Abstract
Background
There is an increasing trend in the use of the internet and smartphone applications to deliver headache interventions. However, their efficacies remain unclear. Moreover, developing such interventions can be cumbersome and increasingly challenging when navigating the regulatory landscape of medical devices. The present study aimed to review the literature on internet- and app-based headache interventions and discuss opportunities and challenges in the process of developing such interventions.
Methods
This narrative review was based on a comprehensive literature search of PubMed. Publications investigating internet- and app-delivered interventions for headache disorders were identified, systematically assessed and summarised.
Results
Internet- and app-delivered interventions were mainly applied to deliver behavioural change techniques and relaxation training, coordinate management plans with patients, and promote medication adherence. Eleven randomised controlled trials, including two large-scale studies, did not demonstrate clear superiority of internet-delivered behavioural therapy over other therapies or waitlist control. Internet-delivered relaxation training trials might be more promising; however, they are old and less robust. Most studies investigating app-delivered behavioural therapy and relaxation training are pilot and feasibility trials, making it difficult to conclude their efficacy. Using the Internet and apps to manage care plans and promote medication adherence seems feasible, resulting in positive impacts on individual disease burden, health care and societal costs. However, these applications require further investigation.
Conclusions
We discussed important aspects of the conceptualisation, design and development of internet- and app-delivered interventions for headaches and provided specific recommendations to develop these interventions. Thereafter, we described and discussed the regulatory pathways for software interventions, which, in most cases, are considered medical devices and are subject to stringent regulations. Adhering to these regulations and creating a usable intervention with the evidence of efficacy established through robust clinical trials is time-consuming and requires medical, technological and regulatory knowledge.
This is a visual representation of the abstract.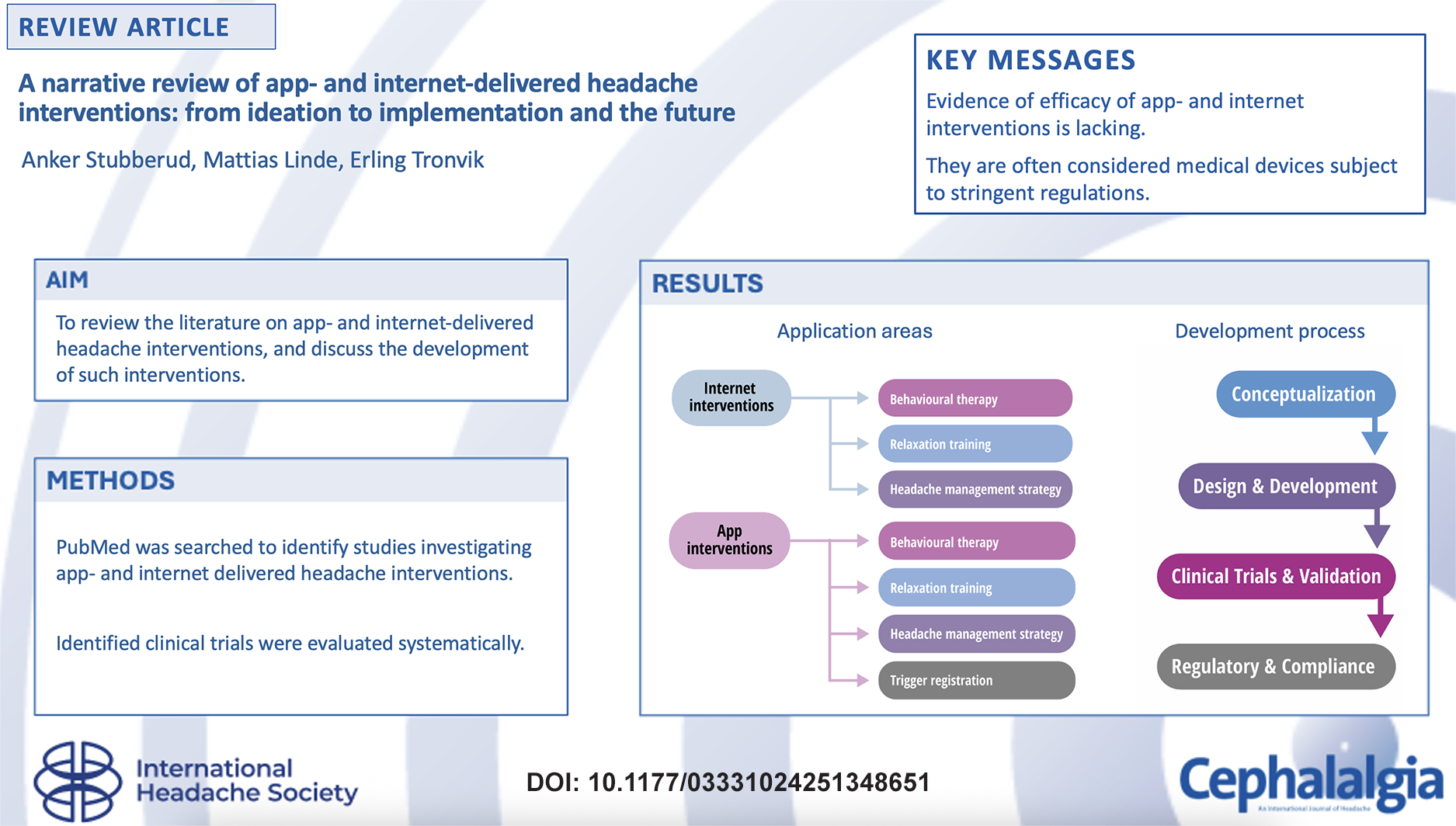
Keywords
Introduction
Headache management is multifaceted, involving pharmacotherapy, behavioural interventions, effective communication, care plans and follow-up with a healthcare provider (1). However, barriers, such as under-prioritisation in government policies, travel and cost, often lead to underutilisation of many treatments, limiting access to care and follow-up. Behavioural interventions (such as biofeedback, relaxation and cognitive behavioural therapy) have not been widely implemented as a result of their time-consuming nature, requirement of trained therapists and costs (2). This has naturally spurred an interest in delivering headache care through the internet and via app platforms (3,4), which seem to facilitate access and