elevated CORT regardless of whether animals were exposed to stress. Studies have shown that acute HPA activity following stress is notably higher in females than in males, mainly due to modulation by the gonadal hormones estradiol and testosterone (68). The prevalence of migraine is higher in females, beginning in puberty (69). Understanding the cause of these sex differences in HPA axis responses to stress may be important for uncovering the factors that contribute to migraine.
Several limitations should be considered when interpreting the results of these studies. The grimace scale is commonly used in preclinical studies to assess the emotional aspect of pain (37,70). Our findings suggest that repeated stress induces a post-stress grimace response, indicating a negative affective component. While grimace and cutaneous hypersensitivity suggest pain, the exact state of the mice remains uncertain, possibly reflecting stress-induced unpleasantness. Consistent with our previous study, no grimace response was observed after SNP injection (35), despite the presence of mechanical hypersensitivity after SNP. Moreover, drug treatment did not consistently reduce elevated grimace scores caused by stress, unlike von Frey results. Other studies have shown that inhibiting amygdala astrocyte activity increased von Frey thresholds but did not affect grimace scores in neuropathic pain (71), suggesting there may be a disconnect in the underlying behaviors that cause mechanical hypersensitivity and grimace responses. Stress-induced hypersensitivity likely involves both sensory and emotional components, which the grimace scale can capture, whereas SNP-induced pain may be driven more by direct nociceptive input without significant affective involvement. Additionally, rodents may naturally suppress grimace responses over time as an adaptive strategy to avoid predation (72). If this suppression extends to stress conditions, repeated noxious challenges could dampen grimace responses, complicating the interpretation of post-stress grimace measures. While our study focused primarily on the effects of HPA signaling and melanocortin systems in both females and males, we did not specifically investigate potential sex differences in the pharmacokinetic profiles of the applied drugs. Pharmacokinetic and pharmacodynamic variations between sexes could potentially influence treatment efficacy and may necessitate sex-specific dosing regimens. Future research should explore these factors to better understand the impact of sex on drug efficacy and dosing in stress-induced migraine models.
The link between stress and migraine is incredibly complex, driven not only by the HPA axis but also by the ANS (73–75), as shown by our previous findings that dural norepinephrine (NE) injection induced allodynia in rats, and NE-stimulated fibroblast media enhanced afferent firing, ERK phosphorylation, and IL-6 release in dural fibroblasts (76). Fully understanding the complexity of stress-induced behavioral hypersensitivity and its relevance to human migraine is beyond the scope of a single study. However, our findings offer valuable insights, showing that increasing ACTH or α-MSH levels, or activating MC4R, can reverse stress-induced migraine-like behavior. This response was also observed with metyrapone or CORT despite no direct involvement of MC4R in CORT signaling. These findings suggest that the activation of one pathway does not exclude the involvement of another, highlighting the potential for modulating both the HPA axis and melanocortin system as a therapeutic strategy for stress-induced migraine attacks. While further research is needed to explore how this might be achieved, it is worth noting that several drugs targeting these systems are already approved for use in humans. These include medications that influence CORT synthesis, GR agonists and antagonists, ACTH therapy, and the MC4R agonist Setmelanotide. Given that these drugs are already available for human use, they could be tested in clinical settings more rapidly than novel chemical entities awaiting FDA approval, offering a promising avenue for developing new treatments for migraine.
Administering metyrapone after stress alleviated stress-induced facial mechanical hypersensitivity in both male and female. A low dose of CORT administration after stress reduced stress-induced facial mechanical hypersensitivity in mice. Post-stress ACTH administration alleviated stress-induced facial mechanical hypersensitivity in male and female mice. Metyrapone attenuated the ability of post-stress ACTH to reduce stress-induced facial hypersensitivity in both female and male mice. Post-stress administration of α-MSH or THIQ alleviated stress-induced facial mechanical hypersensitivity in male and female mice.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-docx-1-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-docx-2-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-docx-3-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-pdf-4-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-pdf-5-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-pdf-5-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-pdf-6-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-pdf-6-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-pdf-7-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-pdf-7-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-pdf-8-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-pdf-8-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-pdf-9-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-pdf-9-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Supplemental Material
sj-pdf-10-cep-10.1177_03331024251352856 - Supplemental material for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice
Supplemental material, sj-pdf-10-cep-10.1177_03331024251352856 for Post-stress modulation of the HPA and melanocortin systems alleviates migraine-like behaviors in mice by Ya-Yu Hu, Hao-Ruei Mei, Shruti Sankar, Abbas Pirwani, Armen Akopian, Christa McIntyre, and Gregory Dussor in Cephalalgia
Footnotes
Acknowledgments
We thank Aiswarya Saravanan for her help with collecting mouse blood samples.
Data availability statement
The data generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
All procedures were approved by the Institutional Animal Care and Use Committee and followed ARRIVE guidelines, the policies of the International Association for the Study of Pain, and the National Institutes of Health guidelines for animal research.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were funded by the National Institutes of Health NS104110, MH126516, and the BBS Internal Research Grant from the University of Texas at Dallas.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