delivery of interventions (5,6). eHealth is a common term used to refer to the use of digital services over the internet to provide health care (3). In its broadest sense, this includes electronic health records, electronic prescriptions, and telehealth with web-based patient–doctor communication. eHealth is also used to describe the delivery of therapies, often psychological or behavioural, through the internet (5). By contrast, mHealth, which could be considered a sub-entity of eHealth, refers to the use of personal mobile or wearable electronic devices in healthcare delivery. mHealth encompasses smartphone apps used to track, monitor and communicate about health, apps used to remotely deliver psychological and behavioural therapies, and the use of wearable tracking and monitoring health and support device-dependent health interventions such as biofeedback (6).
Recently, headache-oriented internet programs and apps have increased rapidly; however, evaluation of the effectiveness of these interventions is limited (7,8). Moreover, developing app and internet interventions requires considerable effort and can be time-consuming, particularly when thorough feasibility and usability testing is implemented (9). Furthermore, the process of establishing such interventions as medical devices (comprising the hallmark defining them as evidence-based clinical tools and separating them from the plethora of commercial and non-evidence-based interventions) is usually challenging and a costly and time-consuming process (5,9). These drawbacks often lead to limited availability of high-quality validated eHealth and mHealth interventions or substandard interventions (5).
This narrative review aimed to (i) review the medical literature on the use of internet- and app-delivered interventions for headaches; (ii) discuss opportunities and challenges in the process of developing internet and app-delivered interventions for headaches, with an emphasis on regulatory pathways; and (iii) discuss the future prospects.
Methods
This narrative review aimed to summarise the literature on app- and internet-delivered headache interventions and discuss the development, implementation and the future of such interventions. This literature review was based on a search of PubMed from its inception to 5 January 2025 using the following queries: ([headache] OR [migraine] OR [tension-type headache] OR [trigeminal autonomic cephalalgia] AND [eHealth OR mHealth OR app OR internet]). Original articles published in the English language investigating app- and internet-delivered interventions for primary and secondary headache disorders according to the International Headache Society criteria were included (10,11). Studies that used internet- or app-delivered interventions were considered eligible, including controlled trials, open-label single-arm trials and pilot studies, as well as studies exploring feasibility, usability and acceptability aspects of app- and internet-delivered headache interventions; however, studies primarily focused on wearable device approaches, such as non-invasive neurostimulation and biofeedback devices, were excluded.
The titles and abstracts of records identified through the literature search were screened by two of the investigators (AS and ML). The full texts of the potentially eligible records were thereafter reviewed in detail. The characteristics of the clinical trials investigating internet- and app-delivered interventions were extracted to standardised data collection forms and summarised. In addition to the database search, the reference lists of relevant reviews on the topic were hand-searched.
We opted to differentiate between internet- and app-delivered interventions because of their potentially different modes of delivery. Consequently, interventions primarily delivered through web-based programs and e-mail services, typically requiring a personal computer and location-bound internet access, were labelled as internet-delivered, whereas interventions using smartphone apps and mobile networks, thus allowing complete mobility of the user, were considered app-delivered.
Results and discussion
In total, 961 studies were identified in the literature search. After screening, 88 records were reviewed in detail, of which 42 were eligible. Hand searching of relevant reviews on the topic led to identifying three additional studies (12–14). Of the 45 studies included in this review, 18 and 12 were clinical trials investigating internet- and app-delivered headache interventions, respectively (15–42). The remaining 15 studies did not describe clinical trials of interventions but explored other relevant and interesting aspects of internet- and app-delivered interventions. The characteristics of the 18 trials investigating internet-delivered interventions, and the 11 trials investigating app-delivered interventions are summarised in Tables 1 and 2.
Overview of internet-delivered interventions.

CBT = cognitive behavioral therapy; HADS-D = Hospital Anxiety and Depression Scale; HIT = Headache Impact Test; ITT = intention-to-treat; MBT = mentalisation-based treatment; MSQ = migraine-specific quality of life questionnaire; QoL = quality of life; RCT = randomised control trials; TTH = tension-type headache.
Overview of app-delivered interventions.

CI = confidence interval; HRV = heart rate variability; MIDAS = migraine disability assessment test; PHQ-ADS = patient health questionnaire anxiety and depression scale; RCT = randomised control trial.
Internet-delivered headache interventions
Previous attempts at pooling scientific evidence regarding the use of internet-delivered headache interventions have been inconclusive (43–45). A 2014 Cochrane review of internet-delivered psychological interventions for chronic pain management, including headache, found insufficient evidence to support the efficacy of such therapies (43). A similar Cochrane review of children and adolescents reached the same conclusion (44). Moreover, a 2016 systematic review identified a wide range of internet-delivered therapies; however, none had an established efficacy (45). In our literature search, we found different combinations of behavioural therapy and relaxation to be the most studied internet-delivered interventions.
Internet-delivered behavioural therapies
We identified 11 trials involving different types of internet-delivered behavioural therapies. Seven small-scale trials failed to demonstrate a clear effect. In a very small study (13), children and adolescents with primary headaches were randomised to receive internet-delivered behavioural therapy or internet-based psychoeducation. No significant differences were found in headache frequency, duration or intensity. In a subsequent trial by the same research group, adolescents were randomised to either receive behavioural therapy, applied relaxation or educational intervention via the internet (30). In the study, 63% of those randomised to behavioural therapy were responders (defined as ≥50% reduction in headache frequency) at post-treatment; however, between-group differences were not observed at follow-up. Another small study of recurrent headaches among adolescents compared specialised treatment with or without family-based behavioural therapy (22); however, no significant differences in the primary outcome of change in headache days were observed. In a small Swedish study (23), adults with episodic migraine were randomised to receive treatment in primary care with or without internet-delivered behavioural therapy. No significant differences in the primary outcome of migraine days were found. In a small Iranian study of 30 women with chronic headaches, internet-delivered mindfulness and bibliotherapy were compared with the usual treatment (29). The experimental treatment improved the quality of life and mental distress; however, no headache-related outcomes were reported. Finally, the results of two studies on the additional effects of therapist-initiated telephone calls (15) and hand massage (19) were negative.
Kleiboer et al. (21) and Sorbi et al. (46,47) developed an internet-based program to support self-management for preventing headache attacks. The program was evaluated in a large randomised controlled trial (RCT), reporting its short-, medium- and long-term effects (14,25,26). In the trial, 368 adults with episodic migraine were randomly assigned to the internet-based program or waitlist control. The program aimed to counteract attacks in the prodromal phase by detecting prodromal features and self-management through relaxation and cognitive-behavioural regulation. The training included eight online sessions and was supported by minimal guidance via e-mail from a trained coach. The primary outcomes measured were migraine frequency and self-efficacy. At the short-term efficacy assessment, three months post-randomisation, both groups showed a 20–25% decrease in migraine attack frequency with a non-significant between-group difference. Those in the intervention group reported greater self-efficacy and internal locus of control (14). At the medium-term follow-up, six months post-randomisation, the intervention group showed a 23% reduction in attack frequency, while the control group showed a 19% reduction, again with no significant between-group differences (25). By contrast, the intervention group showed a significantly lower triptan use, with a 16% reduction. In the long-term follow-up study (26), those initially randomised to waitlist control subsequently received the internet-based program, and outcomes were assessed up to 16 months post-randomisation. It was concluded that the intervention was beneficial for patients with frequent episodic migraines with within-group reductions in migraine attack frequency. However, these long-term findings should be interpreted with caution in the absence of controlled conditions.
In another large study (20), 428 adolescents and adults (aged 14–40 years) with migraine were randomised to receive internet-delivered behavioural therapy focused on psychoeducation, self-monitoring and skills training, headache triggers, or waitlist control. The primary outcome was a ≥50% reduction in monthly headache days at four months post-randomisation. The proportion of responders was similar between the groups, with 17% in the intervention groups and 11.6% in the waitlist control group. Analysis of completers showed the behavioural therapy to be superior, suggesting that low adherence could explain the lack of effect.
By contrast, one RCT, including 189 adults with migraine, compared internet-based cognitive behavioural therapy and self-management with no treatment (16). Participants in the intervention group reported significantly superior improvements in self-efficacy, positive symptom management strategies, and migraine-related depression and stress. However, the absence of data regarding important outcomes, such as headache frequency and intensity, limits the conclusions that may be drawn.
In summary, most RCTs provided no evidence to support the effectiveness of internet-delivered behavioural therapy. Some trials have indicated a positive effect; however, their designs hindered firm conclusions. Further studies based on the growing literature on usability testing are required to improve program functionality (48,49).
Internet-delivered relaxation training
We identified two RCTs that investigated internet-delivered relaxation training. In a pioneering study from Sweden (27), published 25 years ago, approximately 100 adults with unspecified headaches were randomised to receive relaxation and problem-solving interventions through the Internet or waitlist control. In the treatment group, 50% of the participants had a ≥50% reduction in the headache index compared to 4% of the controls. Five years later, a moderate-sized study randomised 139 adults with chronic migraine or tension-type headaches to internet-delivered progressive relaxation, limited biofeedback with autogenic training, stress management, or symptom-monitoring waitlist control (17). Significantly more participants in the internet group (38.5%) had a ≥50% reduction in the headache index compared to the waitlist control group (6.4%). Furthermore, a 35% within-group reduction was observed in medication use among the treated participants. Trials investigating relaxation have demonstrated positive outcomes. However, these findings should be interpreted with caution because of the large dropout rates and analysis of completers only.
Internet-delivered headache management strategies
In addition to behavioural training and relaxation, the internet has been used for headache management strategies. In a non-RCT published in 2017, 663 individuals with medication overuse headache (MOH) were assigned to an all-in-one web-based diary with an alerting and communication system or a paper diary (28). The primary outcome was the number of participants who remained medication overuse-free without relapse during the six-month observation period. In the intention-to-treat analysis, a significantly higher percentage of overuse-free participants was observed in the intervention group (73.1%) than in the control group (64.1%). Moreover, a recently published single-arm, open-label study investigated the effect of an internet-based care plan in 47 adults with chronic migraine and MOH (18). The plan included home-based medication withdrawal, education on the correct drug use and lifestyle issues, prescription of tailored pharmacological prophylaxis, and attendance to six online mindfulness-based sessions. Significant improvements were observed in headache frequency, medication intake, headache impact, and quality of life, with a moderate improvement in pain catastrophising and mild improvement in depression symptoms. Interpreting these results requires caution because no comparative control group was used, and the sample was drawn from a single centre with no control over the prescribed prophylaxis. Finally, in a 2006 RCT (24), adult participants were randomised to either use a web-based computer program before doctor visits to prompt patients to ask questions that led to higher quality of care or to not use it before doctor visits. It was concluded that the intervention may have a positive impact on migraine-specific doctor-patient communication.
App-delivered headache interventions
Regarding internet-delivered interventions, pooled evidence supporting the use of app-delivered interventions is limited (4,8). A 2022 study sought to evaluate the content and quality of commercially available headache management apps (7). Fifty-five apps were identified across the Apple App and Google Play Stores. The study evaluated the characteristics of behavioural change techniques and app quality using the Mobile App Rating Scale (MARS) (50). Almost all apps used behavioural change techniques directed at headache management with demonstrated efficacy, and almost half of all apps used at least five of such techniques. The most commonly used techniques were psychoeducation about the relationship between the identified behaviour and the user's health, psychoeducation informing the user of the likely outcome of engaging and not engaging in the target health behaviour, and information on how to perform the target health behaviour, encouragement, feedback and self-monitoring. The overall quality of the apps, as judged by the MARS, ranged from 2.84 to 4.67 of 5.00, indicating poor to excellent usability. Despite the application of evidence-based therapies, there is a lack of RCTs to determine their efficacy. Another systematic evaluation of commercially available apps for pain management reached the same conclusion (51). Apps generally included evidence-based pain management-specific behavioural change techniques, and the MARS scores ranged from 2.27 to 4.54. However, none of these applications have been evaluated for efficacy in RCTs.
Among the interventional studies identified in the systematic literature search, the same main categories of interventions delivered through the Internet and apps, including behavioural therapy, relaxation, and headache management strategies, as well as apps used for trigger registration.
App-delivered behavioural therapies
Five recent studies investigated the effects of different combinations of app-delivered behavioural training. In 2020, a single-arm open-label pilot study investigated the feasibility and effect of a digital therapeutic self-management tool, including 16 intervention modules for adolescents (n = 40, aged 11–18 years) with migraine (33). The app was considered feasible, and a reduction in monthly headache days from 17.2 to 7.9 was observed after eight weeks. Another small pilot trial investigated the feasibility of an app combining relaxation, biofeedback, education, cognitive-behavioural pain management, sleep and trigger identification among 18 adolescents with migraine (37). During the four-week treatment phase, the app was used for 11 days and eight biofeedback sessions were completed. A 2024 RCT investigated the effect of a therapeutic education program delivered via the Internet or an app compared with general health recommendations in adults with chronic migraine receiving botulinum toxin therapy (31). The education program comprised 15 videos on migraine education and self-management. A significant reduction in headache frequency of 2.9 days was observed in the intervention group compared to the control group. Furthermore, an unblinded RCT of 198 individuals with frequent headaches, including migraine, tension-type headaches and undiagnosed headaches (40). Participants were randomised to receive a smartphone-based coaching intervention designed to improve mental health well-being using various behavioural change techniques or a waitlist control group. The intervention group showed a clinically significant reduction in the Patient Health Questionnaire Anxiety and Depression Scale of 3.9 points, which corresponded to a medium-to-large between-group effect size (Cohen's d = −0.66). Finally, a small pilot study of 12 adolescents with migraine found a mindfulness-based behavioural approach to be feasible, and lead to reductions in headache frequency (32). In summary, few studies have investigated app-delivered behavioural therapies, many of which are small pilot trials; however, larger and more robust studies suggest that app-delivered behavioural therapies may be useful in headache management (31,40).
App-delivered relaxation training
A series of studies was conducted by Minen et al. investigating the mHealth app “RELAXaHEAD” as a progressive muscle relaxation (PMR) preventive treatment for migraine and post-traumatic headaches. The app was developed in a ‘think aloud’ approach of iterative beta testing in people with migraine (52). It includes a headache diary and two PMR sessions of five- and 15-minute durations delivered through an audio file embedded in the app. These studies were designed as feasibility and acceptability studies. In all studies, the application was evaluated as feasible and acceptable. The number of days that the relaxation module was played during 90-day assessment periods ranged from 13 to 22 days, the former being observed in an emergency setting (35) and the latter being observed in an outpatient clinic setting (34). Preliminary efficacy evaluation of the app for migraine demonstrated reductions in Migraine Disability Assessment (MIDAS) scores between 23 and 38 points (35,36), as well as a reduction of four monthly headache days for users with high adherence (34). In the study evaluating RELAXaHEAD for post-traumatic headache, the app was considered feasible and usable (41). However, time constraints, forgetfulness, application glitches and repetitiveness were cited as obstacles to practising PMR.
App-delivered headache management strategies
Smartphone applications have also been used to deliver comprehensive healthcare plans and promote medication adherence. In a 2020 study, 141 employees of a corporate company with migraine or probable migraine received six sessions of individualised telecoaching from a specialised nurse using an app (39). The coaching sessions were used to optimise migraine management through appropriate medical and lifestyle options. The program was delivered as a complementary service, as part of the company's well-being program. The primary outcome was the change in MIDAS scores, in which 54% and 64% reductions were observed after six and nine months, respectively. A cost analysis showed that the participants gained an average of 10.8 working days per year and a positive return on investment. Another pilot study assessed the feasibility, usability, and impact on care teams of an app-based interactive care plan integrated with electronic health records (42). The care plan resulted in fewer downstream face-to-face visits compared to a control group; however, no differences were observed in the rate of telephone contacts or electronic messages with healthcare providers. Finally, a 2018 single-arm pilot trial evaluated an adherence promotion app aimed at improving medication use among 35 adolescents and young adults with migraines (38). A small increase in medication adherence was observed in the population as a whole, and those with lower baseline adherence showed greater improvements.
App used for trigger registration
Another frequently investigated application of headache applications is the collection and analysis of trigger factors, with the subsequent aim of a behavioural change (12,53–56). Migraine triggers are a controversial topic, and a comprehensive review is beyond the scope of this study. An important aspect of app-based trigger registration is that it allows temporally stamped capture of triggers and apps, overcoming many of the typical obstacles in assessing the true causal relationship of triggers, particularly recalling bias and false attribution, both of which are often exaggerated by retrospective reporting (57,58). Notably, in a 2022 study that clearly illustrates this (55), a smartphone app was used to prospectively register triggers and attacks and found that among the average 28 triggers that participants moderately or highly endorsed, only 22 were statistically associated with an increased risk of attacks. This and other similar studies emphasise the awareness of the limitations of trigger mapping in migraine (59).
Development processes, from ideation to implementation
The publication years of the internet-delivered interventions versus app-delivered interventions, indicate a clear shift towards apps, aligning with the increased accessibility of smartphones and mobile networks. Thus, the discussion on development processes and regulatory pathways primarily focuses on app-delivered interventions.
Conceptualisation and design
The conceptualisation of mHealth and eHealth interventions should focus on the target audience, address appropriate health challenges and define the core purposes of the interventions (Figure 1) (5,8). Presently, clinical guidance on how eHealth and mHealth interventions best add value to patient care is limited (60). Therefore, understanding the goal of the intervention under development by researchers and developers is essential. In the initial stages, investigating the market to differentiate interventions from existing solutions and engaging relevant stakeholders are essential.

Flow chart of the eHealth and mHealth development process. FDA = Food and Drug Administration; MDR = Medical Device Regulation.
When designing and developing eHealth and mHealth interventions, user experience and interface (usability) should be prioritised (9). The app should be intuitive, easy to navigate, and visually appealing for the end user, particularly because adherence seems to be associated with more positive outcomes (61,62). A systematic review identified four important aspects influencing mHealth app adherence: app content personalisation to user individual needs; reminder functions; user-friendly and technically stable design; and personal support complementary to the digital interventions (63). Moreover, ensuring usability and accessibility is essential by considering different user-related factors, such as education, functional diversity and payment models (9). Usability can be formally assessed using different scales, where the mHealth App Usability Questionnaire appears to be among the more used (9). Finally, app quality should be ensured through different quality assurance tools such as MARS (49).
In addition to the above general guidance on eHealth and mHealth development, there are some important studies outlining aspects of usability, feasibility and user desires specific to headache interventions (64–66). In a 2020 Modified Delphi study, consumer reviews of 15 headache apps were systematically reviewed (64). The most important features of headache apps were found to be: (i) apps should allow users to track headache characteristics, potential triggers and treatments; (ii) apps must be sufficiently usable; (iii) personalisation and features to assess trends in data are key motivators; and (iv) ease of exporting and viewing of data is critical. Similarly, a retrospective qualitative analysis of the free-text notes inputted into the RELAXaHEAD app revealed four main themes important for users (66): (i) users wish to expand information beyond what can be inputted into specific icons in the app; (ii) monitoring symptoms from related non-headache conditions is essential; (iii) tracking behavioural therapy in addition to medications should be possible; and (iv) users wish to monitor and identify triggers, prevention patterns and relief patterns of their migraines through consistent data tracking in the app. Moreover, in a cross-sectional online survey (65), individuals with migraine were asked how likely they were to pursue different behavioural therapy delivery methods. The respondents were “somewhat likely” to pursue in-person and smartphone-based behavioural therapy, both of which were preferred over telephone-based therapy.
Clinical trials and validation
Following their design and development, eHealth and mHealth interventions must be validated through clinical trials (Figure 1). It is often advisable to start with small-scale trials assessing feasibility, usability and acceptability (67). Indeed, it is evident from this review that pilot trials constitute the bulk of mHealth trials. Nevertheless, one of the major limitations of eHealth, particularly mHealth, is the limited availability of high-quality clinical trials assessing their therapeutic efficacy (68,69). The lengthy time gaps from recruitment to publication, high-cost trial implementation, and rigid RCT protocols often hamper developers from taking these important steps (60). Moreover, early-stage developers and start-ups are often under pressure to demonstrate short-term product growth, which does not align with the traditional pipeline for clinical trials. Nevertheless, eHealth and mHealth should be evaluated thoroughly in well-designed RCTs to ensure their validity and scientific robustness and thereby pave the way for ‘prescribable
By contrast to these challenges, eHealth and mHealth have inherent opportunities in design and technology that could streamline clinical trials, such as quick, inexpensive online recruitment and enrolment through the internet and apps, continuous data collection, and the possibility to follow-up participants remotely. Collectively, these opportunities allow for centralisation and may reduce costs and the need for personnel at the study sites (70).
Notably, achieving a medical device approval (see below) is often dependent on clinical evidence stemming from one's own or others’ clinical trials. Therefore, researchers and developers should familiarise themselves with the intended regulatory pathways during the initial phases of development.
Regulatory pathway for internet and app interventions
eHealth and mHealth interventions are often subject to stringent regulations that ensure safety, efficacy, and data protection. Therefore, an important step in the developmental process is determining whether a device should be classified as a medical device. In the European Union (EU), this falls under the Medical Device Regulation (MDR) (EU 2017/745) (71) and, in the USA, the Food and Drug Administration (FDA) regulates medical apps under the Federal Food, Drug, and Cosmetic Act and its twenty-first Century Cures Act (2016) (72,73). The core definitions are as follows:
− MDR: a ‘Medical Device − FDA: a ‘Medical Device
Notably, neither of these definitions depends on whether the application is used commercially. However, general wellness software does not qualify for medical device status (71,74). Neither does software that provides general information on diseases, symptoms or treatments. Therefore, symptom trackers, such as headache diaries or migraine trigger logs that use the information to make diagnoses, predict outcomes or actively guide clinical management will in many cases be considered medical devices.
Determining the risk level of software medical devices
If the software is defined as a medical device, the next step is to determine the risk level. The MDR categorises devices into Class I (low risk), IIa (moderate risk), IIb (high risk) and III (highest risk) based on their intended use, invasiveness and patient impact. In the USA, the FDA uses a three-class risk-based classification: Class I (low risk), Class II (moderate risk) and Class III (high risk), based on device function, significance in healthcare decisions and potential harm.
Software targeting individuals as medical devices is, in regulatory terms, called either software-as-a-medical device (SaMD) or software-in-a-medical device (SiMD) (71). If the software is integrated into the hardware (for example an insulin pump), then it is a SiMD and is regulated as part of the complete device. If the software works without physical hardware, it is classified independently as a SaMD (Figure 2).

Venn diagram illustrating when software is a medical device. All software with a medical purpose, such as diagnosis, monitoring, prediction or prognosis, is considered a medical device, whether or not it is coupled with hardware. MD = medical device; SaMD = software as a medical device; SiMD = software in a medical device. *List not exhaustive.
Most SaMDs that provide information used for making diagnostic or therapeutic decisions in the EU will most likely be classified as at least Class IIa (moderate risk) unless the decisions could cause severe harm and then require Class III (high risk). In the USA, the Class-level decisions are risk-based, based on the decisions of potential harm to the patient and experiences with similar devices already on the market.
Artificial intelligence (AI) in apps
SaMD incorporating AI can evolve during the learning process, whereas traditional medical device software operates with fixed algorithms. The dynamic nature of AI introduces challenges to regulatory approval, risk management and transparency. All medical devices using AI in the EU will need to comply not only with the MDR, but also with the Artificial Intelligence Act because AI in health care is considered ‘high-risk AI’. The USA does not have a standalone regulation; however, the FDA integrates AI considerations into its existing medical device regulatory framework. Specifically, they emphasise transparency and explainability.
Quality management system
Both the EU and the USA require a Quality Management System for medical devices. These must comply with ISO13485 (in the USA, known as 21 CFR Part 820). Examples of the required elements are risk management handling (ISO14971) and Post-Market Reporting. Cybersecurity and data protection are also important. In the EU, any device handling personal health information must comply with the General Data Protection Regulation and the Health Insurance Portability and Accountability Act.
Regulatory pathway
In the EU, SaMD classes IIa and above need to be assessed by a Notifying Body, which is an independent certification organisation designated by the European Commission to assess and certify medical devices. The device then obtains a CE mark. Clinical data are required for all Class IIa and typically apply to the interventions included in this review. The extent of the data (real-world data to clinical trials) depends on the risk level of the device. In the USA, this work is centralised by the FDA and can reach the market through one of the three pathways. If a Class II SaMD is similar to existing products on the market, a 510(k) pathway can be used. This typically requires benchmark testing, and no clinical data are available. If no apparent similar devices exist, the ‘De Novo’ pathway must be followed, and whether clinical data is needed is risk-dependent. Premarket approval is required for Class III devices, and clinical data are mandatory.
Future prospects
In light of the limited evidence of internet- and app-delivered headache interventions and the above-mentioned challenges of the developmental process, future efforts should (i) clearly identify and address the intended target and outcome of an intervention; (ii) ensure rigorous feasibility and usability testing to ensure adherence and retention; (iii) demonstrate the efficacy of the intervention through well-designed RCTs; and (iv) appropriately navigate the regulatory landscape. Addressing these challenges will be crucial in creating internet and app interventions that aim to be part of routine clinical headache care.
Such achievements can build a foundation for important value propositions in eHealth and mHealth for headache care. eHealth and mHealth have the potential to be cost-effective, broaden the availability of headache services, improve communication with healthcare providers and are potentially green and sustainable (60). Future efforts should also investigate digital twins, patient portals and real-time monitoring (75), as well as incorporate AI using data captured through internet and app platforms to diagnose, prognosticate, forecast and guide treatment (76,77).
Conclusions
The internet and apps are used to deliver behavioural headache therapies, coordinate management plans, and promote adherence; however, presently, no clear evidence of their efficacy exists. Developing internet- and app-delivered headache interventions depends on rigorous feasibility and usability testing, demonstration of efficacy through robust clinical trials, and appropriate navigation of the regulatory landscape. Only when these challenges are overcome will eHealth and mHealth interventions effectively enhance headache management.
Footnotes
Clinical implications
Author contributions
AS was responsible for conceptualisation, literature search and wrote the manuscript draft. ML was responsible for the literature search and wrote the manuscript draft. ET was responsible for conceptualisation and wrote the manuscript draft. All authors reviewed the manuscript for important intellectual content and approved the final version submitted for publication.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: AS: Co-founder and shareholder of Nordic Brain Tech AS, a company developing the app-based biofeedback device Cerebri. Holds a pending patent for Cerebri. Lecture honoraria from TEVA.
ML: Co-founder and shareholder of Nordic Brain Tech AS, a company developing the app-based biofeedback device Cerebri. Holds a pending patent for Cerebri. Lecture honoraria from Abbvie, Eli Lilly, Lundbeck, Novartis, Pfizer, TEVA.
ET: Co-founder and shareholder of Nordic Brain Tech AS, a company developing the app-based biofeedback device Cerebri. Holds a pending patent for Cerebri. Personal fees for lectures/advisory boards: Novartis, Eli Lilly, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Biogen. Consultant for and owner of stocks and IP in Man & Science. Stocks in Keimon Medical AS. Non-personal research grants from several sources, including Norwegian Research Council, KlinBeForsk, Nordic Innovation. Commissioned research (non-personal) from Lundbeck.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by the Research Council of Norway
Ethical statement
This was a literature review, and ethical approval was not deemed necessary.
